Abstract
In the present study, we examined the therapeutic potential of human amnion-derived insulin-secreting cells for type 1 diabetes. Human amniotic mesenchymal stem cells (hAMs) were isolated from amnion and cultivated to differentiate into insulin-secreting cells in vitro. After culture in vitro, the differentiated cells (hAM-ISCs) were intensively stained with dithizone and secreted insulin and c-peptide in a high-glucose-dependent manner. They expressed mRNAs of pancreatic cell-related genes, including INS, PDX1, Nkx6-1, NEUROG3, ISL1, NEUROD1, GLUT1, GLUT2, PC1/3, PC2, GCK, PPY, SST, and GC, and were positive for human insulin and c-peptide. Transplantation of hAM-ISCs into the kidneys of mice with streptozotocin-induced diabetes restored body weight and normalized the blood glucose levels, which lasted for 210 days. Only human insulin and c-peptide were detected in the blood of normalized mice after 2 months of transplantation, but little mouse insulin and c-peptide. Removal of graft-bearing kidneys from these mice resulted in causing hyperglycemia again. Human cell-specific gene, hAlu, and human pancreatic cell-specific genes, insulin, PDX1, GLUT1, GLP1R, Nkx6-1, NEUROD1, and NEUROG3, were detected in the graft-bearing kidneys. Colocalization of human insulin and human nuclei antigen was also observed. These results demonstrate that hAMs could differentiate into functional insulin-secreting cells in vitro, and human insulin secreted from hAM-ISCs following transplantation into type 1 diabetic mice could normalize hyperglycemia, overcoming immune rejection for a long period.
Keywords
Introduction
Type 1 diabetes is incurred by an autoimmune destruction of pancreatic β-cells. Destruction of the insulin-secreting β-cells in the islets of Langerhans leads to hyperglycemia. Transplantation of whole pancreas or allogeneic islet is an ideal treatment for patients with type 1 diabetes. However, tissue donation for transplantation is not readily available. In most cases, treatment depends on insulin pump or multiple daily insulin injections, which do not ensure blood glucose control and prevention of long-term complications. To overcome these problems, cell therapy utilizing insulin-secreting cells as an alternative of pancreatic tissue has been developed.
Embryonic stem cells and induced pluripotent stem cells possess better quality of proliferation and differentiation potential than adult type stem cells. However, problems such as ethical limitation and/or teratoma formation remain to be solved (13,23). Free from these issues, various types of adult stem cells have been widely examined for the cell replacement therapy to treat insulin-dependent diabetes. Included are peripheral blood monocytes (35), fetal liver progenitor cells (44), bone marrow-derived mesenchymal stem cells (BM-MSCs) (1,45), placental stem cells (7), umbilical cord stem cells (9), eyelid adipose stem cells (20), and endometrial stem cells (26). These cells have been shown to differentiate into insulin-secreting cells in vitro and to reduce blood glucose level of hyperglycemia when differentiated cells were transplanted into the animals with experimentally induced type 1 diabetes.
Pancreatic injury, such as pancreatectomy, ductal ligation, or streptozotocin (STZ) treatment, is known to stimulate regeneration of pancreatic islets via self-duplication of preexisting β-cells (38) or with neogenesis of β-cells from non-β-cell progenitors such as the ones located in the pancreatic duct in mice (43). Interestingly, transplantation of BM-MSCs from human (24), mice (14), or rat (5) has been shown to enhance insulin secretion and lower blood glucose levels in hyperglycemic recipient animals with STZ-induced pancreatic tissue damage via the regeneration of recipient pancreatic β-cells. Cotransplantation of islet graft with MSCs also promoted higher graft survival and function than in islet-alone transplantation in recipient animals via enhanced graft vascularization (16,37). However, it is highly likely that regeneration of the recipient cells, regardless whether by self-duplication of preexisting β-cells or by neogenesis from non-β-cell progenitors, may result in the recurrence of immune attack in type I diabetes. In this regard, it is important to verify that restoration of the blood glucose level by cell transplantation is achieved solely by donor cells and not by regenerated host cells when considering the application of therapeutic cells for the treatment of type 1 diabetes. Indeed, many studies observed the presence of human insulin and/or c-peptide in the blood of recipient that has recovered from hyperglycemia following human cell transplantation (23,26,44,45). Nevertheless, most of these studies have not clarified whether the reduction of blood glucose in recipient animals was due to the human insulin secreted from transplanted human cells or by the recipient animal insulin secreted from the regenerated pancreatic β-cells.
