Abstract
Acute ischemic stroke causes a disturbance of neuronal circuitry and disruption of the blood–brain barrier that can lead to functional disabilities. At present, thrombolytic therapy inducing recanalization of the occluded vessels in the cerebral infarcted area is a commonly used therapeutic strategy. However, only a minority of patients have timely access to this kind of therapy. Recently, neural stem cells (NSCs) as therapy for stroke have been developed in preclinical studies. NSCs are harbored in the subventricular zone (SVZ) as well as the subgranular zone of the brain. The microenvironment in the SVZ, including intercellular interactions, extracellular matrix proteins, and soluble factors, can promote NSC proliferation, self-renewal, and multipotency. Endogenous neurogenesis responds to insults of ischemic stroke supporting the existence of remarkable plasticity in the mammalian brain. Homing and integration of NSCs to the sites of damaged brain tissue are complex morphological and physiological processes. This review provides an update on current preclinical cell therapies for stroke, focusing on neurogenesis in the SVZ and dentate gyrus and on recruitment cues that promote NSC homing and integration to the site of the damaged brain.
Introduction
Stroke is one of the leading causes of death worldwide, but approximately two thirds of stroke victims survive (17,83). Neural cell dysfunction in the stroke brain region will cause functional disability. Although stroke is a major cause of death, the greatest health and social burden of stroke is disability (41). Thrombolytic treatment using recombinant tissue plasminogen activator has significantly improved the survival. However, only a small proportion of patients can be rescued by thrombolytic treatments due to a limited treatment window (139). It is known that natural neuronal regeneration can occur in response to brain damage, but in general, neuronal proliferation usually fails to repair the damaged brain completely. Therefore, development of new strategies for treating stroke is essential, and stem cell therapy has shown promising preclinical results (44).
Stem cells have, uniquely, the capacity for endless self-renewal, the potential to differentiate, and depending on their origin, the ability to repopulate various organs with multiple cell types (28). Neural stem cells (NSCs) show tropism to sites of pathology in ischemic stroke (102,124). These remarkable abilities make NSCs an optimal tool for the treatment of stroke that requires a regenerative approach, either by integration into damaged tissue and replacement of missing cells or as delivery vehicles for drugs, growth factors, or functional proteins.
NSC therapy for ischemic stroke can be categorized into endogenous and exogenous types. The mechanisms of NSC migration and homing to the injury site are basic to the application of this therapeutic strategy in neurodegenerative disease. Here, we discuss the features of the normal neurogenic niche in the adult mammalian sub ventricular zone (SVZ) as well as homing and integration of the NSCs.
Presence of NSCs in the Adult Brain
It was once thought that neurogenesis occurred only during embryonic development and that the adult central nervous system (CNS) could not generate new neurons (44). However, extensive studies in recent years have shown that this is not true (130). In fact, neurogenesis can occur in two regions of the adult CNS: the SVZ and the subgranular zone of the dentate gyrus (31,37). The progenitor cells of the SVZ region and the subgranular zone of the dentate gyrus can proliferate, migrate, and differentiate into neurons, astrocytes, and oligodendrocytes. The NSCs derived from these zones show the long-term capacity for self-renewal and multipotency. Additionally, NSCs can also be found in other regions of the brain and peripheral nervous system such as the striatum, spinal cord, neocortex, and the adult human olfactory bulb (95,96,138).
Subventricular Zone and Neural Stem Cells
NSCs have the ability to self-renew, proliferate, and generate multiple cellular lineages both in vitro and in vivo (65). NSCs are present in the CNS throughout life in the SVZ of the adult human brain (108,109). In the SVZ, cells that express glial fibrillary astrocytic protein (GFAP) constitute the genuine NSCs (30). The niche comprises cellular structures, extracellular matrix proteins, and soluble factors (5). The SVZ is one of the main neural stem cell niches.
Subventricular Zone (SVZ)
The SVZ is located on the borders of the lateral ventricles of the adult mammalian brain (3). A layer of ependymal cell lines the ventricular surface and consists of E1 cells, with multiple basal bodies and multiple long cilia, and E2 cells, with two cilia and complex basal bodies that may act as mechanical or chemical sensors monitoring the composition and flow of cerebrospinal fluid (CSF) (88).
