Abstract
Prognosis of patients with heart failure remains poor despite improved conventional and interventional treatment regimens. The improvement of neovascularization and repair processes by administration of bone marrow-derived cells modestly improved the recovery after acute myocardial infarction. However, circulating patient-derived cells are reduced in number and function particularly in chronic heart failure. Therefore, we tested the hypothesis whether the mobilization of circulating mononuclear proangiogenic cells (CPCs) by G-CSF may overcome some of these limitations. In the present pilot study, 32 patients with at least 3-month-old myocardial infarction were randomized to G-CSF alone (G-CSF group) or intracoronary infusion of G-CSF-mobilized and cultured CPCs into the infarct-related artery (G-CSF/CPC group). Primary endpoint of the study was safety. Efficacy parameters included serial assessment of LV function, NT-proBNP levels, and cardiopulmonary exercise testing. G-CSF effectively mobilized circulating CD34+CD45+ cells after 5 days in all patients (408 ± 64%) without serious adverse events. At 3 months, NYHA class and global LV function did not show significant improvements in both treatment groups (G-CSF: ΔLVEF 1.6 ± 2.4%; p = 0.10; G-CSF/CPC: ΔLVEF 1.4 ± 4.1%; p = 0.16). In contrast, target area contractility improved significantly in the G-CSF/CPC group. During 5-year follow-up, one patient died after rehospitalization for worsening heart failure. Eleven patients underwent further revascularization procedures. NT-proBNP levels, cardiopulmonary exercise capacity, and NYHA class remained stable in both treatment groups. The results from our pilot trial indicate that administration of G-CSF alone or G-CSF-mobilized and cultured CPCs can be performed safely in patients with chronic ischemic heart disease. However, only minor effects on LV function, NT-proBNP levels, and NYHA classification were observed during follow-up, suggesting that the enhancement of CPCs by G-CSF alone does not substantially improve intracoronary cell therapy effects in patients with chronic ischemic heart failure.
Keywords
Introduction
Cell-based regenerative approaches offer an attractive option to beneficially interfere with impaired cardiac function, adverse cardiac remodeling and progression of congestive heart failure (23,32). Although most clinical studies published so far used autologous bone marrow-derived cells in patients with acute myocardial infarction or ischemic cardiomyopathy, the harvest of circulating mononuclear proangiogenic cells is an attractive alternative. However, previous studies have also demonstrated that the number and the functional activity of circulating mononuclear proangiogenic cells (CPCs), the formerly so-called endothelial progenitor cells (EPCs), are reduced in patients with coronary artery disease and chronic ischemic heart failure (22,31). Moreover, the function of CPCs was reduced in patients at risk for coronary artery disease or ischemic heart failure such as diabetic patients (28,30). These findings might contribute to the limited efficiency of cell therapy with CPCs in the setting of chronic ischemic heart failure, besides intrinsic effects of the modes and devices used for cell administration to the myocardium (13,27).
Experimental studies have identified several tools to enhance cell functionality and to increase the number of circulating cells by mobilizing factors [for review, see Brunner et al (5)]. One well-established mobilizing cytokine is the granulocyte colony-stimulating factor (G-CSF), which has been used safely for years not only in patients with hematological malignancies but also in healthy bone marrow donors (6,14).
Based on experimental studies showing a favorable effect of G-CSF on infarct healing, neovascularization, and remodeling in animal models of acute myocardial infarction, G-CSF was the first mobilizing cytokine to be investigated in patients with acute myocardial infarction (9,10,25). However, G-CSF as stand-alone therapy without isolation and reinfusion of circulating mobilized cells has not provided significant benefits on left ventricular performance following acute myocardial infarction, although some trends were seen in subgroup analyses (e.g., large infarct size, early treatment) (34). Nevertheless, recent meta-analyses of all available data at least suggest that G-CSF can be applied safely in patients with coronary artery disease (1,7), although initial studies raised safety concerns in patients with cardiovascular disease (19).
Currently, there are no data available investigating the long-term safety and efficacy of G-CSF administration in patients with chronic ischemic heart disease. In addition, the combination of G-CSF mobilization and subsequent intracoronary infusion in the setting of chronic heart failure has been investigated only in few studies so far (11,21), and a long-term follow-up has not been reported yet. Therefore, the aim of the present study was to compare the safety, short- and long-term efficacy of G-CSF administration alone with the combination of G-CSF administration and intracoronary reinfusion of G-CSF mobilized and cultured circulating mononuclear cells in patients with chronic postinfarction heart failure.