Placental tissues are readily available without invasive procedures and contain stem cells exhibiting immunomodulatory properties (28,42) or immunological tolerance (39). These features allow them to be ideal sources for therapeutic cells in regenerative medicine. Human term placenta consists of two tissue layers, chorion and amnion. While chorionic membrane harbors mesenchymal and trophoblast type of stem cells, amniotic membrane consists of epithelial and mesenchymal type of stem cells. Successful in vitro differentiation of amniotic epithelial stem cells into insulin-producing cells has been reported by showing restoration of normoglycemia upon transplantation of differentiated cells into diabetic mice (41). However, since epithelial stem cells are capable of undergoing only two to six passages before senescence (33), they cannot provide enough cell number that is required for the cell replacement therapy of diabetes in humans. In contrast, amniotic mesenchymal stem cells (hAMs) undergo 13 passages or more (22) and can differentiate into insulin-secreting cells in vitro (19). Taken together, these observations suggest that hAMs can be used in cell replacement therapy for the treatment of type 1 diabetes.
In the present study, we aimed to examine the differentiation potential of hAMs into insulin-secreting cells in vitro, which could secrete insulin and c-peptide in a glucose concentration-dependent manner. More importantly, we evaluated whether transplantation of hAMs-derived insulin-secreting cells (hAM-ISCs) into type 1 diabetic mice could normalize hyperglycemia of mice by providing human insulin without inducing the regeneration of mouse pancreatic β-cells in the absence of any immunosuppressants.
Materials and Methods
hAMs Isolation and Culture
Placentae were harvested within 4 h of delivery after cesarean section from the volunteers who had given informed consent. Isolation and culture of hAMs were prepared as described previously (22). In brief, human fresh amnion was mechanically separated from the chorion, and the epithelial cell layer was removed by 0.25% trypsin (Gibco BRL, Gaithersburg, MD, USA) three times each for 30 min. The remaining amnion tissue pieces were treated with 2 mg/ml collagenase A (Roche, Rotkreuz, Switzerland) and 0.05 mg/ml DNase (Roche) to liberate dissociated cells. The cells were cultured in Dulbecco's modified Eagle's medium low glucose (DMEM-LG, Gibco BRL) supplemented with 1% (vol./vol.) penicillin/ streptomycin solution (Gibco BRL), 3.7 mg/ml sodium bicarbonate (Sigma, St. Louis, MO, USA), and 10% (vol./vol.) fetal bovine serum (FBS) (Hyclone, Logan, UT, USA) at 37°C in 5% CO2-humidified atmosphere. After 2 weeks, culture dishes were filled with plastic-adherent, fibroblast-like cells. These cells were subcultured using trypsin/EDTA (Gibco BRL). Adherent cells at passage 3 (p3) uniformly showed fibroblastic morphology. The culture medium was changed twice a week.
Reverse-Transcription Polymerase Chain Reaction (RT-PCR)
RNA was isolated using Tri-reagent (Invitrogen, Carlsbad, CA, USA) according to the manufacturer's instructions. The purity of RNA was assessed by determining the ratio of absorbance at 260 nm to that at 280 nm (>1.8). RT-PCR was performed using a Primus 96 Universal (PeqlabBiotechnologie, Erlangen, Germany). Subsequent PCR reactions were performed using cDNA, primer pairs (Table 1) and PCR mixture (Fermentas, St. Leon-Rot, Germany) according to the manufacturer's instructions. Glyceraldehyde 3-phosphate dehydrogenase (GAPDH) was used as a loading control. Negative control was prepared by an identical manner but without cDNA. RNAs from human embryonic stem cells were kindly donated from Prof. Kim (Korea University, South Korea). Human total RNAs from pancreas (hPAN) and liver were purchased from Ambion (Austin, TX, USA).
