Abstract
Hepatocyte transplantation utilizing induced pluripotent stem cells (iPSCs) or embryonic stem cells (ESCs) has been expected to provide an alternative to liver transplantation. However, it remains uncertain precisely which cell type is the best suited for cell transplantation. In particular, it is unclear whether mature hepatocytes, which have sufficient liver function, or immature hepatic progenitor cells, which have a higher proliferative capacity, will provide a better outcome. The main objective of this study was to investigate the therapeutic efficacy of the transplantation of hepatocytes at various differentiation stages. We utilized transgenic mice that expressed diphtheria toxin (DT) receptors under the control of an albumin enhancer/promoter. ESC-derived endodermal cells, fetal hepatocytes, and adult hepatocytes were transplanted into these mice with experimentally induced lethal acute liver injury caused by DT administration. The transplanted cells were marked by enhanced green fluorescent protein. We evaluated their effects on survival. At 35 days after transplantation, the survival rate of the adult hepatocyte-transplanted group (8/20, 40%) was significantly improved in comparison to that of the sham-operated group (2/25, 8%), the fetal hepatocyte-transplanted group (1/20, 5%), and the ESC-derived endodermal cell-transplanted group (0/21, 0%). The adult hepatocytes proliferated in the recipient livers and replaced a large part of their parenchyma. The transplantation of adult hepatocytes for acute liver failure significantly improved the survival rate in comparison to that of transplantation of immature cells, thus suggesting that ESCs and iPSCs should be differentiated into mature hepatocytes before cell transplantation for acute liver failure.
Keywords
Introduction
Although orthotopic liver transplantation has been one of the most effective therapies for lethal liver diseases, alternative therapies, including cell transplantation, are anticipated because of a chronic donor shortage (7). Some clinical trials have demonstrated that human hepatocyte transplantation was useful for improving liver function and that transplantation of hepatocytes was sufficient to extend the waiting time for liver transplantation (33). These trials, however, still do not appear to resolve the problem of donor shortage, because adult human hepatocytes are quiescent cells, and are difficult to maintain and expand in culture. On the other hand, hepatic progenitor cells (HPCs) or embryonic stem cells (ESCs) have the capacity to proliferate in vitro.
We previously reported that transplantation of fetal HPC-enriched liver cells and ESC-derived endodermal cells could improve the survival rate of lethal liver failure in experimental mice (18,25). At present, cell transplantation using induced pluripotent stem cell (iPSC)- or ESC-derived hepatocytes has been anticipated as a method to resolve the donor shortage. However, it remains unclear which cell type is best suited for cell transplantation. In particular, it is unclear whether mature hepatocytes, which have sufficient liver functions, or immature HPCs, which have a high proliferative capacity, would provide a better outcome. It also remains unclear to what extend iPSCs and ESCs should be differentiated into the hepatic lineage for effective cell transplantation.
Toxin receptor-mediated conditional cell knockout (TRECK) mice were generated previously as an animal model of acute hepatic failure (30). These mice express the human heparin-binding epidermal growth factor-like growth factor (hHB-EGF) precursor, which functions as the diphtheria toxin (DT) receptor, under the control of the albumin enhancer/promoter. Although wild-type mice, which lack the hHB-EGF precursor, are insensitive to DT, the hepatocytes of the transgenic mice are selectively damaged by the administration of DT. The administration of high doses of DT induces lethal liver injury in these mice.
In this study, we transplanted three cell types with different differentiation stages into the transgenic mice with induced lethal liver injury: ESC-derived endodermal cells, fetal hepatocytes, and adult hepatocytes. Our previous study showed that ESC-derived endodermal cells were thought to be the most immature, and that adult hepatocytes were the most mature cells among these three cell types (18,25). We evaluated their effect on mouse survival to determine which cell type is the best suited for cell transplantation for treating acute liver failure.
