Abstract
β-Cell replacement therapy by either whole-organ pancreas or islets of Langerhans transplantation can restore carbohydrate control to diabetic patients and reduces complications associated with the disease. One of the variables inherent in islet transplantation is the isolation of functional islets from donor pancreata. Islet isolations fail to consistently produce good-quality functional islets. A rapid pretransplant assay to determine posttransplant function of islets would be an invaluable tool. We have tested the novel hypothesis that modified oxygen consumption rates (OCR), standardized to DNA quantity (nmol/min-mg DNA), would serve as a pretransplant assessment of the metabolic potency of the islets postisolation. This study compares the ability of current in vitro assays to predict in vivo restoration of normoglycemia in a diabetic nude mouse posttransplantation of adult pig islets. There is known to be a diversity of islet sizes within each preparation. This parameter has not heretofore been effectively considered a critical factor in islet engraftment. Our results suggest a surprising finding that islet size influences the probability of restoring carbohydrate control. Based on this observation, we thus developed a novel predictor of islet graft function that combines the effects of both islet OCR and size. When OCR was divided by the islet index (size), a highly significant predictor of graft function was established (p = 0.0002, n = 75). Furthermore, when OCR/islet index values exceeded 70.0 nmol/min-mg DNA/islet index, an effective threshold of diabetes reversal was observed. This assay can be performed with as few as 1,000 islet equivalents (IEQ) and conducted in less than 60 min. Our data suggest that, using this novel method to assess islet cell function prior to transplantation, OCR/islet index thresholds provide a valuable tool in identifying which islet preparations are most likely to restore glycemic control posttransplant.
Introduction
Despite significant improvements over the past decade (30), the therapeutic value of islet transplantation as a treatment for diabetes remains controversial (26). However, the replacement or regeneration of insulin-secreting β-cells as a means of restoring carbohydrate control continues to be a focus of diabetes research (27). A major consideration for any transplant is the functional quality of the graft, which can be influenced by such variables as the condition of the donor, preservation technique, and ischemic time (15, 28). In islet transplantation, there is the additional variable of damage caused by processes involved in the isolation of the islets from the pancreas including mechanical, enzymatic, and osmotic stresses (29). Currently, there are limited in vitro pretransplant techniques for predicting the posttransplant function of isolated islets. Proposed in vitro methods of assessing the quality of an islet preparation include insulin secretion in response to a glucose challenge (32), ATP-to-ADP ratios (10), action potential (12), and, more simplistically, by viability staining, size, number, or packed cell volume (16). FDA guidelines recommend the diabetic immunodeficient [athymic or severe combined immunodeficient (SCID)] mouse bioassay to test the in vivo function of islets used for clinical transplantation; however, this assay is costly and technically demanding and the results are retrospective (37). A further concern with the nude mouse bioassay is that the minimum number of human “islet equivalents” (IEQ) required to reverse diabetes in the athymic mouse is approximately 2,000 (100,000 IEQ/kg) (7), which is 20-fold in excess of 100 IEQ (5,000 IEQ/kg) required to reverse diabetes as an isograft in mice (31). This discrepancy is mostly likely due to the insensitivity of rodents to both porcine and human insulin (23).
Recently, measuring islets' ability to consume oxygen has been shown to be a valuable tool in assessing the metabolic potency of islet preparations (9, 14, 33). Based on the correlation of oxygen consumption rates (OCR/DNA) with islet function and islet dose per kilogram of recipient body weight, using the mouse bioassay, Papas et al. have developed a pretransplant predictive assay of posttransplant function for a specific islet preparation (20, 21). We have confirmed that OCR and OCR/DNA measurements are of value in predicting pretransplant in vivo capacity of mature porcine islets to reverse diabetes posttransplant. Furthermore, we describe the discovery of a heretofore unknown oxygen consumption rate index that provides a significantly greater predictive measure of islets' ability to reverse diabetes posttransplant.
Materials and Methods
Porcine Pancreatic Islet Isolation
Adult porcine islets were isolated from adult female (>2 years) Yorkshire-Landrace pigs using standardized operating protocols based on a modified Ricordi technique (25), yielding >90% purity from exocrine tissue. Porcine islets were cultured overnight in modified RPMI [10% fetal bovine serum (FBS), 5 mM nicotinamide, 2 mM Glutamax, 1% penicillin–streptomycin (P/S)] at 37°C. After culture, islets were counted and standardized to islet equivalents (IEQ) with a diameter of 150 μm according to established protocols (24), assayed for their functional capacity (as described below), and transplanted into diabetic athymic mice.
