Abstract
Growing evidence has demonstrated that neurogenesis in the subventricular zone (SVZ) is significantly decreased in Parkinson's disease (PD). Modulation of endogenous neurogenesis would have a significant impact on future therapeutic strategies for neurodegenerative diseases. In the present study, we investigated the augmentative effects of human mesenchymal stem cells (hMSCs) on neurogenesis in a PD model. Neurogenesis was assessed in vitro with 1-methyl-4-phenylpyridinium (MPP+) treatment using neural precursor cells (NPCs) isolated from the SVZ and in vivo with a BrdU-injected animal model of PD using 1-methyl-4-phenyl-1,2,3,6-tetrahydropyridine (MPTP). Immunochemical analyses were used to measure neurogenic activity. The number of BrdU-ir cells in the SVZ and the substantia nigra (SN) was significantly increased in the hMSC-treated PD group compared with the MPTP-only-treated group. Double-stained cells for BrdU and tyrosine hydroxylase were notably observed in the SN of hMSC-treated PD animals, and they did not colocalize with the nuclear matrix; however, double-stained cells were not detected in the SN of the MPTP-induced PD animal model. Furthermore, hMSC administration increased the expression of the epidermal growth factor receptor (EGFR) in the SVZ of PD animals, and the coculture of hMSCs significantly increased the release of EGF in the medium of MPP+-treated NPCs. The present study demonstrated that hMSC administration significantly augmented neurogenesis in both the SVZ and SN of PD animal models, which led to increased differentiation of NPCs into dopaminergic neurons in the SN. Additionally, hMSC-induced modulation of EGF seems to be an underlying contributor to the enhancement of neurogenesis by hMSCs. The modulation of endogenous adult neurogenesis to repair the damaged PD brain using hMSCs would have a significant impact on future strategies for PD treatment.
Introduction
Recent studies have demonstrated that the adult mammalian brain has the potential to generate new neurons and to incorporate them into brain areas affected by a disease process (12,22). Neurogenesis in the subventricular zone (SVZ) and the subgranular zone of the dentate gyrus may act as an endogenous repair mechanism (10); thus, modulation of endogenous neurogenesis would have a significant impact on future therapeutic strategies for neurodegenerative diseases such as Alzheimer's disease and Parkinson's disease (PD) (13). Several factors, including neurotransmitters, growth factors, and disease states, have been suggested to play a role in the regulation of endogenous neurogenesis (1). Growing evidence has demonstrated that neurogenesis in the SVZ and hippocampus is significantly decreased in patients with PD and in animal models of PD (3,14) and that neurochemical deficits of dopamine, indirect effects of growth factor release, and α-synuclein accumulation in neural precursor cells (NPCs) of the SVZ by disease process may influence neurogenic activity (5,8,33). In particular, the NPCs in the SVZ express dopamine receptors, and several animal data suggest that dopamine-enhancing drugs increase neurogenic activity through the epidermal growth factor receptor (EGFR) in conjunction with the release of EGF in the SVZ (14,24).
Mesenchymal stem cells (MSCs) are present in adult bone marrow and represent <0.01% of all nucleated bone marrow cells. MSCs are themselves capable of multipotency, with differentiation under appropriate conditions into chondrocytes, skeletal myocytes, and neurons. Moreover, MSCs secrete various cytotrophic factors that, in turn, exert neuroprotective effects (6). We previously demonstrated that human MSCs (hMSCs) had neuroprotective properties in Parkinsonian disorders through a variety of mechanisms, such as antiapoptotic effects, modulation of polyubiquitinated proteins, and anti-inflammatory actions, in addition to possible transdifferentiation of hMSCs into dopaminergic neurons (16,26,27). Furthermore, this cytotrophic effect of MSCs on neurogenic modulation has been studied in animal models of stroke, demonstrating that MSCs promoted neurogenic activity in the SVZ and migration of NPCs into damaged lesions (21). Recently, Cova and colleagues (7) showed that intrastriatal implantation of hMSCs enhanced neurogenesis in the SVZ and induced the migration of neuroblasts toward the lesioned striatum in a 6-hydroxydopamine-induced PD animal model, possibly mediated through sustained expression of brain-derived neurotrophic factor (BDNF). In the present study, we investigated whether intravenous injection of hMSCs would modulate neurogenesis in the SVZ and thus enhance differentiation of NPCs into the dopaminergic phenotype in the substantia nigra (SN) using a 1-methyl-4-phenyl-1,2,3,6-tetrahydropyridine (MPTP)-induced animal model of PD.
