Abstract
Hematopoietic stem cells (HSCs) migrate to injury sites and aid in tissue repair. However, clinical success is poor and is partially due to limited HSC recruitment. We hypothesized that HSC pretreatment with H2O2 would enhance their recruitment to injured gut. As HSCs are rare cells, the number of primary cells obtained from donors is often inadequate for functional experiments. To circumvent this, in this study we utilized a functionally relevant cell line, HPC-7. Anesthetized mice were subjected to intestinal ischemia-reperfusion (IR) injury, and HPC-7 recruitment was examined intravitally. Adhesion to endothelial cells (ECs), injured gut sections, and ICAM-1/VCAM-1 protein were also quantitated in vitro. H2O2 pretreatment significantly enhanced HPC-7 recruitment to injured gut in vivo. A concomitant reduction in pulmonary adhesion was also observed. Enhanced adhesion was also observed in all in vitro models. Increased clustering of α4 and β2 integrins, F-actin polymerization, and filopodia formation were observed in pretreated HPC-7s. Importantly, H2O2 did not reduce HPC-7 viability or proliferative ability. HPC-7 recruitment to injured gut can be modulated by H2O2 pretreatment. This may be through increasing the affinity or avidity of surface integrins that mediate HPC-7 homing to injured sites or through stimulating the migratory apparatus. Strategies that enhance hematopoietic stem/progenitor cell recruitment may ultimately affect their therapeutic efficacy.
Introduction
Evidence suggests that hematopoietic stem cell (HSC) therapy may be an effective treatment option for gastrointestinal disorders, conferring benefit in experimental models of colitis and in Crohn's disease patients (30,40). Administration of murine adult CD34– stem cells (without prior myeloablation) improves clinical scores and significantly reduces mortality in dextran sodium sulfate (DSS)-induced murine colitis (18). Furthermore, celiac patients have increased circulating CD34+ cells when compared to healthy controls (28). Many clinical trials are currently under way examining the role of HSCs in a variety of different organ injuries (34), although the mechanism by which stem cells confer benefit is currently unclear. Roles for cell fusion and anti-inflammatory mechanisms have been suggested (4,38). Regardless of the mechanisms underlying HSC-mediated repair, recruitment to injured organs is a prerequisite event. However, systemic injection, the preferred route of HSC administration, is associated with low levels of stem cell retention in injured tissues and may explain the lack of successful clinical outcomes associated with stem cell therapy (36). Indeed, within infarcted heart, endothelial progenitor cell retention is <5%, with most cells not demonstrating sustained engraftment (1). Furthermore, difficulties remain in obtaining sufficiently large quantities of high-purity HSCs using existing culture conditions. Therefore, identification of methodologies that improve or enhance stem cell recruitment to injured tissue microcirculation is a current high priority for cellular therapies (14,42).
Attempts have been made to enhance progenitor cell recruitment by increasing the local concentration of potent stem cell chemoattractants within ischemic tissue using plasmid DNA encoding for stromal cell derived factor (SDF)-1α (11). However, delivery of vectors to allow for targeted expression of chemokines has been challenging, particularly in clinical settings. Thus, noninvasive techniques not requiring introduction of genetic material are appealing. We previously identified the adhesive mechanisms governing recruitment of the immortalized HSC line HPC-7 to ischemically injured tissue. Adhesion within local microvasculature was shown to depend on site-specific mechanisms, with critical roles demonstrated for α4bβ1 [CD49/CD29; very late antigen-4 (VLA-4)] and αMβ2 [CD11b/CD18; macrophage-1 antigen (Mac-1)] in murine liver and small intestine, respectively. Since integrins play essential roles in mediating stem cell–endothelial interactions, strategies to increase their surface expression and/or increase their adhesive affinity or avidity might be an important approach to improve stem cell recruitment. Indeed, mesenchymal stem cell (MSC) homing to bone marrow (BM) could be enhanced by ectopic α4 integrin expression (21). It is anticipated that such pretreatments will enhance therapeutic benefit through acceleration of tissue recovery and thus improve the effectiveness of cellular therapy.
It is well established that ischemically injured tissues, particularly the gut, release soluble reactive oxygen species (ROS), such as superoxide, hydrogen peroxide (H2O2), and hydroxyl radicals, generated locally by endothelium or by neutrophils accumulating upon reperfusion (3,22). Oxidant injury is strongly associated with increased neutrophil–endothelial interactions. Indeed, H2O2 can modulate neutrophil rolling and adhesion, both in vitro and in vivo, via increased surface expression of αMβ2 by mobilizing it from intracellular stores (5,27). H2O2 has also been demonstrated to significantly increase C-X-C chemokine receptor type 4 (CXCR4) expression on MSCs, resulting in their increased migration toward SDF-1α (23). These studies suggest that development of strategies that enhance stem cell (SC) homing through modulation of adhesion molecule or chemokine receptor expression may be a valuable pursuit. However, no studies have currently determined whether changes in HSC integrin expression or affinity can be induced by ROS pretreatment and, more importantly, whether this is associated with increased retention of these cells within sites of intestinal injury.
Materials and Methods
Animals
Male C57Bl/6 mice (Harlan, Oxon, Oxfordshire, UK) were used for procedures in accordance with the Animals (Scientific Procedures) Act of 1986 (Project License: 40/3336). Anesthetized animals (ketamine/xylazine; IP) underwent carotid artery cannulation to facilitate infusion of labeled HPC-7 cells. Intestinal ischemia-reperfusion (IR) injury was established through superior mesenteric artery occlusion for 45 min and subsequent tissue reperfusion.