The human SVZ is quite different from that of other species because of the presence of a hypocellular gap and a prominent “astrocytic ribbon” in the lateral wall of the lateral ventricles (110). Cell–cell interactions occur in the wall of the lateral ventricle, in the fashion of a “pinwheel architecture,” which refers to the organization of the ependymal lining where processes of astrocytes, in contact with ventricles, are surrounded by ependymal cells (88). However, this organization is not present in the ventricular wall of nonneurogenic regions, suggesting an important role for this intercellular interaction and for contact with the CSF in neurogenesis.
NSCs in the adult rodent SVZ are present in a vascular niche, where they interact closely with blood vessels and with other cells (88,119). Endothelial cells comprise vascular structures that contribute to the SVZ neurogenic niche by releasing soluble factors such as pigment epithelium-derived factor (PEDF), brain-derived neurotrophic factor (BDNF), erythropoietin (EPO), and vascular endothelial growth factor (VEGF) (Fig. 1) (63,70,107,112,120). Extracellular matrix proteins also play an important role in maintaining the SVZ neurogenic niche. Tenascin C, laminin, and collagen type 1 are expressed in the extracellular matrix of the SVZ (Fig. 1) (40,84). These extracellular matrix structures may concentrate growth factors and may be important in the regulation of the microenvironment in the SVZ (84).

Schematic of subventricular zone (SVZ) and dentate gyrus neural progenitor cell (NPC) neurogenesis in the adult brain. (1) Growth factors such as epidermal growth factor (EGF), basic fibroblast growth factor (bFGF), and hepatocyte growth factor (HGF) contribute to the maintenance of self-renewal and proliferation of neural stem cells (NSCs) in the SVZ. (2) Contribution to the SVZ neurogenic niche by endothelial cells composing vascular structures are soluble factors such as pigment epithelium-derived factor (PEDF), brain-derived neurotrophic factor (BDNF), erythropoietin (EPO), and vascular endothelial growth factor (VEGF). (3) Extracellular matrix proteins also play an important role in maintaining the SVZ neurogenic niche. Tenascin C, laminin, and collagen type 1 are expressed in the extracellular matrix of the SVZ. (4) Notch 1 is required for maintenance of the reservoir of adult hippocampal stem cells. (5) CNS injury where factors such as stromal cell-derived factor-1 (SDF-1), leukemic inhibitory factor (LIF), and interleukin (IL)-6 are overexpressed. These factors are important for NSC recruitment and homing.
Soluble factors participate in the maintenance of the SVZ neurogenic niche. Ependymal cells produce noggin, which regulates NSC differentiation (73). Growth factors are important players in the regulation of NSC proliferation and self-renewal. Epidermal growth factor (EGF), basic fibroblast growth factor (bFGF), and hepatocyte growth factor (HGF) contribute to the maintenance of self-renewal and proliferation of NSCs in the SVZ (Fig. 1) (42,91,113).
Dentate Gyrus and NSCs
The presence of NSCs has also been confirmed in the subgranular zone (SGZ) at the dentate gyrus–hilus interface (61,62,100) and is linked with mood and hippocampal function (32,86). Neurogenesis persists in the hippocampus of rats up to 11 months of age (16). However, hippocampal neurogenesis is drastically reduced in aged rats. The neuronal markers NeuN and calbindin-D28k are expressed in NSCs migrating into the SGZ. Stem cells can be isolated from hippocampus-adjacent regions of the subependyma, but the adult dentate gyrus does not contain a population of resident neural stem cells. Indeed, although the lateral ventricle and other ventricular subependymal regions directly adjacent to the hippocampus contain neural stem cells that exhibit long-term self-renewal and multipotentiality, separate neuronal and glial progenitors with limited self-renewal capacity are present in the adult dentate gyrus (118). However, other researchers found considerable capacity for self-renewal and multipotential progenitors in the dentate gyrus (39,97,98).