Islet Viability Staining
Three aliquots of 100 IEQ per isolation were stained with acridine orange/ethidium bromide to assay viability based on membrane integrity (2). Each aliquot was assessed using a fluorescence microscope with a combined filter of 647 nm/535 nm.
Glucose Stimulation Index
The ability of isolated islets to secrete insulin in response to glucose was measured as previously described (3, 36). Briefly, aliquots of 100 IEQ (three replicates per isolation) were incubated in 1.5 ml of RPMI containing 2.8 mmol/L glucose (low) for 1 h at 37°C. Simultaneously, 100 IEQ (three replicates per isolation) was incubated with 1.5 ml of fresh RPMI containing 20 mmol/L glucose (high) for 1 h at 37°C. Supernatants from all cultures were removed to quantify porcine insulin levels measured by ELISA (Mercodia AB) according to the manufacturer's instructions. The DNA content of each islet aliquot was measured using Quant-iT PicoGreen dsDNA kit (Molecular Probes, Eugene, OR). The quantity of insulin secreted (in nanograms per milliliter) was standardized to the amount of DNA presented (in milligrams). The ratio of the mean concentrations of insulin secreted at high to low glucose levels produced a stimulation index (high/low).
Islet Index
The islet index was derived during quantification by dividing the total number of islet equivalents [defined as a volume of islet tissue equal to that of a sphere with a 150-μm diameter (24)] by the actual number of islets (range, 50–400+ μm). Thus, a greater islet index indicates a greater proportion of islets over 150 μm present in the islet isolation. For example, 4,000 islets producing an IEQ count of 2,000 would generate an islet index of 0.5 (2,000/4,000).
Measurement of Oxygen Consumption Rates (OCR)
Islets' OCRs were assessed in triplicate aliquots of 1,000 IEQ (per isolation). Controls assayed in parallel consisted of medium alone and heat-killed islets (1,000 IEQ x 3 incubated for 1 h at 60°C). OCRs were measured using a fiber optic sensor oxygen monitoring system (Instech Laboratories, Plymouth Meeting, PA), which quantifies the decrease in oxygen partial pressure (pO2) over time, as previously described (22). OCRs were standardized to the amount of DNA in each chamber (nmol/ min-mg DNA), determined using a Quant-iT PicoGreen dsDNA kit (Molecular Probes, Eugene, OR).
Validation of the Oxygen Consumption Assay
Islets were cultured overnight in modified RPMI (10% FBS, 5 mM nicotinamide, 2 mM Glutamax, 1% P/S) at 37°C with a 5% CO2 environment. On the day of the experiment, the islets were counted and washed with nonsupplemented RPMI. The islets were then incubated with varying concentrations (0.0, 0.1, 1.0, and 10 mg/ml) of sodium azide (NaAz) (Sigma) for 10 min to inhibit aerobic respiration prior to measuring their ability to consume oxygen. As a negative control, RPMI alone was tested for background oxygen consumption rates. The islets' subsequent OCR measurements were standardized to a percent difference from non-NaAz-treated cells. Each OCR measurement per NaAz treatment was performed in triplicate with 1,000 IEQ per assay. The experiment was repeated three times. Similar procedures were performed using βTC6 and HeLa cells cultured in DMEM and supplemented with 10% fetal calf serum and 100 μg/ml P/S.
Islets In Vivo Function: Nude Mouse Bioassay
To correlate in vitro assay measurements with the ability of islets to restore normoglycemia in vivo, islets were transplanted into diabetic nude mice. Diabetes was chemically induced in 15- to 20-g male athymic Balb/c nude mice (Charles River, Wilmington, MA) by intraperitoneal injection of 200 mg/kg streptozotocin (STZ) (Sigma Chemicals), freshly reconstituted in citric acid/ citrate buffer (pH 4.5–4.7). Diabetes was defined as two non-fasting blood glucose readings of >18 mmol/L at least 2 days apart.
