Abstract
Recent studies have identified adipose tissue as a new source of mesenchymal stem cells for therapy. The purpose of this study was to investigate the therapy with adipose-derived stromal cells (ASCs) in a rat model of healed myocardial infarction (MI). ASCs from inguinal subcutaneous adipose tissue of male Wistar rats were isolated by enzymatic digestion and filtration. Cells were then cultured until passage 3. Four weeks after ligation of the left coronary artery of female rats, a suspension of either 100 μl with phosphate-buffered saline (PBS) + Matrigel + 2 × 106 ASCs labeled with Hoechst (n = 11) or 100 μl of PBS + Matrigel (n = 10) was injected along the borders of the ventricular wall scar tissue. A sham-operated group (n = 5) was submitted to the same surgical procedure except permanent ligation of left coronary artery. Cardiac performance was assessed by electro- and echocardiogram. Echo was performed prior to injections (baseline, BL) and 6 weeks after injections (follow-up, FU), and values after treatment were normalized by values obtained before treatment. Hemodynamic measurements were performed 6 weeks after injections. All infarcted animals exhibited cardiac function impairment. Ejection fraction (EF), shortening fractional area (SFA), and left ventricular akinesia (LVA) were similar between infarcted groups before treatment. Six weeks after therapy, ASC group showed significant improvement in all three ECHO indices in comparison to vehicle group. In anesthetized animals dp/dt+ was also significantly higher in ASCs when compared to vehicle. In agreement with functional improvement, scar area was diminished in the ASC group. We conclude that ASCs improve cardiac function in infarcted rats when administered directly to the myocardium.
Introduction
Heart failure is a leading cause of hospitalization and death in industrialized countries (7) and is reaching epidemic proportions. Myocardial ischemia is characterized by a decrease or interruption of coronary blood flow and is the main cause of heart disease and cardiomyocyte (CM) loss. Despite the presence of resident stem cells (1), myocardial tissue has a limited regenerative capacity and CM loss results in scar formation and a decrease in mechanical heart function (4,30). Over the past decade, cell therapy has emerged as a new approach to repair the ischemic heart (reviewed in 22,43).
Since 1999, when the first report that bone marrow mesenchymal stem cells (BMSCs) could differentiate into cardiomyocytes in vitro (27), research in regenerative medicine has advanced exponentially (10,22). Adult-derived stem cells present advantages for the treatment of heart diseases since they can be used in autologous transplantation and are not tumorigenic (6). In this context, several types of adult stem cells such as bone marrow (BMSCs), skeletal myoblasts, endothelial progenitor cells, and cardiac stem cells have been studied extensively for cardiac repair (9,15,19,21,48).
In recent years, interest has rapidly grown in the plasticity and therapeutic potential of stromal cells isolated from adipose tissue (ASCs) (16,55). Adipose tissue (AT) is a source of mesenchymal stem cells particularly attractive due to their ease of collection, clonogenic, and angiogenic potential and proliferative capacity. Compared with other tissues, AT contains a great number of MSCs, which are easily cultured and have a high capacity for rapid expansion in vitro. Moreover, ASCs have been reported as less immunogenic and immunosuppressive than bone marrow cells, opening the opportunity for allogeneic transplantation (14,31,35,41,46,50,54,55).
Recent studies have reported the improvement of heart function and the reduction of cardiac remodeling after administrating ASCs to treat myocardial infarction (8,20,23,24,28,34). Although heart repair by cell therapy is gaining increasing importance in modern cardiology, solid evidence of a beneficial effect of transplanted cells is not yet rooted. Our study aimed to evaluate the therapeutic potential of stem cells from adipose tissue in the healed infarct model in rats.
Materials and Methods
Animals
All procedures were performed in accordance with the Revised Guide for the Care and Use of Laboratory Animals published by the American Physiological Society (3) as attested by the competent institutional board. Singenic female and male Wistar rats were exposed to a 12-h light/ dark cycle (lights on 07:00–19:00 h) and had access to both water and standard rat chow ad libitum.