Adult neurogenesis is dynamic and is thought to consist of proliferation of nestin-expressing NSCs, transiently amplifying progenitors (TAPs), maturation of doublecortin (DCX)-expressing neuroblasts, and survival of adult-born neurons, which integrate into hippocampal circuitry (33,55). Each stage is discretely regulated by a variety of intrinsic and extrinsic factors, and modulation of adult SGZ neurogenesis by myriad stimuli is the focus of intense research (34,144). However, interaction between NSCs, TAPs, and the neurogenic microenvironment or neurogenic niche needs to be clarified. Recently, it has been reported that Notch 1 is required for maintenance of the reservoir of adult hippocampal stem cells (Fig. 1) (2).
Source of Neural Stem Cells
The availability of NSCs is the first consideration in treatment of neurological diseases. Stem cell therapy for ischemic stroke can be categorized into endogenous and exogenous types.
Endogenous Neural Stem Cells
Multipotent NSCs can be isolated from the adult rodent CNS and cultivated in vitro (113,136) as well as in adult human brain tissue. Autologous transplantation of NSCs is a potential therapy for stroke. In a rat model of spinal cord injury, autologous transplantation of NSCs derived from SVZ was found to be feasible (103). However, there is very little experimental data regarding adult NSC transplantation in models of stroke. One study investigated the effects of adult-derived exogenous NSC transplantation in the cortex of ischemic stroke in adult rats (143). The researchers used histology and magnetic resonance imaging (MRI) to analyze the transplanted NSC survival and migration, and they observed some functional recovery. In another study of embolic stroke, neural progenitor cells isolated from the SVZ of the adult rat were labeled by superparamagnetic particles and intracisternally transplanted into the adult rat 48 h after stroke (51). It was found that MRI could identify the cerebral tissue destined to undergo angiogenesis after cell therapy. Studies show that new neurons are produced in the SVZ after a stroke (7,60). New cells migrate and integrate into the damaged tissue, expressing neuronal markers and acquiring a morphological phenotype that resembles neurons after experimental occlusion of the middle cerebral artery (7). Other studies have shown an increase in neurogenesis after ischemic stroke (99,137,142). These studies provide evidence of the regenerative capacity present in the injured brain, but this potential does not seem to be sufficient and different manipulations may be necessary to potentiate endogenous regeneration.
Manipulation of endogenous neurogenesis and “encouragement” of migration to sites of injury are important in the therapeutic use of endogenous NSCs. Chemotactic and humoral factors produced during disease serve as homing cues that cause NSC migration to the site of injury (35). Further studies are needed to exploit the endogenous regenerative ability of the mammalian brain.
Exogenous Neural Stem Cells
Exogenous NSC therapy for stroke is used primarily to reconstruct destroyed neuronal circuits. These cell grafts may exert a neuroprotective effect by secreting survival-promoting neurotrophic factors either intrinsically or after the introduction of therapeutic transgenes (44).
NSCs can be obtained from multiple tissues—skin (64,71,133), embryonic stem cells (36), embryonic NSCs (140), bone marrow and adipose-derived mesenchymal stem cells (27,38,123), fetal stem cells (26), peripheral blood stem cells (122), olfactory ensheathing cells (125), induced pluripotent stem cells (6,19), and fetal and adult nervous systems (4,116,135).
Genetic manipulation of isolated NSCs may be necessary to obtain sufficient numbers of stem cells and achieve feasibility in NSC-based therapy (94). Telomerase reverse transcriptase (TERT) and myelocytomatosis viral oncogene homolog (Myc) were used to establish NSC lines (56,115). However, Myc could cause genetic instability of an established cell line.
It is essential to thoroughly characterize a cell line before starting human treatment, since isolated NSCs can become tumorigenic after serial passaging and transplantation (121,126). With further research, it may become possible to develop reliable and thoroughly characterized NSC lines for use in the treatment of neurodegenerative diseases.
Neural Stem Cell Homing and Recruitment
How NSCs home to injury sites is important for cell-based regenerative therapy. NSCs are highly migratory and seem to be attracted to injured brain areas such as ischemic regions (20). While stem cell homing to bone marrow has been widely studied (89), the molecular basis of stem cell pathotrophism is not well understood. Further identification of the mechanisms involved would pave the way for the development of treatments to enhance endogenous mobilization of stem cells in disease states, perhaps using small molecules. NSCs express a wide variety of receptors that may enable them to respond to many chemotactic signals that emanate from brain pathologies. Chemokine and cytokine production is a common feature of many brain lesions, including stroke, which suggests that these factors could be important in mediating the responses of stem cells to injuries.