Patients and Methods
Patients
Between September 2002 and March 2004, a total of 32 patients with coronary artery disease and a previous myocardial infarction at least 3 months prior to cell therapy with a well-demarcated left ventricular (LV) regional wall motion abnormality were enrolled into this pilot study. Patients had to be at least 18 years old, suffering from chronic ischemic heart disease, and receiving constant state-of-the-art pharmacotherapy for at least 3 months prior to enrolment. Exclusion criteria were acutely decompensated heart failure, advanced renal failure, chronic inflammatory diseases, and documented or suspected hematological diseases or malignancies. The ethics review board of the Goethe-University of Frankfurt/Main, Germany, approved the protocol (file number 159-02). The study was conducted in accordance with the Declaration of Helsinki. Written informed consent was obtained from each patient.
Study Design
The study flowchart of this single center study included a 5-day course of G-CSF subcutaneous injections (5 or 10 μg/kg) after 1:2 randomization for G-CSF stimulation alone (n = 9, G-CSF group) or G-CSF stimulation and consecutive intracoronary reinfusion (n = 23) of the mobilized and cultivated CPCs into the coronary artery supplying the most dyskinetic LV area (G-CSF/CPC group). Coronary angiography and LV angiography were performed in both groups at baseline and after 3 months, with concomitant percutaneous coronary intervention (PCI) if indicated (Tables 1 and 2). Due to the initially raised safety concerns at the time of study initiation, we performed a dose escalation of G-CSF and carefully monitored putative adverse effects. Thus, the first 12 patients were treated with the lower dose of 5 μg/kg G-CSF. Since this was well tolerated and no unexpected adverse side effects were observed, the following 20 patients received high-dose G-CSF injections.
Patient Characteristics
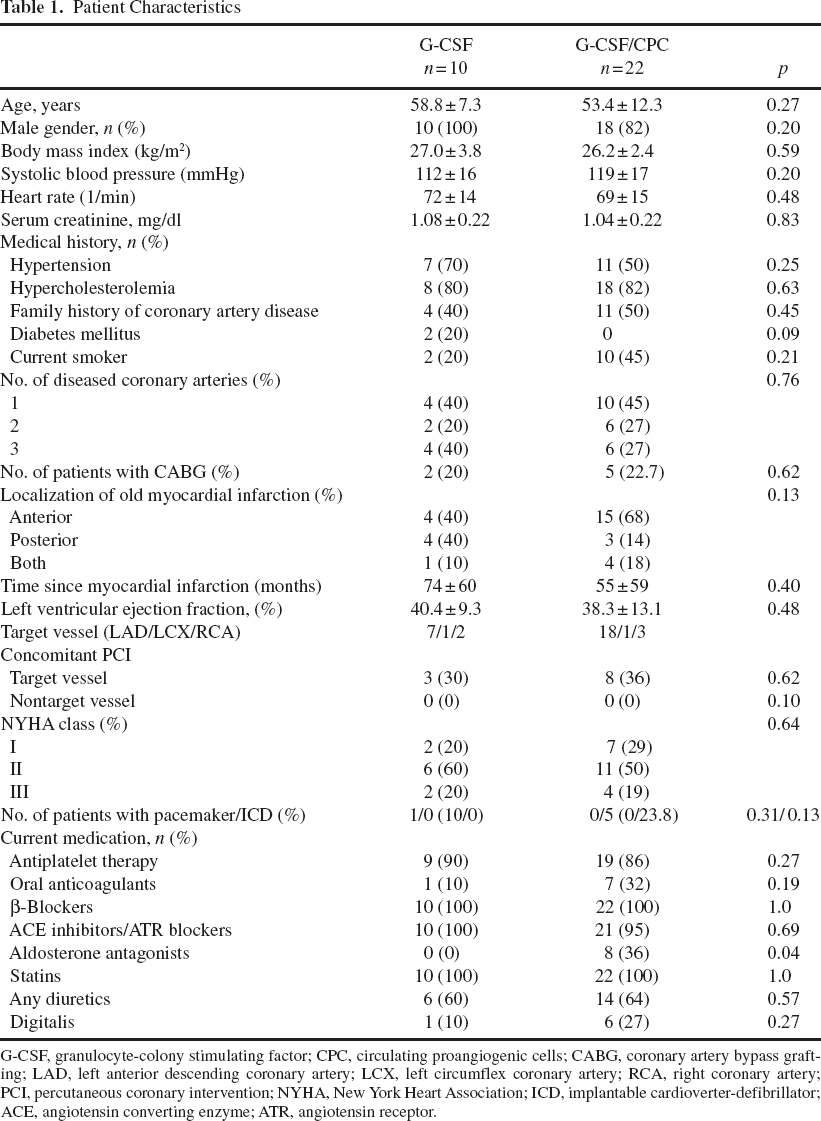
G-CSF, granulocyte-colony stimulating factor; CPC, circulating proangiogenic cells; CABG, coronary artery bypass grafting; LAD, left anterior descending coronary artery; LCX, left circumflex coronary artery; RCA, right coronary artery; PCI, percutaneous coronary intervention; NYHA, New York Heart Association; ICD, implantable cardioverter-defibrillator; ACE, angiotensin converting enzyme; ATR, angiotensin receptor.
