Abstract
The results of uncontrolled, open-label clinical trials indicate that reconstructive cellular therapies have the capacity to produce meaningful functional improvements in patients with brain disorders. However, the transplantation of fetal cells has not progressed to viable best practice treatment for any brain disorder. A conceptual approach, referred to as the Repair Model, has served as a useful heuristic for initiating research in the field and guiding the development of new practices. Analysis of evidence for the treatment of Parkinson's disease indicates that recovery following neural grafting is a complex process influenced by factors beyond the replacement of neurons. An alternative approach, the Composite Brain Model, is outlined to address limitations of the Repair Model. A hierarchical, open-system model is proposed, which aims to track the interactions between the grafted cells, the host brain, and the environment. The Composite Brain Model emphasizes the importance of the interactions between the patient, their physical and social environment, and the provision of rehabilitation during recovery. It is proposed that the Composite Brain Model is useful in providing an alternative perspective for research, theory building, and practice.
Keywords
Introduction
The remarkable scientific discoveries and technical developments in the contemporary biomedical sciences generate strong expectations of therapeutic advances by clinicians, patients, and the community (21). However, many health problems including brain disorders are recalcitrant to emerging treatments and remain unrelenting sources of suffering, disability, and death (55,57). Unless viable new treatments are introduced, projections indicate that the prevalence of degenerative brain disorders such as stroke, Parkinson's disease (PD), and Alzheimer's disease (AD) will increase significantly in the aging populations of developed countries (18,36).
Stem cells are seen as having an enormous potential for treating a host of diseases, including those associated with heart, liver, and pancreatic failure (24). Many of these cell lines are now being developed in preclinical laboratory research and evaluated in phase 1 and 2 clinical trials (5,41). A very promising line of research is the development of cellular therapies for brain disorders; a program that includes the grafting of cells to replace neurons lost through illness or injury (49). The “holy grail” sought by researchers is the discovery of an ideal cell type that, when grafted, will effectively restore the brain to its premorbid form and enable meaningful functional recovery (36,47,52).
After three decades of laboratory and clinical research, cellular therapy has not emerged as “best practice” for any of the targeted brain disorders (2,4,50). There is a degree of scepticism among funding providers regarding the viability of neural reconstruction in producing greater therapeutic benefits than alternative innovate approaches. In the case of PD, there are rival interventions being tested, such as deep brain stimulation (DBS), improved pharmaceutical interventions, and neuroprotection (31,37).
A further challenge to the advancement of neural reconstruction is the problem of the effective translation of preclinical research into safe and effective interventions. Numerous researchers, including Dobkin (21) and Döbrössy and colleagues (23), have examined difficulties in translating evidence from animal models to patients due to differences in anatomy, physiology, neuropathology, and species-specific behaviors. An extension of this analysis is that a key difference between laboratory and clinical studies is the need to account for the experiences and values of the patients living with brain disorders in their physical and social environments. It argued that apart from the biological and technical problems associated with translation, there are also conceptual and methodological inadequacies impeding the progress of clinical research. The aims of the present article are as follows:
to critically evaluate the rationale, strengths, and limitations of the currently used research heuristic, Repair Model;
to propose an alternative “Composite Brain Model” for explaining recovery following neural grafting;
to discuss the potential applications of the alternative model to advance theory and clinical research, particularly the integration of rehabilitation into the practice of reconstructive cellular therapies.
The Repair Model
There are several reasons why neural grafting for PD was selected to provide the evidence for discussing the strengths and limitations of the Repair Model. First, the focal neurological impairment characterizing this disease has been clearly identified as damage to the nigrostriatal dopaminergic system. Although PD is a far more complex disorder than damage to a single neural pathway, the relative specificity of the neuropathology is seen as more amenable to cellular therapies than more diffuse conditions such as Huntington's disease (HD) or Alzheimer's disease (AD) (9,11,45). Second, there are thoroughly investigated animal models that enable preclinical work for ensuring the relatively safe transplantation of cells in PD patients (48). Finally, by the early 2000s, there were publications in peer-reviewed journals reporting on outcomes for more than 300 patients (17,65). Therefore, there is a relatively large body of evidence for enabling the present critical discussions of the recovery process in patients with PD.
