Abstract
Mesenchymal stem cells (MSCs), which are poorly immunogenic and have potent immunosuppressive activities, have emerged as a promising candidate for cellular therapeutics for the treatment of disorders caused by abnormal immune responses. In this study we investigated whether human umbilical cord-derived mesenchymal stem cells (hUC-MSCs) could ameliorate colitis in a trinitrobenzene sulfonic acid (TNBS)-induced colitis model. TNBS-treated colitic mice were infused with hUC-MSCs or vehicle control. The mice were sacrificed on day 1, 3, and 5 after infusion, and their clinical and pathological conditions were evaluated by body weight, colon length, and histological analysis. The expression levels of proinflammatory cytokine proteins in colon were examined by ELISA. The homing of hUC-MSCs was studied by live in vivo imaging and immunofluorescent microscopy. hUC-MSCs were found to migrate to the inflamed colon and effectively treated the colitic mice with improved clinical and pathological signs. The levels of IL-17 and IL-23 as well as IFN-γ and IL-6 were significantly lower in the colon tissues of the hUC-MSC-treated mice in comparison with the vehicle-treated mice. Coculture experiments showed that hUC-MSCs not only could inhibit IFN-γ expression but also significantly inhibit IL-17 production by lamina propria mononuclear cells (LPMCs) or splenocytes of the colitic mice or by those isolated from normal animals and stimulated with IL-23. Systemically infused hUC-MSCs could home to the inflamed colon and effectively ameliorate colitis. In addition to the known suppressive effects on Th1-type immune responses, hUC-MSC-mediated modulation of IL-23/IL-17 regulated inflammatory reactions also plays an important role in the amelioration of colitis.
Introduction
Crohn's disease (CD) and ulcerative colitis (UC), the two major forms of inflammatory bowel diseases (IBD), are characterized by a chronic inflammation of the gastrointestinal tract in genetically susceptible individuals. The precise etiopathogenesis of IBD is still not completely understood, but it is generally believed that genetic defects and environmental factors lead to deregulated immune responses to normally harmless commensal bacteria and luminal antigens in the mucosal microflora (40). CD4+ T cells and the cytokines they produce play a critical role in the course of IBD. An earlier study suggests that CD is most closely associated with T helper 1 (Th1)-type immune responses, whereas UC appears to be more consistent with Th2-like responses (13). Blocking the p40 subunit of interleukin-12 (IL-12), a key regulator of Th1 cell development, can prevent the onset of colitis and effectively treat ongoing disease in animal models, and administration of anti-IL-12 monoclonal antibody may result in clinical improvement in patients with active CD (35). More recent studies, however, have provided strong evidence linking IL-23 with IBD. IL-23 is a heterodimeric cytokine composed of a unique p19 subunit and the p40 chain shared with IL-12. IL-23 is essential for the proliferation and maintenance of the newly identified Th17 cells that characteristically produce IL-17, IL-6, tumor necrosis factor-α (TNF-α), and IL-22. The inflamed mucosa of IBD patients contain high levels of IL-17 and IL-23 (12,13) and patients with CD have increased numbers of circulating and intestinal Th17 cells that readily produce interferon-γ (IFN-γ) and IL-17 upon stimulation with IL-23 (22). A genome-wide association study showed that IL23R variants are associated with CD (8). Monoclonal anti-IL-23p19 prevented and treated active colitis, with downregulation of a broad array of inflammatory cytokines and chemokines in the colon (10).
Mesenchymal stem cells (MSCs) are fibroblast-like, plastic-adherent cells capable of self-renewal and differentiation into cells of the mesoderm lineage, including bone, fat, and cartilage (3). MSCs apparently resides in all organs and tissues including bone marrow, muscle, brain, skin, adipose tissue, and umbilical cord (4). An emerging body of data indicates that MSCs are not only poorly immunogenic but also have potent and broad immunosuppressive activities. MSCs can inhibit the activation and proliferation of T lymphocytes in a major histocompatibility complex (MHC)-independent manner (43). MSCs can also regulate the immune response indirectly through modulation of professional antigen presenting cells such as dendritic cells (18). Although the mechanisms of this effect are not fully understood, both cell-to-cell contact and soluble factors secreted by MSC are likely involved (6).
We have previously established a protocol for isolation and expansion of MSCs from umbilical cord, and found human umbilical cord (hUC)-MSCs share most characteristics with bone marrow-derived MSCs (20,30). In this study, we used the trinitrobenzene sulfonic acid (TNBS) colitis model, which mimics some features of CD (38), to study whether hUC-MSC infusion could ameliorate the experimental colitis by modulating the response of inflammatory immune cells and downregulating the expression of proinflammatory cytokines. In addition to the modulatory effects of hUC-MSCs on Th1-type immune responses, we show that the potent suppression of IL-23/IL-17-regulated inflammatory reactions also plays an important role in the amelioration of colitis.
