Abstract
Pancreatic islet transplantation is a promising method for curing diabetes mellitus. We proposed in this study a molecularly engineered islet cell clusters (ICCs) that could overcome problems posed by islet transplantation circumstances and host's immune reactions. A gene containing highly releasable exendin-4, an insulinotropic protein, was delivered into single islet cells to enhance glucose sensitivity; thereafter, the cells were reaggregated into small size ICCs. Then the surface of ICCs was modified with biocompatible poly(ethylene glycol)-lipid (PEG) (C18) for preventing immune reactions. The regimen of ICCs with low doses of anti-CD154 mAb and tacrolimus could effectively maintain the normal glucose level in diabetic mice. This molecularly engineered PEG-Sp-Ex-4 ICC regimen prevented cell death in transplantation site, partly through improving the regulation of glucose metabolism and by preventing hypoxia- and immune response-induced apoptosis. Application of this remedy is also potentially far-reaching; one would be to help overcome islet supply shortage due to the limited availability of pancreas donors and reduce the immunosuppressant regimens to eliminate their adverse effects.
Introduction
Transplantation of isolated pancreatic islets has shown a tremendous potential for curing patients with type I diabetes (23,25), but the fate of transplanted islets faces many challenges such as a hypoxic condition and aggressive host's immune reactions. To immunologically protect transplanted islets, several attempts have been made. The “Edmonton protocol,” which used several immunosuppressive drugs, has generated tremendous hope for diabetic patients, although insulin independence was difficult to maintain over time (26). Other attempts included the use of biomaterials in which islets were entrapped with semipermeable membrane made with alginate and hydrogel (8,20,28). However, the encapsulation of islets incurred cell death due to hypoxia caused by the relatively large diameter of capsule and nonspecific inflammation initiated caused by the impurity of biomaterials (6,7).
Furthermore, hypoxia of the transplanted islets at the transplantation site has been demonstrated to involve with the early loss of islet graft function (3,5). Transplanted islets with smaller size would be less susceptible to immune reaction (4) and to ischemia and starvation because of adequate oxygen and nutrient supplies in the core of the islets (18,21). To overcome these problems, islet cell clusters (ICCs), which are the small aggregates of islet single cells, have been proposed as a good candidate for replacing islets (10). However, it has been reported that a glucose-stimulated insulin release from ICCs was lost because of the disruption of communication between extrinsic signaling molecules on islet cell surface (2,14,15).
It is known that exendin-4 (Ex-4), a glucagon-like peptide-1 (GLP-1) analogue, can ameliorate the function of endocrine pancreatic islets and attenuate the hypoxia-induced apoptosis and inflammation (1,9,19). In the previous study, we designed a highly releasable Ex-4 protein using a secretion signal peptide (Sp), thereby highly improving hypoxia-induced apoptosis (13). Therefore, the transfer of an Ex-4-encoding gene into ICCs might improve the viability of these cell clusters as well as β-cell function of an insulin secretion. In addition, we also chemically modified the islet surface with biocompatible polyethylene glycol (PEG) via amide bond to further protect transplanted islets against immune reactions (12,29). This surface modification of islets could synergistically protect the transplanted islets with a low dose of immunosuppressive drugs against immune reactions (17).
In this study, we developed a convergent system of PEG-Sp-Ex-4 ICCs by physically and genetically engineered ICCs (Fig. 1). To this end, Sp-Ex-4 gene was first transferred into single islet cells, which were then aggregated to form Sp-Ex-4-transduced ICCs (Sp-Ex-4 ICCs). The surface of Sp-Ex-4 ICCs was physically camouflaged with PEG-lipid. We evaluated the functionality and viability of PEG-Sp-Ex-4 ICCs in vitro and in vivo. We also evaluated whether the molecularly engineered ICCs could be protected against hypoxia-induced apoptosis. In particular, when molecularly engineered ICCs were accompanied with low doses of anti-CD154 mAb and tacrolimus, their survival rate was evaluated in a xenograft model.
A schematic representation of the newly developed protocol using polyethylene glycolsecretion signal peptide-exendin-4 islet cell clusters (PEG-Sp-Ex-4 ICCs). GLP-1, glucagon-like peptide-1; APC, antigen presenting cell; IL-2, interleukin-2.
Materials and Methods
Preparation of Lentiviral Vector for Ex-4 Gene Transduction Into ICCs
The Ex-4 cDNA was chemically synthesized and inserted into the pβ vector at the EcoRI and XbaI sites. The DNA fragment encoding the secretion signalpeptide (Sp) from Ex-4 was also chemically synthesized and inserted into pβ-Ex-4 at the KpnI and EcoRI sites to create pβ-Sp-Ex-4. The Sp-Ex-4 fragment was amplified from pβ-Sp-Ex-4: The forward primer sequence was 5′ CGGAATTCATGAAGATCATCCTGTGGCTG-3′ and the reverse primer sequence was 5′-GCGATATCTCACG ATGGCGGAGG-3′. The forward primer sequence of the β-actin for HEK 293T cells was 5′-TGGCACCACACC TTCTACAATGAGC-3′, and the reverse primer was 5′-GCACAGCTTCTCCTTAATGTCACGC-3′; for rat islet, the forward primer was 5′-CACCCGCGA GTACAACCTTC-3′ and the reverse primer was 5′-C CCATACCCACCATCACACC-3′.
