Abstract
Ischemia-reperfusion and immunosuppressive therapy are a major cause of progressive renal failure after kidney transplantation. Recent studies have shown that administration of bone marrow mesenchymal stem cells (MSCs) improves kidney functional recovery in the acute phase of post ischemia-reperfusion injury. In the present study, we used an original model of renal ischemia-reperfusion in immunosuppressed rats (NIRC) to investigate the effects of bone marrow MSCs on progression of chronic renal failure and the mechanisms potentially involved. Left renal ischemia-reperfusion (IR) was induced in unilateral nephrectomized Lewis rats. After IR, rats were treated daily with cyclosporine (10 mg/kg SC) for 28 days. MSCs were injected into the kidney at day 7 after IR. At day 28 after IR, kidneys were removed for histomorphological, biochemical, and gene expression analysis. The effect of conditioned media from MSCs on epithelial–mesenchymal transition was studied in vitro on HK2 cells. Our results show that, as compared to untreated NIRC rats, rats treated by intrarenal injection of MSCs 7 days after IR displayed improvement in renal function, reduction of interstitial fibrosis, and decrease in chronic tubule injury. These effects were associated with a decrease in interstitial α-SMA accumulation and MMP2 activity, markers of fibroblast/fibroblast-like cell activation, and renal remodeling, respectively. Finally, experiments in vitro showed that MSC-conditioned medium prevented epithelial–mesenchymal transition induced by TGF-β in HK2 cells. In conclusion, our results show that, in immunosuppressed animals, a single intrarenal administration of MSCs reduced renal fibrosis and promoted the recovery of renal function.
Introduction
Renal ischemia-reperfusion occurs in different clinical situations including kidney transplantation, cardiopulmonary bypass, aortic bypass surgery, accidental trauma, sepsis, and hydronephrosis (15). Within the first hours after ischemia-reperfusion, acute renal failure represents the major clinical event associated with significant morbidity and mortality (11,20). Despite the progress in the field of intensive care medicine over the past years, approximately 12.5% of acute renal failure survivors are dialysis dependent (rates range widely from 1–64%, depending on the patient population), and 19–31% of them have chronic kidney disease (18). Chronic renal dysfunction after ischemia-reperfusion is usually associated to tissue damage characterized by interstitial fibrosis and tubular atrophy (IFTA) (37). In the case of kidney transplantation, IFTA can be due to various immunological and nonimmunological insults (13). The immunological factors have been and continue to be under intensive investigation (14). Among the nonimmunological causes, renal toxicity induced by chronic administration of calcineurin inhibitors such as cyclosporine (CsA) seems to play an important role in progression of kidney damage (19,28).
At present, strategies to prevent and/or reverse chronic renal dysfunction after ischemia-reperfusion are one of the major challenges in the field of nephrology.
During the last years, cell therapy has shown promising therapeutical properties in the field of renal diseases (1,4,7,12,23). In the case of post ischemia-reperfusion acute renal failure, others and we have shown that administration of MSCs in rodent improves the recovery of renal function and stimulates angiogenesis and tubular regeneration (24,27,40).
Post ischemia-reperfusion chronic kidney disease (CKD) is characterized by the accumulation of extracellular matrix and usually results in a loss of function when normal parenchyma is replaced by fibrotic tissue. We have recently demonstrated that bone marrow MSCs strongly prevent and reverse fibrosis in a rodent model of post ischemic heart failure (26). Indeed we showed that intracardiac administration of bone marrow MCSs in the chronic phase of ventricular remodeling decreases the extracellular matrix accumulation and improves cardiac function (26). In addition, we demonstrated that this effect was related to the inhibition of fibroblast activation by paracrine factors secreted by bone marrow MSCs (26).
Based on these results, we aimed to investigate the effects of bone marrow MSCs on progression of post ischemia-reperfusion renal remodeling and dysfunction and the mechanisms potentially involved. With this scope, we used an original rodent model that we have recently designed (6), mimicking the development of chronic renal disease occurring in clinic after ischemia-reperfusion and immunosuppressive therapy.
Using this model, we investigated (i) whether cell-based therapy using bone marrow MSCs directly injected in the kidney prevents or reduces the development of fibrosis and (ii) the impact of paracrine factors secreted by bone marrow MSCs on epithelio-mesenchymal transition (EMT) of kidney proximal tubular cells in vitro.
Materials and Methods
Animals
Female Lewis rats weighing 180–200 g (Harlan, Gannat, France, http://www.harlan.com) were housed in an air-conditioned room with 12-h light and dark cycles in an environment controlled at a temperature of 22 ± 2°C and a relative humidity of 45%. Animals occupied standard cages with free access to food and water ad libitum. All experiments reported were conducted in accordance with the European Communities Council Directive (86/609/EEC) for experimental animal care and were approved by a local animal care and use committee.
Surgery Protocols
Unilateral Nephrectomy
Under anesthesia with isoflurane/oxygen inhalation (3%:97%), rats were subjected to right nephrectomy. The kidney was isolated (the adrenal gland remained intact), legated with 4–0 silk suture, and excised.