cDNA Pairs Used in the Present Study
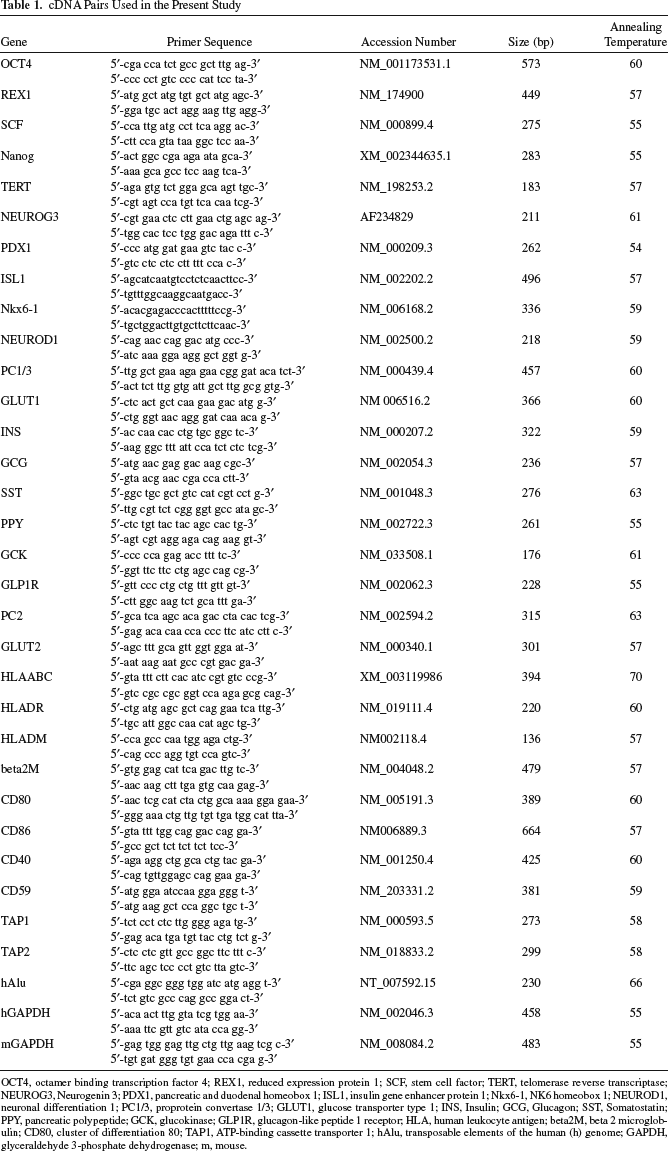
OCT4, octamer binding transcription factor 4; REX1, reduced expression protein 1; SCF, stem cell factor; TERT, telomerase reverse transcriptase; NEUROG3, Neurogenin 3; PDX1, pancreatic and duodenal homeobox 1; ISL1, insulin gene enhancer protein 1; Nkx6-1, NK6 homeobox 1; NEUROD1, neuronal differentiation 1; PC1/3, proprotein convertase 1/3; GLUT1, glucose transporter type 1; INS, Insulin; GCG, Glucagon; SST, Somatostatin; PPY, pancreatic polypeptide; GCK, glucokinase; GLP1R, glucagon-like peptide 1 receptor; HLA, human leukocyte antigen; beta2M, beta 2 microglobulin; CD80, cluster of differentiation 80; TAP1, ATP-binding cassette transporter 1; hAlu, transposable elements of the human (h) genome; GAPDH, glyceraldehyde 3-phosphate dehydrogenase; m, mouse.
Flow Cytometry
To examine cell surface antigens, undifferentiated hAMs at p4 were incubated with primary antibodies for 20 min at 4°C. After washing, cells were analyzed by fluorescence-activated cell sorter (FACS) Calibur apparatus (Beckman Coulter, Brea, CA, USA). Monoclonal antibodies for flow cytometry were purchased from the following companies: cluster of differentiation 14 (CD14), CD31, CD73, CD90, human leukocyte antigen (HLA)-ABC, and HLA-DR from BD Bioscience (Franklin Lakes, NJ, USA), TRA-1-60, stage-specific embryonic antigen (SSEA)-3, SSEA-4 from Millipore (Temecula, CA, USA), and CD105 from Invitrogen.