Inflammation is a key player in the homing and recruitment of NSCs to sites of CNS injury where factors such as stromal cell-derived factor-1 (SDF-1), leukemic inhibitory factor (LIF), and interleukin (IL)-6 are overexpressed (14,46,124,128) (Fig. 1).
SDF-1 and CXC Chemokine Receptor-4 (CXCR4)
Mutual, reciprocal SDF-1/CXCR4 interaction in NSCs and the ischemic stroke region has been reported (46,85,114,131). During steady-state homeostasis, CXCR4 is expressed by hematopoietic cells and also by stromal cells, which are the main source for SDF-1 in the bone marrow (BM). Stress will increase SDF-1 and CXCR4 levels, which stimulates recruitment of immature and maturing leukocytes from the BM reservoir to damaged organs as part of host defense and repair mechanisms (1,57). Astrocytes and endothelial cells of an ischemic area in the brain increase the expression of SDF-1 and trigger constitutive expression and activation of CXCR4, the receptor for SDF-1, resulting in NSC migration toward ischemic brain explants (49). Exposure to SDF-1 enhances migration and proliferation of Lewis x [stage-specific embryonic antigen 1 (SSEA-1); LeX]-positive NSCs, which can differentiate into cells with neuronal phenotypes after transplantation (22). SDF-1 is upregulated in traumatic brain injury (50), suggesting a recruiting mechanism for NSCs to traumatic lesions in the brain.
Leukemic Inhibitory Factor and Interleukin-6
LIF is a member of the IL-6 cytokine family, which also includes ciliary neurotrophic factor (CNTF) and IL-6, among others (13,45). In the nervous system, endogenous LIF expression is very low under normal physiological conditions, but it is systematically induced following ischemia (23,128). Relevance of this rise of LIF has been emphasized in the regenerating adult olfactory epithelium where it is necessary for the lesion-induced proliferation of neuronal progenitors (15). Exogenous LIF will promote NSC self-renewal in vivo (14), suggesting that the very rapid and transient overexpression of endogenous LIF observed after lesion could constitute a signal that recruits and amplifies NSCs, making their regenerative potential available for brain repair. Similar results have been obtained with IL-6, another inflammatory cytokine, which decreases the proliferation of neuroblasts and enhances NSC self-renewal, as demonstrated in a transgenic mouse model of IL-6 overexpression (134). IL-6 may also be involved in the differentiation of NSCs into neurons (12,13).
Integrins
Integrins are heterodimeric transmembrane molecules consisting of α and β subunits that mediate cell adhesion and migration (21,48). Homing of progenitor cells such as NSCs to ischemic tissue may follow the paradigm of mature leukocytes migrating to inflammatory tissue (29,82,106,119). We have shown that peripheral blood stem cell (PBSC) intracerebral transplantation can significantly improve neurological function following chronic cerebral ischemia in rats, accompanied by increased local cortical cerebral blood flow and β1-integrin expression in the ischemic hemisphere (122). The neurological improvement in this study was blocked by β1-integrin inhibitor (synthetic RDG peptide) (26,122). These results reveal that β1-integrin is necessary for neuroplasticity after intracerebral stem cell transplantation, possibly through the enhanced angiogenesis by homing of stem cells to ischemic sites (26,72,79,122).
Whether β1-integrins play the same cellular role or whether different cell types use distinct mechanisms for homing remains to be determined. Further studies are needed to elucidate whether there is a synergism between other adhesion molecules and their counterligands in the multistep recruitment of NSCs to ischemic tissue (72,79). Modulation of these integrins may provide novel opportunities for treating cerebral ischemic disease (Fig. 1).
Neural Stem Cell Therapy for Stroke
Extensive ischemic injury is a neurological disorder caused by multiple factors such as hypoxia, which is a common cause of neurological disability in adults and children. This disorder is characterized by extensive damage of cerebral parenchyma, resulting in formation of a cystic cavity and consequently loss of neural cells and their connections. It leads to the death of multiple neuron types, as well as oligodendrocytes, astrocytes, and endothelial cells (75). Neuronal plasticity and reorganization of neural circuitries contribute to varying degrees of spontaneous recovery, but the majority of patients exhibit persistent motor, sensory or cognitive impairments. At present, there are no effective clinical therapies.