Cumulative Clinical Events During 5-Year Follow-Up
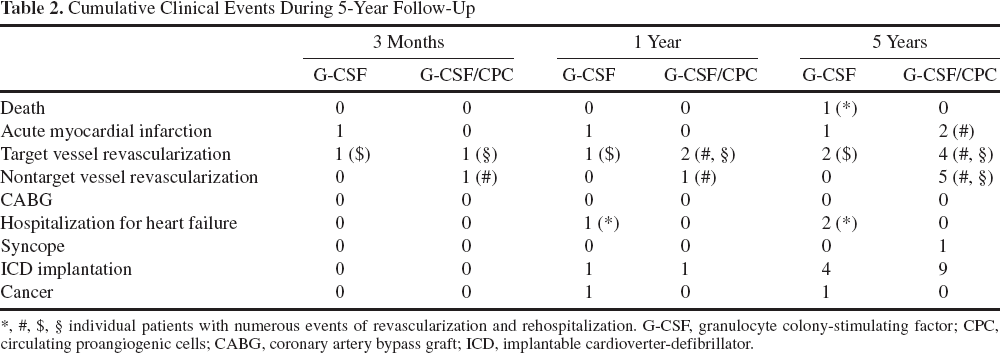
individual patients with numerous events of revascularization and rehospitalization. G-CSF, granulocyte colony-stimulating factor; CPC, circulating proangiogenic cells; CABG, coronary artery bypass graft; ICD, implantable cardioverter-defibrillator.
Primary endpoints of this pilot trial were safety and feasibility, and secondary endpoints included global and regional LV function and volumes after 3 months, determined by both LV angiography and magnetic resonance imaging (MRI). Clinical parameters like functional New York Heart Association (NYHA) class, cardiopulmonary exercise testing, and N-terminal prohormone of brain natriuretic peptide (NT-proBNP) serum levels were obtained during a 5-year follow-up period. There was no control- or placebo-treated group. Figure 1 illustrates the study course.

Flowchart of the study. Patients were randomized 1:2 to stimulation with granulocyte colony-stimulating factor (G-CSF) alone or G-CSF stimulation and reinfusion of mobilized, ex vivo cultured circulating proangiogenic cells (CPCs). Both groups underwent left ventricular (LV) angiography at baseline after G-CSF treatment and 3 months later. Because of safety concerns, the first 12 patients received lower dosages of G-CSF (5 μg/kg bodyweight). After reassessment of data, the following 20 patients received 10 μg/kg bodyweight G-CSF.
G-CSF Stimulation and Fluorescence-Activated Cell Sorting (FACS)
During the 5-day period of G-CSF administration, daily physical examinations and electrocardiography (ECG) were performed. Leukocyte counts and cells for fluorescence-activated cell sorting (FACS) were obtained at day 0, prior to the first dose until the last day of G-CSF stimulation. On day 0 and day 5 of stimulation, a volume of 100 μl peripheral blood was incubated for 15 min in the dark with monoclonal antibodies against CD34 (Becton Dickinson), followed by phycoerythrin-conjugated secondary antibody (PE), with the fluorescein isothiocyanate (FITC)-labeled monoclonal antibodies against human CD45 (Becton Dickinson) and the PE-conjugated monoclonal antibody against human CD133 (Milteny). After incubation, cells were lysed, washed with phosphate-buffered saline, and fixed with 2% paraformaldehyde prior to FACS analysis.
