Abstract
The development of cell-based therapies opens up new avenues for treating a myriad of diseases of the central nervous system (CNS). While significant effort is being directed toward development of patient-specific, autologous transplantable cells, at present, the majority of cell transplantation studies performed clinically utilize allografts. In this context, the issue of graft rejection and immunoprotection is of key importance. In this study, we transplanted mouse glial-restricted progenitors into immunodeficient, immunocompetent, and immunosuppressed mice and monitored their survival noninvasively using bioluminescence imaging (BLI). With the use of serial BLI, we evaluated both the prevalence and dynamics of cell rejection. We demonstrate that allografts in immunocompetent mice were rejected at a rate of 69.2% (n = 13) indicating that graft tolerance is possible even without immunosuppression. Immunosuppression using a combination of rapamycin and FK506 or cyclosporin failed to fully protect the grafts. FK506 and rapamycin treatment resulted in a slight improvement of immunoprotection (22.2% rejected, n = 9) compared to cyclosporin A (55.6% rejected, n = 9); however, the difference was not significant. Notably, immunohistochemistry revealed leukocytes infiltrating the graft area in both rejecting and nonrejecting immunocompetent animals, but not in immunodeficient animals. The induction of an inflammatory process, even in surviving allografts, has implications for their long-term survival and functionality.
Keywords
Introduction
Stem cell-based therapy has tremendous potential to provide a cure for a wide range of disorders, including diseases of the central nervous system (CNS). A variety of mechanisms that mediate cell-dependent effects have been investigated (18). Despite the significant effort directed toward the development of patient-specific, autologous transplantable cells, at present, much of the clinical work is being carried out with allogeneic transplants (16,19,20,26). Rather disappointing outcomes from those studies may be attributable, in part, to a suboptimal control of the host immune response (17,34). Immune rejection of allografts has been repeatedly shown (6,38), while syngeneic transplantations do not seem to be subject to this process (5). Although the CNS is often described as an immunopriviliged site, it does not protect allogeneic stem cells, and it has now been recognized that modulation of the host response towards the graft is of critical importance (13).
Overall, studies on immunosuppression in neurotransplantation are limited, and there is no consensus regarding the optimal drug regimen (3). Cyclosporine A has been used most extensively and showed some beneficial effect, although the efficacy has not been directly compared to other drugs (2,31,40). In one study, a triple-drug scheme was shown to be more effective than cyclosporine A alone, but at a cost of significant adverse events (43). Among double-drug regimens, a combination of FK506 and rapamycin was shown to be the most effective for cells transplantated into the muscle (36). Recently, the combination of FK506 and rapamycin has also been successfully used as an immunosuppressive treatment for stem cell transplantation to the CNS without significant adverse events (42,46). The search for an appropriate immunosuppressive strategy is of critical importance to maximize the functional effects of cell therapy in neurological disorders.
A range of cell lineages have been employed in neurorestorative therapies, depending on disease characteristics (10). Myelin disorders require glial precursors capable of oligodendrocyte generation (4). In transgenic immunodeficient, myelin-deficient mice, robust graft survival and remyelination has been achieved (44). In addition, the role of remyelination has also recently been recognized as a viable approach for therapy of CNS diseases that are not primarily associated with selective white matter damage. Remyelination was proven to be useful for treatment in a rodent model of stroke (33) and has reached the level of clinical trials for spinal cord injury (35). Thus, we decided to employ glial progenitors for the evaluation of various immunosuppressive regimes.
Due to its simplicity, robustness, and high sensitivity, bioluminescence imaging (BLI) has been widely used for noninvasive serial monitoring of biological processes in small animals (32). This method is based on an ATP-dependent enzymatic reaction and as a consequence imaging signal specifically and acurately report on cell viability. It is widely employed in oncology (27), whole organ transplantation (10), biosensor production (30), infection (9), and stem cell transplantation (7). These advances encouraged us to use BLI for our neurotransplantation studies; when glial precursors constitutively express the luciferase enzyme, BLI can provide information on the time course of the location, survival, and growth of transplanted cells (25). With the use of BLI, we were able to demonstrate graft acceptance in some but not all immunocompetent recipients glial allografts, that the onset of graft rejection occurs between one and two weeks after transplantation, and if cells survive this critical period, they will not be rejected at later time points. Immunodeficient mice, such as the rag2-/- mutant, are invaluable in studying graft–host interactions. These animals accept allo- or even xenografts (42), which makes them important positive controls that enable the identification of cell or transplantation procedure-related failures.