Many preclinical studies have used NSCs to treat ischemic stroke animal models. Most animal models of ischemic stroke entail occlusion of arterial blood supply to the brain by surgical techniques (26,122). Researchers have used these models to study the response of endogenous neurogenesis to stroke and found that hypoxic injury to the brain leads to increased neurogenesis in the SVZ (7,52,99). Moreover, studies of the cues that increase neurogenesis and migration of these NSCs to the site of injury showed that SDF-1 and angiopoietin-1 contribute to the homing process of newly formed neural progenitors to the site of injury (92). Stroke-induced neurogenesis is maintained in the aged rat brain (24). There is also evidence in humans for enhanced SVZ cell proliferation and neuroblast formation after stroke (54,81,87). In mice, ependymal cells lining the lateral ventricle participate in the neurogenic response to stroke by producing new neuroblasts, but their survival is poor (18).
Different stem cells and their derivatives of rodent and human origin can survive, differentiate into neurons, and restore function after transplantation in the stroke-damaged rodent brain (9,78). Intracerebral implantation of NSCs after ischemia is a feasible method of administration of regenerative therapy (53,141).
Genetically modified [overexpression of BDNF, nerve growth factor (NGF)] NSCs also have been shown to improve function in a mouse stroke model (68,127). In addition, overexpression of either VEGF or the antiapoptotic factor v-akt murine thymoma viral oncogene homolog 1 (Akt1) in human NSCs promotes angiogenesis and increases neuronal survival, respectively, enhancing the functional improvements in stroke-damaged mice (66,67).
Intravenously administered NSCs, previously sorted for the expression of a surface antigen CD49d, home to ischemic lesions in the brain, and thus improve functional recovery after ischemia (43). Intravenously administered human mesenchymal stem cells (MSCs) have been found to reduce stroke-induced deficits in rats, most likely by inducing angiogenesis and improving cerebral blood flow (93). Human embryonic stem (ES) cell-derived MSCs injected intravenously in rats have been shown to migrate to the infarct area, express neuronal and endothelial cell markers, provide neuroprotection, and improve recovery (76). Mouse NSCs delivered intravenously 3 days after stroke in mice have been reported to suppress inflammation and glial scar formation and give rise to delayed neuroprotection and improved functional recovery, starting 18 days after the insult (10). These findings suggest that there is an extended time window for neuroprotection using NSCs.
To study the different therapeutic effects of the administration of embryonic versus adult NSCs, rats were subjected to middle cerebral artery occlusion treated with either type of stem cell. It was found that ESCs survive longer and in larger proportions than adult NSCs and that the administration of either cell line decreased the infarct size (129). Transplanted human NSCs can survive and differentiate into neurons that express different neuronal markers (25). Taken together, these data suggest that NSC transplantation is a potential regenerative therapy for stroke.
Clinical Trials of Stem Cell Transplantation for Stroke
Currently, stem cell therapy in stroke patients is in its infancy. Several small clinical trials with delivery of stem cells in stroke have been completed (78). Slight improvements were detected in some patients after cells from an immortalized human teratocarcinoma cell line were implanted into ischemic or hemorrhagic infarcts in the basal ganglia and cerebral cortex (58,90). However, no substantial clinical improvements were detected after intravenous injection of autologous MSCs in ischemic patients (11). Several clinical trials using intravenous or intra-arterial infusion of autologous bone marrow-derived and other stem cells in stroke patients are ongoing or planned (www.clinicaltrials.gov). One trial will use conditionally immortalized NSCs isolated from human fetal cortex to treat stroke patients. Conditionally immortalized NSCs have been shown to decrease motor impairments in a rat stroke model (105), possibly by promoting angiogenesis and improving cerebral blood flow.