Materials and Methods
hUC-MSC Isolation, Expansion, and Phenotype Analysis
Umbilical cords for hUC-MSC preparation were obtained from local maternity hospitals with donors' written consent and approval by the institutional review board of the Chinese Academy of Medical Science and Peking Union Medical College. hUC-MSC isolation and in vitro expansion were according to the procedures described previously (30). Briefly, the cord was cut into 1–2-mm3 pieces and digested with 0.075% collagenase II (Sigma) and 0.125% trypsin (Gibco) with gentle agitation at 37°C. The digested mixture was then passed through a 100-μm filter to obtain cell suspensions. Cells were cultured in Dulbecco's minimum essential medium (DMEM)/F12 (1:1) (Gibco) supplemented with 10% fetal bovine serum (FBS) (HyClone), 100 U/ml penicillin-streptomycin, 1% glutamine, and 10 ng/ml epidermal growth factor (EGF; Sigma). Nonadherent cells were removed by changing the medium after 3 days. Cells at passages 5–8 (–20–25 cell doublings) were used for experiments. Surface marker expression was analyzed by flow cytometry using the following antibodies: phycoerythrin (PE)-conjugated human monoclonal antibodies against CD105, CD73, CD29, CD44; fluorescein isothiocyanate (FITC)-conjugated human monoclonal antibodies against CD45, human leukocyte antigen (HLA)-A,B,C, HLA-DR, CD34. Mouse isotype-matched antibodies served as controls. All antibodies were purchased from BD Biosciences (San Diego, CA). Human umbilical vein endothelia cells (HUVEC) were isolated from umbilical cord by 0.25% trypsin/EDTA digestion for 15 min, and cultured in M199 media (Gibco) containing 20% FBS, 10 ng/ml vascular endothelial growth factor (VEGF; Sigma), and 20 ng/ml EGF supplements.
For flow cytometry analysis, adherent cells were detached by treating with 0.05% trypsin-EDTA, neutralized with FBS-containing culture medium, and disaggregated into single cells by pipetting. The cells were incubated with mAbs for 30 min at 4°C, washed twice with PBS, resuspended in 0.5 ml PBS, and immediately analyzed using a FACSCalibur flow cytometer (Becton Dickinson). At least 2 × 105 cells were used for each sample and cell Quest software was used for data analysis.
Mice
Eight-week-old male BALB/c mice weighing approximately 20 g were purchased from the Institute of Experimental Animals, Chinese Academy of Medical Sciences. The mice were maintained on a 12-h light/12-h dark cycle under pathogen-free conditions and had a standard diet and water. Tissue collection for research and experiment protocols involving animals were approved by the local ethical committee and the institutional review board of the Chinese Academy of Medical Science and Peking Union Medical College.
Induction of Colitis
Colitis was induced by TNBS (Sigma-Aldrich) according to the method described previously (34). Briefly, BALB/c mice were lightly anesthetized after a 24-h fast and then administered TNBS using rectal enema (i.r.) via a 3.5 French (F) catheter equipped with a 1-ml syringe. The catheter was inserted into the rectum until the tip was advanced to 4 cm proximal to the anal verge. TNBS (100 mg/kg) dissolved in 50% ethanol was slowly introduced into the colon through the cannula and mice were held in a vertical position for 30 s to ensure the distribution of the reagent within the entire colon and cecum. Control mice were administered only with 50% ethanol solution.
Infusion of hUC-MSCs
Immediately after the TNBS treatment, the mice were randomly divided into groups and injected intravenously with either 1 × 106 hUC-MSCs in 100 μl PBS per animal or with 100 μl PBS alone. The injection was repeated once on the following day. For cell tracking studies, cells were washed with PBS, incubated with the fluorescent carbocyanine CM-DiI dye (Molecular Probes) at a concentration of 1 ng/μl into 1 ml of cell suspension for 5 min at 37°C and 15 min at 4°C, and then washed twice with PBS and resuspended at 107 cells/ml in PBS. The recovery rate of this procedure was 80–90% viable cells.
Assessment of the Severity of Colitis and Histological Examination
To examine the severity of colitis, the body weights were measured every day. The mice were anesthetized and sacrificed on day 1, 3, and 5. The entire colon was removed from the cecum to the anus and flushed with saline. Colonic inflammation and damage were analyzed by histological examination. Colon samples were fixed in PBS containing 4% formaldehyde. Paraffin-embedded sections (5 mm) were stained with hematoxylin and eosin. Colonic inflammation and damage were assessed according to the histopathologic grading system of Macpherson and Pfeiffer (32). In brief, 0: no signs of inflammation; 1: very low level of inflammation; 2: low level of leukocyte infiltration; 3: high level of leukocyte infiltration, high vascular density, thickening of the colon wall; 4: transmural infiltrations, loss of goblet cells, high vascular density, thickening of the colon wall. All scoring was performed by the same individual under blind conditions.
Measurement of Myeloperoxidase Activity
Myeloperoxidase (MPO) activity, an index of inflammatory response, was assayed in colon tissues as described previously (24). MPO activity in the colon homogenate was determined using a Myeloperoxidase Assay Kit (CytoStore Inc., Calgary, Alberta, Canada) according to the manufacturer's instructions. Total protein concentrations were measured using a bicinchoninic acid (BCA) Protein Assay Kit (Pierce Co., Rockford, IL) for calibration.