To evaluate the differentiation efficiency of hAMs into hAM-ISCs, hAM-ISCs were fixed with 10% formaldehyde for 10 min. After washing with phosphate-buffered saline (PBS, Invitrogen) containing 0.1% Triton X-100 (Sigma), cells were permeabilized with PBS containing 0.5% Triton X-100 for 15 min. Following washing with PBS three times, cells were incubated with polyclonal anti-human insulin antibody (Cell Signaling Technology, Boston, MA, USA) for 20 min at 4°C. After washing, cells were labeled with fluorescein isothiocyanate (FITC)-conjugated goat polyclonal secondary antibody (Abcam, Cambridge, MA, USA).
Differentiation of hAMs Into Insulin-Secreting Cells In Vitro
Cells were induced to differentiate into insulin-secreting cells according to the previous method (20). Briefly, hAMs at p3–p4 were plated on 48-well dishes (Nunc, Rochester, MN, USA) at 5 × 103 cells/well and then cultured in control or insulinogenic differentiation medium consisting of 10 mmol nicotinamide (Sigma) and 4 nmol activin A (Peprotech, Rocky Hill, NJ, USA), and 10 nmol glucagonlike peptide-1 (GLP-1, Sigma). For the first week of culture, they were grown in DMEM-HG containing 10% FBS, and during the subsequent 2 weeks, they were cultured in DMEM-LG containing 10% FBS.
Measurement of Insulin and c-Peptide
To measure the amount of human insulin and c-peptide secreted in vitro, cells were treated with DMEM-LG containing 0.5% bovine serum albumin (BSA, Sigma) for 12 h, washed with PBS, and then stimulated by DMEM-HG for 2 h at 37°C. The amount of insulin and c-peptide, a peptide fragment released from the proinsulin molecule during processing into mature insulin, in media was measured using the human insulin and c-peptide ELISA kit (Mercodia, Uppsala, Sweden) according to the manufacturer's instructions. For the measurement of human insulin and c-peptide levels in serum, blood were taken by cardiac puncture from mice, which were fasted overnight and then intraperitoneally injected with glucose (1.5 g/kg body weight), and serum levels were measured. A mouse pancreatic cell line, MIN6N8, and human dermal fibroblasts (hDFs, Lonza) were similarly cultured as in the above except that 1 × 105 cells were seeded per well of 48-well dishes. Mouse insulin and c-peptide in mouse serum were determined using mouse-specific insulin ELISA kit (Mercodia) and c-peptide ELISA kit (Yanaihara, Shizuoka, Japan).
Transplantation of hAM-ISCs Into STZ-Induced Type 1 Diabetic Mice
Female C57BL/6 mice (16–18 g, 8 weeks) were purchased from the Samtako Bio Korea (Osan, South Korea). They were maintained in accordance with the policies of the Institutional Animal Care and Use Committee of Seoul Women's University. Mice that need to be sacrificed before spontaneous death were killed by cervical dislocation as recommended by the University Committee. Type 1 diabetes was induced by a single intraperitoneal injection of STZ (180 mg/kg body weight, Sigma) in 100 mM citrate buffer (pH 4.5). After 7 days, blood glucose level was measured using an ACCUCHEK Active (Roche Diagnostics, Basel, Switzerland) following a 12-h fast. Diabetic mice with glucose level >350 mg/dl were randomly allocated to a saline-injected sham group of 12 mice, a hAMs transplant group of 12 mice, and a hAM-ISCs transplant group of 16 mice. Untreated 12 normal mice were used as a control group. A total of 1.5 × 106 cells were loaded using a PE50 polyethylene tubing (Becton Dickinson, Sparks, MD, USA) and were transplanted underneath the kidney capsule using a Hamilton syringe (Fisher, Pittsburg, PA, USA) at 14 days after STZ injection. No immunosuppressive agent was given to mice throughout the experiment. Undifferentiated hAMs and differentiated hAM-ISCs were obtained by culture of hAMs at p3 for 3 weeks in the control or differentiation medium, respectively. Blood glucose level was measured after a 12-h fast. Nephrectomy of the graft-bearing kidney was performed 6 weeks later, and blood glucose was monitored until death (n = 2). Survival curves were prepared using the Kaplan–Meier method and analyzed for a significant difference by the log-rank Mantel–Cox test using SPSS 12.0 statistical software (SPSS, Chicago, IL, USA).