Cells
Investigating HSC homing intravitally has been hampered by the limited numbers of primary HSCs obtainable from individual mice (16). Therefore, we utilized an immortalized HSC line, HPC-7, generated by transfecting embryonic SCs with the murine LIM-homeobox gene LH2 (32). Hematopoietic progenitor cell lines immortalized in this manner display characteristics of primary HSCs, including surface expression of common murine HSC markers (c-kit+, CD34–, Lin-) (32) and similar behavior at a molecular level (41). Interestingly, bone marrow-derived HSC lines generated by LH2 transfection are able to reconstitute hematopoiesis when injected into lethally irradiated hosts (33). HPC-7 cells also express adhesion molecules present on primary HSCs, and we have used them to model hepatic HSC recruitment (15). Cells were maintained at a density of 1 × 106 cells/ml in Stem Pro 34 SFM with medium supplement (Life Technologies, Paisley, Renfrewshire, UK), 100 ng/ml recombinant mouse stem cell factor (rmSCF; Life Technologies), l-glutamine, penicillin, and streptomycin (all PAA, Yeovil, Somerset, UK). Density was checked daily, and medium was supplemented to maintain the correct cell density. Medium was changed completely every 2-3 days. Cells were fluorescently labeled with 5 μM (5/6)-carboxyfluorescein diacetate, succinimidyl ester prior to use (CFSE; Life Technologies).
HPC-7 cells were pretreated with vehicle control (PBS), an intestinal injury conditioned medium (ICM), or H2O2 (10 μM, 100 μM, or 1 mM) prior to use. To create an ICM, 20 mm of IR-injured proximal jejunum or terminal ileum was excised, incubated in 3 ml of Stem Pro for 24 h, and subsequently homogenized using a electronic tissue homogenizer (Power Gen 125; Fisher Scientific, Loughborough, Leicestershire, UK). The resultant suspension was centrifuged and sterile filtered. HPC-7 cells were pretreated with the ICM for 30 min (2 × 106 cells/ml). H2O2 was added to some HPC-7 cells for 1 h at various concentrations (10 μM, 100 μM, and 1 mM). For experiments involving blockade of integrins, some HPC-7 cells were also incubated for 30 min with 80 μg/ml of a NA/LE (no azide, low endotoxin) rat anti-mouse anti-β2 integrin antibody (GAME-46; BD Pharmingen, Oxford, Oxfordshire, UK), a LEAF (low endotoxin, azide free) rat anti-mouse anti-a4 antibody (R1-2; Cambridge Biosciences, Cambridge, Cambridgeshire, UK), or a LEAF rat IgG control (RTK2758; Cambridge Biosciences). H2O2 concentrations in ICM were analyzed using the PeroXOquant peroxide assay kit (Thermo Fisher Scientific).
To examine the effects of H2O2 on cell survival, 1 × 106 HPC-7 cells were treated with varying concentrations of H2O2 for 1 h. Cells were subsequently transferred to fresh growth medium and cultured for a further 24 h. At this point, viable cell counts were obtained by counting trypan blue exclusion on a hemocytometer. Viability (%) was calculated as V = [100/(L + D)]×L, where L = live cells, D = dead cells, V = viability. To examine the effect of H2O2 on longer-term HPC-7 function, cells were treated with H2O2 and subsequently differentiated into megakaryocytes by incubating for 5 days with 150 ng/ml thrombopoietin (TPO; Peprotech, London, UK). Differentiation was analyzed by examining ploidy using flow cytometry (see below).
To examine the effects of H2O2 on integrin clustering, cells were treated for 1 h with PBS or 100 μM H2O2 at 37°C. Cells were subsequently fixed in glutaraldehyde for 1 h, washed, and resuspended in 100 μl of PBS with 0.1% bovine serum albumin (BSA; Sigma-Aldrich, Dorset, UK). Fc receptors were blocked with CD16/32 antibody (clone 93; Biolegend, London, UK) for 30 min. Cells were then incubated for 1 h on ice with either LEAF rat anti-mouse α4 integrin or LEAF rat anti-mouse β2 integrin at a dilution of 1:12.5. Subsequently, cells were treated for 1 h with secondary antibody—goat anti-rat IgG (Alexa 488 conjugated; Cambridge Biosciences)—at a concentration of 1:50 on ice. Unbound antibody was removed by washing and cells settled onto lysine-coated cover slips. Cells were imaged at ×63 magnification using scanning confocal microscopy (Leica Microsystems, Wetzlar, Germany). Areas of clustering were identified using the “Find Maxima” plugin in the ImageJ software package (NIH, Bethesda, MD, USA). Maxima plots were produced by identifying regions of maximal intensity within a specified tolerance level (noise tolerance: 40) to separate clusters from background.
Frozen Tissue Static Adhesion Assay
We modified the Stamper-Woodruff adhesion assay (35) to determine HPC-7-endothelial interactions in frozen whole intestinal tissue. Jejunal and ileal samples (20 mm) were isolated from sham and IR-injured animals, cut open along the antimesenteric border, and snap-frozen in liquid nitrogen within 5 min postexcision. The luminal surface was cryostat sectioned to remove 60 μm from the mucosa and thus expose vascular endothelium. Samples were transferred onto glass slides, washed, and fixed in acetone. CFSE-labeled HPC-7 cells (1 × 105) were added to the mucosal surface for 20 min. Slides were washed, fixed, coverslipped, and then imaged microscopically (Olympus BX-61, Olympus, Southend-on-Sea, Essex, UK). Five frames from each section were selected following a predefined pattern. Images were obtained, and adherent HPC-7 cells counted using in-house blinded counting software (available from http://www.bham-mrg.co.uk).
ICAM-1 and VCAM-1 Static Adhesion Assay
Plates (96-well) were coated with 10 μg/ml rmI-CAM-1 (intercellular adhesion molecule 1) Fc chimera, rmVCAM-1 (vascular cell adhesion molecule 1) Fc chimera (R&D Systems, Abingdon, Oxfordshire, UK), or 10 mg/ml BSA (negative control). Wells were then washed, blocked using 10 mg/ml heat-denatured BSA, and 5 × 104 HPC-7 cells were added for 20 min. All HPC-7 cells were blocked with 1 μg/ml CD16/32 function-blocking antibody to prevent nonspecific Fc receptor binding. Some HPC-7 cells were functionally blocked with anti-β2 and anti-α4 blocking antibodies prior to use. After fixation, wells were imaged microscopically, and adherent HPC-7 cells counted.