The recombinant lentivirus vector contained three components (1:1:1 ratio): the transfer vector, pHR'CMV-Sp-Ex-4-pgk-eGFP, controlled by the pgk promoter; the lentivirus packaging plasmid pCMV8.91 encoding Gag, Pol, Tat, and Rev proteins; and the envelope plasmid pMD.G expressing the vesicular stomatitis virus glycoprotein (VSV-G) envelope protein. Lenti-Sp-Ex-4 were produced in 293T cells by transient Lipofectamine Plus® (Invitrogen Co., Carlsbad, CA) cotransfection. Forty-eight hours after transfection, the culture supernatant containing the virion particles was harvested and clarified with a 0.45-μm-membrane filter (Nalgene Nunc, ThermoFisher Scientific, Rochester, NY) and stored at a deep freezer. The virus titer was determined by transducing HEK 293T and HeLa cells with the viral preparation and examining the enhanced green fluorescent protein (eGFP) expression using fluorescence-activated cell sorting (FACS) analysis. The eGFP expression was observed using a fluorescence microscope (Nikon eclipse TE2000U, Tokyo, Japan). Fourteen hours after transduction, the total RNA was extracted from the cells using the RNeasy Mini Kit (Qiagen, Inc., Valencia, CA). The purified RNA was re verse-transcribed into cDNA by reverse transcription (RT) reaction (90 min at 37°C). Then, the DNA was synthesized from the cDNA in the presence of heat stable Ex-Taq polymerase (Takara Bio, Inc., Tokyo, Japan), sequence-specific DNA primers, and dNTPs mix.
Preparation of Genetically Engineered ICCs: Sp-Ex-4 ICCs
Outbred Sprague–Dawley (SD) male rats, 7 weeks, weighing 230–250 g, were purchased from Orient-Bio, Inc. (Gyeonggi, South Korea) and were housed in ventilated cages under specific pathogen-free condition at our institution. All animal procedures were approved by the Institutional Animal Care and Use of Committee, which is certified by the Institute of Laboratory Animal Resources of Seoul National University. SD rats were anesthetized by intraperitoneal injection of pentobarbital. The long ventral midline was incised, and the common bile duct near the hilus of the liver was cannulated and injected with collagenase. The digested tissue was separated using discontinuous Ficoll™ PM400 (Amersham Biosciences) density gradient centrifugation and then handpicked (16). Pancreatic islets were dissociated into single cells by suspending them in 0.25% of trypsin–EDTA (Gibco) solution. The dissociated single cells were transduced with a Sp-Ex-4-expressing vector at multiplicity of infection (MOI) of 12.5 for 5 h. After the incubation, single cells containing the Sp-ex-4 gene were recultured with 6 × 105 single cells in a 12-well microplate. After 6 days of spontaneous reassociation, aggregated islet single cells were considered as ICC. Flow cytometry analysis (BD FACSCalibur™; BD Biosciences) was used to count the number of cells expressing GFP. Alternatively, cells were analyzed by fluorescent microscopy (Nikon Instrument). Secreted Ex-4 from the Sp-Ex-4-transduced ICCs containing media was used to determine Ex-4 levels using Ex-4 EIA kit (Phoenix Pharmaceuticals).
Glucose Sensitivity of Sp-Ex-4 ICCs
Samples of each group was washed with normal Hank's balanced salt solution (HBSS) and added with 1 ml of Krebs–Ringer bicarbonate buffer (KRBB) containing a solution of low glucose concentration (2.8 mM) for 30 min in a humidified incubator. After starvation, the medium was exchanged with the same buffer to measure glucose-stimulated insulin secretion during 2-h incubation. Then, the medium was again exchanged with KRBB solution containing 28 mM glucose. The secreted insulin in the medium was measured using an ultrasensitive mouse insulin ELISA kit (Millipore, MA). Insulin levels were determined by absorbance at 450 nm and calculated based on appropriate standard curves.
Effect of Ex-4 on Preventing Hypoxia-Induced Apoptosis
The evaluation of live/dead fluorescence imaging was carried out to compare the viabilities of ICCs and islets using the Living/Dead Viability/Cytotoxicity Kit (Molecular Probe, Invitrogen Co.). On the other hand, the viabilities of islets, ICCs, and Sp-Ex-4 ICCs were quantitatively calculated by carrying out cell counting kit-8 assay (Dojindo Molecular Technologies), which was not affected by GFP produced by Sp-Ex-4 ICCs. CCK-8 allows sensitive colorimetric assays for the determination of the number of viable cells in cell viability assays. In my experiment, 100 IEQ of cells were cultured in 96-well culture plate for 24 h at 37°C under the humidified atmosphere containing 5% CO2. Then, addition of CCK-8 solution allowed quantitative evaluation of live cells in the medium. The absorbance value at 450 nm was normalized by the amount of DNA retrieved from each well. Then the relative cell viability was calculated in each cell type by comparing the absorbance value of experimental samples and freshly isolated islets. Then, ICCs were placed in a 12-well culture plate and incubated for 24 h either at normoxic condition (20% O2) or at hypoxic condition (1% O2). Caspase-3/7 Glo® assay solution (Promega, Madison, WC) was used to measure the activity of both caspase-3 and caspase-7. The light intensity was proportional to the amount of caspase-3 or caspase-7 activities.