Immunocytochemistry
hAMs cultivated in Lab-Tekchamber slides (Nunc) were washed with PBS and then permeabilized in 0.5% Triton X-100 for 10 min. Endogenous peroxidase activity was inactivated with 3% hydrogen peroxide (DakoCytomation, Carpinteria, CA, USA) for 15 min at room temperature (RT). Then the slides were incubated in a blocking solution, PBS containing 2% BSA, for 1 h at RT. After washing with PBS containing 0.1% BSA, cells on slides were treated with mouse anti-human insulin monoclonal antibody (1:500, Abcam) or mouse anti-human c-peptide monoclonal antibody (1:500, Monosan, AM Uden, the Netherlands) for 1 h at RT. Cells were then incubated with biotinylated goat anti-mouse or anti-rabbit IgG (Vectastatin ABC kit, Vector Laboratories, Burlingame, CA, USA) followed by horseradish peroxidase-conjugated streptavidin (Vector Laboratories) for 20 min, respectively, at RT. Immunoreactivity was visualized using 3,3′-diaminobenzidine tetrahydrochloride (DakoCytomation) and counterstained with Mayer's hematoxylin (Sigma). Negative control was prepared by an identical manner but without primary antibody. Cells were photographed under a microscope using a bright-field illumination (Axioskop2+, Carl Zeiss, Oberkochen, Germany).
Immunohistochemistry
Graft-bearing kidneys and pancreata were removed from normal and hAM-ISCs-transplanted mice at 8 weeks or 6 months posttransplantation. Tissues were fixed with 4% paraformaldehyde, embedded in paraffin, and cut into 4-μm sections followed by permeabilization with 1% Triton X-100 in PBS (PBST) for 10 min. For antigen retrieval, tissue sections immersed in 10 mM citrate buffer solution (pH 6.0) were placed in microwave for 20 min. For graft-bearing kidney tissue sections, the sections were incubated in 1% BSA in PBS at RT for 20 min. Then the sections were incubated overnight at 4°C with a mixture of two primary antibodies, mouse anti-human nuclei monoclonal (1:100, Chemicon, Temecula, CA, USA), and rabbit anti-human insulin polyclonal antibodies (1:100; Abcam) in PBST containing 1% BSA. After washing, they were incubated with another mixture of two secondary antibodies, Texas red-conjugated anti-rabbit sheep IgG (1:200, Abcam) and FITC-conjugated anti-mouse goat IgG (1:200, Abcam) for 1 h at RT. Cell nuclei were visualized by incubating for 1 min with 1 μg/ml 4′-6′ diamidino-2-phenylindole (DAPI) in PBS. Fluorescence imaging was visualized by confocal microscope (Nikon, Tokyo, Japan). For pancreatic tissue, the sections were incubated in 0.1% horse serum at RT for 20 min. Then the sections were incubated with anti-mouse insulin monoclonal antibody (1:100, Abcam) for 1 h at RT, biotin-labeled anti-mouse/rabbit house antibody and then horseradish peroxidase-conjugated streptavidin (Vectastatin ABC Kit). Counterstaining was done with Mayer's hematoxylin. Some of the sections were stained with Mayer's hematoxylin/eosin for histochemistry. Imaging was visualized by microscope (Axioskop2+).
Dithizone (DTZ) Staining
After differentiation of hAMs into insulin-secreting cells for 3 weeks in vitro, cells were stained by 100 μg/ml DTZ (Sigma) solution at 37°C for 15 min.