Materials and Methods
Animal Study
To create an MPTP-induced animal model of PD, C57BL/6 adult male mice aged 16 weeks (Samtako Bio Korea, Seoul, South Korea) were injected with MPTP (20 mg/kg IP, four times a day at 2-h intervals, each group, n = 5). Control mice (n = 5) were injected with saline alone using the same administration method. At 3 days after MPTP injection, hMSCs were injected into the tail vain (1 × 106 cells/200 ml). At 4 days after MPTP injection, bromodeoxyuridine (BrdU; 50 mg/kg IP) was injected consecutively for 5 days. Histopathological analyses were compared among three groups of mice (control group, MPTP-only group, and hMSC treatment in MPTP group). The in vivo study design is illustrated in Figure 1. The animal work was approved by the institutional animal care and use committees of Yonsei University.
In vivo study design. At 3 days after 1-methyl-4-phenyl-1,2,3,6-tetrahydropyridine (MPTP) injection, human mesenchymal stem cells (hMSCs) were injected into the tail vain (1 × 106 cells/mouse). At 4 days after MPTP injection, bromodeoxyuridine (BrdU; 50 mg/kg IP) was injected consecutively for 5 days. The mice were perfused at 8 (early time) and 30 days (late time) after MPTP injection.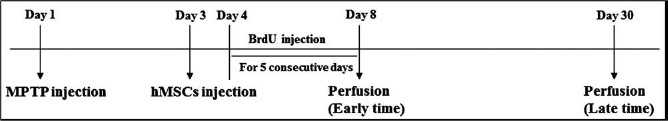
Isolation of hMSCs
Bone marrow aspirates (10 ml) were obtained from the iliac crests of human donors (n = 3, aged 25–40). The mononuclear cell layer was isolated by Ficoll–Hypaque, washed in PBS, plated in 100-mm polystyrene plastic culture dishes, and cultivated in low-glucose Dulbecco's modified Eagle's medium (Gibco-BRL, Grand Island, NY) containing 10% fetal bovine serum (Hyclone, Irvine, CA) and 1% penicillin/ streptomycin (P/S, Sigma, St. Louis, MO) in a humidified incubator at 37°C under 5% CO2. Nonadherent cells were removed after 24 h. When these primary cultures reached 80% confluence, the cells were harvested using 0.25% trypsin and subcultured. At passage 6, hMSCs were injected into the mice via the tail vain (1 × 106 cells/mouse).
Tissue Preparation
For immunohistochemistry, the mice were perfused with saline solution containing 0.5% sodium nitrate and heparin (10 U/ml) and fixed with 4% paraformaldehyde dissolved in 0.1 M phosphate buffer (PB, ~50 ml/mouse) at 8 (early time) and 30 days (late time) after MPTP injection. The brains were removed from the skulls, postfixed overnight in buffered 4% paraformaldehyde at 4°C, and stored in a 30% sucrose solution for 1–2 days at 4°C until they sank. They were then sectioned to obtain 30-μm coronal sections. The 30-μm coronal sections were stored in tissue stock solution (30% glycerol, 30% ethylene glycol, 30% thrice-distilled water, 10% 0.2 M PB) at 4°C until required.
Antibodies
Antibodies and dilutions used in this study were mouse anti-tyrosine hydroxylase (TH) (1:2,000 dilution for brain tissue; 1:7,500 dilution for cell culture; Pel-freez, AR), rabbit anti-TH (1:2,000 dilution for brain tissue; 1:7,500 dilution for cell culture; Chemicon, Temecula, CA), mouse anti-nuclear matrix (NuMA, a nuclear marker) (1:100 dilution; Calbiochem, San Diego, CA), rabbit anti-Ki67 (1:500 dilution; Abcam, Cambridge, UK), mouse anti-BrdU (1:200 dilution; Roche, San Francisco, CA), rabbit anti-EGFR (1:100 dilution; Imgenex, San Diego, CA), and CD105, CD90, CD44, CD34, and CD45 (1:100 dilution; Chemicon, Temecula, CA) for immunochemistry and immunofluorescence. The different primary antibodies were codetected by immunofluorescence using goat anti-mouse IgG Alexa Fluor-488 (green) and goat anti-rabbit IgG Alexa Fluor-594 (red) (1:200; Molecular Probes, Eugene, OR).