Renal Ischemia-Reperfusion and Cyclosporine Treatment
Seven days after uninephrectomy, rats were anesthetized with isoflurane/oxygen inhalation (3%:97%), and ischemia was induced by clamping the left renal vessels for 45 min using atraumatic vascular microclamps (Arex, France). After clamp removal, the left kidney was inspected for restoration of blood flow. During surgery, the animals were kept at a temperature of 37 ± 1°C on a thermostatically controlled table with a rectal temperature probe (Harvard Instruments) under anesthesia. Twenty-four hours after ischemia-reperfusion (IR), all rats were treated daily with cyclosporine A (Sandimmune, 10 mg/kg SC) for 28 days. The animals subjected to nephrectomy, ischemia-reperfusion, and cyclosporine treatment are designed as NIRC group (Nephrectomy + Ischemia-Reperfusion + Cyclosporine). Control-operated animals were subjected to the same surgical procedure without clamping the renal pedicle. At the end of the treatment, rats were euthanized, and blood samples were collected from aortic abdominal artery and centrifuged (10 min, 8,000 rpm). Plasma was stored at −80°C until following analysis. The left kidney was isolated and fixed in 4% paraformaldehyde solution (24–48 h) for following histological analysis or snap frozen in liquid nitrogen and stored at −80°C for RNA and protein extraction.
Mesenchymal Stem Cell Injection
Seven or fourteen days after renal ischemia-reperfusion injury, two different groups of animals (n = 8 for each group) were anesthetized by isoflurane/oxygen inhalation (3%:97%) and subjected to MSCs intraparenchymal injection (NIRC + MSCs group; 3 × 106 cells) or not (NIRC group; culture medium alone). The injection has been performed in three different areas in the medial longitudinal axis of ischemic kidney (3 × 106 cells per kidney). According with our previous results (27), MSCs were pretreated with melatonin (24 h, 5 mM), in order to preserve them from early death after graft. Then cells were extensively washed with phosphate buffer solution, trypsinized, and labeled with Quantum Dot Fluorescent Nanocrystals (Qtracker 655 labeling kit, Invitrogen) according to manufacturing instructions. An appropriate number of cells were resuspended into 30 ml of α-MEM for each injection. MSCs were visualized by fluorescent microscopy on tissue paraffin section (4 mm) of kidney collected at the moment of sacrifice.
Rat Bone Marrow Mesenchymal Stem Cell Isolation and Culture
MSCs were obtained from bone marrow of male Lewis rats (Harlan, France) weighing 180–200 g. Bone marrow from femur cavities was flushed with α-minimal essential medium (α-MEM; ABCYs, Paris) containing 10% fetal calf serum (FCS) and 1% penicillin/streptomycin (Invitrogen, Carlsbad, CA), and the cell suspension was centrifuged (1,200 rpm, 5 min). Then cells were plated in T175 culture flasks (200,000 cells/cm2). Nonadherent cells were removed after 72 h, and MSCs were recovered by their capacity to adhere highly to plastic culture dishes. As previously described (27), the cells were also phenotyped by positive expression of MSCs markers (CD90, CD29, and CD106) and negative for hematopoietic cells (CD34), vascular cells (CD31), or B-cell markers (CD45). MSCs were then routinely cultured and were used from the passage 3 to the passage 6 for the experiments.
Renal Function
Plasma creatinine (μmol/L) and urea (mmol/L), which reflects renal function, were measured with a Luminex 100 IS system (Luminex Corporation, USA).
Quantification of Renal Fibrosis and Tubular Dilatation
For light microscopic investigations, renal tissue specimens were fixed in 4% paraformadehyde solution and embedded in paraffin. Paraffin sections (4 mm) were stained with hematoxylin/eosin (H&E) and Sirius Red (Sigma Aldrich, France). Three different histological preparations obtained from each animal (n = 8 animals per group) were semiquantified microscopically using MorphoExpert software. Tubular dilatation was measured on H&E-stained tissue slides, and the percentage of fibrosis was estimated by Sirius Red-stained surface compared to total kidney tissue area.
Immunofluorescence
Immunodetection of α-smooth muscle actin (α-SMA) was performed in kidney paraffin-embedded sections. Slides were deparaffinized, rehydrated, and submitted to Tris–EDTA antigen retrieval solution (pH 9; 30 min, 95°C). The endogenous peroxidase activity was blocked with 3% hydrogen peroxide and nonspecific antigen sites were saturated with TBS containing 1% of nonfat dry milk. Then slides were incubated with mouse monoclonal anti-α-SMA (1:1,000, 2 h at room temperature; clone 1A4, Sigma-Aldrich, France), washed with TBS containing 0.1% of Tween 20 and finally incubated with Alexa Fluor 568 (A11019, Invitrogen, France) for 45 min. Pictures were captured by fluorescence microscopy (magnification: 200x).