Surface Modification of Sp-Ex-4 ICCs (PEG-Sp-Ex-4 ICCs)
PEG-lipid (C18) (molecular weight, 20,000 Da) was added to the Sp-Ex-4 ICC suspension and then incubated at 37°C under the humidified atmosphere containing 5% CO2. After 1 h of incubation, Sp-Ex-4 ICCs grafted with PEG-lipid (C18) were washed twice with the culture media, followed by a 1-day culture. Fluorescein isothiocyanate (FITC)-labeled PEG-lipid was used to evaluate the distribution of grafted PEG-lipid (C18) on the surface of Sp-Ex-4 ICCs. The PEG-Sp-Ex-4 ICCs were visualized using a confocal scanning laser microscope (Carl Zeiss-LSM510).
Transplantation and Intraperitoneal Glucose Tolerance Test (IPGTT) of PEG-Sp-Ex-4 ICCs in Diabetic Mice
Mice became diabetic 3 days after the intraperitoneal injection of streptozotocin (Sigma) dissolved in citrate buffer (pH 4.5). Diabetes was diagnosed by hyperglycemia (above 350 mg/dl of nonfasting glucose level) over three consecutive measurements. A mixed solution of ketamine (50 mg/ml) and xylazine (23.32 mg/ml) was used to anesthetize transplant recipients. The left kidney was exposed through a lumbar incision, and capsulotomy was performed on the caudal outer layer of the left kidney, after which, ICCs were injected. Transplantation was considered as successful if the blood glucose level returned to normoglycemia (80–200 mg/dl) and was maintained for more than 2 consecutive days.
To evaluate the synergistic effect of the surface modification with PEG-lipid (C18) in combination with immunosuppressive drugs, anti-CD154 mAb (0.2 mg) and tacrolimus (0.2 mg/kg) (Astellas Pharma) were injected into the C57BL/6. Anti-CD154 mAb was administered every 2 days up to day 6 post-transplantation and tacrolimus was given daily into the intraperitoneal cavity of each recipient. Then the left kidney, bearing the transplanted ICCs, was removed. If the blood glucose level was rapidly increased above 300 mg/dl, the function of grafted ICCs could be confirmed. IPGTT at day 15 of transplantation was performed to evaluate the glucose sensitivities of both ICCs and PEG-Sp-Ex-4 ICCs. Examined groups were fasted at least for 6 h before the experiment with free access to water prior to the glucose tolerance test. A solution of 20% d-glucose (Sigma) was injected into the peritoneal cavity, and the change of the blood glucose was measured from the tail vein at different time intervals.
Immunohistochemistry
After nephrectomy, the incised left kidneys bearing the grafts were fixed in 4% paraformaldehyde for 24 h. They were embedded in paraffin and sectioned at 4 μm. The tissue sections were deparaffinized in xylene and then rehydrated in serially graded alcohol. Frozen tissues were cut into sections and stained with hematoxylin and eosin and immunostained with mouse CD4+ (1:100), CD8a+ (1:100), or CD20+ (1:150) mAb (Abcam) using a conventional avidin–biotin–peroxidase method. The presence of insulin and Ex-4 was detected using mouse monoclonal anti-insulin (Abcam) and Ex-4 (Phoenix Pharmaceuticals) at a 1:50 and 1:10,000 dilutions in a humidified chamber.
Statistical Analysis
All values are expressed as means ± SEM. The “n” numbers in the Results section relate to the number of animals or samples used for the experiment. All the collected data were analyzed using the unpaired t test or ANOVA one-way test with post hoc test (Bonferroni) (Sigma Plot, Systat Software, Inc.). A value of p < 0.05 was considered to be statistically significant.
Results
Design of Sp-Ex-4-Transduced Islet Cell Cluster (Sp-Ex-4 ICCs)
Isolated pancreatic islets were dissociated into islet single cells in 0.25% trypsin–EDTA solution. On day 6, following static incubation in the culture media, islet single cells (Fig. 2A, second panel) were again aggregated, thereby forming ICCs as round-shaped clusters (Fig. 2A, third panel), whose average size was in the range of 60–90 μm (Fig. 2B). There were no marked differences between pancreatic islets and ICCs regarding the general morphological aspects of the endocrine cells, the structure of organelles, and their secretory granules. The β-cell secretory granules of both samples displayed an electron dense core, as confirmed by TEM analysis. Cell borders were clearly defined, and the peripheral cells formed a continuous smooth boundary. The component cells were also closely packed (Fig. 2C, D). Lentiviral vector was constructed as described in the Methods section (Fig. 3A). The signal peptide was added to the vector in order to enhance the Ex-4 expression level. Transduction of Sp-Ex-4 was confirmed using Western blot and fluorescent microscopy (Fig. 3B). Lentiviral vector successfully transferred Ex-4 gene to rat ICCs in vitro. Transduction efficiency of single islet cells was 14.3% ± 3.7% (Fig. 3C), and GFP was expressed in the whole region of ICCs 4 days following transduction (Fig. 3B). The Ex-4 secretion rate from Sp-Ex-4 ICCs was about 0.4 ng/DNA/day, which was much higher than that from Sp-Ex-4 transduced islets (0.05 ng/DNA/day) since the transduction efficiency of single islet cells was much higher than that of islets (Fig. 3D). The recovery yield of ICCs, after dissociation of islets and their reaggregation, was about 75% based on IEQ. And the recovery yield of Sp-Ex-4 ICCs was about 50%.