Endothelial Static Adhesion Assay
Immortalized endothelial cells (ECs) were obtained as previously described (2) from the colon of the Immorto-mouse (Charles River, Wilmington, MA, USA) and expanded at 33°C in d-valine modified Eagle's medium (MEM; Promocell, Heidelberg, Germany) containing 10% fetal bovine serum (FBS), l-glutamine, penicillin and streptomycin (all PAA), nonessential amino acid solution (Promocell), MEM vitamin solution (Life Technologies), and 10 units/ml murine interferon-γ (IFN-γ; Peprotech). For experiments, endothelial cells were de-immortalized by transfer into Dulbecco's MEM (DMEM) containing 10% FBS, l-glutamine, penicillin, and streptomycin (all PAA). Endothelial cells were cultured to confluence on amino-propyltriethoxysilane (APES; Sigma)-treated glass coverslips in 24-well plates. ECs were activated with 100 ng/ml tumor necrosis factor-α (TNF-α; Peprotech) for 4 h, washed, and incubated with 1 × 105 CFSE-labeled HSCs for 20 min; nonadherent cells were washed off with PBS. Some HPC-7 cells were functionally blocked prior to use. Coverslips were fixed, mounted, and imaged microscopically. Adherent HSCs were counted from each x10 field of view using in-house observer blind counting software.
Intravital Imaging
Upon reperfusion, the mucosal villi were exposed by cutting (2 cm) along the ileal antimesenteric border and visualized using an inverted intravital microscope (Olympus IX81). At 30 min of reperfusion, 2 × 106 CFSE-labeled HPC-7 cells were administered arterially. Some HPC-7 cells were pretreated with function-blocking antibodies or control IgG antibody. For analysis, one randomly preselected field of view was monitored every 5 min for 60 min. Free-flowing HPC-7 cells were classified as visible cells trafficking through the field of view, but nonadherent. Cells were considered firmly adherent if they remained stationary for ≥30 s. In addition to the continuous ileal monitoring, sections of jejunum and lung were isolated at the end of the experiment. The number of HPC-7 cells within these tissues was quantitated ex vivo by calculating the mean number of CFSE-labeled cells from five fields of view. Data were stored digitally and analyzed off-line (Slidebook, Intelligent Imaging Innovations, Denver, CO, USA).
Flow Cytometry
HPC-7 surface expression of α4 [fluorescein isothiocyanate (FITC)-conjugated R1-2; Cambridge Biosciences] and β2 integrin (FITC-conjugated M18/2; Cambridge Biosciences) was determined using flow cytometry. HPC-7 cells (1 × 105) were pretreated for 1 h with PBS or 100 μM H2O2, fixed, and then resuspended in 100 μl of PBS containing 5% FBS to block nonspecific binding. Cells were incubated with fluorochrome-conjugated primary antibody (dilution 1:50) prior to performing flow cytometry. Intracellular F-actin levels were identified by staining with FITC-conjugated phalloidin (Sigma). HPC-7 cells were stimulated with H2O2 or vehicle for 1 h, fixed, permeabilized with 0.1% Triton X-100 (Sigma), and incubated for 60 min in the dark with 1 μl of FITC-phalloidin. Cells were diluted 1:1 in PBS prior to flow cytometric analysis. Apoptosis was identified by binding of Annexin V (Ann V) to phosphatidylserine residues on the exterior of apoptotic cells. HPC-7 cells (1 × 105) were treated for 1 h with H2O2 at varying concentrations and then resuspended in 100 μl of Ann V binding buffer supplemented with FITC-Ann V (eBioscience, Hatfield, Hertfordshire, UK) prior to flow cytometric analysis. Following TPO treatment, ploidy was identified by changes in population size and characteristic peaks identified by propidium iodide staining. Cells were incubated in PBS containing 1.5% BSA and 4 μl of FITC-conjugated anti-mouse glycoprotein VI (GPVI; JAQ1; Emfret Analytics, Eibelstad, Germany) at 1:50. Cells were washed, resuspended in 500 μl of PBS, and subsequently fixed by dropwise addition of 2.5% neutral buffered formalin (Sigma). Cells were incubated for 45 min on ice, washed with PBS, and resuspended in PI medium on ice for 30 min [PI medium: PBS containing 2 mM MgCl2, 0.05% saponin, 0.01 mg/ml propidium iodide, and 10 units/ml RNase (all from Sigma)]. Cells were subsequently examined using flow cytometric analysis.
8-Oxo-2′-Deoxyguanosine Staining for Oxidative Damage
8-Oxo-2′-deoxyguanosine (8-oxo-dg) is generated as a result of oxidative stress. To assay for 8-oxo-dG content in the gut, 10-μm sections of frozen sham and IR-injured ileum were immobilized on a glass microscope slide. Sections were washed, blocked for 1 h with blocking solution, and then incubated with a 1:100 dilution of anti-8-oxo-dG overnight at 4°C (2E2; Trevigen, Gaithersburg, MD, USA). Sections were then washed and subsequently incubated for 30 min with FITC-conjugated anti-mouse IgG at 1:50 (F2883; Sigma-Aldrich). Following dehydration, sections were mounted and visualized microscopically. Images were rotated to align villous orientation. Microscope and normalization settings were identical between samples to ensure comparability. To examine cellular 8-oxo-dG levels, HSCs were incubated with varying concentrations of H2O2 for 1 h. Cells were washed, fixed, and subsequently resuspended in 0.1% Triton-X100 for 10 min. Cells were further washed and incubated with 8-oxo-dG at a concentration of 1:100 for 1 h. Cells were incubated with secondary antibody (FITC-conjugated anti-mouse IgG; Sigma-Aldrich) at 1:50 for 30 min and subsequently analyzed by flow cytometry.
Electron Microscopy
HPC-7 cells treated with either vehicle or 100 μM H2O2 for 1 h were fixed with 2.5% glutaraldehyde and subsequently imaged on a Philips XL30 FEG ESEM (field emission high-resolution scanning electron microscope; FEI, Eindhoven, the Netherlands).
Statistics
For the analysis of serial data, the area under the curve (AUC) was calculated for each experiment, the mean obtained for each individual experiment and grouped values compared using a one-way ANOVA followed by Bonferroni's multiple comparison posttests (GraphPad, San Diego, CA, USA). Serial data are better analyzed with this methodology as other statistical tests may reduce power by disregarding the correlation between time points in individual subjects (29). All other data were compared by one-way ANOVA followed by Bonferroni's multiple comparison posttests. Single comparisons were examined using unpaired Student's t tests. Results were considered significant when p < 0.05.