Determination of mRNA Gene Expression
Total RNA was isolated from frozen rat tissue using RNeasy kit (Qiagen, France) according to manufacturer's protocol. RNA content was measured by a NanoDrop detector (ND-1000 spectrophometer). RNA quality was evaluated by Experion™ RNA HighSens Analysis Kit (Bio-Rad, USA). cDNA was synthesized from equal amounts of RNA (1 μg) using the SuperScript™ II Reverse Transcriptase (Invitrogen, CA). Real-time PCR (ABI Prism 7000 HT Sequence Detection System) for type I, type III, and type IV collagen was performed using SYBR Green PCR Master Mix (Applied Biosystems, Foster City, USA), with the following primer pairs: type I collagen forward GCTTGATCTGTATCTGCCAC/ reverse CACTCGCCCTCCCGTTT; type III collagen forward ACAGCAGTCCAATGTAGATGAATT/reverse CCCGAGTCGCAGACACATATT; type IV collagen forward ATTCCTTTGTGATGCACACCAG/reverse AAG CTGTAAGCATTCGCGTAGTA.
The reaction mixture was preheated at 95°C for 10 min, followed by 40 cycles (95°C/15 s and 60°C/1 min). Each assay, carried out in triplicate, was normalized by amplifying the housekeeping cDNA glyceraldehyde-3-phosphate dehydrogenase (GAPDH; forward primer: TGTTCTAGAGACAGCCGCATCTT/reverse primer: CACAGCCTTGGCAGCACC) and β-actin (forward primer: CTAAGGCCAACCGTGAAAAGAT/reverse primer: A GGGACAACACAGCCTGGAT) from the same cDNA sample, and the relative gene expression was calculated by the comparative Ct method (2-ΔΔCt).
Western Blot Analysis
Kidney tissue samples and cell monolayer were homogenized in lysis buffer (10 mM Tris pH 7.5, 150 mM NaCl, 1 mM EDTA, 1 mM EGTA, 0.1% SDS) with a protease inhibitor cocktail (Sigma). Homogenates were centrifuged (10,000 rpm, 10 min, 4°C) to eliminate tissue and cells debris, and resulted supernatants were used to electrophoresis. The protein concentration was measured using Bio-Rad Protein Assay (Bio-Rad Laboratories, Ivry-sur-Seine, France). Equal amounts of protein for each sample (10 and 30 μg for α-SMA and matrix metalloprotease 2 (MMP2)/ E-cadherin expression, respectively) were denaturated for 10 min at 95°C with Laemmli buffer and were separated by sodium dodecyl sulfate polyacrylamide gel electrophoresis (SDS-PAGE; 10% acrylamide) using Kalleidoscope Precision Plus Protein™ as molecular weight standards (Bio-Rad). Gels were transferred onto polyvinylidene fluoride (PVDF) membranes. The membranes were blocked with TBS-0.2% Tween + 2% bovine serum albumin (BSA; Sigma) at room temperature for 2 h and probed with polyclonal mouse anti-α-SMA (1:7,000, Sigma), monoclonal mouse anti-MMP2 (1:500; TebuBio) or monoclonal mouse anti-E-cadherin (E-cadh; 1:500; BD Biosciences) overnight at 4°C. The membranes were washed three times with TBS-0.2% Tween and incubated for 30 min with the appropriate secondary antibody (1:10,000) conjugated with peroxidase (ECL™ anti-rabbit, ECL™ anti-mouse, or ECL™ anti-goat, GE Healthcare, Little Chalfont, GB). Detection was performed by ECL reaction (Invitrogen). Membranes were reblotted with polyclonal goat anti-GAPDH (1:1,000; Santa Cruz Biotechnology, Santa Cruz, CA). Protein bands were quantified by densitometry using ImageJ software, and results were expressed as protein of interest/GAPDH ratio.
Gelatine Zymography Analysis
Pro and active forms of MMP2 (gelatinase A) were analysed by gelatin zymography. Protein concentrations of different samples were measured using the Bio-Rad Protein Assay (Bio-Rad Laboratories, Ivry-sur-Seine, France). The samples containing an equal amount of total proteins (30 μg) were mixed with loading buffer [75 mM Tris–HCl (pH 6.8), SDS 1.5%, glycerol 1.65% (v/v), bromophenol blue 0.025%] and loaded onto 8% SDS-PAGE containing 0.8% bovine gelatine A. After electrophoresis, gels were incubated with 2.5% TritonX-100 for 2 h followed by incubation in an activation buffer [50 mM Tris–HCl (pH 7.5), 150 mM NaCl, and 1 mM CaCl2] overnight at 37°C until enzymatic degradation of the substrate took place. Gels were stained with an aqueous solution [Coomassie blue (0.25%; R-250 Brilliant Blue, Sigma, St. Louis, MO), ethanol (50%), and acetic acid (10%)] and then destained with an aqueous solution [acetic acid (10%) and ethanol (50%)]. Gelatinolytic bands were observed as clear zones against the blue background. MMPs are dissociated from their inhibitors during the electrophoresis, and their refolding after removal of SDS allows the measurement of the activity not only of their active forms but also of their artificially activated latent forms (pro-MMP). The intensity of the bands was quantified by gel densitometry using the “Image J” software (version 1.42q; National Institutes of Health, USA).