Cell Culture
ASCs were isolated by a method based on density and differential adhesion, as described by Zuk et al. (56). Inguinal subcutaneous adipose tissue of male singenic Wistar rats (12–16 weeks; 300–450 g) was washed with sterile phosphate-buffered saline (PBS) to remove contaminating debris and red blood cells. Washed tissue was minced, and cells were isolated by enzymatic digestion (0.1% type II Collagenase) in PBS for 45 min at 37°C with gentle agitation. Enzyme activity was neutralized with Dulbecco's modified Eagle's medium (DMEM), and cells were centrifuged for 10 min at 200 × g at room temperature. The supernatant, containing mature adipocytes, was aspirated. Pellet was filtered through 80-μm filters, and cells were washed in PBS three times, counted in Neubauer chamber, and checked for viability using 0.4% trypan blue. Cells were then plated at a density of 5 × 104 /cm2 in 150 cm2 cell culture flasks. Culture medium used was DMEM-high glucose, containing 20% fetal bovine serum (FBS), 2 mM l-glutamine, 100 U/ml penicillin, and 100 mg/ml streptomycin. Following 24-h incubation, nonadherent cells were removed by PBS washing, and the cultures were maintained at 37°C in normoxic conditions (95% air, 5% CO2). Medium was changed twice a week, and cells were trypsinized when reaching 80% confluence. Cells in passage 3 were used for this study.
Cell Characterization
All characterization of ASCs was done at third passage. ASCs were submitted to population doubling time (PDT) assays, mesodermal lineage differentiation, and immunophenotypic characterization. Cultured ASCs were harvested with 0.25% trypsin and suspended as single cells at a concentration of 5 × 105 cells in 500 μl phosphate-buffered saline (10 mM, pH 7.4). To verify that cultured cells were MSCs (mesenchymal cells), the cells were subject to flow cytometry (FACS CANTO, BD Instruments, CA) after labeling with CD45-peridinin chlorophyll protein cyanine 5.5 (PerCP-CY5.5; BD Pharmingen™), CD34-phycoerythrin (PE; Santa Cruz Biotechnology), CD11b-fluorescein isothiocyanate (FITC; Caltag Laboratories), CD29-FITC (BD Pharmingen™), and CD90-FITC (Caltag Laboratories). Isotype IgG1 immunoglobulin served as control. In addition, these cells were cultured in the presence of osteogenic medium (10 nM dexamethasone, 10 mM β-glycerophosphate, 50 μg/ml l-ascorbate 2-phosphate, and 10 nM 1α,25-dihydroxyvitamin D3; BIOMOL Research Laboratories, Inc., Plymouth Meeting, PA) and adipogenic medium (DMEM-low glucose supplemented with 15% fetal calf serum, 50 U/ml penicillin, 50 mg/ml streptomycin, 5 × 10−4 M de indomethacin, 10 μg/ml human insulin, e 10−7 M dexamethasone (Sigma-Aldrich), and stained with Red alizarin and Oil Red O, respectively, to confirm differentiation.
Myocardial Infarction and ASC Transplantation
Myocardial infarction (MI) was induced by left coronary artery (LCA) ligation as previously described (36) in 40 female Wistar rats (8–10 weeks; 200 –250 g). Briefly, rats were anesthetized with isoflurane (Merck), and after exposing the heart, LCA was permanently occluded with a 6–0 silk suture, then the chest was closed with silk stitches, and rats were allowed to recover. Sham-operated (SHAM, n = 5) animals underwent the same procedure, but the coronary ligature was left untied. Twenty-eight percent of the animals (14 of 50) died during surgery or 24 h after surgery. Four weeks after MI induction, 2 of 36 animals had left ventricular akinesis (LVA; see below) below 20% and were excluded. Four weeks post-MI rats were submitted to another thoracotomy and received an intramyocardial injection of 100 μl of 2 × 106 ASCs in Matrigel™ (BD Biosciences) (n = 11) or 100 μl of vehicle (PBS/Matrigel™ solution) (n = 10). Six weeks after treatment, the animals were submitted to functional evaluation and then killed by cervical dislocation under anesthesia for histological evaluation. The hearts of both groups were excised and processed for histology (see below). Another group comprising ASC-(n = 9) and VEHICLE- (n = 4) injected animals was used to track the injected cells by PCR detection of the Y chromosome at different time points after cell injection.