Preparation of CPC and Cell Infusion Procedure
Cell culture and infusion protocols were previously described (4). Briefly, in patients assigned to intracoronary reinfusion of mobilized CPCs, a total of 270 ml peripheral blood was drawn after administration of the last G-CSF dose on day 5. Following Ficoll density gradient centrifugation, mononuclear cells were suspended in X-vivo 15 medium (Biowhittaker) supplemented with 1 ng/ml carrier-free human recombinant vascular endothelial growth factor (VEGF; R&D), 0.1 mol/l atorvastatin (provided by Pfizer), and 20% human serum drawn from each individual patient. Cells were seeded at a density of 6.4 × 105 cells/mm2 in fibronectincoated dishes (Roche). After 4 days of cultivation, cells were detached with 0.5 mmol/l EDTA, washed twice, and resuspended in a final volume of 10 ml X-vivo 10 medium. The resulting cell suspension contains a heterogeneous population of progenitor cells. More than 90% of the cells show endothelial characteristics, as demonstrated by Dil-acetylated low density lipoprotein (LDL) uptake. After resuspension, cultured cells were intracoronarily infused into a target coronary vessel supplying the most dyskinetic left ventricular area using the stop flow technique.
Clinical Follow-Up
Clinical data, medication, and laboratory data were prospectively collected by study nurses. Follow-up visits were scheduled at 3 and 12 months and at 5 years and were performed by physicians. Annual telephone contacts were performed and documented by study nurses, physicians reviewed the data.
In case of unwillingness to undergo a follow-up in our outpatient clinic, patients and/or primary physicians were contacted by telephone.
Cardiopulmonary Exercise Testing
Cardiopulmonary exercise testing was performed according to a standard protocol on a bicycle ergometer with 4 min unload, start at 50 W and stepwise increase by 25 W every 3 min until patients' maximal effort. A 12-lead electrocardiography (ECG) was monitored continuously; blood pressure was measured every 3 min. Minute ventilation, oxygen (O2) uptake, and carbon dioxide exchange (VCO2) were analyzed by breath-by-breath gas exchange measurement (Jaeger Oxycon, Hoechberg, Germany). For further analysis, peak oxygen uptake (peak VO2), oxygen pulse, oxygen uptake efficiency slope (OUES), and the slope of minute ventilation and CO2 elimination (VE/VCO2) were calculated as previously described (16).
NT-proBNP Serum Levels
Blood for serum analysis was collected from every patient prior to cardiopulmonary exercise testing and coronary angiography at baseline and at the different follow-up visits. NT-proBNP was analyzed using a one-step enzyme immunoassay based on electrochemoluminescence technology (Elecsys 2010, Roche Diagnostics).
LV Angiography
LV angiograms were obtained at baseline procedure and at follow-up after 3 months. Quantitative analysis of paired LV angiograms was performed as previously published (4). For analysis of regional contractility and target vessel revascularization during follow-up, as target coronary vessel we defined the coronary artery supplying the predominant dyskinetic left ventricular area in both treatment groups, irrespective whether patients were assigned for cell infusion into the target vessel or not.
Magnetic Resonance Imaging (MRI)
Cardiac MRI was performed at baseline and at 3 and 12 months follow-up in a subset of patients without implanted pacemakers/cardioverter-defibrillators (ICD) and without claustrophobia, by use of a 1.5-T MRI system (Magnetom Sonata, Siemens Medical Solutions, Erlangen, Germany).
All images were acquired by use of a phased-array body surface coil with 4–12 elements during breath holds (maximum: 12 s) and were ECG-triggered. Cine images with a slice thickness of 6 mm were detected throughout the entire left ventricle by use of contiguous 2D True-FISP sequences. The typical in-plane resolution was 2.2 × 1.3 mm2.
Functional and morphologic MRI measurements such as ventricular volumes, mass, function, and ejection fraction were measured for the left ventricle using standard techniques and analyzed with a commercial workstation (Qmass MR version 6 1.6, Medis Medical Imaging Systems, Netherlands). The LV mass index (g/m2) was calculated as the fraction of myocardial mass and body surface area. The MRI analysis was performed by an experienced physician and reviewed and confirmed by a second physician with several years of experience with both of the independent observers being blinded to patient identity and clinical profile. A senior observer then adjudicated any disparities in analysis.
Statistical Analysis
The Kolmogorov–Smirnov test was used to test for normal distribution of all variables. Continous variables are presented as mean ± standard error, if not stated otherwise. All comparisons of functional parameters within groups were performed referring to baseline levels with the paired non-parametric Wilcoxon signed-rank test. Comparisons between groups were performed with the nonparametric Mann–Whitney U test. Categorical variables were compared with the chi-square test. For comparison of multiple groups, ANOVA test was performed with Tukey-HSD post-hoc test. Statistical significance was assumed if p was <0.05. Statistical analysis was performed using SPSS (version 17.0).