Intraperitoneal Glucose Tolerance Test (IPGTT)
IPGTT was performed in fasting unanesthetized mice at 6 weeks after transplantation. After baseline blood glucose measurements, mice were received an intraperitoneal injection of 1.5 g of glucose per kilogram body weight. Tail vein blood glucose concentrations were measured again 30, 60, 90, 120, and 150 min using an ACCUCHEK Active.
Statistical Analysis
Data were expressed as mean ± SEM. Statistical significance was analyzed by one-way ANOVA followed by Scheffe's test using SPSS 12.0. A value of p < 0.05 was considered to be statistically significant.
Results
Stem Cell Properties of hAMs
After the first subculture, plastic-adherent hAMs exhibited uniform, fibroblastic morphology. Flow cytometric analyses of hAMs at p4 showed that they were strongly positive for CD73, CD166, CD105, and CD90, weakly positive for SSEA-4, and negative for CD14, CD31, TRA-1–60, and SSEA-3 antigens (Fig. 1A). The hAMs also showed distinct expression of genes including octamerbinding transcription factor 4 (OCT4), Nanog, stem cell factor (SCF), reduced expression protein 1 (REX1), and telomerase reverse transcriptase (TERT) that are specific to embryonic stem cells and many somatic stem cells (Fig. 1B).

Stem cell-related properties of hAMs. (A) Flow cytometry results showing that hAMs at p4 were strongly positive for CD73, CD166, CD105, CD90, and HLA-ABC, weakly positive for SSEA-4, and negative for CD14, CD31, TRA-1–60, SSEA-3, and HLA-DR. (B) Expression of stem cell-related genes by hAMs. CD73, cluster of differentiation 73; HLA, human leukocyte antigen; SSEA-4, stage-specific embryonic antigen-4; FITC, fluorescein isothiocyanate; PE, phycoerythrin; APC, allophycocyanin; hAMs, human amniotic mesenchymal stem cells; hESCs, human embryonic stem cells; NC, negative control. See Table 1 for gene definitions.
Immunophenotypes of hAMs
RT-PCR analysis of the expression of selected immune-related genes in hAMs showed three different expression patterns during ex vivo expansion (Fig. 2A). Genes of β-2-microglobulin (β2M), CD59, ATP-binding cassette transporter 1 (TAP1), and TAP2 were consistently expressed from the primary until p13, but HLA-DM, CD80, and CD86 were not expressed in any passage. The other group of genes showed gradually decreasing expression during ex vivo expansion. HLA-ABC gene was expressed from primary culture until p12, but not expressed later; CD40 gene was expressed until p11, but not expressed since then. HLA-DR gene was expressed only in earlier passages until p3 and not expressed thereafter. Immunocytochemical analyses also showed that hAMs at p4, p8, and p13 did not exhibit expression of HLA-DR protein (Fig. 2B). In contrast, hAMs showed gradual reduction of expression of HLA-ABC protein from p4 until p13 unlike the consistent mRNA expression during the same period.
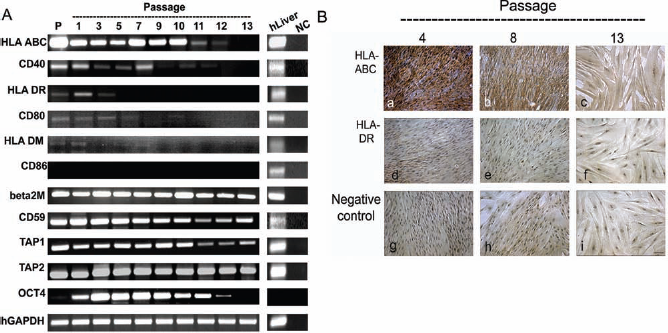
Immunogenic properties of hAMs. (A) Immune-related gene expression profile by hAMs cultured from primary culture until p13 in vitro. See Table 1 for gene definitions. (B) Immunocytochemistry of hAMs against HLA-ABC and HLA-DR. Scale bar: 100 μm. P, primary culture; hLiver, human liver total RNA; NC, negative control.