Results
Pretreating HPC-7 Cells with Intestinal ICM Enhances Their Adhesion to Activated Endothelium and Frozen Intestinal Tissue In Vitro (Fig. 1)
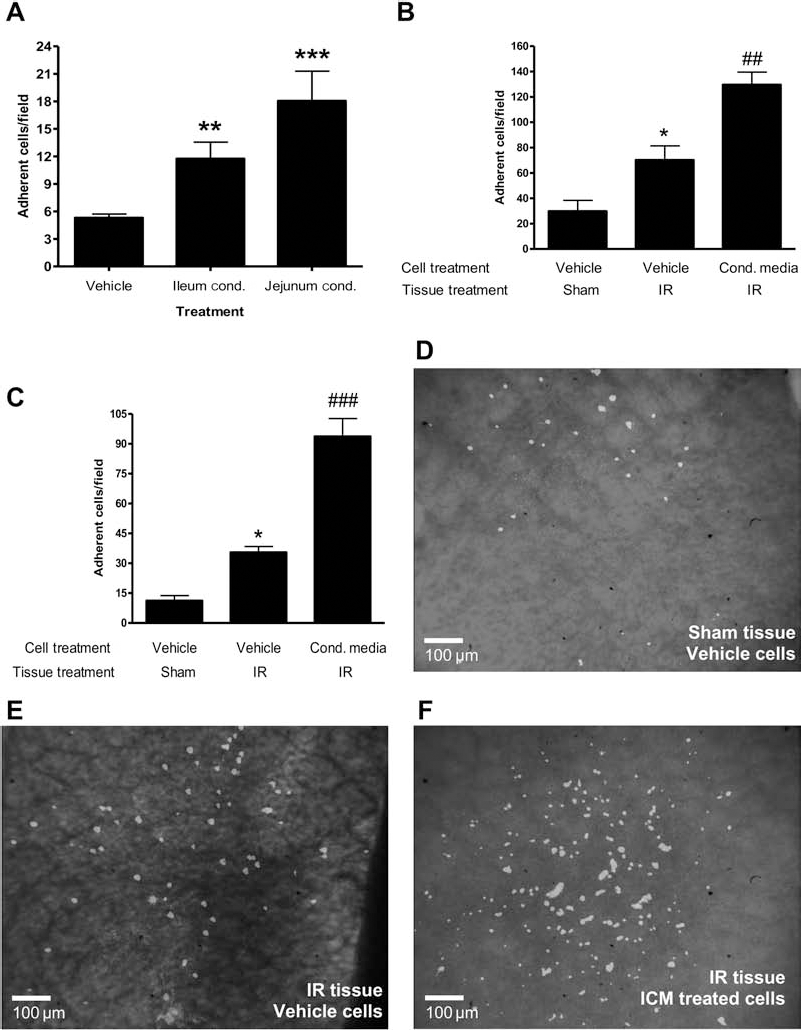
Pretreating HPC-7 with intestinal ICM enhances their adhesion to endothelial cells and frozen intestinal tissue in vitro. (A) Immortalized hematopoietic stem cell line HPC-7 adhesion on tumor necrosis factor-α (TNF-α)-activated murine colonic endothelium was enhanced following pretreatment with ileal or jejunal conditioned medium when compared to vehicle-treated cells. HPC-7 adhesion to ischemia-reperfusion (IR)-injured (B) ileal and (C) jejunal tissue was higher than on sham sections, but was further increased by pretreatment with ileal or jejunal ischemic injury conditioned medium (ICM). Representative images of (D) sham ileum with vehicle-treated HPC-7, (E) IR-injured ileum with vehicle-treated HPC-7, (F) IR-injured ileum with ileal ICM-treated HPC-7. Scale bars: 100 μm. Results presented as mean adhesion ± SEM (n ≥ 3 endothelial assays for A; n ≥ 6 tissue samples for B, C). **p < 0.01, ***p < 0.001 versus vehicle; ##p < 0.01, ###p < 0.001 versus vehicle cells/IR sections.
Adhesion of HPC-7 cells to colonic endothelium was significantly enhanced following pretreatment with either an ileal (p < 0.01) or jejunal (p < 0.01) ICM (adherent cells/field: vehicle: 5.30 ± 0.41; ileum ICM: 11.75 ± 1.80; jejunum ICM: 18.05 ± 3.24) (Fig. 1A). Furthermore, significantly (p < 0.05) more HPC-7 cells adhered to IR-injured ileum sections compared to sham ileum (adherent cells/field: sham sections: 29.89 ± 8.46; IR sections: 70.25 ± 11.07) (Fig. 1B). Adhesion to IR-injured ileum could be further significantly (p < 0.01) increased by pretreating HPC-7 cells with ileum ICM (adherent cells/field: vehicle: 70.25 ± 11.07; ileum ICM: 129.60 ± 9.91) (Fig. 1B, D–F). Similar data were obtained with jejunum sections; significantly (p < 0.05) more HPC-7 cells adhered to IR-injured jejunum sections compared to sham jejunum sections (adherent cells/field: sham section: 11.19 ± 2.53; IR section: 35.45 ± 2.92) (Fig. 1C). Treating HPC-7 cells with jejunum ICM significantly (p < 0.001) enhanced adhesion further compared to vehicle-treated cells (adherent cells/field: vehicle: 35.45 ± 2.92; jejunum ICM: 93.63 ± 9.01) (Fig. 1C).
Intestinal ICM Contains H2O2
Immunohistochemical labeling of gut sections using an anti-8-oxo-dG antibody revealed significant oxidative damage in IR-injured gut sections when compared to sham control sections (Fig. 2A, B). Since the ICM was generated from homogenized gut tissue, the ICM was also assayed for H2O2 concentration. Significantly (p < 0.001) higher levels were observed in ICM compared to sham tissue medium (H2O2 concentration: sham CM: 27.31 ± 1.81 μM; IR ICM: 64.44 ± 2.67 μM) (Fig. 2C).