Materials and Methods
Animals
Alb-TR1-2(+/+)/C57BL6 TRECK mice, which were homozygous for the albumin enhancer/promoter-driven DT receptor alleles, were generated as described previously (30). The green fluorescent protein (GFP) transgenic/ C57BL6 mice, which expressed GFP under the cytomegalovirus enhancer-β-actin promoter and globulin-poly-A-tail (CAG) promoter (29), were purchased from CLEA Japan, Inc. Animals were housed at Kyoto University, and all animal experimental procedures were performed according to the Animal Protection Guidelines of Kyoto University.
Isolation of GFP-Positive Fetal Hepatocytes for Cell Transplantation
Fetal hepatocytes were obtained from E13.5 fetal livers of GFP transgenic mice and were enriched through formation of cell aggregates as described previously (25,39). Briefly, after the digestion of E13.5 fetal liver tissues by 0.5% collagenase medium, fetal liver cells were subjected to floating culture to form cell aggregates on Petri dishes in RPMI 1640 medium (Sigma-Aldrich Co., Ltd., St. Louis, MO) supplemented with 10% fetal calf serum (FCS; ICN, Aurora, OH), penicillin G (Meiji Seika, Tokyo, Japan), streptomycin (Meiji Seika), insulin (Gibco, Grand Island, NY), and hepatocyte growth factor (R&D Systems Co., Ltd., Minneapolis, MN). The cell aggregates were selected by gravity sedimentation and were inoculated on type I collagen-coated culture plates (ASAHI Glass Co., Ltd., Chiba, Japan). After 24 h of incubation, the aggregates adhered to the plates and extended as monolayer colonies. After removing the hematopoietic cells by washing the aggregates twice with phosphate-buffered saline (PBS), the adherent cells were incubated with a 0.25% trypsin-ethylenediaminetetraacetic acid (EDTA) solution (Sigma-Aldrich) for 3 min. The dissociated cells were suspended in a Ca2+-free Hank's balanced salt solution containing 0.5 mM ethyleneglycol-bis-(β-aminoethylether)-N,N,N,N-tetraacetic acid (HBSS-EGTA) at a density of 5×106 cells/ml.
Isolation of GFP-Positive Adult Hepatocytes for Cell Transplantation
Adult hepatocytes were isolated by a modified two-step collagenase perfusion technique as described previously by our laboratory (2). Briefly, after laparotomy of anesthetized 8- to 12-week-old GFP transgenic mice, their portal veins were cannulated with 25-gauge needles. Through these needles, the livers were preperfused with 20 ml of 37°C HBSS-EGTA. Subsequently, the livers were perfused with 30 ml of 37°C collagenase solution containing 0.3% dispase II (Sanko Junyaku Co., Ltd., Tokyo, Japan), 0.3% collagenase type II (Gibco), 150 mmol/L NaCl, 5.4 mmol/L KCl, 0.34 mmol/L NaHPO4, 0.1 mmol/L MgSO4, 5.0 mmol/L CaCl2, 4.2 mmol/L NaHCO3, 5.6 mmol/L glucose, 10 mmol/L HEPES, 0.01% deoxyribonuclease, and 0.005% trypsin inhibiter (all of the chemical reagents described above were purchased from Wako Pure Chemical Industries, Ltd., Osaka, Japan). Then, the livers were excised, minced with scissors, and incubated at 37°C for an additional 20 min in the collagenase solution. After filtering thorough 50 N polypropylene mesh, the suspension was centrifuged three times at 50 x g for 4 min. Cells from the pellets were plated onto 35 mm type I collagen-coated dishes (Asahi glass) at a density of 1 × 104 cells/ml. The culture medium consisted of RPMI 1640 medium (Sigma-Aldrich) containing 10% FCS and penicillin G/ streptomycin. After 24 h of incubation, the adherent cells were washed twice with PBS and incubated with a 0.25% trypsin-EDTA solution for 5 min. The dissociated cells were counted and suspended in HBSS-EGTA at a density of 5×106 cells/ml.