Immunohistochemistry and Immunocytochemistry
The 30-μm coronal brain sections and cultured NPCs were washed twice in PBS and incubated in 0.2% Triton X-100 for 30 min at room temperature (RT). Then they were rinsed three times with 0.5% bovine serum albumin (BSA) in 1 x PBS for blocking. After blocking, they were incubated overnight at 4°C with primary antibody. The brain sections were then rinsed three times in 0.5% BSA in 1 x PBS (10 min/rinse) and incubated with the appropriate biotinylated secondary antibody and avidin–biotin complex (Elite Kit; Vector Laboratories, Burlingame, CA) for 1 h at RT. Bound antibodies were visualized by incubating with 0.05% diaminobenzidine–HCl (DAB) and 0.003% H2O2 in 0.1 M PB. The brain sections and cultured NPCs were rinsed with 0.1 M PB for DAB inhibition. For BrdU immunohistochemistry, the section tissues before blocking were incubated with 0.6% H2O2 in 1 x PBS for 10 min and rinsed three times in 1 x PBS. After rinsing, they were incubated with 50% formamide in 2 x SSC DW (standard saline citrate distilled water) for 1–2 h at 65°C and rinsed three times in 2 x SSC DW. After rinsing, the brain sections were incubated with 2 N HCl in DW for 30 min at 37°C and rinsed three times in 1 x PBS. They were then incubated in 0.2% Triton X-100 for 30 min at RT, and routine immunohistochemical procedure was then followed. Immunostained cells were analyzed by brightfield microscopy.
Immunofluorescence Double Labeling
The 4-μm sections of embedded paraffin tissues were rinsed three times with 0.5% BSA in 1 x PBS for blocking. After blocking, they were incubated overnight at RT or 4°C with the primary antibodies TH+NuMA (to detect dopaminergic neurons differentiated from hMSCs) and TH+Ki67 (to detect dopaminergic neurons differentiated from proliferating cells, i.e., NPCs). The brain sections were then rinsed three times in 0.5% BSA in 1 x PBS (10 min each rinse) and incubated with secondary antibody: goat anti-mouse IgG (Alexa Fluor-488, green) and goat anti-rabbit IgG (Alexa Fluor-594, red) for 1 h at RT. Brain tissues were then washed and mounted using Prolong Antifade Kit (Molecular Probes). Stained tissues were viewed using an Olympus I X 71 confocal laser scanning microscope (Olympus, Tokyo, Japan). To analyze the localization of different antigens in double-stained samples, immunofluorescence images were created from the same tissue section and merged using interactive software.
Cell Cultures of NPCs and SH-SY5Y
A parasagittal section was taken from the medial surface of each hemisphere of postnatal day 1 Sprague–Dawley rat (male and female, n = 28; Samtako Bio Korea, Seoul, South Korea), and a wedge of tissue was microdissected from the portion of the lateral ventricle that included the anterior part of the SVZ (23). The tissues were incubated in Ca2+-, Mg2+-free Hank's balanced salt solution (CMF-HBSS) for 10 min and 0.01% trypsin in CMF-HBSS for 9 min at 37°C. Cultures were rinsed twice in Dulbecco's modified Eagle's medium supplemented with 10% fetal bovine serum, 6 mg/ml glucose, 204 μg/ml L-glutamine, and 100 U/ml P/S to inhibit trypsin, and they were then dissociated into single cells by trituration. The mononuclear cell layer was plated in 100-mm polystyrene plastic culture dishes and cultivated in Dulbecco's modified Eagle's medium (Gibco-BRL) containing 10% fetal bovine serum (Hyclone) and 1% penicillin/streptomycin (P/S, Sigma) in a humidified incubator at 37°C under 5% CO2. Nonadherent cells were removed after 24 h. When these primary cultures reached 20% confluence, the cells were harvested using 0.25% trypsin and subcultured. SH-SY5Y cells (neuroblastoma cell lines, ATCC, USA) were maintained in Dulbecco's modified Eagle's medium supplemented with 10% fetal bovine serum (FBS). Cells were cultured in 25 cm2 flasks and passaged by using trypsin–EDTA solution. Cells were subcultured into individual a Transwell 1 day before the experiment.