Cell replacement therapy, also referred to as reconstructive or restorative neurosurgery, can be seen as a branch of the broader program of transplanting organs, tissues, and cells for patients with a variety of degenerative illnesses or severe injuries. The conceptual framework currently guiding reconstructive neurosurgery is referred to as the “repair” or “reparative” model. The fundamental objective of this approach is to restore the brain to a premorbid state by replacing the lost cells (7). The model suggests that if appropriate replacement cells can be identified, harvested, and correctly positioned, then the impaired brain can be restored to its premorbid, structural, and functional state. Barker (7) proposed that several criteria must be fulfilled if the neural transplant is to “actually” repair the brain, including that all lost cells must be replaced and normal neural circuitry is reestablished.
In addition to describing the desired outcomes, the Repair Model can also be stated as a heuristic for conducting cellular replacement therapy. The six steps illustrated in Table 1 reflect the method by which researchers conduct the practice of neural repair. The apparent simplicity of the process shown in Table 1 does not adequately represent the true challenges and level of expertise required to achieve the desired outcomes for each stage (28).
The Repair Model as Applied to Parkinson's Disease

Finally, the Repair Model can be applied in identifying the parameters for the multiple and interacting variables that produce optimal outcomes. Polgar and colleagues (65) proposed a “path model,” using multiple regression to track the time sequence of the recovery process. The flowchart for the hypothesized sequence of events or “paths” representing the stages of recovery is shown in Figure 1. The first step in the model represents the input variables, including patient characteristics (e.g., age, duration of illness), graft parameters (e.g., cell source, quantity of cells), and surgical protocols (e.g., the targeted nuclei, levels of immunosuppression).
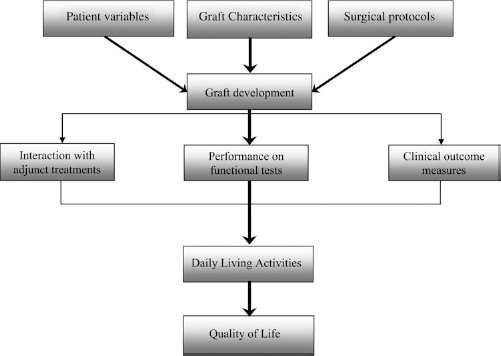
Path model for recovery. Based on Polgar et al. (65).
The next stage of graft development is pivotal as it is assumed that the changes characterizing recovery are directly related to the survival, growth, and functional integration of the grafted cells within the host brain (14). Fluorodopa positron emission tomography (F-18 PET) is most commonly used to evaluate levels of dopamine (DA) turnover (26,58). Tracking the time-dependent associations between increases in DA turnover and clinical improvements shown at subsequent levels of the path model serves as an empirical means for gradually improving therapeutic efficacy. The difficulty with this approach is the inadequacy of the database for conducting meta-analyses including multiple regression analysis. There is a high degree of variability both between and within the results of different research groups; therefore, large sample sizes are required to ensure adequate statistical power (61).
A more realistic approach has been to identify correlations between specific input and recovery variables reported in specific publications (4,7,17,60). There is now a multicenter project, “TransEUro,” which was launched to coordinate research into neural grafting (2). This collaborative project is expected to make best use of current knowledge regarding optimal parameters for implementing control trials for rigorously evaluating the safety and efficacy of neural transplantation for PD (37).
Unlike palliative treatments, such as pharmacotherapy, the replacement of dopaminergic neurons offered the hope of a cure for PD (40). The Repair Model provided a valuable heuristic for initiating the research program for reconstructive cellular therapies. The notion of restoring the brain as closely as possible to its premorbid state was the most logical approach to initiate the research program for neural transplantation.
Limitations of the Repair Model
In the 1990s, the results of a number of uncontrolled, open-label safety and efficacy trials were reported, indicating the survival, growth, and integration of grafted dopaminergic cells and significant improvements in the functioning of patients with PD (35). Preliminary synthesis of the evidence indicated large overall effect sizes on measures of recovery for motor functioning and DA turnover (17,65). However, there were concerns about the validity of the uncontrolled studies that provided the evidentiary bases for estimating efficacy (1). For this reason, there was great anticipation of the results of two double-blind, placebo-controlled randomized trials (RCTs) aiming to establish the efficacy of neural grafting (29,30,54).