Here, we report on the use of BLI as a tool to evaluate the efficacy of frequently used immunosuppressive regimens compared to positive (immunodeficient rag2-/- mice) and negative (immunocompetent, nonimmunosuppressed BALB/c mice) controls for allograft rejection. To this end, allogeneic mouse glial-restricted progenitor cells (GRPs) expressing firefly luciferase under a constitutive cytomegalovirus (CMV) promoter were transplanted into the corpus callosum of adult mice. The transplantation site was selected based on expected application of glial progenitors for treatment of demyelinating lesions. Serial BLI was performed over a period of 3 weeks to evaluate cell survival and the kinetics of immune rejection, and these results were compared with histopathology. To our knowledge, this is the first report to demonstrate, noninvasively, the dynamics of immune rejection of glial precursor cells grafted into the white matter. This study may provide a basis for future studies examining the nature of immune rejection in the CNS, as well as a real-time method for comparing immunosuppressive regimens.
Materials and Methods
Cell Preparation
Mouse GRPs were derived from FVB mouse spinal cord at E13.5. Cells were immortalized using lentivirus encoding the SV40 large T-antigen and selected with puromycin. Subsequently, cells were transduced with a lentivector encoding the firefly luciferase under a constitutive CMV promoter and clonally selected based on the intensity of BLI. Cells were maintained in serum-free Dulbecco's modified Eagle's medium (DMEM)-F12 supplemented with N2, B27, bovine serum albumin, and basic fibroblast growth factor (bFGF), as described elsewhere (22).
Animals
Adult female BALB/c (Jackson Labs) and rag2-/- (Taconic) mice (15–20 g) were used in these experiments. Animals were housed under an artificial light–dark (12/12 h) cycle and had access to food and water ad libitum. All experimental procedures were in accordance with the guidance provided in the Rodent Survival Surgery manual and were approved by our institutional animal care and use committee.
Cell Transplantation
Anesthesia was induced with 5% isofluorane gas and maintained throughout the surgical procedure with a mixed flow of 2% isofluorane and 98% oxygen delivered to at a rate of 2 L/min. For brain injections, animals were stabilized in a stereotactic frame. A 5.0-mm incision was made along the midline of the skull. The bregma was localized and a 1 mm2 burr hole was drilled 2 mm to the right of the bregma. Cells suspended in cell culture medium at a concentration of 1 × 105 cells/μl were loaded into a Hamilton syringe with an attached 31-gauge needle. The needle was lowered 1.2 mm ventral to the surface of the brain, and cells were injected into the corpus callosum. A total of 3 × 105 cells were injected over a period of 3 min, and the syringe was removed 1 min after the completion of cell delivery to minimize back flow.
Immunosuppresive Regimens
Animals were divided into four groups: (1) BALB/c mice receiving daily injections of saline (n = 13); (2) BALB/c mice immunosuppressed with a combination of FK506 and rapamycin (1 mg/kg each, n = 9); (3) mice treated with cyclosporine A (10 mg/kg, n = 9); and (4) rag2-/- animals receiving daily injections of saline (n = 10). All injections were IP, and treatment started 1 day before transplantation.
Bioluminescent Imaging
Animals were imaged twice weekly over a period of 3 weeks, beginning on the day after cell transplantation. Anesthesia was induced with 5% isoflurane and maintained with 2% isoflurane/98% oxygen. Luciferin was administered intraperitoneally at 150 mg/kg. Animals were placed inside a Xenogen IVIS 200 optical imager (Caliper LifeSciences, Mountain View, CA) and imaged 13, 15, and 17 min after luciferin injection. The acquisition time ranged from 1 to 60 s, depending on the signal intensity. BLI data were used to generate cell survival curves. Animals with signal at or below the level of background luminescence were considered to have rejected the transplanted cells. For calculation of the graft rejection rate, data are presented for each time point as the percentage of animals in each group that rejected the cells.
Histopathology
Mice were transcardially perfused with 0.01 M phosphate-buffered saline (PBS), followed by 4% paraformaldehyde (PFA). Brain tissue was removed, postfixed in 4% PFA for 24 h, cryopreserved with 30% sucrose solution, and frozen. Thirty-micron-thick coronal tissue sections were cut using a cryostat and immunostained with primary antibodies against the cluster of differentiation 45 (CD45; 1:100), CD4 (1:100), CD8 (1:200) by Serotec, Kidlington, Oxford, for evaluation of an immune response, proliferating cell nuclear antigen (PCNA; 1:100, Santa Cruz) to visualize cell proliferation and against luciferase (luc; 1:1000, AbCam, Cambridge, MA) to assess graft survival. Secondary antibodies included goat anti-rabbit conjugated to Alexa Fluor-488 (green) and goat anti-mouse IgG conjugated to Alexa Fluor-594 (red) (1:200, Molecular Probes).