Assessment of Expression of Cytokine Proteins
The mice were sacrificed on day 1, 3, and 5 after colitis induction and segments of distal colon were dissected out and snap frozen in liquid nitrogen. The colons were homogenized with 0.5 ml of PBS containing 1% Triton X-100 with protease inhibitor cocktail (Amresco). Then the homogenates were centrifuged at 12,000 × g for 15 min at 4°C, and the supernatants were collected and assayed for protein concentration. Total protein concentrations of the tissue supernatants were measured using a BCA Protein Assay Kit (Pierce Co.) for calibration. Quantitative analysis of IFN-γ, IL-16, IL-23, IL-17, transforming growth factor-β (TGF-β), and IL-10 in the supernatants was performed by enzyme-linked immunosorbent assay (ELISA) using commercially available kits (R&D Systems, Abingdon, Oxon, UK), according to the manufacturer's instructions. The IL-23 ELISA kit uses a p19-specific capture antibody and a p40-specific detection antibody, and will therefore only detects the IL-23 p19/p40 heterodimer.
Isolation of Lamina Propria Mononuclear Cells and Splenocytes
Colonic lamina propria mononuclear cells (LPMCs) and splenocytes were isolated from TNBS-induced colitic mice. LPMCs were isolated and prepared according to published methods (42). Briefly, mice were killed and the gut was flushed with PBS, and the intestine was then cut open longitudinally and dissected into segments of 1–2 cm. The epithelium was removed by incubation at 37°C in PBS supplemented with 0.37 mg/ml EDTA for 15 min. The tissue was subsequently further digested in RPMI-1640 containing 0.15 mg/ml collagenase (Sigma) for 75–90 min at 37°C on a shaking platform. LPMCs were finally isolated from the interface of a Ficoll gradient. The cells were washed twice and suspended at a cell concentration of 1 × 106/ml.
In Vitro Coculture Experiments and Transwell Coculture
To investigate whether hUC-MSCs could inhibit the secretion of the proinflammatory cytokines by LPMCs or splenocytes of TNBS-induced colitic mice, hUC-MSCs were irradiated (30 Gy) and cocultured with the LPMCs or splenocytes isolated from TNBS-induced colitic mice in various ratios and collected at different time points. The cells were suspended in RPMI-1640 medium containing 2 mM glutamine, 100 IU/ml penicillin, 100 μg/ml streptomycin, and 10% fetal bovine serum. Cell suspension (200 μl) was seeded into 96-well, flat-bottomed microtiter plates (Corning Inc., Costar). To further examine the effect of hUC-MSCs on Th1 and Th17 cells, LPMCs or splenocytes from healthy BALB/c mice were isolated, stimulated with rIL-12 (Peprotech, 10 ng/ml), or rIL-23 (eBioscience, 20 ng/ml) and then cocultured with hUC-MSCs. To examine whether the effect of hUC-MSCs on LPMCs is cell-cell contact dependent, LPMCs from healthy normal BALB/c mice were cultured with rIL-12 or rIL-23 in the lower chamber of a 24-mm diameter Transwell plate with a 0.3-μm pore size membrane (Corning Inc., Costar). hUC-MSCs were seeded onto the Transwell membrane of the inner chamber 1–2 h before the beginning of the culture. The concentrations of IFN-γ and IL-17 in the wells were determined by ELISA (R&D Systems, Abingdon, Oxon, UK). Results are presented as means ± SD obtained from triplicate cultures.
Real-Time RT-PCR
For measurement of mRNA levels, total RNA was extracted from colon samples using Trizol Reagent (Invitrogen, Paisley, UK). After chloroform extraction and isopropanol precipitation, the RNA pellets were dissolved in diethylpyrocarbonate-treated water. Total RNA was quantified by measuring the absorbance at 260 nm using a spectrophotometer. RNA was reverse transcribed using a commercial kit (Invitrogen) containing 0.5 mg oligo(dT)18 primer, 40 U of RNasin (Takara Biotech, Japan), and 200 U of Moloney murine leukemia virus (M-MLV) reverse transcriptase, according to the protocol provided with the enzyme. The resulting cDNA was amplified using the SYBR Green Real-Time PCR Kit (ABI) and detected on an ABI 7500 system with the following amplification conditions: 95°C for 10 min and then 40 cycles of 95°C for 15 s and 60°C for 60 s. All experiments were performed at least three separate times. Gene expression was calculated relative to the housekeeping gene glyceraldehyde 3-phosphate dehydrogenase (GAPDH) using the AACt algorithm. The primer sequences were as follows: retinoid-related orphan receptor gamma thymocyte isoform (RORyt), CAGGAGCAATGGAAGTCGTC and CCGTGTAGAGGGCAATCTCA; GAPDH, CACTTGAAGGGTGGAGC and GGGCTAAGCAGTTGGTG.
In Vivo Imaging
hUC-MSCs were labeled with CM-DiI and tracked using the whole body cooled charge-coupled device (CCD) camera system (IVIS 200 Series Imaging System; Xenogen, Alameda, CA). The distribution and fluorescence intensity of CM-DiI-labeled cells were monitored every 5 days after MSC injection. Mice were placed into the light-tight chamber of the CCD camera system, and a gray scale body surface reference image (digital photograph) was taken under weak illumination. Photons emitted from CM-DiI-labeled cells within the animal body were quantified using the software program “Living Image” (Xenogen) as an overlay on Igor (Wavemetrics, Seattle, WA) (9). Mice examined for quantification of CM-DiI-labeled hUC-MSC growth were imaged from the ventral position. Mice were anesthetized with isoflurane consistently during the experimental procedure.