Human MSCs Isolation and Conditioned Medium Collection
Mesenchymal stem cells were obtained from bone marrow aspirate from the iliac crest of five patients (four male, ages, 35, 38, 39 and 55 years; one female, age, 51 years) after informed consent according to institutional guidelines; human MSCs have been prepared by the “Etablissement Français du Sang” according to the Good Manufacturing Practice rules. The procedures for MSC preparation have been validated by the French Health Products Safety Agency.
For the primo-culture (P0), total bone marrow cells were cultured (5×104 cells/cm2) in complete medium: α-MEM + 10% fetal calf serum + 10 μg/ml of ciprofloxacin. After 3 days in culture, nonadherent cells were harvested and medium was replaced by fresh complete medium. Then the culture is feed 2 times a week until confluence or after a maximum of 21 days. Then the cells (mainly MSCs) are detached by trypsin + EDTA and counted using trypan blue exclusion. They are cultured once in the same medium at 1×103 cells/cm2 until confluence or for a maximum of 21 days (first passage, P1). They are then detached and counted. The MSCs at P0 or P1 are cryopreserved until their use.
For conditioned medium, collection human MSCs raising 80% of confluence were washed and then replaced in the complete medium under normal conditions for 124 h. Supernatants of MSCs were collected, centrifuged at 1,200 rpm for 2 min to eliminate cell residues, and finally preserved at −80°C until its following utilization for experiments on human kidney tubular epithelial cell line (HK2).
HK2 Culture and Treatment
Human kidney tubular epithelial cell line (HK2) was obtained from the American Type Culture Collection (Rockville, MD, USA). Cells were maintained at 37°C in an atmosphere of 95% air and 5% CO2 in DMEM containing 10% fetal bovine serum (PAA, Linz, Austria) and 1% penicillin/streptomycin (Gibson BRL, USA). After digestion with 0.25% trypsin (Invitrogen, Carlsbad, CA) and 0.02% EDTA, 1.0 × 105 cells were grown in six-well plates; then cells were stimulated for 72 h with recombinant human transforming growth factor-β1 (TGF-β1; 10 ng/ml; eBioscience, San Diego, USA) dissolved in normal culture medium or in MSC-conditioned medium. For experiments, cells were divided into four groups based on different experimental conditions as follows: control in normal culture medium, TGF-β1 (10 ng/ml)-treated cells in normal culture medium, control cells in MSC-conditioned medium, TGF-β1 (10 ng/ml)-treated cells in MSC-conditioned medium. Images (200x magnification by phase contrast microscopy) of cells were collected at 72 h to evaluate morphological changes, and cell monolayers were washed with ice-cold phosphate buffer and preserved at −80°C before protein extraction as previously described.
Statistical Analysis
All data were expressed as means ± SEMs. Groups of data were compared with an analysis of variance (ANOVA) followed by post hoc tests (Bonferroni's posttest). Values of p < 0.05 were considered as significant.
Results
Effects of MSCs on Kidney Function
As already described (6), our animal model consisted of a right nephrectomy, ischemia-reperfusion injury on the left kidney, and cyclosporine treatment for 28 days (NIRC group). MSCs, obtained from bone marrow of male Lewis rats, were directly injected into renal parenchyma at 7 or 14 days after ischemia-reperfusion injury.
In order to evaluate the effect of MSC injection on kidney function, we measured the creatinine and urea plasma levels on blood samples collected 7, 14, and 28 days after ischemia-reperfusion injury, and we compared the results obtained in untreated animals (NIRC group) with those of the animals subjected to MSC injection (NIRC + MSCs).
At these experimental conditions, NIRC rats presented a significantly increase in plasma creatinine (Fig. 1A) and urea (Fig. 1B) compared to control group, reaching the maximum value 7 days after IR.