In Vitro Differentiation of hAMs Into Insulin-Secreting Cells
After differentiation culture for 3 weeks, some of hAMs formed clusters of which cells were strongly stained with DTZ, a zinc-chelating agent known to selectively stain pancreatic β cells (Fig. 3A). RT-PCR analyses revealed that both expression of β-cell-related genes, including insulin (INS), pancreatic and duodenal homeobox 1 (PDX1), NK6 homeobox 1 (Nkx6-1), neurogenin 3 (NEUROG3), insulin gene enhancer protein 1 (ISL1), neuronal differentiation 1 (NEUROD1), glucose transporter type 1 (GLUT1), GLUT2, proprotein convertase 1/3 (PC1/3), PC2, and glucokinase (GCK), and expression of pancreas-related genes, including pancreatic polypeptide (PPY), somatostatin (SST), and glucagon (GCG), were prominent in the hAM-ISCs, while undifferentiated hAMs showed little expression of these genes, except NEUROD1, GLUT1, PC2, and GCK genes (Fig. 3B). To determine whether these hAM-ISCs could secrete insulin in a high-glucose-dependent manner, we performed human insulin ELISA. Undifferentiated hAMs secreted 15.3 ± 1.4 and 16.5 ± 4.3 pg/ml, respectively, in response to 5.5 or 25 mmol glucose stimulation (Fig. 3C). In contrast, hAM-ISCs secreted 20.8 ± 3.8 and 102.4 ± 6.5 pg/ml, respectively. Under the same condition, human dermal fibroblast cells as a negative control secreted 5.0 ± 0.2 and 4.5 ± 0.2 pg/ml of insulin, respectively. Human c-peptide ELISA showed similar results as that undifferentiated hAMs secreted 18.9 ± 6.0 pg/ml to 5.5 mmol glucose and 29.0 ± 3.0 pg/ml to 25 mmol glucose stimulation, whereas hAM-ISCs released 29.3 ± 3.0 and 239.3 ± 47.7 pg/ml, respectively (Fig. 3D). Human fibroblasts secreted 29.5 ± 2.3 and 27.6 ± 4.8 pg/ml of c-peptide responding to a 5.5- or 25-mmol glucose challenge, respectively. When an insulinoma cell line MIN6N8 was similarly examined using mouse-specific ELISA kit, they secreted 3.8 ± 1.1 and 12.5 ± 1.9 ng/ml of mouse insulin and 0.8 ± 0.1 and 4.2 ± 0.1 ng/ml of mouse c-peptide upon 5.5 or 25 mmol glucose challenging, respectively (Fig. 3E). Immunocytochemical studies showed that undifferentiated hAMs were almost negative against both anti-human insulin and c-peptide antibodies, whereas hAM-ISCs, particularly cells in cluster-like structure, were strongly positive to both antibodies (Fig. 3F). However, flow cytometry analysis revealed the insulinogenic differentiation efficiency of 8.7 ± 0.2% (Fig. 4A), as shown in double immunofluorescence imaging against DAPI and anti-human insulin antibody (Fig. 4B). These results indicate that hAMs could successfully differentiate into insulin-secreting cells, hAM-ISCs, in vitro.

Differentiation of hAMs into insulin-secreting cells in vitro. Cells at p3–4 were cultivated in differentiation medium for 3 weeks and then analyzed for the β-cell characters. (A) Cells cultured in control medium (control) exhibited little staining with DTZ, whereas those in a differentiation medium (hAM-ISCs) showed intense staining. (B) Expression profile of β-cell-related genes by hAM-ISCs. hPan, human pancreatic mRNA; NC, negative control. See Table 1 for gene definitions. (C, D) ELISA analyses of insulin (C) and c-peptide (D) in response to 5.5 or 25 mmol glucose challenge. Results were obtained from three independent experiments. b indicates a significant difference (p < 0.05). (E) Mouse insulin and c-peptide level of MIN6N8 cell line in response to 5.5 or 25 mmol glucose stimulation. (F) Immunocytochemistry of hAMs (control) and hAM-ISCs against anti-human insulin or anti-human c-peptide antibody. Scale bar: 200 μm. hAM-ISCs, human amniotic mesenchymal stem cell-derived insulin-secreting cells.