For treatment, cultured NPCs at passage 3 were plated on 12-mm round Aclar plastic coverslips or 24-well culture plates at a density of 1.0 × 105 cells/coverslip/well. Cells were incubated in a humidified incubator at 37°C and 5% CO2 for 24 h. At 2 days, the cultures were pretreated with MPP+ (100 μM) for 24 h. After MPP+ was washed off, primary NPC cultures were then cocultured with 3 × 104 hMSCs or 1 × 105 SH-SY5Y cells/well for 24 h using a Transwell. The cocultured cells were used for immunochemistry, cell proliferation assay, and ELISA assay.
EGF Elisa and MTS Assay
The MPP+-stimulated NPCs were cocultured with vehicle or hMSCs using a Transwell, and the level of EGF was determined in the supernatant of MPP+-treated NPCs. The supernatant was stored in a fresh tube, and a sandwich ELISA was performed according to the manufacturer's instructions (Alpco, Salem, NH). The MPP+-stimulated NPCs were cocultured with vehicle or hMSCs using a Transwell, and an MTS cell proliferation assay was performed. Each well of cocultured cells was incubated in DMEM with 20% AQueous One solution regent at 37°C for 1 h, and the absorbance was recorded at 490 nm using a 96-well plate reader. The MTS assay was performed according to the manufacturer's instructions (Promega, Madison, WI).
Stereological Cell Counts
Unbiased stereological estimations of the total number of stained cells in the SN and SVZ were made using an optical fractionator as previously described, with some modifications (17). This sampling technique is not affected by tissue volume changes and does not require reference volume determinations (31). The sections used for counting covered the entire SN from the rostral tip of the pars compacta back to the caudal end of the pars reticulata and the lateral wall of the SVZ. This generally yielded eight to nine sections in a series. Sampling was performed using the Olympus CAST-Grid system (Olympus Denmark A/S, Albertslund, Denmark), using an Olympus BX51 microscope connected to the stage and feeding the computer with the distance information in the z-axis. The SN and SVZ were delineated at 1.25x objective. A counting frame (60%, 35,650 μm2) was randomly placed onto the first counting area and systematically moved through all counting areas until the entire delineated area was sampled. Actual counting was performed using a 40x oil objective. Guard volumes (4 μm from the top and 4–6 μm from the bottom of the section) were excluded from both surfaces to avoid the problem of a lost cap, and only the profiles that came into focus within the counting volume (with a depth of 10 μm) were counted. The total number of stained cells was calculated according to the optical fractionator formula (30).
Statistical Analysis
Data are expressed as mean±SE. Comparisons between groups were made using the Student's t test (paired) or oneway analysis of variance (ANOVA) followed by a Dunnett post hoc test. Values of p < 0.05 were considered statistically significant. Statistical analyses were performed using commercially available software (SPSS version 10.0; SPSS, Inc., Chicago, IL).
Results
Characterization of hMSCs
Fluorescence-activated cell sorting (FACS) analysis demonstrated that hMSCs expressed CD29, CD44, CD73, and CD105, positive markers for hMSCs, and did not express CD14, CD34, CD45, and CD106, negative markers for hMSCs (Fig. 2A). Immunofluorescent labeling showed that hMSCs were positive for CD44, CD90, and CD105 and negative for CD34 and CD45 (Fig. 2B).
Flow cytometric analysis (A) and immunofluorescent labeling of hMSCs (B). Scale bar: 100 μm.