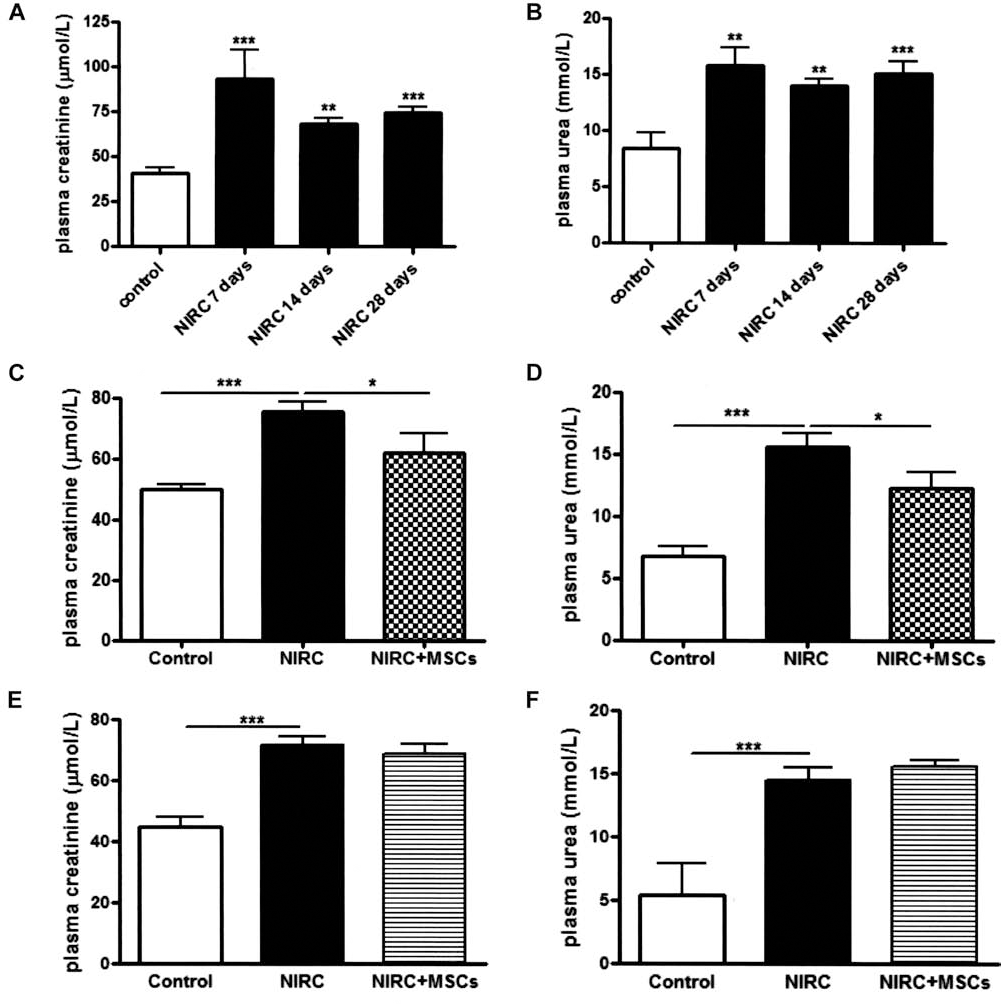
Effect of MSC injection on kidney function. (Top) Plasma creatinine (A) and urea (B) in NIRC animals at 7, 14, and 28 days after IR. (Middle) plasma creatinine (C) and urea (D) levels determined 28 days after IR injury from animals subjected to MSC injection (NIRC + MSCs) 7 days after IR. (Bottom) Plasma creatinine (E) and urea (F) levels determined 28 days after IR injury from animals subjected to MSC injection (NIRC + MSCs) 14 days after IR compared to untreated animals (NIRC). *p < 0.05, ***p < 0.001 (n = 8 for each group). NIRC, nephrectomy + ischemia-reperfusion [IR] + cyclosporine treatment; MSCs, mesenchymal stem cells.
The injection of MSCs 7 days after IR ameliorated kidney function as shown by the significant decrease in plasma creatinine (Fig. 1C) and urea (Fig. 1D) as compared to NIRC rats at 28 days after IR.
When MSC injection was performed 14 days after IR, we did not observe any improvement in kidney function (Fig. 1E, F).
Prior to MSC graft into renal parenchyma, cells were labeled with fluorescent inorganic nanocrystals (Quantum Dot), a reliable labeling agent for tracking exogenous MSCs in histological sections. As shown in Figure 2A, cells were visualized (in red) by fluorescence microscopy on kidney sections from MSC-treated rats (NIRC + MSCs) sacrificed at 28 days after ischemia-reperfusion injury; cells were visualized in the three different areas of injection sites for each animal. The counterstaining with hematoxylin/eosin (Fig. 2B) allowed us to localize the injection sites.

Tracking of MSCs after renal intraparenchymal injection. (A) Representative image (scale bar: 100 μm) of QD fluorescence labeling of MSCs injected 7 days after IR and recovered 28 days after IR on kidney histological preparations. (B) Representative image (scale bar: 100 μm) of hematoxylin/eosin staining within the injection site; cells (labeled in brown) have been localized in the three different injection areas for each treated kidney (n = 8 for each group).
Effect of MSCs on the Development of Kidney Fibrosis
In order to evaluate the extent of kidney fibrosis and of tubular dilatation, tissue sections obtained from kidney collected 28 days after ischemia-reperfusion injury of untreated (NIRC group) or MSC-treated (NIRC + MSCs) or control animals were stained with Sirius Red and hematoxylin/eosin staining. Examination of kidney sections showed a significant increase in collagen accumulation (Fig. 3A, B) and in tubular dilatation (Fig. 3C, D) of NIRC rats compared to control animals. The injection of MSCs 7 days after IR injury (NIRC + MSCs group) significantly reduced the extent of fibrosis (Fig. 3A, B) and also the tubular dilation (Fig. 3C, D) as compared to NIRC animals.