Electrocardiogram (ECG)
Animals were anesthetized with ketamine and xylazine (50 and 5 mg/kg, IP, respectively). Rats were placed in the supine position, and electrodes connected to surgical needles were inserted subcutaneously in order to record the classical six limb leads (L1, L2, L3, aVR, aVL, and aVF) as previously described (45). Angle of left ventricular depolarization vector (âQRS) and the presence of Q wave in L1 were determined from recordings obtained at 50 mm/s with a sensitivity of 20 mm/mV (Cardimax FX-2111, Fukuda Denshi). The data for âQRS were represented as frequency of animals with âQRS larger than 90° (right axis deviation) or between 0 and 90° (normal axis). âQRS > 90° and presence of Q wave in L1 were used as evidence of infarction as previously reported (32). Presence of Q wave was analyzed 48 h after infarction, and the other ECG parameters were evaluated 28 days after infarction surgery (1 h before treatment) and 6 weeks after cell or vehicle injection.
Echocardiographic Assessment (ECHO)
Echocardiographic examination was performed blind by the same operator, following the American Society of Echocardiography (ASE) recommendations. We used an echocardiographic color system (Megas/Esaote) equipped with a 10-MHz electronic-phased-array transducer. Short-axis two-dimensional views of the left ventricle (LV) were taken at the level of the papillary muscles to obtain the M-mode recordings. Ejection fraction was measured as EF% = (EDV – ESV) / EDV, where the end-diastolic and -systolic volumes (EDV and ESV, respectively) were calculated by the method of Teichholz. In order to estimate LV global function, we calculated LV diastolic and systolic area (LVDA and LVSA, respectively) to determine shortening fractional area {SFA (%) = [(LVDA-LVSA)/ LVDA] × 100}. Additionally, the total and the akinetic perimeter of the LV in diastole (LVTP and LVAP, respectively) were determined to calculate percentage of LV akinesis [%LVA= (LVAP/LVTP) × 00]. Parameters were evaluated 28 days after infarction surgery (1 h before treatment) and 6 weeks after cell or vehicle injection.
Left Ventricular Pressure Measurements in Anesthetized Rats
One day after the last echocardiogram measurement, animals were anesthetized with ketamine/xilazine (50 and 5 mg/kg, IP, respectively) and the right carotid artery was cannulated with a PE10 catheter. One extremity of the catheter was inserted into the left ventricle, and the opposite end was connected to a pressure transducer (MLT0380/D, ADInstruments) coupled to a Power Lab400 acquisition system (ADInstruments) and LV pressure acquisition started. The left ventricular parameters evaluated were left ventricular end-diastolic pressure (LVEDP), left ventricular end-systolic pressure (LVESP), left ventricular developed pressure [(LVDP) = LVESP – LVEDP], cardiac index of left ventricular contractility (dP/dt+, rate of pressure rise) and index of left ventricular relaxation (dP/dt-, rate of pressure decay). All values were measured using Chart 4 software (PowerLab, ADInstruments).
Histological Study
After all cardiac functional analyses were performed, the animals were sacrificed and had their hearts excised for histological analysis. After being arrested in diastole by KCl (30 mM) perfusion, the hearts were fixed by immersion in 4% paraformaldehyde in phosphate buffer. Ventricles were sectioned in four slices from apex to base (A, B, C, and D, respectively). Infarct size determination was performed by computer-assisted planimetry (ImageJ version 1.27z, National Institute of Health, USA) as the mean percentage of the circumference occupied with the scar tissue on each section, after staining with Picrosirius (Sirius red) for collagen scar evaluation.
Polymerase Chain Reaction for Identifying Y Chromosome
The polymerase chain reaction (PCR) amplification was used to identify the Y chromosome to detect male transplanted cells. Left ventricular tissue from the injection site was isolated and stored in liquid nitrogen. Genomic DNA templates (150–300 ng/μl) from different groups were used in PCR reactions (20 μl) with rat sex-determining region Y (Sry) primers according to the protocols published by Dvorianchikov et al. (11). Primer sequence is as follows: forward primer, 5′-CGTGAAGTTGCCTCAACAAA-3′ and reverse primer, 5′-TCTGGTTCTTGGAGGACTGG-3′ (GenBank: NM_012772).PCR assays were performed in triplicate. Control reactions were performed using glyceraldehyde 3-phosphate dehydrogenase (GAPDH) primers with the following sequences: forward, 5′-GGTGAAGGTCGGTGTGAAC-3′ and reverse, 5′-CGTTGATGGCAACAATGTC-3′ (GenBank: NM_017008.3). PCR products were separated by electrophoresis on a 1.5% agarose gel with 1x tris-aminomethane tris-acetate-ethylenediaminetetraacetic acid buffer, stained with ethidium bromide (EtBr), and photographed under ultraviolet light.