The outcomes of the two trials were disappointing and were viewed as a setback to the progress of the neural transplantation program (34). The results obtained in the two RCTs (26,58) did not reflect a reduced effect size due to the subtraction of the scores for the placebo surgical controls, but rather the overall failure of the grafted groups to improve from baseline (63). This outcome is in contrast with the statistically and clinically meaningful improvements found in PD patients following neural grafting. However, benefits were shown in some of the subgroups on select outcome measures such as improvements in patients under 60 on United Parkinson's Disease Rating Scale (UPDRS) (25) or the stabilization of UPDRS scores of patients with four or more grafts per putamen (58). The two RCTs were valuable for drawing attention to the challenges inherent in neural transplantation for PD: most troublingly, the risk of late-onset, off-medication dyskinesias (60).
Relying on the Repair Model as a heuristic has not produced the benefits anticipated 30 years ago (27). A heuristic device is a conceptual tool used to guide a problem-solving process. As such, the Repair Model is not evaluated as true or false, but rather its usefulness is assessed by the extent to which it leads to the desired goals. In accordance with a realist epistemology, a heuristic works because it correctly depicts, at least up to a point, the true nature of the world (16). Therefore, the Repair Model needs to be examined in light of the evidence for recovery processes in patients following neural grafting.
Changed Anatomy
There are numerous studies that demonstrated the survival, growth, and reinnervation of the neostriatum by grafted dopaminergic cells. Postmortem analyses indicated the robust survival of the grafted cells and the dense reinnervation of the targeted striatal regions (4,46,51,58). However, the histopathological studies also disclose the unprecedented anatomical form of the grafted basal ganglia. Up to four embryonic mesencencephalic grafts are distributed in each of the host putamina, resulting in a “blended” structure, which has been referred to as a “nigrostriatum” (64). The decision to graft DA-rich cells into the neostriatum rather than the anatomically appropriate substantia nigra was a justifiable strategy in initiating neural transplantation (6). As Dobkin (21) reflected, the promotion of axonal growth over long distances is quite difficult in the comparatively large human brain. It was more important to ensure that the dopaminergic projections formed appropriate synapses rather than to attempt to recreate the brain in its original form.
It is evident that cellular therapies do not “actually” reconstruct the impaired brain in the sense of restoring it to its premorbid state. Rather, in PD, the grafted brain has a novel anatomical appearance clearly distinguishable from a normal brain by the ectopic location of the new dopaminergic cells (9). It follows that clinical benefits following grafting are not due to the seamless replacement of damaged CNS modules, as assumed under the Repair Model, but rather to the initiation of a process that may lead to the functional reorganization of the impaired brain.
Host Neural System
There is a significant positive correlation between increases in dopaminergic activity and the degree of recovery following grafting (33,35). Secondary data analysis indicated a correlation of ρ = 0.66, p < 0.05, between measures of DA turnover and degree of functional recovery (65). However, there are patients who have surviving grafts, without demonstrating functional recovery (26,34,58). The available evidence indicates that the survival, growth, and effective functioning of the graft are a necessary but insufficient conditions for recovery in PD. Significant increases in striatal dopaminergic activity were not associated with meaningful improvements on outcome measures (58). The researchers concluded that, despite the high level of survival, the DA-rich cells failed to provide benefit to patients (10). Improvements were reported on UPDRS outcomes in younger patients but not in older (over 60) patients even though both groups presented with increased DA turnover (26). These results imply that the state of the host neural system has a decisive influence on the recovery process following transplantation.
The neural degeneration characteristic of PD continues following transplantation. F18-flurodopa PET scans demonstrate continuing decreases of DA in ungrafted striatal regions (13,33). Also, recent postmortem studies indicated that some, but not all, of the grafted cells developed pathological changes characteristic of PD (46,51). Braak and Del Tredici (12) suggested that these pathological changes were likely to be the outcome of interactions between the host neuropathological processes and the transplanted DA cells. Brain disease and injury result in diffuse, multisystem pathology in the neural networks constituting the human brain (9,11). A limitation of the Repair Model is that it is inadequate for representing the complex and continuous interactions between the grafted neurons, the host neural system, and pathological processes.
Neural Development
Functional recovery following neural grafting is understood to be a time-dependent process, as reflected in the multiple postgraft evaluations of patients at regular time intervals up to trial end point (26,58,60). Functional improvements emerge over time, correlated with the growth and integration of the cells with the host neural network (13).