Following porcine islet isolation and a 24-h culture period, 4,000 IEQ was transplanted into the left renal capsule of the diabetic nude mice, under 1.5% isoflurane anesthetic. Prophylactic mice received buprenorphine (0.05–0.10 mg/kg) analgesia, which was subsequently administered as needed postoperatively. Blood glucose was monitored using a mini glucometer (Freestyle) on days 0 (pretransplant), 1, 4, and 7, followed by weekly blood glucose recordings. Animals in which diabetes was reversed were nephrectomized, and blood glucose levels were monitored on at least two separate occasions to confirm a restoration of hyperglycemia. All animal studies were conducted in compliance with the guidelines of the Canadian Council on Animal Care and the protocols approved by the Animal Care and Veterinary Services Committee of the University of Western Ontario
Statistical Analysis
The individual predictive ability of viability staining, glucose stimulation index islet size (islet index), oxygen consumption rates, and oxygen consumption/islet index (OCR/II) were assessed using a logistic regression model with probability of diabetic reversal as the response. The extent to which an assay was of significant value in predicting reversal or nonreversal was established using the receiver operating characteristic curve (ROC) methodology (1, 4). A ROC plot is obtained by calculating the sensitivity and specificity of each observed data value and plotting sensitivity (true positive) against 1 – specificity (false positive) with diabetes reversal being the true positive response. The effectiveness of an assay prediction is then quantified by the area under the ROC curve, with the area of 0.5 indicating a random predictor and 1.0 indicating a perfect predicator. In addition, the overall predictability of each model was quantified using Nagelkerke's correlation coefficient (r2) for logistic regression (18). A value of p = 0.05 was used to declare the statistical significance of an index being of value in predicting probability of diabetes reversal posttransplant. All statistical analyses were performed using SAS 9.2 software (SAS Institute, Cary, NC).
Results
The data reported here were generated from porcine islet isolations, each of which was tested for in vivo function by transplantation into the kidney capsule of four to six diabetic athymic mice per isolation. Mice presenting with streptozotocin-related toxicity, either at the time of transplant or immediately posttransplant, were excluded from the analysis. Similarly, mice that had blood glucose levels less than 18 mmol/L at the time of transplant were also excluded. At the end of the observation period (100 days), all normoglycemic mice were nephrectomized to confirm the restoration of hyperglycemia. Postnephrectomy glycemic control was abolished in all cases.
Viability Staining Islets
Viability stains are based on dye exclusion, an indicative marker of membrane integrity. Cells that have intact membranes will exclude DNA-chelating dyes from entering the cells, while cells that have damaged plasma membranes will stain with the DNA-chelating dye. However, exclusion of these stains (viability) fails to assess metabolic function. Twelve islet isolations were tested in a total of 45 transplants for the ability of viability staining to predict in vivo reversal of diabetes. ROC analysis showed that this assay is not significantly better than chance at predicting reversal with an area under the curve (AUC) of 0.59 ± 0.09 with 95% confidence limits of 0.42–0.76 (Table 1). Islet's staining >90% viable results in a restoration rate of diabetes of only 43.1%. Logistic regression analysis (Fig. 1A) shows that this assay is not useful as a predictor of islet function (p = 0.30, r2 = 0.024).

Logistic analysis correlating standard islet viability assays to diabetes reversal rates in nude mice (y-xis) following adult porcine islet transplantation. (A) Islet viability based on fluorescence dye exclusion (p = 0.30, r2 = 0.024, n = 45). (B) The ability of islets to secrete insulin in response to glucose was not predictive of in vivo function (p = 0.23, r2 = 0.027, n = 53). (C) Islet size marginally influenced the ability of islets to reverse diabetes posttransplant (p = 0.12, r2 = 0.04, n = 73). (D) The ability of islets to consume oxygen indicated a trend to determining transplant function compared to other viability assays tested (p = 0.004, r2 = 0.11, n = 77).
Statistical Analysis Correlating In Vitro Islet Functional Assays to Reversal of Diabetes

A.I., actual islet number; AO, acridine orange; AUC, area under the curve ± SEM; EtBr, ethidium bromide; IEQ, islet equivalent (volume of islet tissue equal to that of a sphere having a 150-μm diameter); II, islet index (IEQ/A.I); OCR, oxygen consumption rates; ROC, receiver operating characteristic; SE, standard error.