Effect of hMSCs on Loss of Dopaminergic Neurons in MPTP-Induced Animal Models
To determine the effects of hMSCs on the loss of dopaminergic neurons, brain tissue in the SN was immunostained with TH, a marker for dopaminergic neurons. As expected, there was a marked increase in the loss of TH-ir in MPTP-induced PD animal models; however, hMSC treatment markedly attenuated the loss of TH-ir cells in MPTP-induced PD animal models (Fig. 3A). Stereological analysis revealed that the number of TH-ir cells in the SN was significantly higher in the hMSC-treated group than in the MPTP-only-treated group (5,680±105 vs. 2,990±36, p < 0.05) (Fig. 3B and Table 1).
Effect of hMSCs on survival of dopaminergic neurons in MPTP-induced Parkinson's disease (PD) animal models. hMSC treatment markedly attenuated the loss of tyrosine hydroxylase (TH)-ir cells in MPTP-induced PD animals (A), and stereological analysis revealed that the number of TH-ir cells in the substantia nigra (SN) was significantly higher in the hMSC-treated group than in the MPTP-only-treated group (B) (n = 5; *p < 0.05). Scale bar: 100 μm. The Number of Cells in the Control and MSC-Treated Groups MSCs, mesenchymal stem cells; MPTP, 1-methyl-4-phenyl-1,2,3,6-tetrahydropyridine; TH, tyrosine hydroxylase; BrdU, bromodeoxyuridine; EGFR, epidermal growth factor receptor; SN, substantia nigra; SVZ, subventricular zone. Comparison between control and MPTP. Comparison between MPTP and MPTP + hMSCs. Comparison between control and MPTP + hMSCs.

Effect of hMSCs on Neurogenesis in the SVZ of MPTP-Induced PD Animal Models
To determine the effects of hMSCs on regulation of NPC proliferation, brain tissue in the SVZ was immunostained with BrdU, a marker for cell proliferation. At 8 and 30 days after MPTP injection, the number of BrdU-ir cells in the SVZ was prominently decreased in MPTP-induced PD animals; however, hMSC treatment in MPTP-induced PD animals significantly attenuated the neuronal loss of BrdU-ir cells in the SVZ at both early and late times (Fig. 4A). Stereological analysis revealed that the number of BrdU-ir cells in the SVZ was significantly increased in the hMSCs-treated group compared with the MPTP-only-treated group at late time (14,640 ± 703 vs. 8,560 ± 341, p < 0.01), whereas the difference in BrdU-ir cells between these groups was not significant at early time (Fig. 4B and Table 1). The number of NPCs in the SVZ did not differ significantly between the hMSCs-treated PD and control groups in both the early and late periods after BrdU injection.
Effect of hMSCs on neurogenesis in the subventricular zone (SVZ) of MPTP-induced PD animal models. hMSC treatment prominently increased the number of BrdU-ir cells in the SVZ of MPTP-treated PD animal models (A). Stereological analysis revealed that the number of BrdU-ir cells in the SVZ was significantly greater in the hMSCs-treated group compared with the MPTP-only-treated group at 30 days after MPTP injection (B) (n = 5; *p < 0.05, **p < 0.01). Scale bar: 100 μm.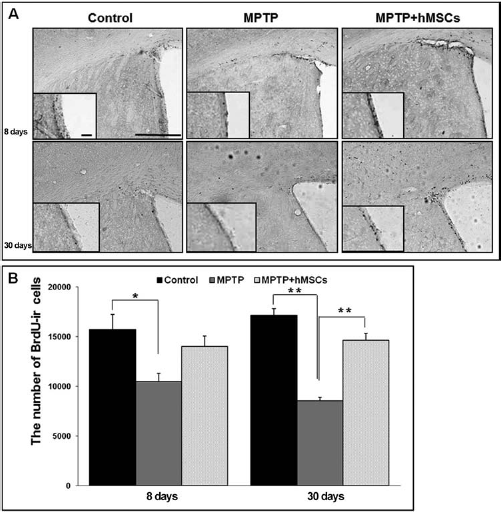
Effect of hMSCs on Neurogenesis in the SN of MPTP-Induced PD Animal Models
BrdU-ir cells in the SN were more frequently observed in the hMSCs-treated group than in the MPTP-only-treated or control groups in both the early and late periods after BrdU injection (Fig. 5A). Stereological analysis revealed that the number of BrdU-ir cells in the SN was significantly greater in the hMSCs-treated group compared with the MPTP-only-treated group in the late period of BrdU injection (1,660 ± 177 vs. 580 ± 163, p < 0.05) (Fig. 5B and Table 1). BrdU-ir cells in the early period of BrdU injection tended to be higher in the hMSCs-treated group than in the MPTP-only-treated group; however, this difference did not reach statistical difference (Table 1).