The processes by which the transplanted brains develop as an integrated biological system are unprecedented in human embryonic development (59). The challenge for understanding the recovery process is that the 6.5- to 9-week postgestation cells are implanted into damaged and progressively decaying brains of older patients (28). At the time of surgery, these cells were taken from donors before having undergone the normal stages of development including maturation, synaptic development, and pruning. The postnatal emergence of motor behaviors in infants closely correlates with the development of neural networks, including the corticostriatal and other basal ganglia circuits (42). The formation of cell assemblies is shaped by feedback from infants' movements within the configuration of the environment. It is unclear how these intricate neural networks that develop over a lifetime can be replicated by the insertion of immature cells (58).
At the time of transplantation, the surviving neural networks of the host brain have undergone significant changes in response to injury or disease processes. Neural networks, particularly cortical structures involved in higher perceptual and cognitive functions, have precise, intricate organization. As such, networks involved in learning depend on the development (formation and shedding) of synaptic connections over time (42). In PD, surviving basal ganglia and thalamic neurons undergo substantial changes to compensate for the gradual reduction of dopamine in the neostriatum. It remains to be understood how these disease-induced adaptations influence graft–host interactions and, ultimately, the emergence functional improvements. It is hard to see how the requirement of “recreating all neural circuits” (7) can be realistically achieved.
Unexplored Psychological Changes
The fact that the neural circuitry of reconstructed brains is not “actually” identical to those of healthy brains raises the possibility of new kinds of behaviors and experiences. This possibility is relevant to the issue raised by Moreira and Palladino (54) that the grafting of neurons in one part of the brain could influence neural activity in other regions including those involved in the integration of higher psychological functioning. The graft-induced modifications in higher functions include changes in conscious experience of self and notions of personal identity in human participants. However, there have been no reliable reports of significant long-term changes in the sense of “self” or “identity” in patients with PD following neural grafts. The lack of evidence is consistent with the postcommissural putamen, the region of the neostriatum that is most commonly targeted and primarily functions to control movement and motor learning (28).
This discussion has not come to a satisfactory conclusion in that the quantitative methods associated with the Repair Model may not be sufficiently sensitive for identifying subtle changes in patients' behaviors and experiences. Researchers rely foremost on standardized tests and evaluation protocols for data collection (20). Unexpected behavioral outcomes are reported only when they constitute adverse side effects such as “late-onset, l-DOPA-induced dyskinesias” or transient but clinically significant hallucinations (19,26,59). Therefore, beneficial physical and psychological changes experienced by patients may not be reported by researchers, and thus more subtle changes experienced by patients remain undocumented.
Qualitative data collection such as in-depth interviews would generate preliminary data regarding changes in subjective states and help to clarify apparently irrelevant behaviors (53). The rigorous use of qualitative or interpretive methods has a long history in medicine and health sciences (15,66). For instance, the content and meaning of delusions and hallucinations may provide an additional dimension for understanding outcomes from patients' perspectives.
Environmental Factors
The interaction of neonates with their environments is an essential condition for the postnatal development of functional neural circuitry and adaptive behaviors. Similarly, the posttransplantation environment of the patient may influence the integration of immature cells and the functional reorganization of the brain (21–23). There is now sufficient evidence based on animal studies to hypothesize that exercise, environmental enrichment, and skills training increase neurotrophin levels, enhance the growth of synapses, and improve recovery in a variety of animal models of brain injury (21). However, there is uncertainty about the extent to which tasks and activities that improve recovery in animal models are applicable to designing programs for patients with neural transplants (23). Apart from biological differences, we need to take into consideration that human participants are capable of constructing and interpreting the meaning of their actions and social environments (54). The Repair Model as shown in Figure 1 is not a useful conceptual tool for guiding research to identify the optimal conditions for graft x Environment interactions in humans. It will be argued in the next section that an open-system model is required to take into account the influences of the environment on patient as a person.
The Composite Brain Model
Basic Assumptions
The “Composite Brain Model” is based on the assumption that neural transplantation produces brains that are unprecedented in mammalian and therefore human evolution. The creation of novel anatomical structures and associated neural circuitry changes the physiology of transplanted brains and the actions and experiences of the graft recipients. The following six propositions represent the Composite Brain Model, as first outlined by Polgar and colleagues in 1999 (63):
Composite Brains are constituted of two distinct but interacting neural systems: the host brain and the neural graft. The overall output (i.e., changes in the responses and experiences of patients) is the product of the interaction between the two systems.
The two neural systems are in a dynamic balance: the nature of the balance changing over time. It follows that the Composite Brain is never in a final form; rather, it is continually transforming as the graft and host systems change and interact with each other.