Results
Patient Characteristics
The baseline patient characteristics are summarized in Table 1. Patients of the 5 and 10 μg/kg G-CSF group did not show significant differences in their baseline characteristics (data not shown). The median time since the last myocardial infarction was 36 months (range 3–192 months). Baseline left ventricular ejection fraction (LVEF) was only moderately impaired (median: 39%, range 20–64%), as well as median baseline NT-proBNP serum levels were moderately elevated (523 pg/ml, range 13–3,963 pg/ml). No patient demonstrated clinical signs of acute cardiac decompensation.
Safety and Efficacy of G-CSF Administration in Chronic Ischemic Heart Failure Patients
After 5 days of treatment with low and high dose G-CSF, leukocyte counts were significantly elevated (Fig. 2A). Moreover, circulating CD34+CD45+ and CD34+CD133+ progenitor cells were significantly increased in both groups to a similar extent (Fig. 2B, C). Due to the similar extent in progenitor cell mobilization in the two groups, the groups were pooled for further analysis.
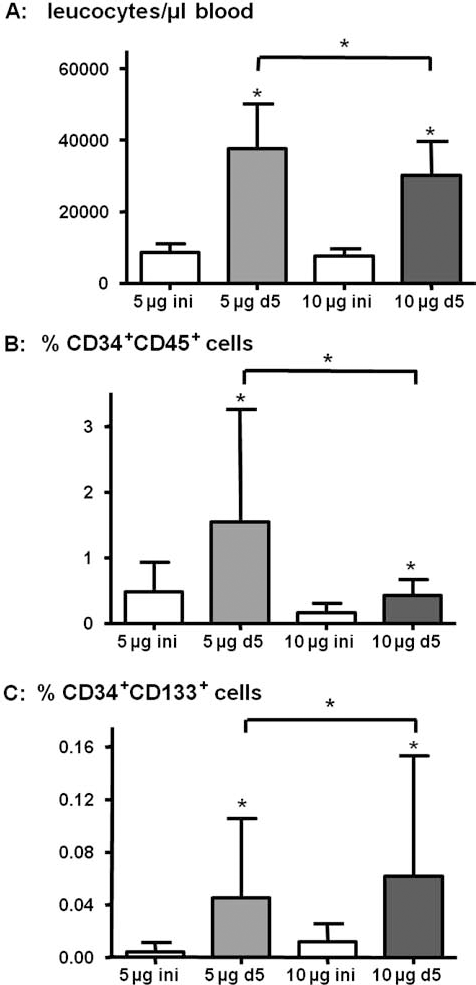
Results of white blood count (A) and fluorescence-activated cell sorting (FACS) analysis for CD34+CD45+ (B) and CD34+CD133+ cells (C) normalized to the number of gated events in the leukocyte gate in patients from the 5 and 10 μg/ kg G-CSF group at baseline (ini) and after G-CSF (d5). Data are presented as mean ± standard deviation. ∗p < 0.05 before versus after 5 days G-CSF mobilization and between the 5-μg and 10-μg G-CSF mobilization group as indicated.
During the 5-day course of G-CSF stimulation, mild cephalgies and episodes of mild to moderate bone and muscular pain could be resolved easily with painkillers. No patient developed chest pain episodes or clinical signs of decompensated heart failure. No novel ischemia-related ECG changes were observed during G-CSF treatment and after intracoronary CPC infusion. Likewise, Troponin T levels remained unchanged. Moreover, no specific G-CSF-mediated severe complications like spleen rupture or acute thrombogenic events due to leucocytosis and increased coagulability occurred.
One patient developed gastric bleeding the night before planned intracoronary CPC infusion, which was therefore withhold. This patient had a history of several peptic ulcers requiring interventional gastro-duodenoscopies years before and was treated again successfully with clipping of peptic ulcers, and the further course was uneventful. As he received the full 5-day dosage of G-CSF, he was followed up and analyzed in the G-CSF group.
In the remaining 22 patients of the G-CSF/CPC group, intracoronary infusions of 29 ± 12 × 106 CPCs (range 9–56 × 106 cells) were successfully performed without any procedural complications. During further in-hospital course, no ischemia-related ECG changes or Troponin T elevations were recorded.
Clinical Events
No patient was lost to follow-up. Table 2 summarizes the clinical events of patients in both treatment groups during 5-year follow-up. At 5 years, one patient of the G-CSF group died due to progressive heart failure. This patient also required rehospitalization for heart failure at 12 months and 3 years after study therapy.
In addition, three patients experienced an additional myocardial infarction. One patient of the G-CSF group suffered from a non-ST elevation myocardial infarction (NSTEMI) in a nontarget vessel with unsuccessful revascularization attempt at 3 months, and two patients of the G-CSF/CPC group experienced an ST elevation myocardial infarction (STEMI) of nontarget vessels at 4 months and 3 years, which were successfully revascularized.