Effect of hMSCs on neurogenesis in the SN of MPTP-induced PD animal models. BrdU-ir cells in the SN (arrows) were more frequently observed in the hMSCs-treated group than in the MPTP-only-treated or control groups in both the early and late periods after BrdU injection (A). Stereological analysis revealed that the number of BrdU-ir cells in the SN was significantly greater in the hMSCs-treated group than in the MPTP-only-treated group in the late period of BrdU injection (B) (n = 5; *p < 0.05). Scale bar: 100 μm.
hMSCs Enhanced Differentiation of NPCs Into Dopaminergic Phenotype in the SN
To evaluate whether hMSCs would augment dopaminergic differentiation from NPCs, brain tissue in the SN areas was double immunostained with Ki67, a marker for cell proliferation, and TH, a marker for dopaminergic neurons, at 22 days after last BrdU injection. Ki67 and TH-ir cells were not detected in the SN of the MPTP-induced PD animal model; however, double-stained cells were notably observed in the SN of hMSCs-treated PD animals (Fig. 6A). On stereological analysis, the number of cells double stained with Ki67 and TH in the SN was 189 ± 52, which roughly corresponded to 10% of the total number of Ki67-ir cells at 22 days after last BrdU injection (Fig. 6B). Furthermore, these double-stained cells did not colocalize with NuMA (Fig. 6C), a marker for human-specific cells, which indicates that these cells did not originate from the hMSCs injected via the tail vein.
Enhanced differentiation of NPCs into the dopaminergic phenotype in the SN by hMSCs. Brain tissue in the SN areas was double immunostained with Ki67 and TH at 22 days after last BrdU injection. Ki67 and TH-ir cells were not observed in the SN of MPTP-induced PD animal model; however, double-stained cells were notably observed in the SN of hMSCs-treated PD animals (A). On stereological analysis, the number of double-stained cells in the SN was 189 ± 52, which roughly corresponded to 10% of the total number of Ki67-ir cells (B). These double-stained cells did not colocalize with antinuclear matrix (NuMA), a marker for human-specific cells (C). Arrows indicate double-stained cells with TH and Ki67.
Modulatory Effect of hMSCs on Expression of EGFR in the SVZ
EGF is known to enhance proliferation of NPCs in the SVZ by binding to the EGFR that is present on activated B cells and rapidly dividing C cells (11). To evaluate the effects of hMSCs on the expression of EGFR, the area of the SVZ was immunostained with EGFR. The expression of EGFR was prominently decreased in the SVZ of MPTP-treated PD animals compared with controls; however, hMSC treatment notably increased the expression of EGFR in the SVZ of MPTP-treated PD animals (Fig. 7A). Stereological analysis showed that the number of EGFR-ir cells in the SVZ was significantly lower in MTPT-treated PD animals compared with controls (the early period: 4,240 ± 524 vs. 2,360 ± 222, p < 0.05; the late period: 3,200 ± 105 vs. 1,200 ± 183, p < 0.01), whereas hMSC treatment led to a significant increase in EGFR-ir cells of MPTP-treated PD animals the late period of BrdU injection (2,400 ± 138 vs. 1,200 ± 183, p < 0.01) (Fig. 7B and Table 1).
Modulatory effect of hMSCs on expression of epidermal growth factor receptor (EGFR) in the SVZ. The expression of EGFR was prominently decreased in the SVZ of MPTP-treated PD animals compared with that of the controls; however, hMSC treatment notably increased the expression of EGFR in the SVZ of MPTP-treated PD animals (A). Stereological analysis showed that hMSC treatment led to a significant increase in EGFR-ir cells of MPTP-treated PD animals at 22 days after last BrdU injection, that is, day 30 (B) (n = 5; *p < 0.05, **p < 0.01). Scale bar: 100 μm.