Statistics
The number of animals per experimental group was estimated based on expected continuous measure of outcomes. Surprisingly, no scores were recorded between good graft survival and complete rejection, and as a result, we received binary outcomes. Accordingly, we calculated the appropriate magnitude of effect measure such as relative risk (RR) and the number of animals needed to avoid “bad” outcomes (NNT) as well as estimated sample size in each group to reach necessary power and demonstrate statistical significance.
Results
In Vitro Characterization of GRPs
Immunocytochemistry against firefly luciferase demonstrated that nearly 100% of transduced, clonally expanded GRPs expressed the reporter gene (Fig. 1A). Expression of luciferase was confirmed up to 20 passages indicating stable expression that was not subject to silencing. The in vitro bioluminescence imaging signal was measured in luciferase-expressing GRP cells plated at six serial dilutions, ranging from one cell to 105 cells. BLI of cultured GRP cells demonstrated that detectable signal could be seen even for single cells (Fig. 1B, top). Quantification of BLI signal showed a linear correlation with the number of cells (Fig. 1B, graph).
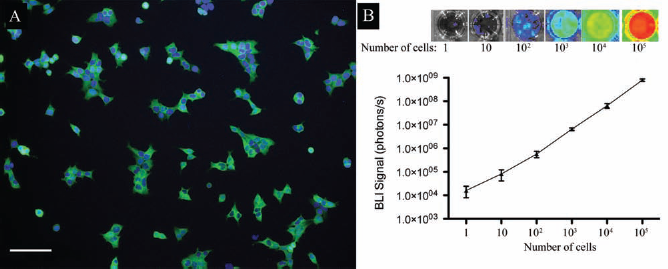
Characterization of luciferase-expressing glial-restricted progenitors (GRPs). (A) Immunocytochemistry against firefly luciferase (green) reveals positivity in nearly 100% of GRPs. Nuclei are counterstained with DAPI (blue). Scale bar: 100 μm. (B) Bioluminescence of cultured GRP cells (top) visualizes the cell number-dependent signal intensity; the graph (bottom) represents quantification of photon signal with a linear correlation (R2 = 0.999).
BLI Monitoring of Allograft Survival and Rejection
BLI was used to noninvasively monitor transplanted GRP cell survival in immunocompetent BALB/c mice, immunosuppresed BALB/c mice receiving either cyclosporin A or a combination of FK506 and rapamycin, or immunodeficient rag2-/- mice. Imaging was performed twice weekly, beginning at 1 day after GRP cell transplantation into the corpus callosum. Using BLI data for individual animals, cell survival curves were generated to determine the percentage of animals in each group in which the transplanted GRP cells were either accepted or rejected (Fig. 2). As expected, in all rag2-/- mice, BLI signal was detected throughout the study indicating complete graft survival (100% of animals) (Fig. 2A). Hence, by 16 days of observation, signal continued to increase representing GRPs that continue to proliferate (Fig. 2A, top). In the immunocompetent, nonimmunosuppressed group, the BLI signal was detected in all mice and initially increased in all animals, indicating good survival and expansion of the allograft. The onset of the rejection process (decrease in BLI signal) occurred 9–13 days after transplantation, and by day 16, rejection was complete. Notably, 30.8% of animals in this group accepted the graft, and in these animals, the BLI signal continued to rise at a pace similar to rag2-/- hosts. When evaluating effect of immunosuppressive drugs, we found that none of the treatments were fully protecting the grafts. FK506 and rapamycin treatment resulted in 77.8% surviving grafts (Fig. 2C), whereas cyclosporine treatment resulted in 44.6% surviving grafts (Fig. 2D). While the difference between the treatment groups was not significant we found that animals immunosuppressed with combination of FK506 and rapamycin were almost twice less likely to reject the graft than those treated with cyclosporin A (RR = 1.75), and only three animals needed to be treated with FK506/rapamycin to avoid one graft rejection. Power sample calculations revealed that such difference between these two groups resulting from binary outcome would be statistically significant if each group included as many as 33 animals.