Differentiation of ESCs Into Endodermal Cells
We obtained ESC-derived α-fetoprotein (AFP)-producing endodermal cells as described previously (16,18). Briefly, ESCs that expressed the hybrid gene of enhanced GFP (EGFP) and a hygromycin resistance cassette under the control of AFP promoter were differentiated into endodermal cells by sequential administration of all-trans retinoic acid, basic fibroblast growth factor, and hepatocyte growth factor. The AFP-producing cells were selected by 3 days of growth with hygromycin. These procedures enabled us to obtain the AFP-producing endodermal cells with approximately 90% purity. In addition, since the LacZ gene was introduced into the transgenic ESCs, they could be detected in the recipient livers by β-galactosidase (β-gal) immunostaining after transplantation. After the 10-day differentiation protocol, the differentiated cells were washed twice with PBS and incubated with a 0.25% trypsin-EDTA solution for 3 min. The dissociated cells were counted and suspended in a HBSS-EGTA at a density of 5 × 106 cells/ml.
Cell Transplantation and Diphtheria Toxin Administration
Male 8- to 12-week-old Alb-TR1-2(+/+) transgenic mice were used as recipients for cell transplantation. Under general anesthesia, we injected 0.2 ml of a cell suspension containing 1×106 cells per mouse via the inferior splenic pole using 29-gauge needles. In order to prevent bleeding and cell suspension leakage, the injection sites were ligated.
For the survival analysis, we administered DT intraperitoneally twice a week from the day of the cell transplantation for 4 weeks. The DT was purified as described previously (30). A total of 40 ng/kg DT was administered in each injection, except for the last administration (day 28), when a lethal dose of 10,000 ng/kg of DT was administered.
Survival Analysis
Eighty-six Alb-TR1-2(+/+) transgenic mice were divided into four groups: the first group (n = 20) received the adult hepatocyte transplantation (adult hepatocyte transplanted group), the second group (n = 20) received the fetal liver cell transplantation (fetal liver cell transplanted group), the third group (n = 21) received the ESC-derived endodermal cell transplantation (ESC-derived endodermal cell transplanted group), and the fourth group (n = 25) was injected with 0.2 ml HBSS-EGTA (sham-operated group). Liver injury was induced by administration of DT as mentioned above. The observation period was 35 days. Kaplan–Meier analysis with the log-rank test was used for the survival analysis, and values of p < 0.05 were considered to be statistically significant.
Histological and Immunohistological Analyses
The harvested livers were fixed with 4% paraformaldehyde at 4°C overnight. After incubation in 30% sucrose solution in PBS, the livers were embedded in optimum cutting temperature compound (Sakura Finetechnical Co., Ltd., Tokyo, Japan), frozen in liquid nitrogen. Then the samples were sectioned at a 5-μm thickness using a Cryostat (Leica, Nussloch, Germany). After antigen retrieval using a Target Retrieval Solution (Dako, Glostrup, Denmark), nonspecific binding was blocked with 1.4% bovine serum albumin (Sigma-Aldrich) dissolved in 0.1% saponin (Wako Pure Chemical Industries) in PBS. The sections were incubated with primary antibodies as follows; rabbit polyclonal anti-GFP antibody (Molecular Probes, Inc., Eugene, OR), goat polyclonal anti-mouse albumin antibody (Bethyl Laboratories, Inc., Montgomery, TX), goat polyclonal anti-cytokeratin19 (CK19) antibody (Santa Cruz Biotechnology, Inc., Santa Cruz, CA), rat anti-mouse CD34 antibody (BD Biosciences, Franklin Lakes, NJ), mouse monoclonal anti-β-gal antibody (Promega, Madison, WI), and goat polyclonal anti-mouse proliferating cell nuclear antigen (PCNA) antibody (Santa Cruz Biotechnology) at 4°C overnight. Each primary antibody was diluted at 1:100.
After washing, the stained sections were incubated with either Alexa 488-conjugated donkey anti-rabbit IgG, Alexa 488-conjugated goat anti-mouse IgG, Alexa 555-conjugated donkey anti-goat IgG, or Alexa 555-conjugated goat anti-rat IgG for 2 h at room temperature. Each secondary antibody was diluted at 1:500. After washing, the stained sections were covered with Vectashield mounting medium with 4,6-diamidino-2-phenylindole (Vector Laboratories, Burlingame, CA). Hematoxylin–eosin (H&E) staining was then performed according to a standard protocol.