Direct Effect of hMSCs on Survival of NPCs In Vitro
To investigate the effect of hMSCs on survival of NPCs obtained from the SVZ, the NPCs were cocultured with SH-SY5Y cells or hMSCs using a Transwell after MPP+ treatment and analyzed immunocytochemically in a 24-h incubation period. The MPP+ administration or coculture with SH-SY5Y cells induced marked loss of NPCs compared with the controls, whereas coculture with hMSCs showed a significant reduction in the loss of NPCs compared with MPP+ alone (Fig. 8A). Stereological and MTS analyses also showed that the survival of NPCs after MPP+ administration was significantly higher in the hMSCs treatment group than in the MPP+-only or SH-SY5Y cells treatment group (Fig. 8B, C). To evaluate the effect of hMSCs on the modulation of EGF, we determined the level of EGF in the medium of MPP+-treated NPCs. The level of EGF was significantly higher in the medium cotreated with hMSCs compared with the MPP+-only or SH-SY5Y cells-treated medium (Fig. 8D).
Direct effect of hMSCs on survival of neural progenitor cells (NPCs) in vitro. To investigate the effect of hMSCs on the survival of NPCs obtained from the SVZ, the NPCs were cocultured with hMSCs or SH-SY5Y cells using a Transwell after MPP+ treatment. The MPP+ administration or coculture with SH-SY5Y cells induced marked loss of NPCs compared with the controls, whereas coculture with hMSCs resulted in a significant reduction in the loss of NPCs (A). Stereological and MTS analyses showed that the survival of NPCs after MPP+ administration was significantly higher in the hMSC treatment group than in the MPP+-only or SH-SY5Y cells treatment group (B and C). The level of EGF in the medium of NPCs cotreated with hMSCs was significantly higher compared with the change of MPP+-only treatment or coculture with SH-SY5Y cells (D). (n = 3; *p < 0.05, **p < 0.01). Scale bar: 100 μm.
Discussion
The present study demonstrated that hMSC administration significantly augmented neurogenesis in both the SVZ and SN of PD animal models, which led to increased differentiation of NPCs into dopaminergic neurons in the SN. Furthermore, hMSCs-induced EGF modulation appears to be one of underlying contributors to the enhancement of neurogenesis by hMSCs. Our data suggest that the modulation of endogenous adult neurogenesis to repair the damaged PD brain using hMSCs would have a significant impact on future strategies for PD treatment.
Growing evidence has suggested that the neurogenic activity in the SVZ and dentate gyrus is significantly decreased in PD (3,14) and that dopaminergic modulation of the NPCs through a nigrostriatal pathway is an important contributor to the neurogenesis of PD (5). Additionally, the PD-related microenvironment of α-syncline is known to decrease neurogenesis via disturbed notch-1 expression as well as increased induction of apoptosis in NPCs of the SVZ (8). Our study demonstrated that hMSC treatment had augmentative effects on neurogenesis against MPTP-induced NPC loss in the SVZ, showing 13% and 35% increases in the survival of BrdU-positive cells at early and late periods of BrdU injection, respectively. This result is well in accordance with previous studies that demonstrated the neurogenic effects of MSCs in animal models of PD and ischemic stroke.
Several lines of evidence have suggested that endogenous dopaminergic neurogenesis occurs at a low level in the SN of healthy mice and increases after introduction of neurotoxin, although the results were not consistent due to methodological differences among the studies (immunohistochemical staining, BrdU dose, or section thickness) (2,13). The present study demonstrated for the first time that hMSC administration significantly increased the number of BrdU-ir cells in the SN of MPTP-treated PD animal models. In particular, dopaminergic phenotypes incorporated with the Ki67 label were notably discovered in the SN of hMSCs-treated PD animals. Because MSCs can transdifferentiate into cells exhibiting dopaminergic phenotypes, we further determined the origin of these double-merged cells using NuMA staining, a marker for human-specific cells. These cells did not colocalize with NuMA, indicating that they did not originate from hMSCs injected via the tail vein; instead, they probably originated from NPCs. However, our data did not provide information on whether hMSC-induced cell proliferation in the SN may occur in situ from resident NPCs or progeny migrating from the SVZ. Nevertheless, our study indicates that along with the neuroprotective properties of hMSCs on dopaminergic neurons, enhancement of dopaminergic differentiation from the NPCs by hMSCs plays a role in the sparing effects of dopaminergic neurons in the SN of PD animals.