Effect of MSC injection on renal interstitial fibrosis and tubular dilatation. (A) Representative images (scale bar: 100 μm) of Sirius Red staining on kidney histological preparation from control and NIRC- and MSC-treated (NIRC + MSCs) animals. (B) Quantification of interstitial fibrotic tissue by Morpho Expert software analysis; results are expressed as mean ± SEM of percentage of red-stained surface compared to total kidney area. (C) Representative images (scale bar: 100 μm) of hematoxylin/eosin staining on kidney histological preparation from control and NIRC and MSC-treated (NIRC + MSCs) animals. (D) Quantification of tubular dilatation by Morpho Expert software analysis; results are expressed as mean ± SEM of percentage of tubular lumen occupied area compared to total kidney surface. **p < 0.01, ***p < 0.001 (n = 8 for each group). NIRC, nephrectomy + ischemia-reperfusion + cyclosporine treatment; MSCs, mesenchymal stem cells.
This histological pattern was also reflected in tissue mRNA levels of extracellular matrix (ECM) components. According to the decrease in extracellular matrix deposition described in histological results, the injection of MSCs 7 days after IR significantly reduces type I (Fig. 4A), type III (Fig. 4B), and type IV collagen (Fig. 4C) mRNA expression as compared to NIRC rats.

Effect of MSCs on collagen mRNA expression. Renal mRNA expression of type I (A), type III (B), and type IV (C) collagens from control and NIRC- and MSC-treated (NIRC + MSCs) animals. Results are normalized to reference genes GAPDH and actin and referred to control animal expression levels. *p < 0.05, **p < 0.01, ***p < 0.001 (n = 8 for each group). NIRC, nephrectomy + ischemia-reperfusion + cyclosporine treatment; MSCs, mesenchymal stem cells; GAPDH, glyceraldehyde 3-phosphate dehydrogenase.
α-Smooth muscle actin (α-SMA) protein is normally expressed in physiological conditions as a vascular structures component. In contrast, α-SMA is also expressed by activated fibroblasts and tubular cells undergoing to epithelial–mesenchymal transition during kidney remodeling. At our experimental conditions, control animals exhibited a specific vascular distribution of α-SMA protein (Fig. 5A). By contrast, a diffused and nonorganized α-SMA protein labeling is observed in peritubular and interstitial area of tissue sections from NIRC animals (Fig. 5B). This histological feature was already described during renal remodeling after IR. As observed for collagen accumulation, MSC-grafted animals (NIRC + MSCs) showed an α-SMA labeling perivascular-like pattern (Fig. 5C) similar to that observed in control animals. According to immunohistological results, Western blot analysis showed an α-SMA protein increase in NIRC rats compared to control animals. MSC injection significantly reduced α-SMA protein amount in total kidney homogenates as compared to NIRC animals (Fig. 5D).
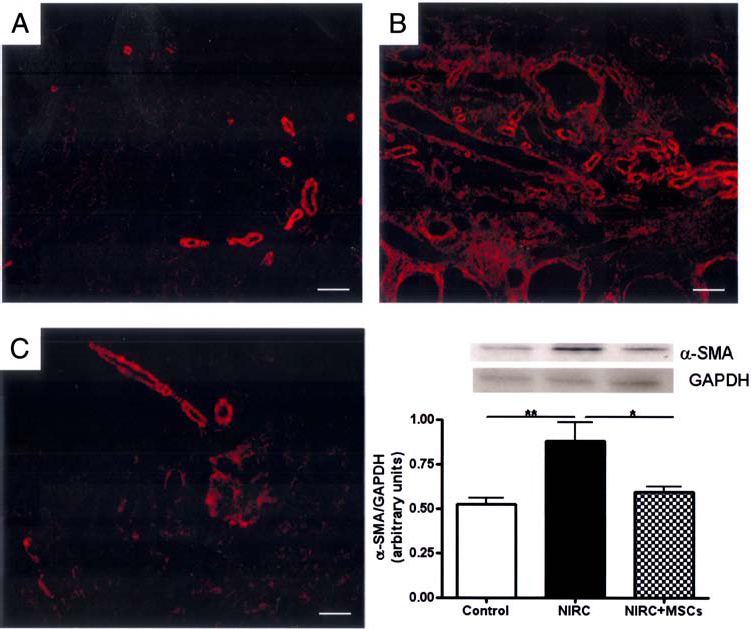
Effect of MSCs on α-SMA protein expression. Representative images (scale bar: 50 μm) of immunofluorescence labeling for localization of α-SMA protein on kidney histological preparations from control (A) and NIRC- (B) and MSC-treated (C; NIRC + MSCs) animals. (D) Quantification of α-SMA protein expression by Western blot in total kidney homogenate obtained from control and NIRC- and MSC-treated (NIRC + MSCs) tissues; results are expressed as mean ± SEM of α-SMA/GAPDH ratio of densitometry arbitrary units. *p < 0.05, **p < 0.01 (n = 8 for each group). NIRC, nephrectomy + ischemia-reperfusion + cyclosporine treatment; MSCs, mesenchymal stem cells; α-SMA, α-smooth muscle actin.