(A) Morphology of islets, dispersed single cells, and reaggregated ICCs, respectively. Scale bars: 50 μm. (B) Size distributions of reaggregated single cells. Transmission electron m icroscope (TEM) image of (C) islets and (D) ICCs. Scale bar: 2 μm. (A) A schematic diagram of the novel lentiviral vector constructs. The insertion of signal peptide sequence (Sp) is shown for enhanced exendin-4 (Ex-4) expression. (B) (Top) Photographs of GFP expression throughout HeLa, HEK 293T, islets, and ICCs observed at day 4 of transduction using a fluorescence microscope (NIKON, Eclipse TE2000S) and NIKON digital camera system. Each of the left and right panels represents the image of green fluorescent protein (GFP)-expressing cells and the corresponding visible optical image. Scale bars: 50 μm (islets) and 50 μm (ICCs). (Below) An RT-PCR analysis for the expression of Sp-Ex-4 mRNA in HEK 293T cells transduced with lentivirus. Lane 1: Sp-Ex-4 expression plasmid vector. Lane 2: HEK 293T cells transduced with Mock lentivirus. Lane 3: HEK 293T cells transduced with Sp-Ex-4 lentivirus. Lane 4: islets transduced with Mock lentivirus. Lane 5: islets transduced with Sp-Ex-4 lentivirus. (C) An analysis of transduction efficiency of Sp-Ex-4 into single islet cells using flow cytometry. (D) Ex-4 secretion profile from (○) Sp-Ex-4-ICCs, (•) ICCs, (A) Sp-Ex-4 transduced islets, and (▾) control islets for 12 days after 5 h of transduction. (E) Influence of Sp-Ex-4 transduction in the glucose stimulated insulin release of ICC under static incubation condition. The functionality of the transduced islets was evaluated by monitoring the amount of insulin secreted from the islets in a Krebs–Ringer bicarbonate buffer (KRBB; pH 7.4), containing low (2.8 mM) or high (28 mM) glucose concentration for 2 h, respectively. (F) The stimulation index (SI) of islets, ICCs, and Sp-Ex-4 ICCs. The data are expressed as the mean ± SD (n = 5). *p < 0.001, comparing the values of Sp-Ex-4-transduced islets and Sp-Ex-4-transduced ICCs. **p < 0.001, comparing the values of ICCs and Sp-Ex-4-transduced ICCs. ***p < 0.05, comparing the values of ICCs and Sp-Ex-4-transduced ICCs.

Glucose Sensitivity of Sp-Ex-4 ICCs
Downregulation of β-cell function was observed after the aggregation of single islet cells into ICCs (Fig. 3E). ICCs that were transduced with Sp-Ex-4 gene significantly enhanced the amount of secreted insulin at high glucose stimulation (123.6 ± 5.9 ng/DNA/2 h) compared to the control islets (64.8 ± 10.9 ng/DNA/2 h). On the other hand, ICCs showed a similar insulin release level in both high (55.3 ± 14.4 ng/DNA/2 h) and basal (50.1 ± 8.5 ng/DNA/2 h) glucose stimulations. Therefore, the stimulation with high glucose concentration resulted in a robust release of insulin in Sp-Ex-4 ICCs that was twofolds higher than the basal levels, whereas the nonengineered ICC group was not responsive to high glucose stimulation, as reflected by its insulin concentration level, which is lower than that of Sp-Ex-4 ICCs. Since the glucose sensitivity of a cell is determined by the capacity to control the amount of insulin at high and low glucose concentrations, stimulation index (SI) is used to calculate the glucose sensitivity of Sp-Ex-4 ICCs. There was a twofolds increase in glucose sensitivity of Sp-Ex-4 ICCs when compared to the nonengineered ICCs, for which SI was 1.
Viability of Sp-Ex-4 ICCs Under a Hypoxic Condition
To examine the long-term effect of Ex-4 gene insertion on cell viability, we cultured the cell for 25 days under a static incubation condition. Surprisingly, the viability of Sp-Ex-4 ICCs was constant and remained without any decrease until day 25 of transduction (Fig. 4A, B). On the other hand, the viability of islets was continuously decreased with time up to 21.0% ± 6.5% at day 25 of incubation. Even though ICCs could improve the cell viability, its viability was still decreased with time up to 54.5% ± 4.9% at day 25 of incubation. In terms of relative cell viability under a hypoxic condition (1% oxygen) in vitro, it was observed that the viability of Sp-Ex-4 ICCs was also constantly maintained (103.5% ± 3.5%). However, the viability of ICCs and islets was decreased as much as 68.7% ± 6.2% and 55.4% ± 8.5%, respectively, compared from the viability of cells in a normoxic condition (Fig. 4C). In the hypoxic condition in vitro, when the caspase activity in cells were measured and compared from completely dead cells, islets and ICCs showed significantly increased caspase activities from normoxia to hypoxia, such as 82.8% ± 14.2% and 53.6% ± 10.4%, respectively. However, the caspase activity in Sp-Ex-4 ICCs was only 30.1% ± 13.3% in the hypoxic condition (Fig. 4D).