The novel anatomy and physiology of the Composite Brain may result in unforeseen actions and experiences that are not characteristic of persons with either healthy or impaired brains.
The subjective experiences of patients with neural grafts, as revealed through interpretive research, are essential for understanding the psychological and physical changes occurring during the recovery process.
Balance and functional integration between the graft and host neural systems is shaped by the interaction between the graft recipients and their physical and social environments.
There are a number of stages of recovery over time following neural transplantation; the observed behavioral recovery may have different explanations across the stages.
The above propositions can be transformed into a systems model of structures and processes, representing the recovery process (Fig. 2).
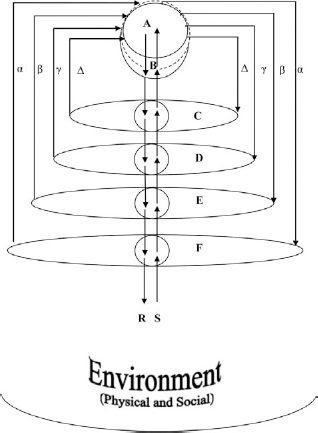
Composite brain model. A, graft; B, target neural structure; C, subsystem incorporating B; D, functional system incorporating system; E, brain as a whole; F, person with neurological disorder; S, physical stimuli/communications; R, responses and actions; α, β γ, Δ, reciprocal connections between graft and the neural system.
Structures
Processes
It is assumed that there are multiple and reciprocal interactions between the different levels of the hierarchical system shown in Figure 2. Changes at all levels of the host CNS are determined by an interaction between the environment and the graft. The following represents some, but by no means the entirety, of the interactions that take place during recovery following transplantation.
Direct Interactions Between the Host and the Grafted Cells
There are multiple reciprocal interactions between the graft (A) and the recipient (F). Somatic interactions (a) include immunological responses and the formation of blood vessels in the brain to supply the new cells. The formation of possible direct connections with the host CNS are represented by the symbols β, γ, and Δ. The proximal relationship between (A) and (B) define the graft milieu. The interactions between the graft and the neurons and glial cells of the host begin immediately after transplantation and have decisive consequences for the efficacy of the procedure. Biological interactions include the release of nerve growth factors, the exchange of neurotransmitters, and the formation of synapses. In the case of PD, the growth of axons in grafted cells (A) form spinal dendrites of intrinsic striatal cells (B).
Interactions Between Multiple Levels
Increased DA input enables the striatal cells to modulate the activity of neurons located in the globus pallidus (C) and subsequently the thalamus and premotor and motor cortex (D). The modified motor output is expected to improve the person's (F) movements (R) in their environment. In time, the neurons of the motor and premotor cortices (D) project axon collaterals to the implanted striatal neurons (A) completing the circuit.
Interaction Between Patient and Environment
“Environment” encompasses both the physical and socially constructed aspects of the patient's context. The physical stimuli and social reinforcers (S) arising from the environment shape the patients' responses and experiences (R) according to the principles of learning. The modification of responses, including both meaningful communications and disordered movements, depends on changes in the recipient (F/E). The organized patterns of neuronal activity of the brain are assumed to be the physical bases for mental activity by contemporary neuropsychologists (42).
Influence of Cortical Activity on Grafted Structure
In the case of transplanting in PD patients, patterns of cortical activity (E) are directly conveyed to the neostriatum (B) and the grafted cells (A) through new projections ((3) and indirect multisynaptic projections through (C). These projections connect with the circuits previously described, providing the basis for hypothetical cell assemblies.
The above examples are a selective overview of the interactive processes initiated by the transplantation of cells and the experiences of the patients. These hypothesized processes are actualized according to the brain disorder being treated and the type and location of the cells selected for grafting. Some of the changes may be stable, depending on learning and neural plasticity, as discussed in the next section.
Applications to Research
The Composite Brain Model serves as a heuristic for generating research problems and questions from a different perspective to the Repair Model.
Constructing New Brains
In postulating that the healthy brain is not, in principle, restored to its original state by neural grafting, the application of the Composite Brain Model encourages looking at alternate ways for creating functional neural circuits for providing best possible outcomes for patients. Mukhida and colleagues (56) suggested that the ectopic placement of grafted dopaminergic neurons do not normalize the overall functioning of the basal ganglia system. An alternative approach to neural reconstruction involves modifying other components of the basal ganglia system. There is strong evidence that lesioning or the high-frequency stimulation (DBS) of nuclei, such as the subthalamic nucleus (STN), is effective for the symptomatic treatment of PD (70). The grafting of inhibitory “GABA”-rich cells in this circuit may produce the best functional outcomes in some subgroups of PD patients (3,57). The Repair Model constrains the research questions that can be asked about reconstructing neural circuits. Optimal anatomical solutions for restoring function may involve the transplantation of cells to locations other than natural sites.