Statistical Analysis
All data are expressed as mean ± SEM. Unpaired or paired Student's t test was performed to compare two groups, depending on the experimental situation. Oneway ANOVA was used to compare difference between groups followed by Bonferroni post hoc test. When more than one time point is included, two-way ANOVA was applied followed by Bonferroni post hoc test. Values were considered significant when p < 0.05.
Results
Characterization of ASCs
ASCs were adherent and fibroblast-like and showed significant expansion in culture (Fig. 1A). Cell growth was exponential, and population-doubling time was 1.177 days (n = 4) (Fig. 1B). Moreover, ASCs differentiated into osteoblasts as demonstrated by alizarin staining after 4 weeks in culture and adipocytes as demonstrated by oil red staining after 21 days, indicating that the cells are multipotent (Fig. 2). Furthermore, the cells expressed mesenchymal but did not express hematopoietic markers (Fig. 3). We found that 86% of the cells were negative for CD45; 98.4% were negative for CD34, and 97.8% were negative for CD11b. In contrast, 99.7% of the cells were positive for CD29, and 99.8% were positive for CD90.
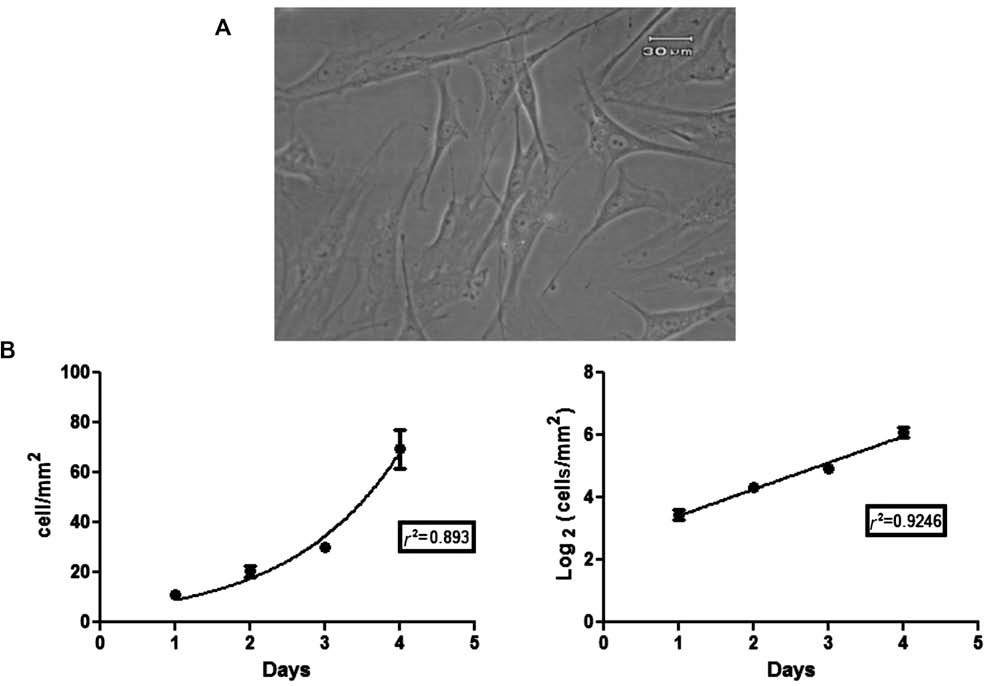
ASC, morphology, and growth in vitro. (A) Cultured adipose-derived stromal cells (ASCs), after three passages, exhibit a spindle-shaped fibroblastic morphology. Scale bar: 30 μm. (B) Growth profile of ASCs during 4 days in culture (left). Growth rate of ASCs in semilogarithmic scale allows for calculation of the time that the cells take to duplicate the initial population (population doubling time, PDT).