Cell viability assessment for 25 days under static incubation condition. (A) Results of CCK-8 assay for (○) ICCs, (•) islets, and (▾) Sp-Ex-4 ICCs under static incubation condition. (B) Live/dead cells imaging assay. ICCs and islets are treated with calcein AM (green) and ethidium homodimer-1 (red), where green and red indicate live and dead cells, respectively. ICCs (left; scale bar: 50 μm) and islets (right; scale bar: 100 μm) were observed and photographed under a fluorescence microscope (NIKON, Eclipse TE2000S). (C) Cell viability test and (D) caspase activity in islets, ICCs, and Sp-Ex-4 ICCs under hypoxia. Sp-Ex-4-transduced and untransduced cells were cultured under normoxic condition (20% O2 and 5% CO2), followed by 24 h of incubation in hypoxic condition (1% O2 and 5% CO2). Data are expressed as mean ± SD (n = 5). *p < 0.05, comparing values for Sp-Ex-4-transduced ICCs and ICCs.
Transplantation of Sp-Ex-4 ICCs in Diabetic Nude Mice
To clarify the effect of transduction of SP-Ex-4 gene on the β-cell function and viability in hypoxia and apoptotic conditions, these effects of Sp-Ex-4 ICCs were compared to those of nonengineered ICCs in immunodeficient athymic (BALB/c) nude mice. IEQ is a unit that defines the insulin secretion capacity based on islet diameter (22). In this study, the same equivalent factor used to count islets was used to quantify the amount of ICCs required for transplantation. If the blood glucose level was maintained in the normal range, the kidney in which ICCs or Sp-Ex-4 ICCs were transplanted was removed at day 30 to confirm whether the maintained normoglycemia was due to the transplanted ICCs. To achieve euglycemia, 500 IEQ of ICCs were not sufficient, but 1,000 IEQ of ICCs were critical for controlling blood glucose level (Fig. 5A, B). On the other hand, 500 IEQ of Sp-Ex-4 ICCs produced better outcomes by decreasing the number of islet grafts required to restore normoglycemia (Fig. 5C). Overall, 1,000 IEQ of ICCs and 500 IEQ of Sp-Ex-4 ICCs exhibited a similar graft survival rate after transplantation (Fig. 5D), indicating the beneficial effect of Ex-4 in preventing hypoxia- induced cell death at transplantation site and in increasing β-cell function by maintaining normoglycemia. Fifteen days after transplantation, IPGTT was performed to evaluate the functionality of the graft. Both 1,000 IEQ of ICCs and 500 IEQ of Sp-Ex-4 ICCs transplanted groups exhibited a glucose clearance similar to the normoglycemic control mice (Fig. 5E).
Monitoring of blood glucose levels of BALB/c nude mice transplanted with ICC and Sp-Ex-4 ICC grafts for 40 days. (A) 500 IEQ of ICC recipients (n = 6), (B) 1,000 IEQ of ICC (n = 5), and (C) 500 IEQ Sp-Ex-4 ICC recipients (n = 6). On the following nephrectomy (islet graft removal at day 30), all recipients became hyperglycemic. (D) Graft survival rate after transplantation of ICCs and Sp-Ex-4 ICCs. (•) 500 IEQ of ICCs, (▾) 500 IEQ of Sp-Ex-4 ICCs, and (▄) 1,000 IEQ of ICCs. (E) Intraperitoneal glucose tolerance test (IPGTT) of ICC and Sp-Ex-4 ICC transplant mice after the intraperitoneal injection of 20% glucose solution at day 15. (•) Nondiabetic nontransplanted control mice (n = 4), (○) 500 IEQ of Sp-Ex-4 ICCs (n = 5), (▾) 500 IEQ of ICCs (n = 6), and (A) 1,000 IEQ of ICCs (n = 5). Results are expressed as mean ± SEM. *p < 0.05, compares the values of 500 IEQ of ICCs with 500 IEQ of Sp-Ex-4 ICCs, 1,000 IEQ of ICCs, and normal healthy mice.
Histological analysis revealed a significant amount of β-cell mass in Sp-Ex-4 ICCs, indicating the role of Ex-4 in blocking the hypoxia-induced early graft loss after transplantation. Insulin+ cells were stained in Sp-Ex-4 ICCs, indicating that the Sp-Ex-4 ICCs graft played a critical role in maintaining glucose homeostasis after transplantation into the kidney. Ex-4 secretion enhanced β-cell function, which, in turn, produced more insulin to regulate blood glucose level. Ex-4+ cells were only stained in Sp-Ex-4 ICCs (Fig. 6), indicating that the improved glucose regulation was primarily due to the continuous secretion of Ex-4 from the graft.