Fluorescence Microscopy
The mice were killed 5 days after the colitis induction; colons were removed, embedded in Tissue-Tek optimal cutting temperature (OCT) compound, and deposited in liquid nitrogen. Sections at 5μm thickness were placed on slides, fixed in acetone at 4°C for 10 min, and then placed in PBS for 5 min to remove Tissue-Tek OCT. All slides were blocked with 5% BSA in PBS for 30 min. Endogenous peroxidase was blocked with the methanol and hydrogen peroxidase for 20 min. Slides were washed three times for 5 min each in PBS before being probed with a mouse anti-human nuclei monoclonal antibody (Chemicon, MAB1281) overnight in a humidified chamber at 4°C. Slides were washed again, and after incubation for 30 min with an anti-mouse FITC-conjugated secondary antibody (eBioscience), slides were washed three times for 5 min in PBS. To detect the cell nucleus, sections were stained with 4′,6-diamidino-2-phenylindole (DAPI; Sigma-Aldrich) for 5 min and then mounted with glycerol buffered with PBS.
Statistical Analysis
Data are presented as mean ± SD. Means between groups are compared with the unpaired Student' t-test. Bonferroni posttests were used for multiple comparisons. Differences between groups are considered statistically significant when p < 0.05 and highly significant when p < 0.01.
Results
Isolation, Expansion, and Phenotype Analysis of hUC-MSCs
hUC-MSCs were isolated and expanded by in vitro culture as previously described (29,30). Figure 1 shows the immunophenotypes of the hUC-MSCs used in the experiments. The cells expressed high levels of CD44, CD105, CD73, CD29, and HLA-ABC, but no or very low levels of CD45, CD34, and HLA-DR (Fig. 1). These phenotypes were maintained in culture for up to 20 passages. The cells could also be induced to differentiate into osteocytes, adipocytes, and chondrocytes (data not shown).

Immunophenotypic characterization of human umbilical cord mesenchymal stem cells (hUC-MSCs). hUC-MSCs were characterized by flow cytometry using antibodies against CD29, CD34, CD44, CD45, CD73, CD105, human leukocyte antigen (HLA)-A, B, C, and HLA-DR or with isotype-matched Ig as negative controls. Cells were positive for CD29, CD44, CD73, CD105, and HLA-A, B, C, but negative for CD34, CD45, and HLA-DR.
hUC-MSCs Reduce Clinical Severity in TNBS-Induced Colitis
All mice developed consistent clinical signs of colitis within 7 days after TNBS administration, including weight loss, bloody diarrhea, and lassitude. Control mice did not develop any signs of colitis and gained weight over time. TNBS-treated mice lost weight rapidly, which was the lowest on day 3 and then gradually recovered (Fig. 2A). Compared with mice receiving PBS, hUC-MSC infusion reduced the extent of body weight losses, which was significantly different on day 2 (p < 0.05) (Fig. 2A), and the colon length was longer after hUC-MSC infusion, which was significantly different on day 3 and 5 (data not shown).
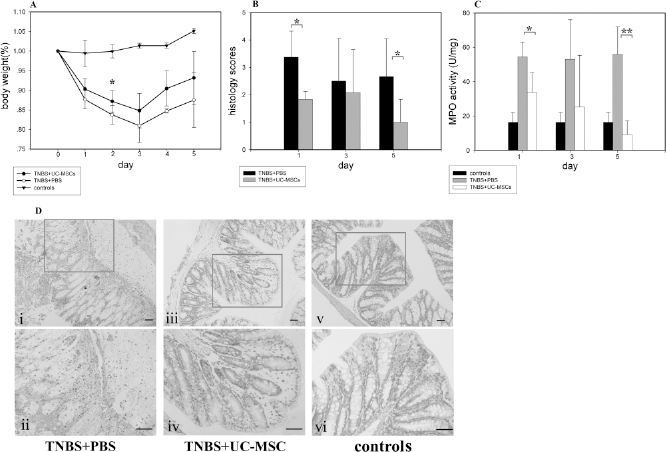
hUC-MSCs ameliorate trinitrobenzene sulfonic acid (TNBS)-induced colitis. TNBS-induced colitic mice were infused with hUC-MSCs or vehicle control on day 0 and 1. Colonic inflammation and damage were analyzed macroscopically and microscopically. The results are presented as means ± SD, n = 6 mice/group (*p < 0.05; **p < 0.01). (A) Daily body weight measurement as a percentage of the starting weight. (B) Histological scores of H&E-stained colon sections. (C) The myeloperoxidase (MPO) activity of colon samples of all treated mice. (D) H&E staining of representative colon sections (Scale bar: 50 μm, original magnification in the top pictures 200×; the bottom pictures 400×).
Since neutrophils are the predominant inflammatory cells in TNBS-induced colitis, MPO activities in colonic tissues were assayed as a measure of neutrophil infiltration. Compare with the PBS-treated colitic mice, the MPO activities in the colon tissues of hUC-MSC-treated animals were significantly lower on day 1 and 5 (Fig. 2B).
Compared with the PBS-treated control group, the histopathologic scores were significantly lower in hUC-MSC-treated group (Fig. 2C). H&E-stained colon slices from the colitic mice treated with PBS showed disordered mucosal architecture, diffused depletion of goblet cells, inflammatory cell infiltration, edema, ulceration, fibrosis, and epithelial cell necrosis (Fig. 2D, i, ii). In contrast, the colonic tissue from hUC-MSC-treated animals showed only thickening of the epithelium, focal depletion of goblet cells, and less inflammatory cell infiltration within the lamina propria (Fig. 2D, iii, iv). As a control, the colons receiving only 50% alcohol showed no lesion of the epithelium, normal mucosal architecture, regular goblet cells, and an ordinary rate of inflammatory cells within the lamina propria (Fig. 2D, v, vi).