The neurogenic activity of hMSCs in our PD models appeared to be mediated by complex mechanisms. First, our study demonstrated that hMSCs modulate the induction of EGF in NPCs of the SVZ. In this study, hMSC administration notably increased the expression of EGFR in the SVZ of MPTP-treated PD animals, and coculture of hMSCs significantly increased the release of EGF in the medium of MPP+-treated NPCs. Like other neurotrophic factors, EGF is known to be an endogenous regulator of SVZ neurogenesis, thus leading to a significant enhancement in proliferation and migration of NPCs in the SVZ (18,28), although the fate of the newly generated cells has been controversial. Additionally, hMSCs possess the ability for the induction of various neurotrophic factors, such as BDNF, nerve growth factor (NGF), insulin-like growth factor (IGF), and EGF (15). In an animal model of ischemic stroke, Wakabayashi et al. (30) demonstrated that hMSCs significantly induced the endogenous expression of EGF in the host cells. In this regard, hMSCs would enhance neurogenic activity in the SVZ of PD models via modulation of endogenous EGF expression. Second, hMSC administration in the present study attenuated dopaminergic neuronal death in the SN of MPTP-treated PD animals. Recent studies have demonstrated that NPCs in the SVZ exhibited dopaminergic receptors and dopaminergic innervation that originated in the SN (14), suggesting the important role of dopaminergic modulation in the neurogenic activity of the SVZ. Adding to the evidence that dopamine agonists augment neurogenesis in animal models of PD (14,32), O'Sullivan et al. (25) very recently demonstrated a positive impact of chronic levodopa use on the number of neural stem cells in the SVZ of patients with PD. Furthermore, a recent study showed that dopamine stimulated the release of EGF from cells in the SVZ and in turn acted on the EGFR to promote cell proliferation (24), suggesting a role of EGF as a key mediator of dopamine-induced neurogenesis in the SVZ. Therefore, increased survival of dopaminergic neurons in the SN by hMSCs would enhance neurogenesis in the SVZ through activation of dopaminergic receptors or EGFR in the NPCs. Finally, our in vitro data showed that hMSC treatment increased survival of NPC MPP+-induced neurotoxicity. We previously demonstrated that hMSCs can restore the balance between neuronal survival and apoptosis through upregulation of cell survival signaling pathways and suppression of apoptotic cell death signaling (26,27). Together, our data suggest that the neuroprotective properties of hMSCs on NPCs of the SVZ also seem to be involved in the modulation of neurogenesis.
Regarding the functional impact of adult neurogenesis, several lines of evidence have suggested that adult neurogenesis plays a regulatory role in olfaction, mood, and memory functions. In particular, Van Kampen and Eckman (29) reported that, in a PD animal model, an increase in neurogenesis following treatment with a dopaminergic drug restored the nigrostriatal dopaminergic projection concomitant with functional motor recovery, even though a debate exists as to whether NPCs from the SVZ would transdifferentiate into nigral dopaminergic phenotypes or migrate into the striatum and then incorporate into host neurons (9). In addition to the molecular and cellular benefits of hMSCs, cell therapy with hMSCs has advantages in clinical applications, such as ease of harvest from bone marrow, variety of administration roots, absence of immunological rejection, and absence of ethical problems. Importantly, our group recently reported that cell therapy with hMSCs in patients with multiple-system atrophy and ischemic stroke was feasible and safe (4,19,20). From a therapeutic perspective, the augmentative effects of hMSCs on endogenous adult neurogenesis along with the neuroprotective properties in Parkinsonian disorders would be applicable clinically in future strategies for PD treatment.
In summary, the present study demonstrated that hMSC administration significantly augmented neurogenesis in the SVZ of a PD animal model and increased differentiation of NPCs into dopaminergic neurons in the SN. Our results suggest that the modulation of endogenous adult neurogenesis to repair the damaged PD brain using hMSCs would have a significant impact on future strategies for PD treatment.
Footnotes
Acknowledgments
This study was supported by a grant of the Korea Healthcare Technology R&D Project, Ministry for Health, Welfare and Family Affairs, Republic of Korea (A084227). The authors declare no conflict of interest.