Histological analysis of transplanted ICC grafts after removal of the left kidney 7 days after transplantation. (Left) H&E stain, anti-insulin+ cells, anti-exendin-4+ cells of 500 IEQ of control ICC transplant grafts; (right) H&E stain, anti-insulin+ cells, anti-Ex-4+ cell of 500 IEQ Sp-Ex-4 ICC transplant grafts survived for 30 days. Asterisk, transplanted islets. Scale bar: 50 μm.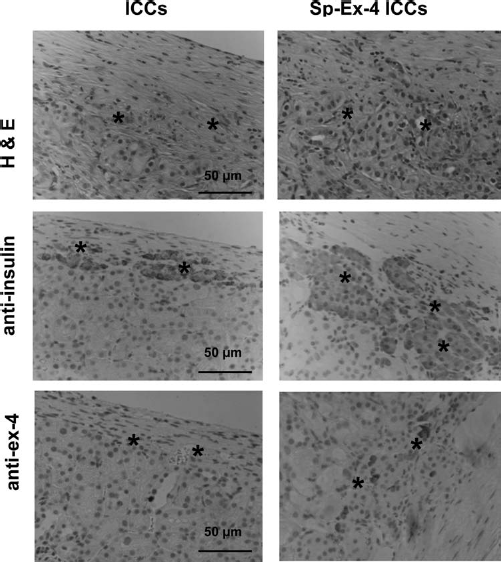
Transplantation of PEG-Sp-Ex-4 ICCs in Diabetic C57BL/6 Mice
To clarify the effect of PEGylation on immune reactions, PEG-Sp-Ex-4 ICCs were compared to non-PEGy-lated Sp-Ex-4 ICCs in immunocompetent C57BL/6 mice. PEG-lipid (C18) was efficiently anchored to the surface of genetically engineered ICCs. Evenly distributed FITC-labeled PEG-lipid on the surface of ICC was visualized (Fig. 7A). Notably, PEG conjugation to the cell (both PEG-ICCs and PEG-Sp-Ex-4 ICCs) did not affect cell viability but was stably attached to the live cells (Fig. 7B). The viability of PEG-ICCs and Sp-Ex-4 ICCs were 104.1% ± 7.5% and 93.7% ± 10.6%, respectively. These grafts were transplanted under the left kidney capsule, and they are subject to immune rejection after transplantation. The respective median survival time of mice receiving 500 IEQ of ICCs and Sp-Ex-4 ICCs were 8.0 ± 2.0 days and 11.0 ± 0.5 days, respectively (Fig. 8A, B). Following the transplantation of PEG-Sp-Ex-4 ICCs, the mean survival time was only 13.0 ± 0.5 days (Fig. 8C). Since 500 IEQ of ICCs failed to restore normoglycemia, the study on PEG-ICCs was omitted. Therefore, prompted by the substantial therapeutic benefits achieved with PEG molecule on the surface of Sp-Ex-4 ICCs, we further examined the utility of this system in delaying graft rejection in the context of kidney transplantation following administration of low doses of immunosuppressive drugs. When we used low doses of anti-CD154 mAb and tacrolimus, the immune reactions were more effectively prevented (Fig. 8D). Notably, all mice groups receiving PEG-Sp-Ex-4 ICCs along with low doses of anti-CD154 mAb and tacrolimus prolonged islet cell survival rate without any hyperglycemia (Fig. 8E, F). All mice became hyperglycemic following the removal of the left kidney bearing islet grafts.
(A) Chemical structure of PEG-conjugated octadecylamine (C18) and confocal laser scanning images of fluorescein isothiocyanate (FITC)-PEG-lipid (C18) modified rat islets. PEG-lipid (C18) was introduced on the surface of islets by hydrophobic interaction. The hydrophobic lipid portion of PEG-lipid was spontaneously incorporated into the lipid bilayer membrane of islets. Scale bar: 50 μm. (B) The effect of PEG conjugation on the viabilities of ICCs and Sp-Ex-4 ICCs. The relative cell viabilities were evaluated by CCK-8 assay. Monitoring blood glucose levels of immunocompetent C57BL/6 mice transplanted with ICC and Sp-Ex-4 ICC grafts for 50 days. (A) 500 IEQ of Sp-Ex-4 untransduced ICC recipients (n = 6), (B) 500 IEQ of Sp-Ex-4 ICC recipients (n = 6), (C) 500 IEQ of PEG-Sp-Ex-4 ICC recipients (n = 6), (D) 500 IEQ of Sp-Ex-4-ICC recipients injected with low doses of anti-CD154 mAb and tacrolimus, and (E) 500 IEQ of PEG-Sp-Ex-4 ICC recipients injected with low doses of anti-CD154 mAb and tacrolimus. (F) Graft survival rate of ICCs, Sp-Ex-4 ICCs, and PEG-Sp-Ex-4 ICCs combined with or without low doses of anti-CD154 mAb and tacrolimus. (•) ICCs, (○) Sp-Ex-4 ICCs, (▾) PEG-Sp-Ex-4 ICCs, (Δ) Sp-Ex-4 ICCs with drugs, and (▄) PEG-Sp-Ex-4 ICCs with drugs (drug concentration: 0.2 mg of anti-CD154 mAb and 0.2 mg/kg of tacrolimus).

Immunohistochemistry
The results of immunohistochemical analyses for each case are shown in Fig. 9. Sp-Ex-4 ICCs exhibited increased lymphocytic infiltration in the kidney tissue at day 10 of transplantation. Although all groups of mice displayed at least some detectable tissue infiltration, there was no evidence of a significantly greater cumulative incidence of lymphocytic infiltrates, relative to ICC-receiving mice. Significantly great numbers of CD4+ and CD8a+ (T-cell marker) cells were detected in the kidney tissue in both Sp-Ex-4 ICCs and ICCs. Compared from unmodified cells, less extensive lymphocytic infiltrates of CD4+, CD8a+, and CD20+ were found in the surface-modified Sp-Ex-4 ICC transplanted kidney. Immune cells were not detected in the central area, although immune cells were gathered severely around the islet. Also immunostaining revealed that insulin+ cells were not detected in the whole area of the transplantation site in unmodified ICCs. We found no partially destroyed PEG-grafted ICCs and thus a decreased total islet cell mass in the transplantation site. On the other hand, insulin+ cells of PEG-Sp-Ex-4 ICCs were detected in the whole area of transplantation site. When anti-CD154 mAb and tacrolimus were administered for the PEG-Sp-Ex-4 ICC receiving group, there was no evidence of CD4+ and CD8a+ with a weak image of CD20+ staining.