Taken together, the above data demonstrated that the hUC-MSC infusion effectively inhibited TNBS-induced tissue inflammation and ameliorated colitis.
hUC-MSC Infusion Suppress Colon and Systemic Proinflammatory Cytokine Production
Because proinflammatory cytokines play an important role in the development of TNBS-induced colitis, a possible mechanism of the hUC-MSC therapy is the suppression of the production of these cytokines in the colon. Consistent with previous reports (14,35), TNBS treatment rapidly induced production of IFN-γ, a typical Th1 cytokine, and its levels continued to rise over time (Fig. 3A). The levels of IL-6 also increased rapidly following TNBS treatment (Fig. 3B). However, the levels of these two cytokines in the colon tissues from TNBS-treated mice that had been infused with hUC-MSCs were significantly lower in comparison with the mice without hUC-MSC infusion (p < 0.05 for IL-6 on day 3; p < 0.01 for IFN-γ on day 5) (Fig. 3A, B). The results indicated that the infusion of hUC-MSCs had an inhibitory effect on the expression of IFN-γ and IL-6 in the colonic tissues, the two cytokines that are classically associated with TNBS-induced colitis.
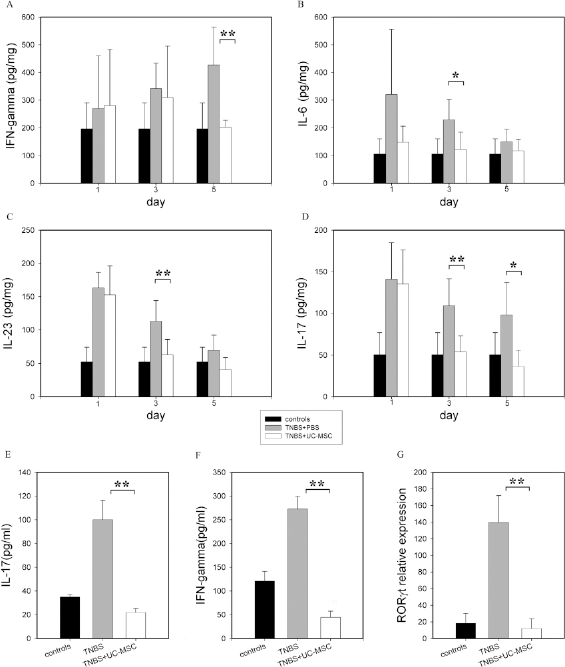
Expression of cytokines in colon tissues. (A–D) The levels of interferon (IFN)-γ, interleukin (IL)-6, IL-23, and IL-17 proteins in the colon extracts were determined by ELISA at the specified time points after colitis induction. The results are presented as means ± SD, n=5/group. Black bar: 50% ethanol treatment group; gray bar: TNBS treatment group; white bar: TNBS and hUC-MSC treatment group. (E, F) The levels of IL-17 and IFN-γ in the sera of colitic mice were determined by ELISA on day 5 (n = 5/group). (G) Colonic samples were analyzed for retinoid-related orphan receptor gamma thymocyte isoform (RORγt) expression by real-time PCR on day 3. Data are presented as means ± SD of two separate experiments in which 5 mice/group were analyzed (*p < 0.05; **p < 0.01).
IL-23 is a proinflammatory cytokine that has been linked with a number of autoimmune pathologies including several forms of intestinal inflammation (16). As shown in Figure 3C, the IL-23 levels were rapidly and dramatically elevated by TNBS treatment (approximately threefold vs. the vehicle control at 24 h). IL-23 is a potent inducer of IL-17 expression in mice, and in line with the increased levels of IL-23, the colon tissues of TNBS-treated animals had dramatically elevated levels of IL-17, which were the highest at 24 h and remained high over the 5-day observation period (Fig. 3D). hUC-MSC infusion, however, altered the expression patterns of the two cytokines, as their levels were significantly lower in the hUC-MSC group (p < 0.01 for IL-23 and IL-17 on day 3; p < 0.05 for IL-17 on day 5) (Fig. 3C, D).
To further investigate the impact of hUC-MSC on cytokine expression, the concentrations of IFN-γ and IL-17 in the sera from colitic mice with or without hUC-MSC infusion were determined by ELISA. Compared with the control mice, TNBS-treated colitic mice had much higher levels of IFN-γ and IL-17 (Fig. 3E, F). In contrast, the concentrations of IFN-γ and IL-17 in sera from colitic mice that had received hUC-MSC infusion were significantly lower (p < 0.01 on day 5), indicating that the systemic inflammatory responses were similarly downregulated by hUC-MSC infusion. The concentrations in the colonic extracts of anti-inflammatory cytokines TGF-β and IL-10 were also determined. Compared with the control, TNBS treatment appeared to result in reduced expression of both cytokines, but the reduction was not statistically different and hUC-MSC infusion did not make an obvious impact on their expression (data not shown).