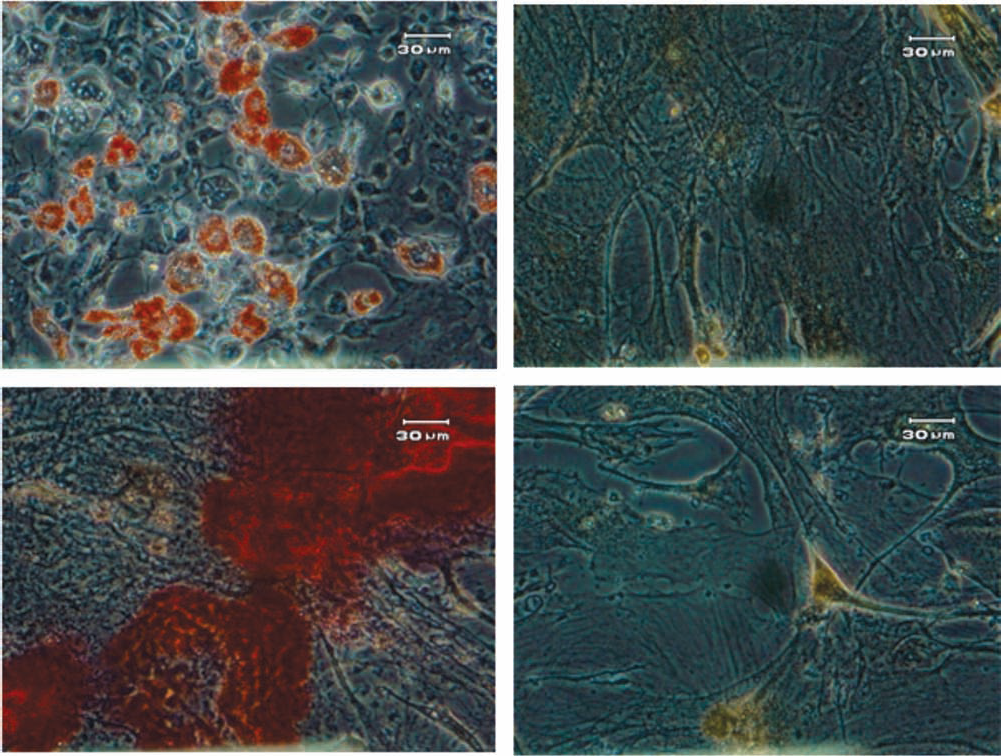
ASC differentiation in vitro. ASCs at passage 3 were cultured with the appropriate induction media and differentiated into adipocytes as shown by staining with Oil Red O (top left) and osteoblasts as shown by staining with Red Alizarin (bottom left). Figures on the right show control cells cultured in standard medium. Scale bar: 30 μm.

ASC phenotypic characterization. Fluorescence-activated cell sorting (FACS) analysis of the ASCs with isotype controls shown in the top row and in the bottom row ASCs expressing high levels of CD29 and CD90 and low levels of CD11b and CD34. Fourteen percent of cells in early passage (third passage in this case) still expressed CD45.
Functional Analyses
Electrocardiogram (ECG)
Infarcted animals examined by ECG (VEHICLE and ASC groups) presented Q wave in L1 and a rightward deviation of the QRS axis (âQRS > 90°) before treatment. The presence of Q wave and rightward deviation are signs of large MI. In contrast, sham-operated animals had no Q wave in L1 and sustained normal QRS axis values between 0 and 90° (Table 1). Six weeks after cell or placebo injection, electrocardiographic parameters in both groups (VEHICLE and ASC) were still typical of infarcted animals, with persistence of Q wave in L1 and a rightward deviation of the QRS axis when compared to sham group (VEHICLE: 143° ± 10.59° and ASC: 134° ± 14.64° vs. SHAM: 64.16° ± 9.27°*; *p < 0.001) (Table 1).
Electrocardiogram Parameters

Mean ± SEM. Presence of Q wave in Lead1; âQRS, entricular depolarization vector angle and depolarization vector. *p < 0.001 adipose-derived stromal cell (ASC) and VEHICLE versus SHAM rats hearts 6 weeks after injection.