Neural Plasticity
The Composite Brain Model does not assume that brain repair is analogous to replacing damaged parts; rather, neural grafting is the means by which brain plasticity is enhanced (62). This concept covers a variety of mechanisms associated with reorganization of the brain in response to environmental changes and injury (32). The signs and symptoms of brain disorders present when residual brain plasticity is insufficient to compensate for severe damage (62).
As shown in Figure 2, it is assumed that there are multiple and reciprocal interactions between the graft, the host CNS, the host as a person, and the environment. For lasting functional benefits, these interactions depend on long-term changes in the host CNS (38,43). Neural plasticity encompasses a variety of mechanisms (8,41), in particular, responses to environmental change (learning) and reorganization of neural networks following brain damage. Evidence based on animal research indicates that exercise, environmental enrichment, and training directly influence the structural and physiological properties of the mammalian brain (21,43). Kolb and colleagues (41) summarized a wide range of environmental influences including drugs, rewards, social play, and stress that affect the synaptic organization of the normal mammalian brain. In particular, these researchers (41) argue that the long-term improvements following both neural transplantation and neurological rehabilitation depend on the enduring changes observed in neural networks.
As discussed in several reviews (23,39), grafts both receive and extend axonal projections from neurologically relevant structures. Increased environmental complexity directly enhances the development of grafted cells, increasing the density of dendritic spines and the size of the cells and the release of neurotrophic factors (22). The overall results of current research using animal models indicate that transplanted neurons express neural plasticity, which is consistent with synaptic changes associate with learning (23).
A variety of research questions can be generated based on the different types of brain plasticity indicated in Figure 2. Research questions are central to improving efficacy in the timing of these different processes and the ways in which they might be enhanced. Further research is needed to identify markers for age-correlated optimal host brain plasticity. The position taken under the Composite Brain Model is that neural transplantation is not about replacing damaged units, but enhancing neuroplasticity.
Personal Meaning
Research informed by the Repair Model is essentially about cells, impaired brains, and behavioral changes, as measured on standardized tests. While psychological factors are addressed using structured, quantitative assessments, there has been little effort to understand the values and personal experiences of the patients undergoing these problematic, experimental transplantation procedures. The collection of qualitative evidence is essential, as it is the patients themselves who are aware of changes should they emerge postgrafting (61). There is scope for qualitative research, since there are many gaps in the literature.
For example, the selection of primary outcome measures does not take into account the perspectives of the patients or their families. Recently, Politis and colleagues (67) used a structured questionnaire to identify the symptoms of PD that were the most troublesome for patients, which did not always coincide with those measured on standardized scales. Evidence is needed to demonstrate that improvements on objective primary outcome measures are also valued by patients (61).
There is no evidence of how patients interpret the outcome of experimental transplantation procedures. For example, there has been no in-depth debriefing of patients who were assigned to sham-operated, placebo control groups in RCTs lasting over 2 years. Risks associated with participation have been evaluated only in terms of severe physical consequences (25,63,68). The experiences of the participants are relevant to evaluating the benefits and burdens associated with trials performed under utilitarian ethical principles.
A possible reason why patients with grafts enrolled in double-blind RCTs demonstrate weak recovery in comparison to those in open studies (34) is that their posttransplantation status is uncertain. There is very little known about how patients and their families interpret their contribution to the posttransplantation recovery process. Some information such as life changes in individual patients, such as returning to work, are included (33); yet there is no accurate evidence of questions such as, “Should I exercise; how often?”; “Should I consult a physiotherapist and a speech pathologist?” The literature is silent on what people think about what is happening to them and what they should do in order to optimize their recovery.
Neurological Rehabilitation and Reconstructive Therapies
Rehabilitation services are routinely provided for patients who undergo demanding reconstructive or neurosurgical procedures. For example, Tassorelli and colleagues (69) reported that rehabilitation procedures tailored to the patients' problems were useful for reducing adverse side effects of deep brain stimulation for PD. Regardless of the theory used to guide research, there is strong clinical justification for posttransplantation rehabilitation. Döbrössy and colleagues (23) noted that there has been very little progress in the development of specialised rehabilitation processes based on recent evidence. The question that remains is “how should we conduct rehabilitation with patients with neural grafts?” (62).