Insulin Secretion in Response to Glucose: Stimulation Index
Islets from 12 different isolations were tested in vitro for their ability to produce insulin in response to a glucose challenge. The results of this assay did not correlate with subsequent reversal of diabetes in 53 transplants with an AUC from ROC analysis of 0.54 ± 0.09 with confidence limits of 0.37–0.71 (Table 1) and no significant (p = 0.23, r2 = 0.027) logistic regression coefficient (Fig. 1B). This lack of correlation may in part have been due to variability inherent in the assay; however, in our hands, the capacity of islets to respond to glucose in vitro was of no value in predicting posttransplant outcome.
Islet Index
The islet index (II) provides an indication of the size distribution of islets following the isolation procedure. It is derived by dividing the number of islet equivalents (IEQ) by the actual number of islets. Logistical regression analysis of 73 transplants from 20 pancreatic isolations, performed to determine whether islet size affects diabetes reversal rates, indicated a surprising finding that nude mice transplanted with the greatest number of small islets became normoglycemic with the highest frequency (Fig. 1C). However, ROC analysis indicates that as a diagnostic test of function, islet size alone is not significantly better than chance, with an AUC of 0.63 ± 0.07 and 95% confidence limits of 0.49–0.76 (Table 1). While these data suggest that smaller islets are more efficient at reversing diabetes, the predictive value is marginal (p = 0.12, r2 = 0.04).
Islet's Oxygen Consumption Rate
In aerobic cellular respiration, oxygen serves as the final electron acceptor from cytochrome a3 of the electron transport chain, present in the inner membrane of the mitochondria. Thus, measuring a cell's ability to consume oxygen (OCR) is an indicative means of assessing the metabolic potency of the mitochondria and overall function of the cell (see Fig. 1D). To confirm the validity of our methodology to correlate cellular oxygen consumption rates with mitochondrial potency, insulin-producing porcine islets were incubated with increasing concentrations of sodium azide (NaAz), a known inhibitor of mitochondrial respiration (17, 38, 39). Reductions in OCRs were assessed by comparing the effect of NaAz on OCR measurements to the OCR capability of nontreated cells. When islets were incubated with 0.1 mg/ml NaAz, a 37.6% reduction in OCR was achieved. Cells treated with NaAz at concentrations of 1 and 10 mg/ml resulted in OCR reductions of value 59.7% and 84.2%, respectively, whereas culture medium alone produced a 94.7% OCR reduction compared to nontreated islets (Fig. 2). Overall, NaAz significantly reduced the islet's ability to consume oxygen in a dose-dependent manner (p < 0.05, p < 0.01, and p < 0.001) (Fig. 2). To further support our observations, we reproduced the above experiment in both βTC6 and HeLa cells. Photomicrographs of βTC6 cells loaded with MTred illustrated the progressive loss of mitochondrial membrane potential induced by increasing doses of NaAz (data not shown).

Pancreatic islets [n = 3 per treatment, 1,000 islet equivalents (IEQ) per triplicate] were incubated with increasing concentrations of sodium azide (NaAz) prior to oxygen consumption rate (OCR) measurements (Nos. 2–4). The subsequent OCR was standardized as a percent difference from non-NaAz-treated cells (no treatment, No. 1). Overall, NaAz significantly reduced the islet's ability to consume oxygen in a dose-dependent manner (*p < 0.05, **p < 0.01, and ***p < 0.001, ANOVA). Real-time oxygen consumption rates (see inset) were monitored by a decrease in partial pressure within the chamber of the oxygen monitor compared to medium alone (No. 5).
It has previously been reported that measuring the oxygen consumption rate of islets can predict the capability of human islets to reverse diabetes in the nude mouse bioassay. Our data confirm this observation for porcine islets. The logistic regression analysis (Fig. 1D) of 20 islet isolations show a superior trend compared to the other assays tested (Fig. 1A–C). ROC analysis of 77 transplants indicates that this assay is indeed significantly better (p = 0.004, r2 = 0.11) than random chance at predicting reversal with an AUC of 0.68 ± 0.07 with 95% confidence limits of 0.54–0.81 (Table 1). OCR/DNA ranged from 27 to 288 nmol/min-mg DNA in this study using porcine islets, which represents a similar range reported when human islets were tested using the same methodology (20).