At 12 months, restenosis had occurred in two of the eight patients (25%) of the G-CSF/CPC group receiving concomitant target vessel percutaneous coronary intervention (PCI) at the time of cell administration. In G-CSF/CPC patients without concomitant PCI, no patient developed restenosis. Thus, the overall restenosis rate of the target vessel was 9% at 12 months after G-CSF exposure and intracoronary CPC infusion. Moreover, the overall revascularization rates including nontarget vessel revascularization procedures following G-CSF exposure were 9% at 3 months, 13% at 1 year, and 34% at 5 years (Table 2). There were no discernable trends in restenosis between groups of high and low G-CSF dosages or treatment groups. No patient required bypass surgery.
During 5 years of follow-up, four patients of the G-CSF group and nine patients of the G-CSF/CPC group underwent ICD implantation, all for primary prevention. In addition, one patient experienced a syncope.
Finally, one patient of the G-CSF group developed an urothelial carcinoma of the bladder at 1 year, which was surgically treated.
Effects on NYHA Class, NT-proBNP Serum Levels, and Cardiopulmonary Exercise Capacity
From baseline to 3 months, NYHA class remained stable in the G-CSF group and in the G-CSF/CPC group (see Fig. 3A, B). However, a significant reduction in NYHA class occurred in the G-CSF/CPC group from baseline to 12 months. During the further observation period, most patients remained stable in NYHA classes I and II.

Individual courses of New York Heart Association (NYHA) class for the G-CSF group (A) and G-CSF/CPC group (B) from baseline to 3 months and 1 and 5 years follow-up. ∗p < 0.05 versus baseline. (C, D) Illustrate the course of N-terminal prohormone of brain natriuretic peptide (NT-proBNP) serum levels of the G-CSF group (C) and the G-CSF/CPC group (D) during 5-year follow-up. Data are presented as mean ± standard deviation.
The clinical course as assessed by NYHA classification was paralleled by similar changes in NT-proBNP serum levels during the study (see Fig. 3C, D). In both groups, NT-proBNP serum levels were reduced nonsignificantly at 3 months. No further reductions were detected at 1-year follow-up, and NT-proBNP serum levels remained rather stable until 5 years of follow-up.
Serial cardiopulmonary exercise testing was performed in five patients of the G-CSF group and in 12 patients of the G-CSF/CPC group. In the G-CSF group, peak VO2, OUES, and VE/VCO2 slope slightly deteriorated at 3 and 12 months after therapy, whereas peak oxygen pulse modestly increased at 3 months with return to baseline at 12 months. However, these effects were not statistically significant (see Table 3). In contrast, in the G-CSF/CPC group, peak VO2, OUES, VE/VCO2 slope, and peak oxygen pulse improved at 3 months and remained stable until 12 months after therapy. Likewise, exercise time remained without significant change in both treatment groups.
Cardiopulmonary Exercise Testing (CPET) Results

Effects on LV Function and Diameters After 3 Months by LV Angiography and MRI
Paired assessment of quantitative LV angiography was available in 26 patients. In three patients, LV angiography could not be assessed quantitatively due to poor image quality. In the G-CSF/CPC group, one patient refused the planned invasive follow-up study, and one patient was only available for telephone contact. In addition, in one patient who developed a rapid progressive glomerulonephritis, angiographic follow-up was cancelled. In additional patients, regional contractility analysis was not possible, thus leaving a total of 26 paired LV angiograms for analysis.
Global LV function and volumes did not change significantly in the G-CSF and G-CSF/CPC group after 3 months (see Table 4). Regional contractility of the infarcted area targeted by intracoronary cell administration improved significantly in the G-CSF/CPC group, but not in the G-CSF group. However, there were no differences between the groups.
Global and Regional Parameter of LV Function, Obtained by LV Angiography
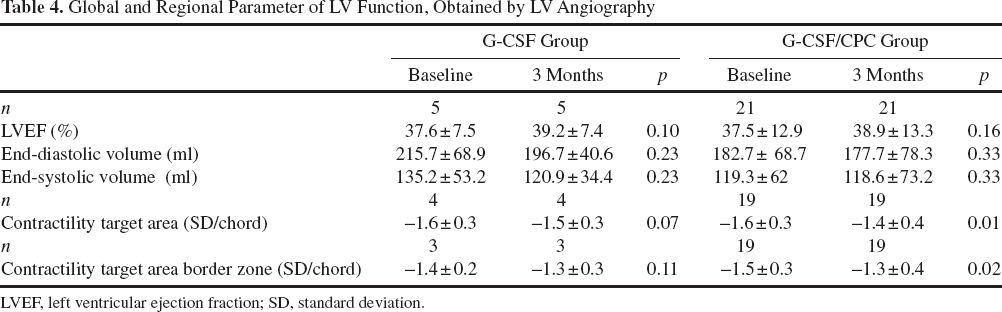
LVEF, left ventricular ejection fraction; SD, standard deviation.