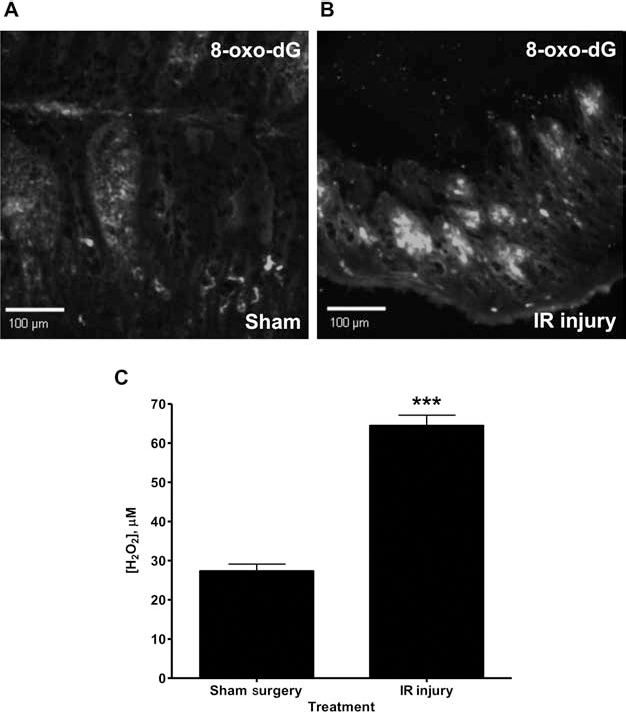
Sections isolated from IR-injured ileum stain intensely for 8-oxo-2′deoxyguanosine (8-oxo-dg). Accumulation of 8-oxo-dg, a marker of oxidative damage, was determined immunohistologically. (A) Sections of sham ileal tissue display relatively low 8-oxo-dG staining, whereas (B) IR-injured ileum displays intense staining for 8-oxo-dg. Villous length in IR sections is shorter than sham sections due to the high susceptibility of the mucosa to reperfusion injury. Images (×10 magnification) represent those obtained from three repeat experiments. (C) Significant increases in H2O2 were observed in media generated from IR-injured gut compared to sham gut media. Scale bars: 100 μm. Results in (C) are presented as mean concentration ± SEM (n = 3 conditioned media samples). ***p < 0.001.
Pretreating HPC-7 Cells with 100 μM H2O2 Enhances Their Adhesion to Activated Endothelium and Frozen Intestinal Tissue In Vitro
HPC-7 cell adhesion to colonic endothelium was significantly (p < 0.001) enhanced following pretreatment with only 100 μM H2O2 (adherent cells/field: vehicle: 5.30 ± 0.41; 10 μM H2O2: 5.13 ± 1.57; 100 μM H2O2: 20.00 ± 5.70; 1 mM H2O2: 4.07 ± 2.20) (Fig. 3A, B). On IR-injured ileum sections, this dose also significantly enhanced HPC-7 cell adhesion (p < 0.01) compared to vehicle-treated cells (adherent cells/field: vehicle cells/IR sections: 72.86 ± 8.67; H2O2-treated cells/IR sections: 147.80 ± 20.01) (Fig. 3C). Furthermore, adhesion of H2O2-treated HPC-7 cells was significantly higher on IR sections than that seen with H2O2-treated HPC-7 cells on sham sections (adherent cells/field: H2O2-treated cells/sham sections: 62.91 ± 5.95; H2O2-treated cells/IR sections: 147.80 ± 20.01) (Fig. 3C). Similar data were obtained with jejunal tissue; on IR-injured sections, 100 μM H2O2 significantly enhanced HPC-7 adhesion (p < 0.01) compared to vehicle-treated cells (adherent cells/field: vehicle cells/IR sections: 35.45 ± 2.92; H2O2-treated cells/IR sections: 86.83 ± 13.62) (Fig. 3D). Adhesion of H2O2-treated HPC-7 was significantly higher on IR sections than that seen with H2O2-treated HPC-7 on sham sections (adherent cells/field: H2O2-treated cells/sham sections: 37.58 ± 10.33; H2O2-treated cells/IR sections: 86.83 ± 13.62) (Fig. 3D).

Pretreating HPC-7 with H2O2 enhances their adhesion to endothelial cells and frozen intestinal tissue in vitro. (A) Pretreating HPC-7 with 100 μM H2O2 significantly enhances their adhesion on TNF-α-activated murine colonic endothelium compared to vehicle control. No increases were observed with lower or higher doses. (B) Representative images of carboxyfluorescein diacetate, succinimidyl ester (CFSE)-labeled HPC-7 adherent on endothelium are shown following pretreatment with vehicle and 10 μM, 100 μM, and 1 mM H2O2. Pretreating HPC-7 with 100 μM H2O2 significantly enhances their adhesion on both (C) sham and IR-injured ileal tissue and (D) sham and IR-injured jejunal tissue when compared to vehicle-treated cells. Scale bars: 100 μm. Results presented as mean adhesion ± SEM (n ≥ 3 endothelial assays for A; n ≥ 6 tissue sections for C, D). ***p < 0.001, vehicle treated versus 100 μM treated (A). **p < 0.01, vehicle cells/IR sections versus H2O2 cells/IR sections; ##p < 0.01, H2O2 cells/sham sections versus H2O2 cells/IR sections (C, D).
Pretreating HPC-7 Cells with 100 μM H2O2 Enhances their Adhesion in IR-Injured Gut In Vivo and is Associated with a Concomitant Reduction in Pulmonary HPC-7 Recruitment
Recruitment of HPC-7 in IR-injured ileum was significantly (p < 0.01) enhanced following pretreatment with 100 μM H2O2 when compared to vehicle-pretreated cells (AUC: vehicle: 529.80 ± 140.80; 100 μM H2O2: 1877.00 ± 503.3) (Fig. 4A, B; videos available online at http://www.bham-mrg.co.uk). Pretreating HPC-7 with 1 mM or 10 μM H2O2 did not enhance recruitment compared to vehicle-treated cells (AUC: vehicle: 529.80 ± 140.80; 1 mM H2O2: 139.40 ± 8.32; 10 μM H2O2: 637.00 ± 107.5) (Fig. 4A, B). Similar results were obtained in the jejunum when examined post-intravital monitoring (adherent cells/field: vehicle: 4.29 ± 0.91; 1 mM H2O2: 0.80 ± 0.35;100 μM H2O2: 18.20 ± 6.50; 10 μM H2O2: 6.52 ± 2.28) (Fig. 4C). Pulmonary recruitment was significantly (p < 0.05) decreased following HPC-7 pretreatment with 100 μM H2O2 (adherent cells/field: vehicle: 92.43 ± 4.97; 100 μM H2O2: 67.10 ± 12.02) (Fig. 4D).