The orphan nuclear receptor RORyt is a key transcription factor that regulates the differentiation and function of both mouse and human Th17 cells (17). Moreover, RORyt is expressed in a subset of mouse lamina propria T cells and is required for expression of IL-17 (17). Therefore, the RORyt gene expression in colon tissues was assessed by real-time RT-PCR. Compared with the untreated control animals, the expression of RORyt in colon samples from colitic mice without hUC-MSC treatment on day 3 was dramatically increased (Fig. 3G). In sharp contrast, the RORyt expression in colon samples from hUC-MSC-treated colitic mice was inhibited. These results suggest that the expression of RORyt is upregulated in lamina propria cells by TNBS treatment, and enhanced RORyt expression, together with TGF-β and the proinflammatory cytokines IL-6 and IL-23, promotes Th17 cell differentiation and IL-17 production. Accordingly, a possible mechanism of hUC-MSC-mediated suppression of Th17 response and IL-17 expression is through downregulating the RORyt expression.
hUC-MSCs Suppress Production of IFN-γ and IL-17 by LPMCs and Splenocytes In Vitro
To more directly test the effect of hUC-MSCs on proinflammatory cytokine expression, in vitro coculture of hUC-MSCs with splenocytes or LPMCs were carried out. Although the splenocytes isolated from colitic mice did not produced measurable levels of IL-6, IFN-γ, and IL-23 during the in vitro culturing without further stimulation (data not shown), the splenocytes expressed low but detectable amount of IL-17 (22.16 ± 2.86 pg/ml). However, the presence of hUC-MSCs in the coculture system suppressed the production of IL-17 in a dose-dependent manner, and the suppression was highly significant when hUC-MSCs/splenocytes ratio was 1:20 or less (Fig. 4A). Similar experiments were also performed using LPMCs isolated from colitic mice cultured alone or cocultured with irradiated hUC-MSCs. As shown in Figure 4B, IL-17 accumulated to a level of 29.81 ± 3.89 pg/ml at 72 h in the supernatant of LPMCs cultured alone, whereas there was no obvious increase in the IL-17 concentrations when LPMCs and hUC-MSCs (1:1 ratio) were cocultured.
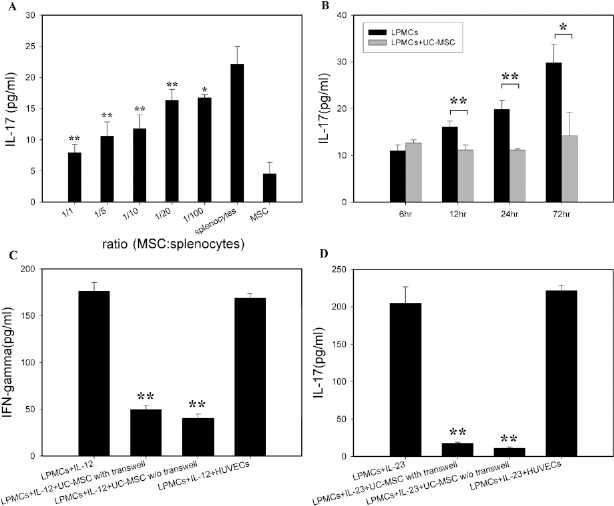
In vitro coculture experiments. The inhibitory effect of hUC-MSCs on IL-17 expression by (A) splenocytes (all different were significant vs. splenocytes) or (B) lamina propria mononuclear cells (LPMCs) of colitic mice. (C, D) LPMCs isolated from normal mice were cultured in the transwell plates to avoid cell–cell contact with hUC-MSCs at ratio 1:1 and stimulated with IL-23 or IL-12 separately. The results are presented as means ± SD of three separate experiments (*p < 0.05; **p < 0.01, t-test and Bonferroni test).
To further examine the suppressive effects of hUC-MSCs on Th17 as well as Th1 cells, LPMCs from normal healthy mice were isolated and cultured alone or together with hUC-MSCs under different stimulation conditions. As expected, large amounts of IFN-γ (176.36 ± 9.14 pg/ml) were produced when LPMCs were stimulated with the Th1 polarizing cytokine IL-12 and cultured alone (Fig. 4C). When LPMCs were stimulated with IL-23, they produced large amounts of IL-17 (204.867 ± 21.8 pg/ml) when cultured alone (Fig. 4 D). However, both IL-12-stimulated production of IFN-γ and IL-23-stimulated production of IL-17 were significantly suppressed by coculturing LPMCs with hUC-MSCs at ratio 1:1, and the suppression was hUC-MSC specific, since human umbilical cord vein endothelia cells had no such effects (Fig. 4C, D). Similarly, the IL-17 expression by IL-23-stimulated splenocytes was also inhibited by hUC-MSCs in a dose-dependent manner (data not shown). Moreover, when coculture experiments were conducted using the transwell system to separate LPMCs from hUC-MSCs by a semipermeable membrane, the inhibition on the expression of either IFN-γ or IL-17 was not significantly affected (Fig. 4C, D), indicating that the effects were largely mediated by soluble factor(s) rather than by direct cell contact.
Collectively, the in vitro experimental data described above were consistent with the findings in the in vivo studies and confirmed the suppressive activity of hUC-MSCs on Th1 cells. More importantly, our results also demonstrated that hUC-MSCs could also potently suppress IL-23-stimulated IL-17 expression. Thus, hUC-MSCs may ameliorate TNBS-induced colitis by downregulation of Th1 type response and by suppression of IL-23/IL-17-mediated colon inflammation.