Echocardiographic Assessment (ECHO)
Twenty-eight days after MI, SFA% was significantly lower in the infarcted groups (VEHICLE: 28.4% ± 9.12%, ASC: 28.0% ± 10.34%, vs. 58.2 ± 4.03% in SHAM group; p < 0.005) when compared to SHAM, but no difference between infarcted groups was observed. Ejection fraction in infarcted groups (VEHICLE and ASC) was also significantly decreased 28 days after MI (VEHICLE: 42.8% ± 2.45%; ASC: 47.09% ± 3.23% vs. 86.80% ± 1.62% in SHAM group; p < 0.005), when compared to SHAM group, but again no significant difference between infarcted groups was detected. Also, the percentage of LV akinesis at the level of the papillary muscles twenty-eight days after MI did not show difference between infarcted groups (VEHICLE: 64.6% ± 7.55% and ASC: 72.1% ± 5.07%), while SHAM group did not show any akinetic perimeter in the left ventricle. These data suggest a similar magnitude of cardiac impairment in both infarcted groups before treatment (Table 2). In one animal of the VEHICLE group, we could not perform the echo due to excessive scar accumulation around the heart after infarct surgery that precluded a viable echo window. There were significant differences in EF between ASC and VEHICLE groups (Fig. 4A) and LVA (Fig. 4B) at 6 weeks after cell or vehicle injection when we normalized the parameters by their baseline values.
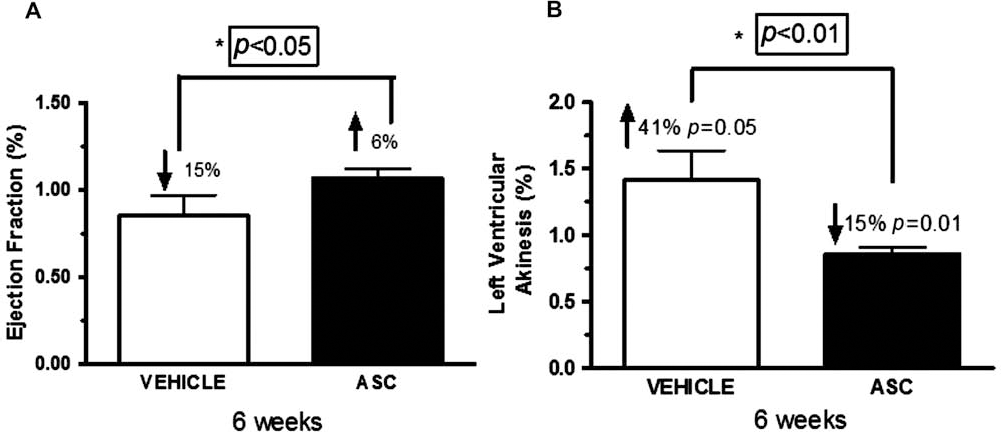
Normalized ejection fraction (EF) and left ventricular akinesis (LVA). (A) Vehicle group showed a 15% reduction in EF%, while cell-treated group showed a 6% increase in EF%. (B) Left ventricular akinesis increased by 41% in the vehicle group while decreasing by 15% in the cell-treated group. Measurements after cell or vehicle injection were normalized to their baseline values for each animal, and then mean values ± SEM were calculated. *p < 0.05 versus VEHICLE, 6 weeks after injection. VEHICLE (n = 9) and ASC (n = 11).
Echocardiogram Parameters

Mean ± SEM. SHAM (n = 5), ASC (n = 11), and VEHICLE (n = 9) 28 days after left coronary artery (LCA) ligation. EF, ejection fraction; SFA, shortening fraction area; LVA, left ventricular akinesis.
p < 0.005 ASC and VEHICLE versus SHAM rat hearts.
Left Ventricular Pressure Measurements in Anesthetized Rats
Left ventricular pressure (LVP) parameters in anesthetized rats were measured 6 weeks after intramyocardial injection of cells or vehicle and are shown in Table 3. Intramyocardial injection of ASC improves cardiac contractility, as measured by dp/dt+, when compared to vehicle administration (p < 0.05). In relation to other LV parameters, no significant differences were observed between groups although all parameters showed a tendency to improve in the ASC group compared to the VEHICLE group.
Hemodynamic Analysis–6 Weeks After Therapy
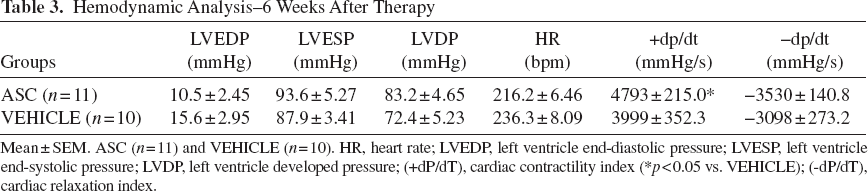
Mean ± SEM. ASC (n = 11) and VEHICLE (n = 10). HR, heart rate; LVEDP, left ventricle end-diastolic pressure; LVESP, left ventricle end-systolic pressure; LVDP, left ventricle developed pressure; (+dP/dT), cardiac contractility index (*p < 0.05 vs. VEHICLE); (-dP/dT), cardiac relaxation index.