Oxygen Consumption Rates Standardized to Islet Index
The ability to predict the in vivo functional capacity of an isolated islet is likely to be multifactorial; however, the optimal multifactorial effects have not yet been discovered. Based on our unexpected finding of a trend toward improvement in graft function with smaller islets, we attempted to combine, in a novel test, the influence of islet size in relation to the islet's ability to consume oxygen and found this combination successful. Thus, we have standardized the results of our OCR/DNA tests with the islets index (II), thereby generating an OCR/II ratio. Logistic regression analysis of pretransplant OCR/II indexes from 20 islet isolations shows a strong correlation (p = 0.0002) with diabetes reversal rates in 75 nude mouse transplants (Fig. 3A). Comparison of the regression lines between Figures 1D and 3A provides a visual guide to the beneficial effect of combining the OCR/ DNA test with the islet index. This analysis also provides a useful mathematical relationship between the probability that islets will function posttransplant and any OCR/II index. The equation that describes this relationship is given by Pr (reversal) = 1/[1 + exp(2.4030 − 0.033 * OCR/islet index)]. ROC analysis confirms the value of this test (Fig. 3B and Table 1). Results from the 75 transplants produce an AUC of 0.79 ± 0.06 with 95% confidence limits of 0.67–0.90 (p = 0.0002, r2 = 0.24).

To account for the influence of islet size on their ability to consume oxygen, the OCR/DNA was standardized to their islet index generating OCR/II. (A) Logistic regression analysis showed robust correlation with diabetes reversal rates (p = 0.0002, r2 = 0.24). (B) Receiver operating characteristic (ROC) analysis confirms this relationship with an area under the curve (AUC) of 0.79 (p = 0.0002, n = 75 transplants).
Transplant recipients in this study were reclassified into two groups according to their OCR/II ranges, either <70 (n = 50) or >70 (n = 25), based on the 50% probability of reversal from the logistic regression curve (Fig. 3A). Islets with an OCR/II value >70 were significantly superior at restoring normoglycemia in diabetic nude mice compared to those mice transplanted with islets having an OCR/II value of <70 (p < 0.0001), indicating that an OCR/II measurement of 70 could be a valuable pretransplant functional threshold (Fig. 4).
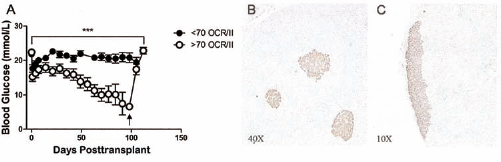
Oxygen consumption rates/islet index measurements correlated to in vivo graft function. (A) OCR/islet index thresholds. Diabetic nude mice transplanted with islets with >70 OCR/II (n = 25) show reversed diabetes compared to animals transplanted with islets with <70 OCR/II (n = 50; ***p < 0.001, Student's t test). Arrow indicates time of nephrectomy. (B) Immunohistochemistry of a biopsied porcine pancreas stained with insulin prior to islet isolation (original magnification: 40x). (C) Immunohistochemistry of nude mouse kidney stained for insulin after islet transplant, showing reversal of diabetes and nephrectomy in the >70 OCR/II group (original magnification: 10x).
Discussion
There are many variables that can influence the ability of isolated islets to reverse diabetes. These include, but are not limited to, the source and condition of the donor pancreas, ischemic times, enzyme strength and toxin contamination, isolation technique, postisolation culture conditions, dose of islets implanted, transplant site, and degree of recipient hyperglycemia (8, 11). Here, we report for the first time the discovery of a novel in vitro derived index that provides a highly predictive value for the ability of islet isolation to reverse diabetes in the standard diabetic nude mouse bioassay. Porcine islets provide a stringent surrogate for the clinical environment (35) and demonstrate comparable function in this assay (19). Further, the isolation of adult porcine islets presents several additional challenges due to their inherent fragility compared to human islets (34), suggesting that the numbers of reversals demonstrated here might be at least comparable if human islets had been utilized. In this study, we examined five different in vitro metabolic potency parameters of islets to assess the ability of these assays to predict in vivo function. Our data have confirmed the observation of Caumo et al. that the measurement of insulin release to an in vitro glucose challenge is of modest statistical value in predicting islet function following transplantation (5). Similarly, pretransplant membrane permeability viability staining is not predictive of reversal of diabetes.