In summary, these small initial human studies cannot be comparable due to differences in target population, type of cells, timing of injection and mode of delivery, but they indicate that stem cell therapy may be technically feasible in stroke patients. Side effects were observed in these clinical trials (58,111,117); therefore, safety is still a principle concern. Epilepsy, risk of bleeding or thrombosis at the site of injection and risk of malignant transformation are the major adverse effects that have been observed. No tumorigenic potential of the cells has so far been observed in two clinical trials (11,59).
Summary
The process of neurogenesis in the mammalian brain and different interactions in the SVZ and dentate gyrus have been revealed in animal studies. This article illustrated the recruitment and integration of NSCs in the stroke mammalian brain. Cell–cell interaction and cell–extracellular matrix interactions are critical to this complex process, including proliferation, differentiation, and migration of NSCs. Growth factors and cytokines contribute to the proliferation, homing, and recruitment of NSCs in the diseased brain.
Future Directions of Neural Stem Cell Therapy for Stroke
Stem cell research could lead to the development of radical new therapies for neurodegenerative diseases that currently lack effective treatments. Over the past few years, there has been continuous progress in developing different kinds of NSCs derived from different origins. Patient-specific cells that may be useful for transplantation can now be produced from induced pluripotent stem (iPS) cells (101). Also, NSCs in the adult brain generate new neurons in response to neurodegeneration (74,142). It has become clear that the characteristics of the pathological environment, such as inflammation, play an important role in the survival, differentiation, and function of both grafted and endogenous cells (47,77,132). Besides cell replacement, stem cells are known to lead to improvements that could also be of clinical value through immunomodulation, trophic actions, neuroprotection and angiogenesis (66,67,69,93,104). Therapeutic approaches using stem cells mainly for neuroprotection by supplying neurotrophic molecules or modulating inflammation will most likely be applied soon in stroke models. The spectrum of stem cell therapy for stroke is illustrated in Figure 2. However, the use of clinical pearls of stem cells to treat stroke will require more basic research so that the mechanisms regulating the proliferation, migration, differentiation, survival, and function of stem cells and their derivatives are better understood and can be effectively controlled.
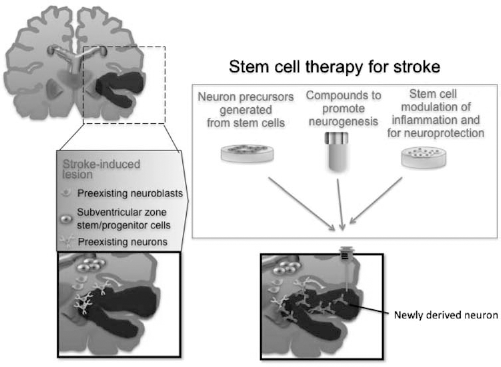
Stem cell therapy for stroke. Ischemic stroke leads to the death of multiple neuronal types, as well as astrocytes, oligodendrocytes, and endothelial cells in the cortex and subcortical regions. Stem cell therapy could be used to recovery damaged neural circuitry by transplanting stem cell-derived neuron precursors. Additionally, compounds could be infused that would promote neurogenesis from endogenous SVZ neural stem cells or stem cells could be injected systemically for modulation of inflammation and neuroprotection.
Once researchers have identified a reliable and consistent source of NSCs, it will be essential to determine whether transplanted cells can integrate into the normal anatomy of the damaged area. Research has shown that NSCs transplanted into rodent brains are capable of integration into neuronal circuitry and have the phenotypical and electrical properties of neurons (8,80). Before clinical application of these cells, preclinical evidence of efficacy and safety of various sources of NSCs is needed. Most stroke patients have few or no therapeutic options and are prepared to receive any new treatments. Harnessing the full potential of NSCs requires much more insight into the mechanisms behind regeneration. Clinical translation of stem cell research into treatment applicable to stroke patients will require the collaboration of scientists and clinicians.
Footnotes
Acknowledgments
This work was supported in part by grants from the Chen-Han Foundation for Education, Taiwan Department of Health Clinical Trial and Research Center of Excellence (DOH99-TD-B-111-004), the Topnotch Stroke Research Center (China Medical University, CMU98-CT-24), the National Science Council (NSC97-2314-B-039-036-MY3, Taiwan), China Medical University (DMR-98-120), and Buddhist Tzu Chi General Hospital (TCRD 99-12). The authors declare no conflict of interest.