Monitoring survival of transplanted GRP cells with bioluminescence imaging (BLI). Mice with transplanted luciferase (Luc)-transfected GRPs were imaged twice weekly. Four experimental groups are presented: (A) immunodeficient, (B) immunocompetent, (C) immunosuppressed with FK506 and rapamycin, and (D) cyclosporine. The quantified photon signal (photon/s) for individual mice is given in the graphs for animals that accepted (blue lines) or rejected (red lines) transplanted cells. Representative bioluminescent images for accepting and rejecting animals are shown above each graph.
Immunohistochemical Validation of BLI and Assessment of Allograft Leukocyte Infiltration
In order to validate the BLI data and to characterize the graft rejection process, immunohistochemical analysis was performed (Fig. 3). Anti-luciferase staining in tissues from grafted animals with a strong BLI signal revealed many surviving cells (Fig. 3A, E, G). No luciferase-positive cells were detected in tissues from mice in which the BLI signal was undetectable (Fig. 3C). No migration of transplanted cells out of graft was observed.

Immunohistochemical characterization of graft site. Immunostaining against firefly luciferase (A, C, E, G, red) and the panleukocyte antigen CD45 (B, D, F, H, green). Shown are the results for immunodeficient rag2-/- (A, B), immunocompetent BALB/c (C–F), and immunosuppressed (FK506/rapamycin) mice (G, H). For immunocompetent mice shown is example with complete rejection (C, D) and surviving grafts (E, F). Staining was positive in all animals that had a detectable BLI signal at the time of sacrifice (A, E, G); consequently, staining was negative for the mice that lost the BLI signal (C). Extensive infiltration with CD45+ leukocytes was observed in all animals (D, F, H) except rag2-/- mutants (B). Proliferation of surviving allografts has been confirmed using anti-proliferating cell nuclear antigen (PCNA) antibody (A, inset, green). Sections were counterstained with DAPI (blue). Scale bar: 200 μm.
In order to visualize the graft rejection process, tissue sections were immunostained for the panleukocyte marker CD45 (Fig. 3) and markers specific for lymphocytes T such as CD4 and CD8 (Fig. 4). As expected, no leukocytic infiltration was observed in immunodeficient rag2-/- mice; CD45-positive cells were extremely rare (Fig. 3B). In animals that rejected the graft, the implantation area was highly infiltrated with CD45 cells, with a characteristic perivascular cuffing (Fig. 3D). Surprisingly, in nonimmunosuppressed BALB/c mice that did not reject the cells, the graft area was infiltrated by CD45 cells, to an extent similar to that observed for graft-rejecting mice (Fig. 3F). Cell proliferation was detected within surviving allografts (Fig. 3A, inset), which is in agreement with amplification of BLI signal observed throughout the study. Treatment with immunosuppressive drugs also failed to prevent infiltration, and CD45-positive cells were quite abundant around the surviving grafts (Fig. 3H). Extensive infiltration of grafts by lymphocytes T was observed in both rejecting and accepting animals (Fig. 4).

Lymphocyte infiltration within surviving and nonsurviving grafts. Immunostaining against CD4 (A, B) and CD8 (C, D) antigens (red) and luciferase (green) in immunocompetent nonimmunosuppressed animals ilustrates a robust infiltration of surviving (A, C) as well as rejected (B, D) allografts by both subclasses of lymphocytes. Scale bar: 200 μm.
Discussion
BLI and Graft Survival
BLI is a powerful tool for the in vivo monitoring of immune-mediated rejection, as well as an effective way to compare immunosuppressive regimens. The fact that rag2-/- mice fully accepted the grafts and that in immunocompetent animals decline of BLI signal started at about 1 week after grafting indicates that indeed BLI signal loss results from graft rejection and not other reasons of cell death. Our findings reveal that none of the immunosuppressive strategies were fully effective in protecting glial allografts. The combination of FK506 and rapamycin was somewhat better than cyclosporin, but the difference was not significant. Of note is the fact that in both immunosuppression groups, BLI signal is higher compared to rag2-/- and saline animals, which may indicate additional neuroprotective and/or graft supporting effects. From the longitudinal graft kinetic data, it appears that cell proliferation in immunosuppressed groups occurs at a lower pace. Potentially, this may be explained by the negative effect of calcineurin inhibitors on cell proliferation as decreased calcineurin activity may result in a lower cell proliferation (24). The majority of immunocompetent, nonimmunosuppressed animals rejected the grafts. However, several animals appeared to accept allogeneic transplants, and these cells continued to expand at a rate similar to that observed in immunodeficient animals. Of note, with immunohistochemistry, we observed leukocytes infiltrating the graft area in both rejecting and nonrejecting immunocompetent animals, but not in immunodeficient animals. The inflammatory process that was observed, even for surviving allografts, raises concerns about their long-term functionality.