A substantial amount of research has been dedicated to improving the protocol for isolating adult porcine islets (6, 40). However, these studies have focused on maximizing islet yields (total IEQ or IEQ per gram of pancreas), often neglecting the critical parameter of in vivo function. This has led to preselection of pancreata that contain a greater concentration of large islets (>150 μm) to maximize IEQ yields (8). In the present study, islet index analysis demonstrated an unexpected trend that suggests that smaller islets are superior in their ability to reverse diabetes. Our findings are supported by the observations by Lehmann et al. that smaller human islets secrete more C-peptide (13) and those by MacGregor et al. that smaller rat islets are more effective than larger islets in restoring carbohydrate control (14); however, these authors did not provide the unique finding that OCR/islet index is a powerful predictor of graft function.
The inhibition studies with NaAz have shown that measuring OCR standardized to the DNA concentration is an effective means of assaying in vitro islet mitochondrial function, validating the work of Papas et al. (20, 21). Furthermore, our in vivo OCR correlation data confirm their observation that measuring pretransplant OCR has posttransplant predictive value with ROC analysis, confirming that this assay provides results that are significantly better than random chance. When rat islets were transplanted into immunodeficient mice using a similar protocol to that employed in this study, diabetes reversal was not obtained for OCR/DNA levels of less than 120 nmol/min-mg DNA and an effective OCR/DNA threshold of 150 nmol/min-mg DNA was established (21). Based on our observations with logistic regression, OCR/ DNA measurements greater than 150 nmol/min-mg DNA resulted in a marked increase in overall islet engraftment and diabetes reversal rates, consistent with similar observations using both human and rat islets (20, 21).
In this study, we have generated a new in vitro metabolic index (OCR/II) that takes into account both an islet's OCR and size. This factor proved to be a statistically significant prognostic method of in vivo islet function with ROC analysis showing an AUC of 0.79 ± 0.06 (p = 0.0002). This is a substantial improvement over the OCR/DNA measurement alone with an AUC of 0.68 ± 0.07. Based on the logistic regression curve, it is possible to derive a threshold value of 70 for this factor, above which there is a high probability of diabetes reversal. From these data, we have been able to derive an equation for predicting the probability of reversal (Pr), where Pr = 1/[1 + exp(2.4030 − 0.033 * OCR/II]. This equation allows the calculation of the probability that an islet isolation will reverse diabetes. Deriving this standardized OCR index, in triplicate, requires approximately 3,000 IEQ, is inexpensive, and can be derived in less than 60 min.
A question that these observations pose is, what action should one take in the event that the OCR/II predicts a low probability of diabetes reversal? In an environment such as xenotransplantation where the supply of pancreata is unlimited, these isolations should be discarded. In the current clinical environment of allografting where the number of donors is limited, such an action is not so lightly to be undertaken. In this study, we transplanted 4,000 IEQ, which is above the minimal threshold required for reversing diabetes with porcine islets in nude mice (23). One can only speculate that had we transplanted a greater number of islets from those isolations with a poor OCR/II index, reversal rates may well have increased. Papas et al. have recently suggested that islet transplant dose should be based on functional tissue (OCR/kg body weight), instead of the quantity of β-cells per kilogram of recipient body weight (21). Thus, a large transplant dose of islets might compensate for inadequacy of in vitro function based on modified islet oxygen consumption indexes. Islet isolations with a high OCR/II index could potentially reduce the required islet dose, thus increasing the efficacy of clinical islet transplantation. The availability of a simple in vitro test of subsequent in vivo function would also facilitate the validation of changes in islet isolation techniques. We would recommend that those involved in clinical islet transplantation correlate the OCR/II with clinical transplant success. This assay may also be of value in predicting the utility of islets derived from alternative sources including, but not limited to, stem cell-derived insulin-producing β-cells.
Footnotes
Acknowledgments
This work was supported by the Canadian Institutes of Health Research (CIHR), the National Research Council of Canada (NRCC)'s Industrial Research Assistances Program (IRAP) in conjunction with Sernova Corp. (London, ON) and London Health Sciences Multi-Organ Transplantation Program (MOTP). The authors declare no conflict of interest.