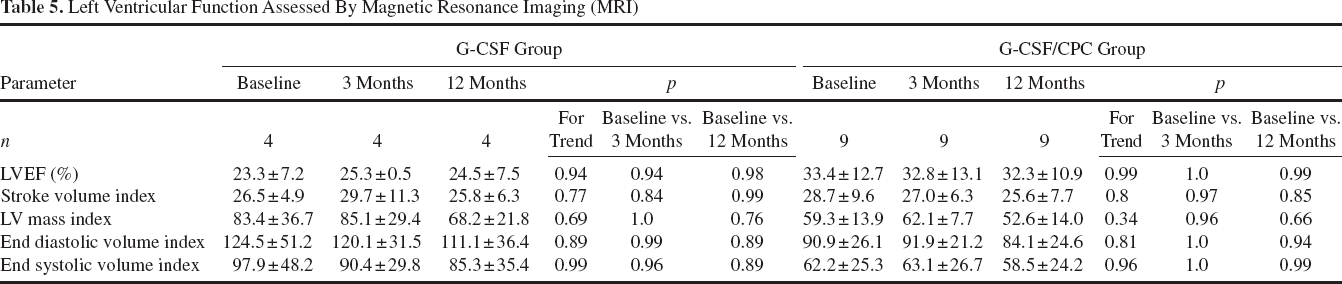
Serial MRI analysis was performed in a subset of 13 patients at baseline, 3 months and 1 year of follow-up (see Table 5). LVEF remained unchanged in both treatment groups after 3 months and 1 year. Likewise, enddiastolic and endsystolic volume indices as well as LV mass indices remained stable within 3 months and 1 year after G-CSF administration.
Left Ventricular Function Assessed By Magnetic Resonance Imaging (MRI)
Discussion
The results of the present pilot trial suggest that G-CSF treatment is safe in patients with old myocardial infarction. However, neither G-CSF application alone nor the intracoronary reinfusion of G-CSF mobilized and cultured circulating mononuclear cells induced a significant improvement of global left ventricular function. Regional left ventricular contractility of the target area increased significantly in the group receiving reinfusion of G-CSF mobilized CPC at 3 months, suggesting that direct application of the cells may indeed provide some positive local effects. However, improvements of clinical symptoms and natriuretic peptide serum levels did not persist at 1 or 5 years after therapy.
Although the findings of the current pilot study are negative with respect to global left ventricular functional improvement, they demonstrate that G-CSF can be safely applied in patients with ischemic heart disease. None of our patients developed severe G-CSF associated complications like splenic rupture or thromboembolic complications, which have been described previously (8,15). Moreover, at the time of initiation of the current study, the study of Kang et al. suggested that G-CSF administration may be associated with enhanced restenosis development in patients with acute myocardial infarction (20). Although our patient cohort is rather small, the cumulative revascularization rate of 13% at 1 year corresponds well with established revascularization rates in stable coronary artery disease patients (29), and we did not observe an excess of adverse cardiovascular events over 5 years. Moreover, in the meantime, several other studies applied G-CSF alone in patients with acute myocardial infarction, and a recent meta-analysis including 385 patients revealed no evidence for an increased restenosis development imposed by G-CSF administration (1). Apart from the safety of G-CSF application, this meta-analysis revealed a beneficial effect on global left ventricular function in the subgroup with baseline ejection fraction below 50% as a marker of larger myocardial infarctions. This is in parallel with the short- and long-term results obtained by intracoronary infusion of bone marrow-derived mononuclear cells (24,26), which also demonstrated greater improvement of LV function in patients with more depressed baseline LVEF. But in contrast to the results of the intracoronary bone marrow cell (BMC) application in Repair-AMI, G-CSF treatment in subgroups was effective if administered early (<37 h) after acute myocardial infarction, whereas the intracoronary approach revealed significant improvement only if treatment was performed between 4 and 7 days post AMI. Likewise, the Leuven trial (18) with intracoronary BMC application within 24 h post-MI failed to improve global LV function. However, the trials that are included into the meta-analysis were performed in patients with acute myocardial infarction that received G-CSF administration only without additional cell application. Therefore, it is difficult to extrapolate these pure G-CSF effects to the chronic setting with additional intracoronary application of the mobilized cells after culture. Moreover, it is anticipated that in the setting of chronic post-infarction heart failure, homing and mechanisms of functional improvement differ from the acute event.