There are several research groups looking into translating evidence from laboratory research into effective rehabilitation programs for patients with intracerebral or spinal cord grafts (21,23,43). However, there appears to be only one published peer-reviewed RCT reporting on the interaction between rehabilitation and intracerebral transplantation (44). In this trial, 18 patients were randomly assigned; one group (n = 14) was transplanted with manufactured cells (human teratocarcinoma, NT2) and underwent rehabilitation, while the control group (n = 4) was provided with rehabilitation only. There were no statistically or clinically significant benefits reported in this study. However, as no detailed description was provided for the rehabilitation program offered to the patients (23), the results of this RCT are of very limited use.
Döbrössy and colleagues (23) argued that some of the current strategies for conducting neurological rehabilitation are relevant to devising programs suitable for patients with intracerebral transplants. The Composite Brain Model is an appropriate conceptual framework for advancing research and the development of effective practices in this area. The model shown in Figure 2 can be simplified as follows:
graft ↔ Host CNS ↔ Person with graft ↔ Environment
The research question that is generated is how to optimize the above parameters for ensuring the best possible responses in patients undergoing rehabilitation. Rehabilitation is a systematic way of manipulating the patients' experiences and responses to the environment. The patient takes an active role in the recovery process (15). Strategies for effective neurological rehabilitation draw on psychosocial influences, such as the commitment of the patient to the treatment and the support networks available to elicit, shape, and maintain functional improvements. Berlucchi (8) noted that the effectiveness of rehabilitation depends on the active involvement of patients in maintaining motivation and commitment to the aims and outcomes of the program.
The environment includes both physical stimuli and the multitude of interpersonal communications that serve as reinforcers for shaping the patients' behavior. Döbrössy and colleagues (23) suggested that research with animals indicated that patients need to “learn how to use the graft” in order to achieve the highest possible level of recovery. The purpose of rehabilitation is to help the patients to acquire strategies that will enable them to make the best possible use of the graft-induced changes in the plasticity of specific neural networks and the overall plasticity of the brain.
The identification of the functional changes that respond to environmental reinforcers is an important step in program development (23). As discussed previously, in addition to using standardized tests for assessing changes in the signs and symptoms of diagnosed brain disorders, rehabilitation providers also need to identify and negotiate with patients the criteria for realistic behavioral improvements. The central challenge is to design rehabilitation programs that are directly relevant to patients. The best approach to rehabilitation is by no means obvious; current techniques often aim to circumvent neural damage, while the goal of rehabilitation following neural grafting is to engage the damaged system to ensure optimal integration.
The obvious limitation of the present theoretical approach is the lack of evidence for the processes indicated in Figure 2. It is beyond the scope of the present review to provide a detailed outline for rehabilitation programs effectively combining transplantation with rehabilitation. When such programs are implemented, they will reconceptualize the practice of neural reconstruction, with the transplantation of new cells being seen as initiating a sequence of changes in neural plasticity that can be shaped by environmental influences.
Conclusion
The research program driven by the Repair Model focuses on identifying appropriate cell lines and determining the quantity and location required for transplantation. Functional improvements are assessed using standardized tests specific to each brain disorder. While the Repair Model has been very useful, several deficiencies regarding the scientific validity and heuristic value of this approach were identified in the present paper. It is evident that neural grafting does not restore impaired brains to their premorbid states but rather initiates a process for creating novel types of brains. The Composite Brain Model assumes that changes in neural circuitry provide new possibilities for action and experience. The surgical placement of cells is seen as the beginning of a process whereby recovery is mediated by the improved plasticity of the host brain. Neurological rehabilitation is essential for integrating new cells into functional circuits for reinstating optional functioning in patients.
At present, there is no reliable evidence that either the physical and social environment or the actions and experiences of patients have causal effects for improving the recovery process. Therefore, the Composite Brain Model is an alternative perspective—a heuristic for planning research to produce evidence that may lead to a functional systems theory in a biopsychosocial framework.
Footnotes
Acknowledgments
I would like to thank Ms. Joanna Ng, Prof. Greg Murphy, Ms. Melissa Buultjens, and Dr. Leila Karimi for their support in preparing the manuscript. The authors declare no conflict of interest.