Immunohistochemistry of islet grafts. (A) ICCs, (B) Sp-Ex-4 ICCs, and (C) PEG-Sp-Ex-4 ICCs were transplanted into C57BL/6 diabetic mice. Islet grafts were retrieved at day 10 of transplantation. (D) Sp-Ex-4 ICCs and (E) PEG-Sp-Ex-4 ICCs were transplanted into the C57BL/6 diabetic mice, which were treated with low doses of anti-CD154 mAb (0.2 mg) and tacrolimus (0.2 mg/ kg). The grafts were retrieved at day 50 of transplantation. Paraffin-fixed islet grafts were stained with H&E, anti-insulin, and anti-exendin-4 and immunostained with anti-CD4+, anti-CD8a+, and anti-CD20+. Lymphocytic infiltrates of ICCs and Sp-Ex-4 ICCs after transplantation (10 days after transplantation). Arrow, immune cells staining; asterisk, ICCs. Scale bar: 50 μm.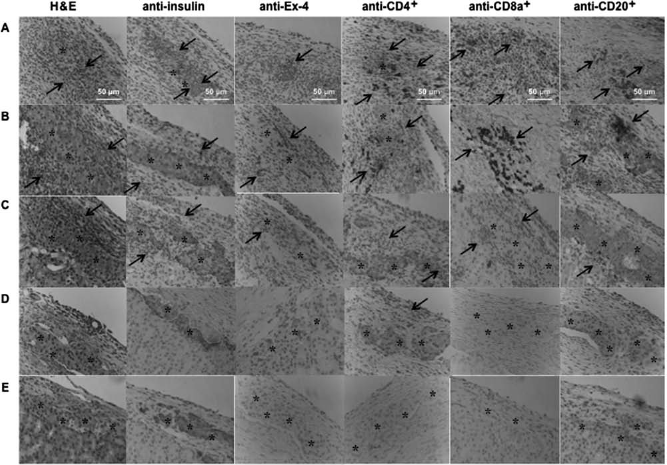
Discussion
We developed a clinically applicable PEG-Sp-Ex-4 ICC to increase the therapeutic potential of cell therapy. There are several advantages of PEG-Sp-Ex-4 ICCs. First, genetically transduced Ex-4 could improve the insulin secretion from β cells. Second, due to the small size of reconstructed ICCs, the hypoxia was prevented in the central area of the ICCs, thereby reducing hypoxia-induced cell death. Also, the secreted Ex-4 further prevented hypoxia-induced apoptosis of ICCs. In addition, due to the hydrodynamic volume and repulsion properties of the surface-anchored PEG, the immunogenicity of PEG-Sp-Ex-4 ICCs could be attenuated, thereby reducing the clinically required dose of immunosuppressive drugs.
The capacity of insulin secretion from the ICCs was important for successful ICC construction. In view of normal pattern of insulin secretion into islets, the ICCs showed abnormal insulin secretory responsiveness to different concentration of glucose, even though they had normal capacity of insulin secretion. The contact between adjacent pancreatic endocrine cells is likely to be one of the factors affecting the basal insulin secretion level. Halban et al. (11) studied the possible importance of contact between adjacent pancreatic islet cells for the control of insulin release. They demonstrated the elevated basal insulin release in isolated islet cells, whereas cultures with cell contacts displayed lower basal insulin release. The restoration of lower insulin release seems to be interrelated with the contact between reaggregated cells. However, whether such regulation is a consequence of cell association or a direct communication between cells remains to be established. In our study, we demonstrated that mere association is not sufficient to restore low basal insulin release. To support this assumption, Konstantinova et al. (14) demonstrated the importance of communication between β cells inhibiting basal insulin secretion and for enhancing glucose-stimulated insulin secretion. They found that ephrin A (EphA) forward signaling inhibits basal insulin secretion, and glucose stimulation attenuates EphA forward signaling by dephosphorylation of EphAs. This mechanism enables pancreatic islets to switch from low level of insulin secretion to high levels of insulin secretion upon glucose stimulation. However, the process of dissociation and reaggregation results in disrupting the communication between adjacent β cells, resulting in unsynchronized glucose-stimulated insulin secretion. This finding indicated the importance of signaling events between cells and communication between islet cells in regulating insulin secretion (2,14). Since ICCs had low control capacity of glucose sensitivity, we transduced these single cells with an insulinotropic factor (Ex-4) encoding vector to improve cell sensitivity to glucose. Ex-4 increases glucose sensitivity of islets via stimulating the PI3K pathway to regulate ion channels of β cells. However, the glucose sensitivity of Sp-Ex-4 ICCs was still lower than that of whole islets. Interestingly, when compared with ICCs, the functional activity of Sp-Ex-4 ICCs was remarkably increased, secreting about twofold higher amount of insulin at high glucose concentrations than that of nontransduced ICCs. This result was caused by the insulinotropic action of the expressed Ex-4 protein, whose effect was similar to GLP-1 protein. Therefore, the expression of Ex-4 could be a solution to overcome the glucose responsiveness of the reconstructed ICCs.