Post Mortem Study
Infarct Size
Figure 5A shows two representative slices, at the level of the papillary muscles, from different rat hearts treated with VEHICLE and ASC. Figure 5B summarizes the results of all animals, 6 weeks after cell or vehicle intramyocardial injection. MI size as a percentage of the total left ventricle perimeter was significantly smaller in the ASC versus VEHICLE group (37.86 ± 2.02 vs. 48.28 ± 1.97, respectively, p < 0.01).

Infarct size. (A) Representative slices of left ventricle from vehicle and cell-treated hearts stained with Sirius red. (B) Infarct size as percentage of left ventricular perimeter in VEHICLE and ASC groups. **p < 0.005 versus VEHICLE. ASC (n = 5) and VEHICLE (n = 5).
Survival of Transplanted Cells by PCR Analysis
Left ventricular tissue samples from nine rats in ASC group and four rats in VEHICLE group were used for detecting the sex-determining region Y (Sry gene) by PCR at different time points after cell or vehicle injection (immediately, 1 week, and 6 weeks). All tissues from animals of the ASC group were positive for Sry at all different time points after cell injection (Fig. 6A–C), confirming the presence of the transplanted cells even after 6 weeks after injection. As expected, all tissues of the VEHICLE group were negative for Sry as shown in the figure.

Expression of sex determining region Y (Sry) gene in heart tissue of rats evaluated by RT-PCR. (A) Immediately after injection of ASCs. Lanes 1, 2, 3: ASC-injected animals; lanes 4, 5: vehicle injection; positive control (+); negative control (-); and 1 kb plus ladder (L). (B) One week after injection. Lanes 1, 2: ASCs; lane 3: vehicle; positive control (+); negative control (-); and 1 kb plus ladder (L). (C) Six weeks after injection. Lane 1: vehicle; lanes 2, 3, 4, 5: ASCs; positive control (+); negative control (-); and −1 kb plus ladder (L). The expected PCR product for Sry was 115 bp in size and the internal control glyceraldehyde 3-phosphate dehydrogenase (GAPDH) was 95 bp. All samples were positive to the GAPDH.
Discussion
In the present study, we provide evidence that intramyocardial injection of cells derived from adipose tissue 28 days post MI significantly preserves left ventricular function, as assessed by echocardiogram and direct hemodynamic evaluation. ASCs improved heart function by reducing the degree of fibrosis in infarcted tissue. We also observed, by tracking grafted cells by PCR to detect Sry gene, that ASCs were present in the host tissue up to 6 weeks after transplantation.
Our data confirmed recent reports showing that transplantation of ASCs can favorably affect cardiac function after myocardial infarction either in acute (8,24,47,52) or chronic (28,34,39) phase. However, our study is unique in performing an exhaustive functional and histological cardiac assessment based on electrocardiogram, echocardiogram, and hemodynamic measurements, evaluation of infarct extension and cell survival, as well as a full characterization of the injected ASCs before transplantation in chronically infarcted rats. Bone marrow mononuclear and mesenchymal cells have been the most commonly used source of stem cells for cardiac regeneration, (5,12,13,18,36,37,38,42). Like bone marrow MSCs, ASCs are capable of giving rise to different cell types, such as osteocytes, chondrocytes, and myocytes when properly stimulated (14,17,26,55,56). They also contain a subset of rare cells that are able to spontaneously differentiate into cardiomyocytes (23,39).
ASCs secrete soluble angiogenic and antiapoptotic factors such as vascular endothelial growth factor (VEGF) and hepatocyte growth factor (HGF) and can restore blood flow in vascular ischemic disease (40). Furthermore, ASCs when cocultured with cardiomyocytes decreased the rate of apoptosis via insulin-like growth factor-1 (IGF-1) and VEGF production (44).