Homing and Survival of hUC-MSCs
To study whether hUC-MSCs could be attracted to the TNBS-treated inflammatory colonic tissues and suppress local inflammation, CM-DiI-labeled hUC-MSCs were administered intravenously on the day colitis was induced. The distribution and survival of these cells were then assessed at different time points by using an IVIS 200 Series Imaging System, which allows noninvasive and sensitive cell tracking in live animals (9). Healthy mice were also administered with labeled hUC-MSCs and served as controls. The cells in the colitic mice were initially found largely in the lung and then migrated to and accumulated in the abdominal part and remained there for more than 2 weeks (Fig. 5A). In contrast, cells injected into healthy mice did not accumulate in the abdomen but rather distributed in a dispersed manner (Fig. 5B). The findings in the whole body imaging experiments were further confirmed by fluorescent microscopic studies of colonic tissues, as hUC-MSCs were detected in the colon slices from the colitic mice, but not in the colons of normal mice infused with hUC-MSCs (Fig. 6). These studies thus indicated that, even in the xenotransplantation setting, hUC-MSCs are poorly immunogenic and could home efficiently to and survive in the inflamed colon where they would directly suppress inflammatory and immune responses.

In vivo imaging of hUC-MSC distribution in colonic mice. Cells (1 × 106) were labeled with CM-DiI and injected intravenously into mice on the day colitis was induced and the labeled cells tracked using an IVIS 200 series imaging system. (A) Distribution of hUC-MSCs in the colitis mice from day 1 to 15. (B) Difference in the distribution and localization of hUC-MSCs in the mice with or without colitis on day 10 (ventral and dorsal views). The scale to the right of the images describes the color map for the photon count. (C) Fluorescence intensity of hUC-MSC at the abdominal part of three colitic mice.
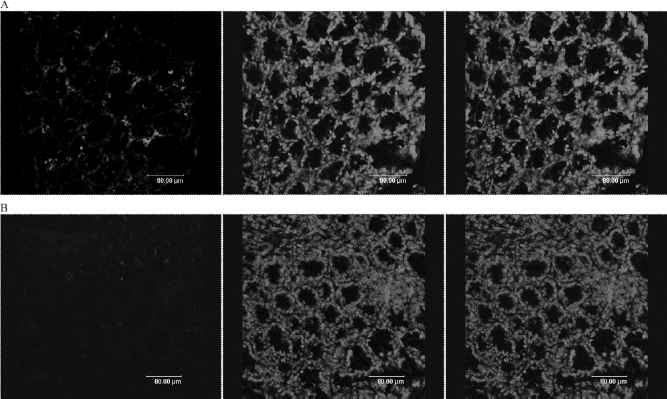
Representative fluorescence microscopic colonic slice obtained on day 5 of (A) TNBS-induced mice infused with MSC and (B) with PBS. Left, the hUC-MSC cells staining with a mouse anti-human nuclei monoclonal antibody and an anti-mouse FITC conjugated secondary antibody; middle, nuclear staining with DAPI; right, merger of the left and the middle images (original magnification in pictures 400×).
Discussion
Because MSCs possess potent immunomodulatory properties, MSC-based therapy is being explored as a novel approach for autoimmune diseases in animal models (14,43) and for acute steroid-resistant graft versus host disease (GvHD) following allogeneic stem cell transplantation in patients (28). In the present study, we investigated whether hUC-MSCs could be used to treat TNBS-induced acute colitis, a T-cell-mediated mouse model for CD. We demonstrated that systemic administration of hUC-MSCs was able to effectively attenuate the clinical and pathological severity of TNBS-induced colitis by several disease parameters, including body weight loss and infiltration of inflammatory cells.
CD4+ Th1 cells and the inflammatory cytokines they express have long been considered as important mediators of CD (36), but more recent studies have provided evidence that the newly discovered Th17 cells are closely involved in the pathogenesis of the disease (22,26). We analyzed the effects of hUC-MSC infusion on the expression of several important cytokines in colon tissues of colitic mice. We found that TNBS treatment induced expression of high levels of IFN-γ, a typical Th1 cytokine, and IL-6 in colitic colon tissues, which have been characteristically associated with this colitis model (14,35). However, this increase was dramatically inhibited by hUC-MSC infusion. Moreover, the in vitro coculture studies showed that the expression of IFN-γ induced by IL-12-stimulated normal LPMCs was also significantly inhibited by hUC-MSCs. MSCs are known to suppress IFN-γ production by Th1 cells in vitro (1); our data are consistent with these studies and indicate that the suppression of Th1 type response contributes to the therapeutic effects of hUC-MSCs. Similarly, a recently published study has demonstrated that human adipose tissue-derived MSCs can also alleviate TNBS-induced colitis through downregulation of Th1-driven inflammatory responses (14).