Analyses of the Proportion of Transplanted Cells in the Recipient Livers
We harvested the transplanted livers on days 1, 2, 7 and 14 from the adult hepatocyte-transplanted, fetal hepatocyte-transplanted, and the ESC-derived endodermal cell-transplanted groups in order to analyze the proportion of transplanted cells in the livers. Three livers were harvested on each of these days from the three groups. GFP immunofluorescence staining was performed in the adult hepatocyte-transplanted and fetal hepatocyte-transplanted groups. β-gal immunofluorescence staining was performed in the ESC-derived endodermal cell-transplanted group. The GFP-positive cells and β-gal-positive cells were counted in 10 high-power fields. All microscopic analyses of the tissue sections were performed using a fluorescence microscope BZ-9000 (Keyence Co., Ltd., Osaka, Japan), and the number of GFP-positive cells and β-gal-positive cells in the transplanted livers was counted using the cell count software program BZ-H1C (Keyence Co.). Statistical significance between these groups was evaluated by Student's t test with Bonferroni correction. A value of p < 0.017 was considered to be significant.
Quantitative Polymerase Chain Reaction
Total RNA was extracted from adult hepatocytes of GFP transgenic mice and fetal hepatocytes of E13.5 fetuses using an RNeasy Mini Kit (Qiagen, Chatsworth, CA) and treated with RNase-free DNase (Qiagen). Total RNA (1 μg) was reverse-transcribed into cDNA with the oligo(dT) 12–18 primer (Invitrogen) using an Omniscript reverse transcription (RT) kit (Qiagen). Quantitative polymerase chain reaction was performed using the TaqMan Universal PCR Master Mix and Assays-on-Demand Gene Expression Assay Mix with specific primers for matrix metalloproteinase-2 (MMP-2), MMP-3, MMP-9, MMP-12, E-cadherin, N-cadherin, and glyceraldehyde-3-phosphate dehydrogenase (GAPDH) (Applied Biosystems, Foster City, CA). The quantification of the given genes was expressed as the relative mRNA level compared after normalization to GAPDH.
Analyses of the Role of E-Cadherin in the Engraftment of Transplanted Hepatocytes
In order to analyze the role of E-cadherin in the engraftment of transplanted hepatocytes, adult hepatocytes with rat monoclonal E-cadherin neutralizing antibodies (Sigma) were transplanted into the same acute liver failure model mice. Thereafter, we compared the proportion of transplanted hepatocytes between the adult hepatocyte-transplanted group and the adult hepatocyte with E-cadherin neutralizing antibodies-transplanted group. Although a cell suspension containing 1 × 106 hepatocytes per mouse was transplanted into mice of both groups, 50 μg E-cadherin neutralizing antibodies per mouse were added to the suspension just before the transplantation of the adult hepatocytes with E-cadherin neutralizing antibodies. The transplanted livers were harvested on days 1 and 7. Five livers were harvested on each of these days from both groups. GFP immunofluorescence staining was performed, and the GFP-positive cells were counted in 10 high power fields. The methods used for cell counting was the same as that described above. Statistical significance between these groups was evaluated by Student's t test. A value of p < 0.05 was considered to be significant.
Results
The Differences in Engraftment Among Adult Hepatocytes, Fetal Liver Cells, and ESC-Derived Endodermal Cells
To compare the ability of the different cells to engraft and proliferate in the recipient livers, we harvested the livers that were transplanted with cells on days 1, 2, 7, and 14 after transplantation from the three groups (Fig. 1). Immunostaining of these samples on days 1 and 2 revealed that the donor cells of the three groups were trapped in the whole recipient livers and formed small clusters (Fig. 1A, B, E, F, I, J). The number of the donor adult hepatocytes was approximately equal to that of the donor fetal hepatocytes and the donor ESC-derived endodermal cells (Fig. 1M). On days 7 and 14, the clusters of the transplanted adult hepatocytes had become larger, whereas the clusters of the fetal hepatocytes and the ESC-derived endodermal cells did not grow (Fig. 1C, D, G, H, K, L, M). Therefore, these results suggest that adult hepatocytes had an advantage in terms of a better proliferative capacity in the recipient livers.