The best cell source for cardiovascular disease is still in debate. Studies that compared adipose and bone marrow in vivo are conflicting. Mazo et al. injected either bone marrow mononuclear cells (BM-MNCs) or ASCs from mice in chronic infarcted rats. They showed that only ASCs improved cardiac ejection fraction (28). However, when mesenchymal stem cells from both adipose and bone marrow were compared in the acute phase of MI, none was effective, probably due to the dramatic cell death after transplantation (52). ASCs constitute the plastic adherent cell fraction isolated from the stromal vascular fraction (SVF), and both have been reported to enhance cardiac ejection fraction after acute MI (2). In the present work, we decided to use mesenchymal cells from adipose tissue based on previous results from our lab showing that intramyocardial injection of MSCs from bone marrow preserved cardiac function (36) while mobilization of bone marrow cells by granulocyte colony-stimulating factor (G-CSF) was ineffective (25,53). Compared with bone marrow, AT is easier to obtain and contains a greater number of MSCs, which are easily cultured and have a high capacity for rapid expansion in vitro, thus making it an ideal source of MSCs. We demonstrated that the population of progenitor cells obtained from rat adipose tissue (ASCs) in passage 3 display markers of MSCs (CD29+, CD90+) and the majority of the population was CD34-, CD45-, CD11-, as reported by Miyahara et al. (34). The mesenchymal phenotype of ASCs was also shown in human cells (29,33), although classical hematopoietic markers varied during cell culture (29).
Miyahara et al. injected ASCs as a cell monolayer to cover the damaged tissue, contrasting to our approach of intramuscular delivery of cells (34). Mazo et al. used mouse cells in chronic infarcted rats (28). Furthermore, some studies used porcine (51) or mice (23,52) as both donor and host of the adipose cells and showed improvement in cardiac function after cell therapy. In this context, our study reinforces the use of ASCs for the treatment of chronic MI and heart failure. The chronic ischemic stage is characterized by collagen deposition, maturation of the scar, and a progressive loss of heart pumping function that can lead to congestive heart failure, as reviewed in Sun (49). Therapeutic options for congestive heart failure are limited, and thus, in accordance with our data, cell therapy using ASCs may provide an alternative that warrants further investigation.
In the present study, transplanted ASCs survived in the host tissue for up to 6 weeks of follow-up. However, we used a qualitative PCR analysis, which does not allow us to know whether cell number remained unchanged after 6 weeks of transplantation. It is reasonable to assume that the number of reminiscent cells is low due to the adverse environment after a myocardial infarction (28,34). Failure of cell survival has been linked to several factors such as inflammatory response, mainly in the acute phase of MI, the hypoxic microenvironment and absence of survival signals. Moreover, the cell injection itself may cause high cell mortality due to mechanical injury and subsequent acute inflammatory response (18). Mazo et al. performed PCR of genomic DNA in order to detect the presence of green fluorescent protein (GFP) and Sry gene and showed no donor cells in cardiac tissue 4 weeks after therapy with ASCs in healed MI. Interestingly, the authors found that, 1 week after transplantation, the benefits in cardiac function were already evident and that less than 0.5% of cells still remained in the tissue (28). The inoculation of ASCs in Matrigel could explain the prolonged survival of cells found in the present work.
Conclusions
In conclusion, our data demonstrated that intramyocardial injection of ASCs in infarcted healed hearts improve cardiac contractility and reduce progressive collagen deposition as shown by diminished infarct size in treated groups.
Footnotes
Acknowledgments
We thank the agencies that funded our project. The present work was supported by CNPq, CAPES and FAPERJ. The authors declare no conflicts of interest.
The following author contributions are recognized: Concept and design (L.L.S.B., J.P.S.W.D.C., P.F.O., R.C.S.G., and A.C.C.D.C.); cell culture (L.L.S.B.); cell characterization (T.K.-B. and L.L.S.B.); myocardial infarction induction (J.P.S.W.D.C. and L.L.S.B.); ECG acquisition and analysis (M.S.C.-A. and L.L.S.B.); echocardiography acquisition and analysis (N.N.R. and L.L.S.B.); hemodynamic procedures and evaluation (P. F.O. and L.L.S.B.); polymerase chain reaction for identifying Y chromosome (V.M.); histology (L.L.S.B.); data analysis and interpretation (L.L.S.B., P. F.O., J.P.S.W.D.C., R.C.S.G., and A.C. C.D.C.); manuscript writer (L.L.S.B., M.S.C.-A., and J.P.S.W.D.C.); and significant manuscript reviewer/ reviser (R.C.S.G. and A.C.C.D.C.).