Pretreating HPC-7 with H2O2 enhances their adhesion in IR-injured gut in vivo and is associated with a concomitant reduction in pulmonary recruitment. (A) Pretreating HPC-7 with 100 μM H2O2 enhances their recruitment in IR-injured ileal microcirculation in vivo, whereas 1 mM reduces recruitment. (B) Representative intravital images of CFSE-labeled HPC-7 adherent in mucosal villi are shown following their pretreatment with vehicle and 10 μM, 100 μM, and 1 mM H2O2. (C) HPC-7 recruitment into injured jejunum is also enhanced following pretreatment with 100 μM H2O2. (D) Reduced pulmonary recruitment was also observed with this pretreatment strategy. Scale bars: 100 μm. Results presented as mean adhesion ± SEM (n ≥ 4 mice for A, C, D). *p < 0.05 versus vehicle-treated HPC-7.
Pretreating HPC-7 with 100 μM H2O2 Enhances Adhesion Through the β2 Integrin Without Increased Expression
Incubating H2O2-pretreated HPC-7 cells with blocking antibodies against β2 or α4 significantly (p < 0.01) reduced adhesion to endothelium in vitro (adherent cells/field: H2O2 alone: 20.00 ± 5.70; +IgG control: 19.49 ± 1.81; +anti-α4: 3.55 ± 0.56; +anti-b2: 3.67 ± 1.33) (Fig. 5A), but only anti-β2 was effective at significantly (p < 0.05) reducing recruitment in vivo in the ileum (AUC: IgG treated: 2241.00 ± 164.20; anti-α4: 1911.00 ± 500.80; anti-β2: 900.00 ± 274.50) (Fig. 5B).

Pretreating HPC-7 with H2O2 enhances adhesion through the β2 integrin without increased expression. (A) Functional blockade of α4 or β2 significantly reduces adhesion of 100 mM H2O2-pretreated HPC-7 on murine colonic endothelium compared to IgG treated cells. (B) Functional blockade of β2, but not α4, significantly reduces the enhanced vivo recruitment of H2O2-pretreated HPC-7 (solid black line: H2O2 and IgG-treated HPC-7; solid gray line: H2O2 treated and α4 blocked; dashed gray line: 100 μM H2O2 and β2 blocked). (C) Expression of α4 or β2 is not enhanced on HPC-7 following pretreatment with 100 μM H2O2 as observed by flow cytometry (filled area: negative control; black line: 100 μM H2O2; gray line: vehicle). (D) H2O2 (100 μM)-pretreated HPC-7 display higher levels of intracellular F-actin staining (phalloidin) compared to vehicle-treated cells and distinct cellular protrusions (bottom, white arrows), whereas vehicle-treated cells (top panel) do not. (E) H2O2 enhances HPC-7 adhesion to vascular cell adhesion molecule 1 (VCAM-1) (gray bars)- and intracellular adhesion molecule-1 (ICAM-1) (black bars)-coated surfaces. Functional blockade of α4 or β2 abolishes H2O2-mediated binding to VCAM-1 and ICAM-1, respectively. (F) Pretreatment of HPC-7 with H2O2 promotes integrin clustering of both α4 and β2 integrins compared to vehicle-treated controls. Maxima represent areas of clustering. Scale bars: 2 μm (D) and 5 μm (F). Results presented as mean adhesion ± SEM [n ≥ 3 separate protein assays for (A); n ≥ 4 mice for (B); n ≥ 3 cell samples for (C) and (D); n ≥ 5 separate protein assays for (E); n = 3 cell samples for (F)]. *p < 0.05, **p < 0.01, ***p < 0.001 versus labeled groups. MFI = mean fluorescent intensity.
Pretreating with 100 μM H2O2 did not enhance HPC-7 expression of α4 or β2 (Fig. 5C), but did result in a significantly (p < 0.05) higher level of intracellular F-actin mean fluorescent intensity (MFI) of phalloidin staining: vehicle: 28.47 ± 0.66; 100 μM H2O2: 34.33 ± 1.99) (Fig. 5D) and the formation of filopodia-like protrusions (Fig. 5D). HPC-7 adhesion to ICAM-1 was significantly (p < 0.01) enhanced following treatment with 100 μM H2O2 (adherent cells/field: vehicle: 2.84 ± 0.29; H2O2: 12.86 ± 1.41) (Fig. 5E). Specific blockade of the β2 integrin attenuated this enhanced adhesion (adherent cells/field: H2O2 + IgG: 15.67 ± 2.49; H2O2 + anti-b2: 2.94 ± 1.06) (Fig. 5E). Similarly, adhesion to VCAM-1 was significantly (p < 0.01) enhanced following H2O2 treatment (adherent cells/field: vehicle: 6.55 ± 2.24; H2O2: 42.21 ± 8.37) (Fig. 5E), and as expected, this enhanced recruitment was mediated by the α4 integrin (adherent cells/field: H2O2 + IgG: 42.00 ± 1.53; H2O2 + anti-α4: 6.63 ± 1.57) (Fig. 5E). Following 100 μM H2O2 pretreatment, both α4 and β2 subunits appeared to cluster on the HPC-7 surface, as seen through identification of maximal expression areas. No clustering was observed on vehicle-treated cells (Fig. 5F).