Green fluorescent protein (GFP) and β-galactosidase (β-gal) immunofluorescent images of the liver sections of adult hepatocyte-transplanted mice, fetal hepatocyte-transplanted mice, and embryonic stem cell (ESC)-derived endodermal cell-transplanted mice on day 1, day 2, day 7, and day 14. Top: Adult hepatocyte-transplanted livers (A–D). Middle: Fetal hepatocyte-transplanted livers (E–H). Bottom: ESC-derived endodermal cell-transplanted livers (I–L). Green fluorescence represents GFP (top and middle) and β-gal (bottom). The arrowheads indicate the transplanted cell cluster (A–L). Scale bars: 100 μm. The proportion of GFP-positive cells in the liver sections of adult hepatocyte-transplanted mice and fetal hepatocyte-transplanted mice and the proportion of β-gal-positive cells in the liver sections of ESC-derived endodermal cell-transplanted mice on days 1, 2, 7, and 14 (M). ∗p = 0.016. ∗∗p = 0.003. The data are expressed as the means ± standard deviation (n = 3).
Engraftment and Proliferation of Transplanted Adult Hepatocytes
For further examination of the behaviors of the transplanted adult hepatocytes in the recipient livers, the livers of the adult hepatocyte-transplanted group were harvested periodically (Fig. 2A–L). On day 1, the transplanted cells formed infarct regions on the surface of the recipient liver (Fig. 2A). Fluorescence microscopy revealed that they formed small clusters that had a few donor cells, obstructing small vessels on day 7 (Fig. 2B, F, J). Moreover, on the livers of the adult hepatocyte-transplanted mice on day 21, the GFP-positive clusters had grown large enough to be visible macroscopically under a fluorescent illuminator (Fig. 2C, G, K). On day 35, after the administration of 10,000 ng/kg DT, almost all hepatic lobules of the recipient livers were positive for GFP. Microscopic examinations showed that the GFP-positive transplanted hepatocytes proliferated extensively, and a large part of recipient liver tissue was replaced by donor cells (Fig. 2D, H, L). These findings indicated that the transplanted adult hepatocytes continued to proliferate in the injured livers. On the other hand, HE staining showed hemorrhage, necrosis, and the disruption of liver lobules in the sham-operated group (data not shown).

The time course analyses of mature hepatocyte-transplanted mouse livers that were harvested on day 1, day 7, day 21, and day 35. First row: Macroscopic images of the harvested livers (A–D). Second row: Macroscopic GFP fluorescent images of the harvested livers (E–H). Third row: GFP immunofluorescent staining of the liver sections (I–L). Fourth row: The immunohistological analyses of adult hepatocyte-transplanted mouse livers on day 35. Green fluorescence represents GFP, and blue fluorescence represents DAPI (M–P). Red fluorescence indicates albumin (M), cytokeratin 19 (CK19) (N), cluster of differentiation 34 (CD34) (O), and proliferating cell nuclear antigen (PCNA) (P). The arrowheads indicate the pink PCNA-positive nuclei. Scale bars: 200 μm (A–H) and 50 μm (I–P).
The Characteristics of Transplanted Adult Hepatocytes in the Recipient Liver
To evaluate the characteristics of the engrafted GFP-positive adult hepatocytes, we performed immunohistochemical assays of the adult hepatocyte-transplanted livers on day 35 (Fig. 2M–P). All of the GFP-positive donor cells were positive for albumin (Fig. 2M). However, these cells were negative for CK19 and CD34 (Fig. 2N, O). These findings showed that the engrafted adult hepatocytes did not differentiate into either bile duct epithelial cells or vascular endothelium. Furthermore, 13.9% of the donor cells were stained with PCNA, indicating that the engrafted adult hepatocytes maintained the ability to proliferate in the recipient livers (Fig. 2P).