The present pilot study revealed in line with the aforementioned results of clinical trials in patients with acute myocardial infarction (2,34), that G-CSF therapy alone failed to improve global left ventricular function in patients with stable ischemic heart failure. In contrast, the combined approach of G-CSF administration and intracoronary reinfusion of the mobilized cells had moderately positive effects on left ventricular target area contractility. A comparable approach, but without culturing the mobilized cells, has been used in the MAGIC CELL 3 DES trial (21), where Kang and coworkers randomized 40 patients with at least 14-day-old myocardial infarction to receive either intracoronary infusion of autologous, G-CSF mobilized mononuclear cells from the peripheral blood or no additional treatment. Consistent with the data of our study, no significant difference in global left ventricular function could be detected between the two groups at 6 months follow-up.
The moderate short-term and neutral long-term results of the G-CSF/CPC group are in line with the results of the TOPCARE-CHD trial, where we demonstrated that intracoronary infusion of nonmobilized circulating mononuclear cells is not associated with improved left ventricular function, in contrast to intracoronary infusion of bone marrow-derived mononuclear cells (3). Initially, we hypothesized that the limited effect of circulating mononuclear cells may be related to the low number of infused cells (22 ± 11 × 106 CPCs in TOPCARE-CHD) and thus attempted to increase the number of the infused cells by prior G-CSF treatment. However, after G-CSF pretreatment, the number of cells after 4 days of culture was only modestly increased by 30%. In addition, functional analysis of the G-CSF mobilized and cultured cells revealed a profoundly reduced migratory capacity in vitro and a lower capacity to enhance neovascularization in a mouse hindlimb ischemia model (17). One explanation for these effects is that G-CSF treatment leads to a reduced surface expression of the C-X-C chemokine receptor type 4 (CXCR4), which plays a crucial role not only for the migratory capacity of circulating mononuclear cells but—more importantly—for recruitment and homing of the cells to the target tissue. From a clinical point of view, either direct intramyocardial injection of the mobilized cells or local increase of the physiological chemoattractant stromal cell derived factor-1 (SDF-1), the ligand of CXCR4 in the target area may compensate for this limitation of the G-CSF mobilized cells. Moreover, we cannot exclude that there are further effects of G-CSF on paracrine mechanisms activated in cell therapy of ischemic heart disease (12).
Indeed, Losordo et al. (23) performed intramyocardial injection of G-CSF mobilized, CD34+ peripheral blood mononuclear cells in a 167 patient trial. These patients, suffering from refractory angina, demonstrated improvements in angina frequency and exercise time. Of note, 4.6% of patients experienced NSTEMI during cell mobilization, which may be related to the fact of very severe coronary artery disease, which was not present in our patients.
For future trials, a different observation by Zaruba and colleagues may be of interest (33). They recently demonstrated that the antidiabetic sitagliptin is associated with enhanced cell homing and improved left ventricular function following G-CSF application in animal models with acute myocardial infarction, which is due to inhibition of SDF-1 cleavage. This combined approach of G-CSF mobilization of cells and enhanced homing due to DPP IV inhibition is currently investigated in patients within the SITAGRAMI trial (NCT00650143).
The most important limitations of this pilot trial are the low number of patients. However, when we initiated this trial in 2002, no data were available to perform a power calculation and safety concerns regarding the use of G-CSF in patients with ischemic heart disease gained increasing attention.
In summary, based on the data from our pilot trial, we conclude that, in patients with old myocardial infarction, G-CSF alone or in combination with reinfusion of mobilized and cultured circulating mononuclear cells is safe but does not provide convincing beneficial effects on global left ventricular function, although moderate improvement was seen with respect to regional contractility. Therefore, we plan no follow-up trial with this protocol. Whether improving the impaired homing capacity of G-CSF-mobilized circulating mononuclear cells, for example, by inhibition of DPP IV, will lead to more favorable results, is currently investigated in an ongoing trial in patients with acute myocardial infarction.
Footnotes
Acknowledgments
We greatly appreciate the excellent work of our study nurses Isabel Geweyer, Liane Ziska, and Stephanie Estel, as well as the responsible lab technician Tina Rasper, and the staff from the Institute for Cardiovascular Regeneration, Frankfurt. We also thank Dr. Iris Burck, Department of Radiology, for excellent help in MRI analysis and Ina Ziegler and Inka Diehl for assistance. The authors declare no conflicts of interest.