Treating HSCs with 100 μM H2O2 for 1 h Does Not Activate Cellular Apoptosis
Only HPC-7 treated for 1 h with 1 mM H2O2 displayed significant (p < 0.05) entry into early apoptosis, identified as cells with Ann Vpos/PIneg staining (Ann Vpos/PIneg cells per 1×104 total cells analyzed: vehicle: 214.00 ± 56.32; 1 mM H2O2: 2456.00 ± 986.00; 100 μM H2O2: 287.70 ± 109.30; 10 μM H2O2: 203.00 ± 48.13) (Fig. 6A). Treatment of HPC-7 cells with 1 mM H2O2 does not significantly increase the Ann Vpos/PIpos population when cells are analyzed at 1 h (data not shown). Clear population shifts were visible on scatter plots when comparing PBS-treated cells to 1 mM H2O-treated cells, but not for 100 or 10 μM (Fig. 6B-E).

Treating HPC-7 with 100 μM H2O2 for 1 h does not promote entry of cells into early apoptosis. (A) Treating HSCs with 1 mM H2O2 significantly enhances their entry into early apoptosis [as evidenced by increased Annexin V-positive/propidium iodidenegative (Ann Vpos/PIneg) staining], whereas 100 or 10 μM H2O2 does not. Early apoptotic cells are identified as PIneg/Ann Vhigh and late apoptotic cells are identified as PIhigh/Ann Vhigh. Representative flow cytometry plots are shown of HPC-7 treated with (B) vehicle, (C) 10 μM H2O2, (D) 100 μM H2O2, and (E) 1 mM H2O2. Results in (A) are presented as positive cells per 104 total cells (n = 3 treated cell samples per group). *p < 0.05 versus vehicle.
Treating HPC-7 Cells with 100 μM H2O2 Does Not Promote Cell Death, Senescence, or Function Loss
H2O2 at a dose of 100 mM did not significantly affect cell viability when observed post-H2O2 treatment (e.g., % cell viability at 72 h: vehicle: 85.48 ± 7.55%; 100 μM H2O2: 83.61 ± 3.81%) (Fig. 7A). Furthermore, this dose did not affect the ability of HPC-7 to grow over a 72-h time period (e.g., total viable cells at 72 h: vehicle: 5.73×106 ± 1.84×106; 100 μM H2O2: 5.34×106 ± 3.19×106) (Fig. 7B). In contrast, treatment of HPC-7 with 1 mM H2O2 significantly reduced viability and growth when compared to PBS-treated cells (e.g., % cell viability at 72 h: vehicle: 85.48 ± 7.55%; 1 mM H2O2: 0.51% ± 0.51%; p < 0.001; e.g., total viable cells at 72 h: vehicle: 5.73×106 ± 1.84×106; 1 mM H2O2: 7.00×103 ± 7.00×103; p < 0.01). No long-term loss of function was observed following 100 μM H2O 2 treatment as pretreated HPC-7 were still able to form megakaryocytes. This was confirmed by the generation of a subset of HPC-7 cells large in size and showing typical polyploid DNA staining following incubation with PI (Fig. 7C, D). Even without H2O2 pretreatment, HPC-7 displayed some baseline 8-oxo-dG staining, a marker for oxidative stress. However, increasing levels of intracellular 8-oxo-dg staining were observed with increasing H2O2 concentration (Fig. 7E).

Treating HPC-7 with 100 μM H2O2 for 1 h does not decrease cell viability or growth, nor does it prevent differentiation into cells of hematopoietic lineage. Treating HPC-7 with 100 μM H2O2 does not decrease (A) cell viability or (B) cell numbers counted at 24, 48, or 72 h posttreatment. H2O2 (100 μM)-treated HPC-7 remained functional, since thrombopoietin (TPO) treatment for 5 days could still differentiate them into megakaryocytes. (C) Populations of large cells (megakaryocytes) appear in both vehicle-treated (left) and H2O2-treated (right) populations. (D) Flow cytometry analysis reveals that differentiated cells from both vehicle (left) and H2O2 (right) treatment groups displayed polyploidy, a characteristic feature of megakaryocytes. (E) Vehicle-treated HPC-7 display a small shift for 8-oxo-dG (black line) compared to negative control antibody (filled area), suggesting a degree of basal oxidative stress. (F) Treatment of HPC-7 with 1 mM H2O2 (solid line) causes a larger shift in intracellular 8-oxo-dG staining than seen with 100 μM H2O2 treatment (dashed line). 8-Oxo-dG staining in vehicle-treated cells is also shown (filled area). Results presented as % cell viability (A, n ≥ 3 treated cell samples) and viable cell count (B, n ≥ 3 treated cell samples). Flow cytometry plots representative of three experiments. **p < 0.01 versus vehicle. SSC = side scatter; FSC = forward scatter.
Discussion
This study provides the first evidence that progenitor cell recruitment to the gut is a regulated event and that recruitment achieved by injury alone is not maximal. H2O2, present in appreciable levels in ICM, could enhance HPC-7 adhesion, most likely through increasing β2 integrin binding to ICAM-1, integrin clustering, F-actin polymerization, and filopodia-like protrusion formation. Since the effective H2O2 concentration was not cytotoxic and decreased pulmonary retention, it has the potential to be used clinically as an adjuvant therapy to enhance stem/progenitor cell recruitment.
H2O2 can modify the affinity of leukocyte α4 integrin and promote conversion of both leukocyte β1 and β2 from inactive into active conformations (5,27). Similar events taking place on HPC-7 integrins can explain the enhanced adhesion seen in the current study. H2O2 also activates the nuclear factor k-light-chain-enhancer of activated B cells (NF-kB) pathway, which controls transcription of around 60 genes for inflammatory cytokines and chemokines (31,43). It is therefore plausible that H2O2 triggers cytokine release, which subsequently activates HPC-7 integrins in an autocrine or paracrine fashion. Indeed, we have shown that pretreating HPC-7 with interleukin (IL)-1β (itself a target gene of NF-κB) enhances HPC-7 adhesion to activated endothelium and VCAM-1 (Kavanagh et al., manuscript in preparation). Although H2O2 can mobilize the β2 integrin subunit from large intracellular stores to the surface on neutrophils and eosinophils (13,26), increased HPC-7 integrin expression was not observed. While studies have identified intracellular α4 and β2 in HSCs, it is unclear whether they can be transferred to the surface at a similar speed to that seen in leukocytes (6). Hence, it is possible that the relatively acute treatment time frames used in this study may be insufficient to permit surface integrin upregulation in HSCs.