Survival Analysis
At 35 days after transplantation, the survival rate of adult hepatocyte-transplanted mice (8/20, 40%) was significantly improved (p = 0.0044) in comparison to that of the sham-operated (2/25, 8%), the fetal hepatocyte-transplanted (1/20, 5%), and the ESC-derived endodermal cell-transplanted (0/21, 0%) mice (Fig. 3). There were no significant differences between the other three groups. This result indicates that adult hepatocyte transplantation provided a greater therapeutic effect on acute liver damage.
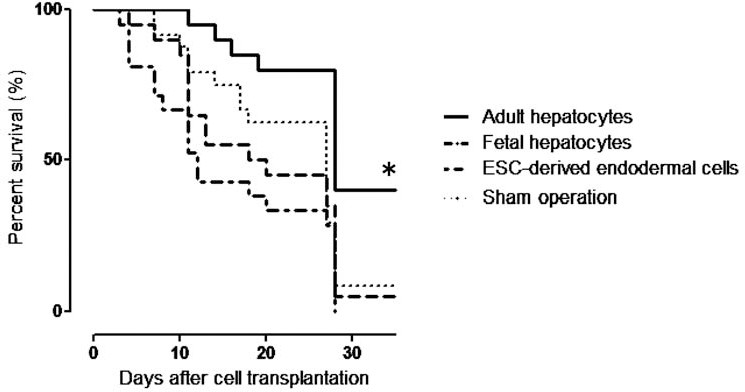
The survival analyses. At 35 days after transplantation, the survival rate of the mature hepatocyte-transplanted group (8/20, 40%) was significantly (∗p = 0.0044) improved in comparison to that of the sham-operated group (2/25, 8%), fetal hepatocyte-transplanted group (1/20, 5%), and ESC-derived endodermal cell transplanted group (0/21, 0%).
Long-Term Survival of Adult Hepatocytes
In order to investigate the long-term survival of adult hepatocytes after withdrawing DT administration, the recipient livers were harvested 3, 6, and 12 months after cell transplantation. Although GFP-positive donor cells could still be detected until 12 months, the number of donor cells had gradually decreased (Fig. 4A–I). PCNA immunofluorescent staining revealed that there were some PCNA-positive donor cells in the recipient livers at 3 months after transplantation (0.57%) (Fig. 4J). However, at 6 and 12 months after transplantation, PCNA-positive donor cells could not be detected (Fig. 4K, L).

Long-term engraftment of adult hepatocytes. The number of transplanted adult hepatocytes gradually decreased, but some cells survived for at least 1 year without diphtheria toxin (DT) administration. PCNA-positive donor cells were detected until 3 months after transplantation. First row: macroscopic images of the harvested livers (A–C). Second row: GFP fluorescent images of the harvested livers (D–F). Third row: GFP immunofluorescent staining of the liver sections (G–I). Fourth row: GFP, PCNA, and DAPI immunofluorescent staining of the liver sections. Green, red, and blue fluorescence represent GFP, PCNA, and DAPI, respectively. The arrowheads indicate the pink PCNA-positive nuclei (J–L). Scale bars: 200 μm (A–F) and 100 μm (G–L).
Differences in Gene Expression Between Adult and Fetal Hepatocytes
To compare the adhesive ability and migration activity between adult hepatocytes and fetal hepatocytes, we performed relative quantitative analysis of the gene expression of MMP-2, MMP-3, MMP-9, MMP-12, N-cadherin, and E-cadherin using real-time PCR (Fig. 5). As a result of this investigation, the gene expression level of E-cadherin in adult hepatocytes was found to be significantly higher than that in fetal hepatocytes (Fig. 5A). Furthermore, the adult hepatocytes expressed a higher amount of MMP-3 mRNA than did the fetal hepatocytes (Fig. 5D). In contrast, the fetal hepatocytes had a tendency to have higher expression levels of MMP-2, MMP-9, and MMP-12 mRNAs than the adult hepatocytes (Fig. 5C, E, F).