Thus, we have shown that BLI is a useful method for monitoring of intracerebral graft survival. As validated by immunohistochemistry, the decline of bioluminescence to background level represented full rejection, with an absence of living cells in living tissues. BLI can also be employed to evaluate rejection after systemic transplantation (39). This imaging modality was found to be useful for detection of graft overgrowth (7). The engraftment efficiency for various routes of cell delivery was also successfully addressed by BLI (23,37,41). Combining BLI with magnetic resonance imaging (MRI) in a multimodality imaging approach may provide complementary data, including information about the precise localization of surviving cells (15). BLI data have also been used to guide immunosuppressive treatment for β-islet transplants. The decrease in BLI signal in islet recipients preceded functional graft failure (hyperglycemia) (12), and the administration of antilymphocyte serum treatment, based on BLI data, dramatically prolonged allograft survival (11).
The necessity of immunosuppression is difficult to avoid in the field of cell therapy both in preclinical and clinical settings. Long-term cell survival is a prerequisite for any substantial therapeutic benefit of stem cell-based therapy, as the cells take weeks or months to fully mature and integrate (42). While there are reports indicating long-term survival of cerebral allografts without immunosuppression (34), a number of studies have reported elimination of nonimmunoprotected grafts by immune system (6,40,45). Autologous cell sources including those generated by using induced pluripotent stem (iPS) cell technology are expected to be immune to graft rejection. However, it has recently been shown that even autologous iPS cell grafts can be immunorejected (1). Although applicability of BLI for graft monitoring is limited to small animals such technology is instrumental in gaining knowledge about basic mechanisms governing graft rejection or tolerance.
Histology Findings
Histological detection of transplanted cells (anti-Luc staining) correlated well with BLI data. Given that >95% of transplanted cells expressed the luciferase gene, the imaging data convey accurate information about the transplanted cells. No leukocytes were found within the graft area in rag2-/- mice, which are incapable of an adaptive immune response; thus, an innate immune response is not sufficient to initiate graft rejection. In contrast, in all immunocompetent recipients, we observed robust immune cell infiltration around the transplantation site and the presence of many CD45+, CD4+, and CD8+ leukocytes. The administration of immunosuppressive drugs was not able to protect the graft against massive inflammatory cell infiltration. Notably, these cells were present in animals in which transplanted cells had been rejected, as well as in those in which cells continued to expand. Thus, excellent graft survival despite significant immune reaction is infrequent, but possible. Further studies are needed to determine what mechanisms are responsible for these observations. We also show here that BLI of luciferase-expressing grafts is a robust approach for the evaluation of transplanted cell survival but is unable to detect a host inflammatory reaction. The histology findings demonstrate the need for concurrent monitoring of the temporal dynamics of inflammatory processes, as leukocyte infiltration may limit the functionality of otherwise well-surviving grafts. Such monitoring may be achieved by using recently obtained light-producing transgenic animal models that express luciferase under leukocyte-specific promoters (14,28,29). In addition, the application of dual-color imaging (8,21) may open up new avenues for the simultaneous detection of transplanted cells and host inflammatory reactions, leading to a more comprehensive understanding of graft–host immunological interactions. This approach may facilitate an explanation of allograft survival versus rejection in immunocompetent, nonimmunosuppressed animals.
The immune response against the graft depends heavily on the concordant expression of histocompatibility antigens but will also likely be affected by other characteristics of transplanted cells such as the use of primary versus immortalized cells. Nevertheless, our data indicate a delicate balance between allograft rejection and acceptance and that an understanding of this phenomenon may result in strategies that allow healthy allografts to be maintained in humans without the need for toxic immunosuppression. Summarized, the robustness and accuracy of BLI for monitoring engrafted cells enabled us to demonstrate the time course and dynamics of allograft rejection and observe the efficacy of immunosuppressive regimens.
Footnotes
Acknowledgments
The study was supported by the Maryland Stem Cell Research Fund (grants MSCRFII0193, MSCRFII0190) and NIH 2RO1 NS045062. M.J. was supported by Kolumb Fellowship from Foundation for Polish Science. The authors thank Mary McAlister for editorial assistance. The authors declare no conflict of interest.