As already described (10), an increase in matrix metalloproteinase 2 (MMP2) activity is associated to pathological kidney remodeling. Western blot analysis and zymography showed that both MMP2 expression (Fig. 6A) and activity (Fig. 6B) increased in NIRC compared to control animals. MSC injection significantly reduced MMP2 activity, without a significant effect on protein amount.
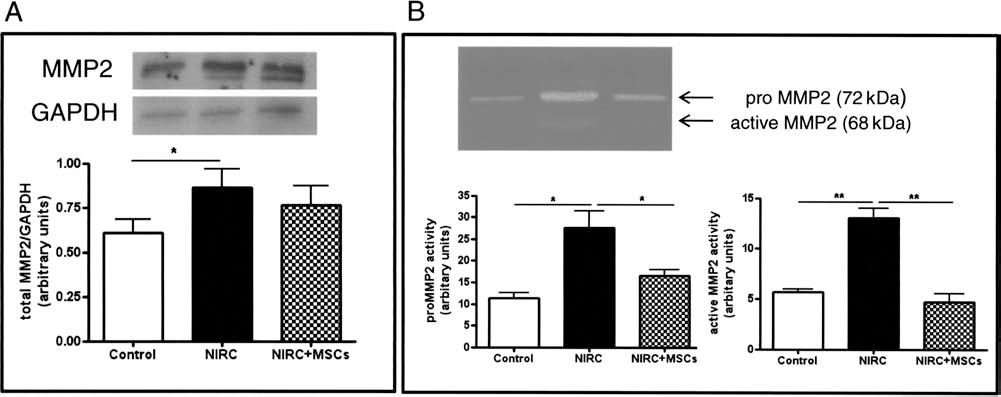
IR associated with cyclosporine induced an increase in MMP2 protein expression and activity: effect of MSCs injection. (A) Quantification of MMP2 protein expression by Western blot in total kidney homogenate obtained from control and NIRC- and MSC-treated (NIRC + MSCs) tissues; results are expressed as mean ± SEM of MMP2/GAPDH ratio of densitometry arbitrary units. (B) Representative image of gelatin zymogram of total kidney homogenate from control and NIRC- and MSC-treated (NIRC + MSCs) animals showing the pro (72 kDa) and the active (68 kDa) forms of MMP2. Densitometry quantification of pro MMP2 and active MMP2 activity; results are expressed as mean ± SEM of arbitrary units. *p < 0.05, **p < 0.01 (n = 8 for each group). *p < 0.05. NIRC, nephrectomy + ischemia-reperfusion + cyclosporine treatment; MSCs, mesenchymal stem cells; MMP2, matrix metalloproteinase-2.
Effect of MSC-Conditioned Medium on TGF-β-Treated HK2 Cells
One of the major mechanisms involved in the development of fibrosis and in the impaired kidney function is the epithelial–mesenchymal transition of proximal tubular cells in the kidney. Different cytokines are largely secreted during pathological conditions; among these, TGF-β seems to play an important role in the stimulation of EMT. This cytokine is at the present the most used in in vitro models of EMT (45).
As already described (22,42,45), we used TGF-β to induce EMT in kidney proximal tubular epithelial cells (HK2), and we studied the effect of MSC-conditioned medium in this in vitro model. Our results showed that TGF-β is able to induce morphological changes in HK2 cells from an epithelial phenotype to a fibroblastic-like appearance (Fig. 7A). TGF-β treatment also promoted a decrease in E-cadherin concomitant to an increase in α-SMA protein expression (Fig. 7B), which are hall-markers of epithelial–mesenchymal transition. Incubation of HK2 cells with conditioned medium (36) of MSCs inhibited the TGF-β-dependent morphological changes of HK2 cells (Fig. 7A), reduced α-SMA and increased E-cadherin protein expression (Fig. 7B).

Effect of MSC-conditioned medium (CM) on TGF-β-induced epithelial mesenchymal transition in HK2 cells. (A) Representative images (scale bar: 100 μm) of contrast-phase microscopically field of HK2 cells in the presence or the absence of TGF-β. (B) Quantification of α-SMA and E-cadherin protein expression by Western blot in HK2 cells in the presence or the absence of TGF-β. Results are expressed as mean ± SEM of α-SMA/GAPDH and E-cadherin/GAPDH ratio of densitometry arbitrary units of three different experiments. *p < 0.05, **p < 0.01. MSCs, mesenchymal stem cells; α-SMA, α-smooth muscle actin; TGF-β, transforming growth factor-β.