The differences in gene expression between adult and fetal hepatocytes measured by quantitative PCR: (A) E-cadherin, (B) N-cadherin, (C) matrix metalloproteinase-2 (MMP-2), (D) MMP-3, (E): MMP-9, and (F): MMP-12. The relative ratios of the values to adult hepatocytes are presented. Each mRNA level was normalized to the glyceraldehyde 3-phosphate dehydrogenase (GAPDH) level. The data are expressed as the means ± standard deviation (n = 3). ∗p = 0.003. The proportion of GFP-positive cells in the liver sections of adult hepatocyte-transplanted mice and adult hepatocyte with E-cadherin neutralizing antibodies-transplanted mice on days 1 and 7 (G). The data are expressed as the means ± standard deviation (n = 5).
The Role of E-Cadherin in the Engraftment of Transplanted Hepatocytes
To elucidate the role of E-cadherin in the engraftment of transplanted hepatocytes, we compared the proportion of transplanted hepatocytes between the adult hepatocyte-transplanted group and the adult hepatocyte with E-cadherin neutralizing antibodies-transplanted group on days 1 and 7 (Fig. 5G). As a result, the adult hepatocytes with E-cadherin neutralizing antibodies tended to demonstrate a lower implantation capacity in the recipient livers than did the adult hepatocytes.
Discussion
Although hepatocyte transplantation had been considered to be a promising alternative to liver transplantation for severe liver diseases, the problem of donor shortage has not been resolved. Recently, the application of ESCs and iPSCs has provided a possible mechanism for resolving this problem, and transplantation using ESC- or iPSC-derived hepatocytes is anticipated to abrogate the need for so many donors. Delgado et al. showed that human hepatoblasts engraft more effectively during cell transplantation than do adult hepatocytes in mouse livers (10). In contrast, Haridass et al. reported that adult human and mouse hepatocytes had a higher repopulation capacity than fetal liver cells or ESC-derived cells did when they were transplanted into immunodeficient albumin-urokinase (AlbuPA) transgenic mice (15). Therefore, it remains unclear to what extent ESCs or iPSCs should be differentiated into the hepatic linage for efficient cell transplantation. In this study, we transplanted three types of cells at various differentiation stages into mice with experimentally induced lethal acute liver injury in order to examine the effects of cell differentiation on cell transplantation.
Adult hepatocytes maintained their ability to proliferate in vivo and efficiently repopulated recipient livers. Furthermore, the transplantation of adult hepatocytes improved the survival rate of severe acute liver failure model mice. On the other hand, the other immature cells did not repopulate livers or rescue mice from liver failure. This indicated that transplantation of mature hepatocytes was more appropriate for acute liver failure than transplantation of immature cells. Therefore, our results suggest that ESCs and iPSCs should be differentiated into mature hepatocytes in vitro before cell transplantation in order to achieve better therapeutic effects on cell transplantation.
Previously, our laboratory reported that transplantation of fetal hepatocytes (25) and ESC-derived endodermal cells (18) into the TRECK mice with acute, lethal liver failure improved their survival. However, in this study, transplantation of the fetal hepatocytes and the ESC-derived endodermal cells could not improve the survival of the mice, although we used the same Alb-TR1-2(+/+)/ C57BL6 TRECK mice and the same donor cells. In this study, the TRECK mice were more sensitive to DT than they had been previously. Therefore, in order to control the mortality, the DT doses were decreased in this study, and the protocol of DT administration had to be changed. The major difference between this study and previous studies was the generation of the TRECK mice. This could explain why the sensitivity to DT was changed. Therefore, it was supposed that there was not enough time and stimulation to promote the proliferation in the livers to allow immature cells to improve the survival in this study.
It is considered that there might be several steps involved in the repopulation of recipient livers by cells transplanted via the portal vein system. First, the transplanted cells should be attached to small portal vessels mechanically by embolization. Second, the cells will adhere to recipient vessels and hepatocytes by adherent molecules. Third, they will infiltrate into the recipient liver parenchyma and then proliferate there (14,22,31). In this study, there were no differences between the number of engrafted cells after adult hepatocyte, fetal hepatocyte, and ESC-derived endodermal cell transplantation on days 1 and 2 after transplantation. However, the engrafted adult hepatocytes continued to proliferate, whereas fetal hepatocytes and ESC-derived endodermal cells did not. These findings suggest that adult hepatocytes have not only higher cellular functions but also a better ability to adhere, infiltrate into recipient parenchyma, and proliferate in vivo, which might lead to the better therapeutic effects observed following the adult hepatocyte transplantation.