An important finding in this study is that TNBS treatment induces expression of high levels of the proinflammatory cytokines IL-23 and IL-17, but the increased expression was significantly reversed by hUC-MSC infusion. Furthermore, we demonstrated that administration of hUC-MSCs had a significant suppressive effect on the expression of RORγt, a key regulator of Th17 cell differentiation and function (17). These data strongly suggest that hUC-MSCs may also have the capability of suppressing Th17 cell response and IL-17 expression. We directly tested this possibility by in vitro coculture experiments and showed that the expression of IL-17 by splenocytes or LPMCs isolated from colitic mice or by IL-23-stimulated normal spleen cells or LPMCs was inhibited by hUC-MSCs. Our data thus demonstrated suppressive activities of hUC-MSCs on Th17 as well as Th1 cells. TGF-β and IL-10 may be involved in the pathogenesis of colitis; however, hUC-MSCs have no effect on TGF-β and IL-10 according to our data.
Accumulating bodies of evidence have identified IL-23 as a pivotal player in the pathogenesis of IBD and other autoimmune diseases (33). In human studies, elevated levels of IL-23 are found in CD patients (13), and genome-wide genetic analysis demonstrates that IL23R variants are closely associated with CD (8). Moreover, genetic deletion or antibody neutralization of IL-23 effectively prevent development of colitis and attenuate active disease in animal models (10,25). IL-23 is primarily produced by activated antigen-presenting cells such as dendritic cells and macrophages (31), and one of its key targets is Th17 cells. IL-23 expands and maintains differentiated Th17 cells (26). Activated Th17 cells characteristically produce large amounts of IL-17, and to a lesser extent, TNF-α and IL-6 (26). IL-17 is a key element in the inflammatory cascade that leads to intestinal inflammation. IL-17 enhances T-cell priming and stimulates the expression of proinflammatory cytokines (such as IL-1, IL-6, and TNF-α), chemokines [such as chemokine ligand 1 (CXCL1), monocyte chemotactic protein-1 (MCP-1), and macrophage inflammatory protein-2 (MIP-2)], and matrix metalloproteases that are involved in mobilizing neutrophils and in driving chronic inflammation (23,44). hUC-MSCs significantly suppressed the expression of IL-23 and IL-17 in vivo, and this inhibitory activity was further attested by in vitro coculture experiments. Thus, in addition to the known suppressive effects on Th1-type immune responses, another possible mechanism of the hUC-MSC therapy is the attenuation of the IL-23/Th-17 axis-orchestrated intestinal inflammation and immune destruction.
Gonzalez-Rey et al. (15) have shown recently that human adipose tissue-derived MSCs also can protect against the dextran sulfate sodium (DSS)-induced experimental colitis by inhibiting Th1-driven inflammatory responses and by upregulating CD4+CD25+FoxP3+ Treg cells, indicating that the therapeutic effects are not specific to the TNBS-induced colitis. It will be interesting to investigate whether downregulation of the Th17-type responses by MSCs plays a role in the DSS-induced model as well. Also, the present study demonstrated that hUC-MSCs could inhibit the acute inflammatory process, but the effect on chronic inflammatory process is still unknown and should be investigated in appropriate models in future studies.
Bone marrow MSCs appear to distribute to a wide range of tissues and organs in a random fashion when transplanted into unconditioned rodents or nonhuman primates (5,37), whereas they specifically home to inflamed or injured tissues in response to the signals upregulated by tissue damage (11). Similar to bone marrow MSCs, hUC-MSCs express a variety of cytokines, chemokines, chemokine receptors, and adhesion molecules, including stromal cell derived factor-1 (SDF-1; CXCL12) and its receptor CXCR4, CD11a and its ligand CD54 [inter-cellular adhesion molecule 1 (ICAM-1)], CD49d and its ligand CD106 [vascular cell adhesion protein-1 (VCAM-1)] [(30) and unpublished results]. We found in the present study that systemically administered hUC-MSCs migrated to the inflamed colitic colon and remained there for several weeks, indicating that hUC-MSCs were responsive to the cues sent by the injured colon tissues and well tolerated even in the xenotransplantation setting. This is not unexpected as hUC-MSCs express constitutively HLA class I but not HLA class II and costimulatory molecules CD80, CD86, and CD40 (41), and a single IV infusion is not sufficient to induce an immune response, consistent with previous xenotransplantation studies (2,39). Indeed, the best therapeutic effects will probably be achieved by the ability of systemically infused hUC-MSCs to migrate to the colitic colon, where they can more efficiently control the excessive inflammatory and immune reactions by direct cell–cell contact and by delivering soluble factors in situ.
In summary, we showed in the current study that systemically infused hUC-MSCs homed to the TNBS-induced inflammatory colon and effectively attenuated colitis. hUC-MSCs not only could suppress the expression of Th1-type cytokine IFN-γ in colitic tissues, but also significantly inhibit the production of IL-23 and IL-17, which are now identified as playing an essential role in IBD. MSCs also have been reported to repair injured intestinal mucosa in Crohn's disease because they are capable of differentiating into interstitial or stromal lineage cells (21). The potent immunomodulatory properties and the potential in tissue repair and regeneration (19) make MSCs an attractive cell therapy candidate for IBD, and promising results from recent clinical trials suggest that MSC transplantation may serve as an effective treatment for patients who have failed to respond to the conventional therapies (7,27). Considering that hUC-MSCs can readily be isolated with no harm to donors and expanded rapidly in large quantities, they provide an excellent choice for future clinical applications.
Footnotes
Acknowledgments
This study was supported by 863 projects from Ministry Science & Technology of China (2011AA0201018), National Natural Science Foundation of China (30900557), and National Basic Research Program of China (2011CB964800). The authors declare no conflicts of interest.