Discussion
Several studies showed the beneficial effects of MSCs administration in the acute renal damage after ischemia-reperfusion (1,2,25,27,35). However, the effect of MSC-based cell therapy in the progression of chronic renal damage and failure following ischemia-reperfusion insult is still uncertain (17). The demonstration that cell therapy may improve the outcome of post ischemia-reperfusion chronic renal dysfunction would be particularly relevant in clinical practice as, despite the improvement of classical therapeutical approaches, a large percentage of patients with acute renal failure undergo chronic disease (18). In order to mimic better the clinical frame of post ischemia-reperfusion chronic renal disease occurring in humans, we used uninephrectomized rats submitted to ischemia-reperfusion and chronic cyclosporine treatment. The use of cyclosporine has a double interest. First, as observed for immunosuppressive therapy in human, cyclosporine aggravates the outcome of renal disease and increases interstitial fibrosis and tubular damage. These effects have been related, in part, to the ability of cyclosporine A to increase secretion of profibrotic TGF-β by proximal tubular cells (8,36). Second, its immunosuppressive properties minimize the previously described immunomodulatory activity of MSCs (3,31,43,44).
Using this model, we showed that a single intrarenal administration of bone marrow MSCs 7 days after ischemia-reperfusion significantly improved renal function and modified renal remodeling. The improvement of renal function was associated to reduced extracellular matrix accumulation as shown by the decrease in collagen staining and mRNA expression measured by histomorphological analysis and semiquantitative RT-PCR, respectively. In addition, MSCs administration also reduced tubular dilation, which is a classical feature of progressive renal failure.
In the kidney, resident fibroblasts and fibroblast-like cells derived from tubular cells via EMT play a key role in renal fibrosis (39). Both cell types express α-SMA that promotes their migration during remodeling process (16,38,41). Our results show that kidneys from NIRC animals presented a diffused distribution of α-SMA protein suggesting the interstitial accumulation of activated fibroblasts and fibroblast-like cells. The injection of MSCs strongly reduced α-SMA expression in the renal interstitium, and as observed in normal kidney, α-SMA was found mainly in the vascular area. These results show that injected MSCs decrease the number of activated fibroblasts and/or fibroblast-like cells.
We have previously shown that paracrine factors secreted by MSCs inhibit activation of cardiac fibroblast in vitro (26). Recently, Semedo and colleagues (32–34) suggested that MSCs might inhibit EMT through immunomodulatory and anti-inflammatory mechanisms. However, it is unlikely that these mechanisms account for the EMT in our ischemia-reperfusion model as animals were treated with the immunosuppressive drug cyclosporine. In addition, we did not observe significant differences in the inflammatory infiltrates and plasma level of proinflammatory cytokines interleukin (IL)-1α, IL-1β, IL-2, IL-6, IL-12, interferon (INF)-β, and granulocyte-macrophage colony-stimulating factor (GM-CSF) between control and NIRC- and MSC-treated animals (data not shown). Therefore, we investigated whether paracrine factors secreted by MSCs may directly prevent EMT in vitro. Our results showed that MSC-conditioned medium prevented TGF-β-induced EMT in proximal tubular cells. Taken together, these observations suggest that inhibition of EMT by MSCs occurs, in part, by a direct activity on tubular cells.
MMP2 plays a major role in renal remodeling (5,29). Indeed, it has been shown that transgenic mice overexpressing MMP2 display fibrotic lesions and functional changes, characteristic of human chronic kidney disease (10). In kidney, MMP2 has important functions that go beyond the degradation of extracellular matrix. Moreover, it has been extensively shown that MMP2 directly stimulate EMT of tubular cells (9) and promote their migration through degradation of tubular basal membrane (30). As observed in other animal models of renal fibrosis (21), we found that MMP2 expression and activity increased after renal ischemia-reperfusion and cyclosporin treatment. Injection of MSCs strongly decreased MMP2 activity in NIRC and prevented degradation of basal membrane. Taken together, these results suggest that MMP2 plays a key role in the antifibrotic effects of MSCs.
Our functional results showed that a single intraparenchymal injection of MSCs improves renal function in an experimental situation mimicking the renal damage after IR and immunosuppression occurring in humans. Interestingly, we found that improvement of renal function was observed only when MSCs were administrated 7 days after ischemia-reperfusion. In contrast, we did not observe any significant amelioration when injection was performed 14 days after IR. These results suggest that the efficacy of MSCs may depend on the degree of renal fibrosis and underline the importance of the choice of the time of administration for the success of cell therapy. This is a critical point to be considered in the view of the clinical transfer of cell therapy protocols.
In conclusion, our results show that a single intrarenal administration of bone marrow MSCs strongly reduced renal fibrosis and promoted the recovery of renal function after IR and immunosuppressive therapy. It is conceivable that cell therapy using intrarenal administration of MSCs may represent a novel therapeutical approach for treatment of chronic renal failure.
Footnotes
Acknowledgments
This work was supported in part by INSERM and grants from the National Research Agency (ANR; grant under the program Physiopathologie des Maladies Humaines, SYNMESCARI Project) and from the European Program FP7-Health-2007-B (STAR-TREK project). We thank the Service d'anatomie et cytologie pathologiques (CHU Rangueil, Toulouse, France), the Service de Zootechnie and GenoToul (INSERM, IFR31, Toulouse, France), and the Laboratoire d'ingénierie cellulaire (EFS Pyrenées- Méditerranée, Toulouse, France). The authors declare no conflicts of interest.