In order to elucidate the mechanisms underlying the higher repopulation ability of the adult hepatocytes, the gene expression levels of some cadherins and MMPs were compared between adult and fetal hepatocytes. E-cadherin has been considered to be a key molecule that controls cell-to-cell adhesion and influences cell migration (10,23,28). On the other hand, MMPs have been thought to play an important role in extracellular matrix degradation during cell migration (21,38). Several studies suggested that the expression of MMPs was correlated with the invasive ability of hepatoblasts and hepatocellular carcinoma cells (1,10,27). However, adult hepatocytes of normal livers expressed only modest levels of MMPs, although hepatocyte transplantation activated hepatic stellate cells to upregulate MMPs expression in the recipient livers (6,10,37,40). In this study, quantitative PCR revealed that E-cadherin was significantly upregulated in adult hepatocytes, whereas the MMPs were not, in contrast to fetal hepatocytes. In addition, transplanted hepatocytes with E-cadherin neutralizing antibodies tended to have a lower repopulation capacity. Therefore, it was suggested that E-cadherin played a more important role in the engraftment of adult hepatocytes in the recipient livers and that this might lead to the higher therapeutic effect for transplantation of the adult hepatocytes.
A large number of donor cells were observed at 3 and 6 months after transplantation of adult hepatocytes. Even at 12 months after transplantation, a small number of donor cells were present. In addition, immunofluorescent staining of PCNA revealed that donor cells still had the ability to proliferate at 3 months after transplantation. Compared with the recipient livers on day 35, however, the number of engrafted donor cells decreased gradually at the later time points. At 12 months, the number of donor cells was so small that we could hardly detect the GFP-positive lobules macroscopically. In general, the lifespan of hepatocytes is considered to be 200–400 days (24). Therefore, it was speculated that the number of donor cells decreased due to cellular senescence in the absence of DT administration and that the remnant recipient hepatocytes proliferated and became predominant in the regenerated livers.
Recent years have seen substantial progress in re search on technologies that induce the differentiation of numerous cell types from ESCs and iPSCs (9,11,19,26,35,36). In the field of liver regenerative medicine, many laboratories have reported the induction of ESCs and iPSCs to differentiate into hepatocytes (3,4). Furthermore, several groups have reported the generation of hepatocyte-like cells from human ESCs (8), human iPSCs (32,34), and murine iPSCs (13). However, it remains difficult to differentiate ESCs and iPSCs into hepatocytes with cellular functions that are comparable to terminally differentiated hepatocytes (5). Nevertheless, the present study revealed that immature cells should be differentiated into mature hepatocytes in vitro in order to provide more effective cell transplantation for acute liver failure.
We previously reported that human ESC-derived AFP-producing cells could differentiate into hepatocyte-like cells that expressed higher hepatocyte functions using coculture with MLSgt20 cells, which were derived from mesenchymal cells residing in murine fetal livers and that this accelerated the hepatic maturation of both murine hepatic progenitor cells and murine ESCs (12,17,20). This method might contribute to sufficient hepatic maturation of ESCs or iPSCs for cell transplantation.
In conclusion, the transplantation of adult hepatocytes for acute liver failure significantly improved the survival rate of mice in comparison to transplantation of immature cells, suggesting that ESCs and iPSCs should be differentiated into mature hepatocytes for the purpose of effective cell transplantation to treat acute liver failure. It was also suggested that E-cadherin played an important role in the donor cell repopulation of recipient livers. Our study might contribute to the realization of effective hepatocyte transplantation utilizing ESCs and iPSCs in the near future. Further experiments will be required to determine a suitable cell source for treating other liver dysfunctions such as chronic liver failure, metabolic liver failure, and liver cirrhosis.
Footnotes
Acknowledgment
The authors declare no conflicts of interest.