Interestingly, while modest H2O2 treatment enhances adhesion to both protein surfaces and endothelium via the β2 and α4 integrins, blocking the α4 integrin does not attenuate H2O2-mediated HPC-7 adhesion in vivo in the injured gut. This is likely a result of endothelial counterligand availability in the IR-injured intestine. Indeed, basal VCAM-1 expression in murine intestine is relatively lower than other tissues such as the brain, heart, and mesentery (10). In addition, work from our lab has identified that the recruitment of naïve untreated HPC-7 cells to the gut following IR also depends on the β2, but not the α4, integrins (Kavanagh et al., manuscript in preparation). Furthermore, when challenged with an intravenous injection of TNF-α, intestinal upregulation of VCAM-1 is observed at 2 h, with peak VCAM-1 expression at 9 h post-TNF-α injection (10). Since HPC-7 is introduced in the current study at time points prior to significant VCAM-1 upregulation induced by IR injury, it is not surprising that their recruitment is mediated by basally expressed endothelial ICAM-1. It is possible that H2O2 pretreatment may be equally effective at increasing HPC-7 retention within those organs in which recruitment depends upon the α4 integrins, such as the liver (15). Hence H2O2 pretreatment may have universal appeal as a strategy for enhancing HPC-7 recruitment within a variety of injured sites.
Unlike other ROS mediators such as hydroxyl radicals and superoxide anions, H2O2 readily diffuses across plasma membranes and can thus act as a potent intracellular signalling messenger (8,25). This may explain why H2O2 was able to induce morphological changes and increase G-actin polymerization to its filamentous form, F-actin. Since cell migration requires the coordinated formation of filopodia at the cell front, the development of such structures prior to administration may underlie the enhanced migratory responses to local chemokines and thus their subsequent adhesion. Furthermore, we provide evidence suggesting that H2O2, through its effects on clustering, was also able to modulate integrin avidity, the increased adhesiveness independent of integrin conformational changes. Evidence is emerging that dynamic reorganization of integrins into microclusters, through F-actin polymerization, is the major mechanism regulating integrin binding strength and is a prerequisite for their activation and ligand binding (39). Changes in levels of cytoskeletal elements and integrin clustering may play a major role in H2O2-mediated HPC-7 adhesion.
H2O2 is frequently used experimentally as a means of generating oxidative damage and apoptosis within cells (7,19). Therefore, there was a possibility that the enhanced HPC-7 adhesion observed was simply due to toxic effects. However, either concentrations higher than 100 mM or longer treatment durations of the same dose are required to induce cellular damage (7,19). In the current study, HPC-7 cells treated for only 1 h with 100 μM continued to proliferate and maintain their differentiation potential. While some increase in 8-oxo-dG expression was seen in these cells, the level of oxidative stress experienced did not appear to be sufficient to trigger apoptosis. Interestingly, MSCs are protected from apoptosis following oxidative preconditioning, suggesting that H2O2 pretreatment could also aid in enhancing HSC survival in ischemic tissues (23). It is also important to note that H2O2 is generated physiologically during inflammation by ECs, smooth muscle, and activated macrophages and neutrophils (3,7). Indeed, resident neutrophils can generate local concentrations of 200–500 μM H2O2 upon stimulation (12). This estimate provides a physiologically relevant source for the effective H2O2 concentration used in the present investigation as circulating HSCs would be exposed to these levels locally. Hence, pretreating HSCs with H2O2 at physiologically relevant concentrations could therapeutically enhance tissue retention, effectively “priming” them for maximal adhesion prior to their systemic delivery.
Pulmonary entrapment is also a major obstacle for systemic stem cell delivery for regenerative purposes, both experimentally and clinically (9). Indeed, patients receiving exogenous HSCs for hematologic disorders experience posttransplant pulmonary injury due to cells becoming trapped in lung capillaries (9,20). In addition to promoting lung damage, pulmonary entrapment significantly reduces the available pool of circulating transplanted HSCs available for recruitment to injured tissues. This study is the first to provide a strategy that not only effectively enhances recruitment into specific sites of tissue injury but also concomitantly decreases their entrapment within lungs. This observation may simply be related to cell availability; that is, enhanced intestinal recruitment means less cells are available for pulmonary entrapment.
In conclusion, although there is increasing evidence that HSCs are beneficial following tissue injury, the efficacy of such therapies is likely to be proportional to the degree of cell recruitment that can be achieved. Enhancing the effectiveness of regenerative processes may therefore depend on identifying and then modulating the adhesive mechanisms that underpin stem cell trafficking. We provide a simple, quick, and effective method for enhancing HPC-7 adhesion into injured gut through modulation of surface integrins. Previous studies have enhanced stem cell recruitment by introducing genes that encode for the potent stem cell chemoattractant SDF-1α within cardiac tissue (37). However, the clinical applicability of such techniques is debatable and may be associated with aggravated tissue injury due to side effects such as SDF-1α-dependent lymphocyte recruitment (24). The current study achieves a similar, if not better, increase in recruitment but offers the benefit of not requiring genetic manipulation and, crucially, does not require manipulation of host tissues to achieve enhanced recruitment. The data in this study may help in the design of future cellular therapies using HSCs. It is anticipated that enhancing HSC recruitment to injured organs may expedite the recovery process and encourage therapeutic success clinically in the field of regenerative medicine.
Footnotes
Acknowledgments
This research was supported by project grants from the British Heart Foundation (PG/08/043/25067) and the Bardhan Research and Education Trust (09-0675). Author contributions: D.P.J.K. designed and performed experiments, collected and analyzed data, and prepared the manuscript; A.I.Y. designed and performed experiments and drafted the manuscript; J.S.A. provided reagents and drafted the manuscript; J.F. drafted the manuscript; N.K. designed experiments, analyzed data, prepared the manuscript, obtained funding, and supervised the project. The authors declare no conflict of interest.