In general, the size of an isolated islet was about 200 μm, which means that the isolated islets were gradually dead due to the limitation of oxygen transport. Therefore, small size of islets would be superior to large islets with respect to the viability. Here we demonstrated that the viability of newly constructed ICCs was significantly higher than that of islets when cultured for 25 days. In addition, when they were cultured under a hypoxic condition, caspase-3 activity was significantly attenuated. These results were correlated with the fact that smaller size islets were highly resistant against a hypoxia-induced apoptosis because their small size permits adequate and rapid transport of oxygen into the core of islets. Furthermore, when Sp-Ex-4 lentivirus was transduced into ICCs, their viability was not affected during a long-term in vitro culture. Also, under a hypoxic condition, the caspase-3 activity in ICCs was not strongly attenuated, which could be attributed to the fact that Ex-4 protein was an antioxidant and antiapoptotic agent and could be contributed to better transplantation outcome.
When Sp-Ex-4 ICCs were xenotransplanted into immunocompetent diabetic mice, their median survival time was not strongly improved when compared from nontransduced ICC xenotransplantation. This result indicated that the overexpressed Ex-4 could not improve the viability of the transplanted ICCs from host's xenograft immune rejection, even though it has antioxidant and antiapoptotic activities as evidenced by the infiltration of extensive immune cells, such as CD4+, CD8a+, and CD20+ immune cells, into the transplanted ICCs. To prevent the direct attack of immune cells to the transplanted Sp-Ex-4 ICCs, we applied the surface modification using biocompatible PEG onto surface of Sp-Ex-4 ICC. In the previous studies, we chemically modified the surface of intact islets via amide bond between the carboxylic group of PEG and an amine group of the collagen layer of islets. However, this chemical modification technology could not be applied as surface modification of Sp-Ex-4 ICCs because the surface of the reconstructed ICCs did not have a collagen layer due to single cell dissociation of islets using collagenase. Therefore, we synthesized PEG-lipid (C18) for the specified surface modification of ICCs and physically anchored the lipid chain into the cellular membrane of Sp-Ex-4 ICCs. As shown in Fig. 7B, when FITC-labeled PEG-lipid was grafted onto the surface of ICCs or Sp-Ex-4 ICCs, the PEG-lipid could be grafted onto the whole surface of Sp-Ex-4 ICCs without affecting their viability. Interestingly, when the PEG-lipid grafted Sp-Ex-4 ICCs (PEG-Sp-Ex-4 ICCs) were transplanted into a xenograft model, immune cells could not infiltrate into the transplanted PEG-Sp-Ex-4 ICCs, meaning that the PEG molecules could attenuate the immunogenicity. However, due to the indirectly occurring immune reactions such as cytokine attacks, PEG-Sp-Ex-4 ICCs were rejected within 2 weeks even though PEG blocked the infiltration of immune cells. Interesting results were obtained when low doses of anti-CD154 mAb and tacrolimus were coadministered to the recipients after xenotransplantation of PEG-Sp-Ex-4 ICCs in order to further block indirect immune reaction initiated from helper T cells. As a result, PEG-Sp-Ex-4 ICCs with low doses of anti-CD154 mAb and tacrolimus completely maintained normoglycemia without any hyperglycemia for 50 days before nephrectomy. Therefore, both low doses of anti-CD154 mAb and tacrolimus and surface-grafted PEG could synergistically improve the viability of PEG-Sp-Ex-4 ICCs in a xenograft model. If we apply this system in allogeneic model, we could observe this comparable result without using several immunosuppressive drugs. In addition, the anti-inflammatory effect of exendin-4 could be more significantly highlighted, contributing to lower the dose of immunosuppressive drugs, which are currently in use. However, the transduction level must be efficient enough to have a meaningful effect on cell viability and functionality. Sp-Ex-4 transduction in islet single cells is proven to be more efficient than in standard nondissociated pancreatic islets, therefore superior in term of the transplantation outcome and in maintaining the cell viability. The use of this strategy can be more effectively applied in human-to-human islet transplantation, contributing to higher success rate in treating diabetes mellitus.
If this platform technology of PEG-Sp-Ex-4 ICCs would be added to the “Edmonton protocol”(24), however, we could expect that the type, dose, and frequency of medications could be effectively reduced without any nephropathy (27).
In conclusion, findings in this study suggested that the construction of PEG-Sp-Ex-4 ICCs could improve the viability and functionality of ICCs and reduce the immunogenicity of ICCs themselves, hypoxia-induced apoptosis, and adverse effects of clinical immunosuppressants. Even though transduced Sp-Ex-4 gene improved β-cell function, further studies are needed to enhance islet single cell communication in order for PEG-Sp-Ex-4 ICCs to be a good candidate for replacing islets as a new transplantation source. As the study by Konstantinova et al. (14) revealed, restoring the islet β-cell signaling events would be important for improving glucose-stimulated insulin secretion. As further work, the cell-to-cell communication in ICCs should be exploited for developing an ideally engineered ICC.
Footnotes
Acknowledgments
This study was supported by a grant from the World Class University (WCU) program (grant no. R31-2008-000-10103), the Converging Research Center Program (grant no. 2009-0081879), and the Basic Science Research Program (grant no. 2010-0027955) through the National Research Foundation of Korea (NRF) funded by the Ministry of Education, Science and Technology, Republic of Korea. The authors declare no conflicts of interest.