Differentiation efficiency of hAMs into hAM-ISCs. (A) hAMs and hAM-ISCs were labeled with DAPI and FITC-conjugated anti-human insulin antibody followed by flow cytometric analysis. (B) Some of both types of cells were visualized under a fluorescence microscope. Arrows indicate hAM-ISCs stained with both antibodies. Scale bar: 100 μm.
Blood Glucose Level of Type 1 Diabetic Mice Could Be Normalized by Transplantation of hAM-ISCs
To examine whether hAM-ISCs could act as therapeutic cells for the type 1 diabetes, we transplanted hAM-ISCs into the kidneys of hyperglycemic mice induced by STZ pretreatment and analyzed their effects. Mice treated with saline (sham, n = 12) began to die at 18 days after transplantation, and all 12 mice died within 45 days (Fig. 5A). Mice transplanted with undifferentiated cells (hAMs, n = 12) similarly began to die at 22 days after transplantation, and all 12 mice died within 55 days. In contrast, 9 of the 16 mice that have been transplanted with differentiated cells (hAM-ISCs, n = 16) survived even for 210 days after transplantation. The rest of the mice began to die as early as 12 days, and all seven mice were dead within 65 days after transplantation. During 70 days of experimental period, mice of both sham group and hAMs group consistently showed high blood glucose level until death (Fig. 5B). In contrast, mice of the hAM-ISCs group exhibited lowered glucose level, and from 35 to 70 days of transplantation, their glucose level was not statistically different from those of normal mice (Fig. 5B). Individual recording of blood glucose level of all 16 mice exhibited by the hAM-ISCs group is shown in Figure 5C. Nine mice (Nos. 1–9) exhibited lowering of blood glucose level reaching to normal level after 7–14 days of hAM-ISCs transplantation, and all of them maintained normoglycemia until 55 days. One mouse (No. 10) exhibited similarly lowered level until 28 days but then showed hyperglycemia and eventually died after 65 days. Other six mice (Nos. 11–16) remained hyperglycemia until death. At 56 days of hAM-ISCs transplantation, graft-bearing kidneys were removed from two mice whose blood glucose level was in a range between 99 and 101 mg/dl. Following nephrectomy, their blood glucose level markedly increased reaching to 424 or 467 mg/dl during the next 7 days and eventually died after 14 and 20 days, respectively. During the experimental period, average body weight of both sham and hAMs group mice gradually decreased until death, that of hAM-ISCs group mice progressively increased similar to that of normal mice (Fig. 5D). However, individual recording of body weight of all 16 hAM-ISCs mice showed that seven mice (Nos. 10–16), which remained hyperglycemic until death, as seen in Figure. 5C, gradually lost body weight throughout the period. The rest of the mice of which blood glucose level was lowered (Nos. 1–9) progressively gained weight (Fig. 5E).

Physiological analyses of normal and type 1 diabetic mice after cell transplantation. Mice were given saline (sham), hAMs, or hAM-ISCs under their left kidney capsules. (A) Survival rate. (B) Blood glucose level. Blood glucose levels of hAM-ISC-transplanted mice for 35–70 days of experimental period were not statistically different from those of normal mice during the same period by one-way ANOVA. Nephrectomy (arrow) was done in two mice, which exhibited normalized glucose level following hAM-ISC transplantation. (C) Individual recording of blood glucose level of 16 hAM-ISC transplanted mice as shown in (B). Nos. 1 and 2 represent hAM-ISC transplanted mice that have been nephrectomized as in (B). (D) Changes of average body weight of mice. (E) Individual recording of body weight of 16 hAM-ISC transplanted mice as shown in (D). Nos. 1 and 2 represent hAM-ISC-transplanted mice that have been nephrectomized as in (B). There was no significant difference between the normal group and hAM-ISCs group based on one-way ANOVA test from 35 to 70 days after transplantation. (F) Glucose tolerance test. Normal mice and mice showing normoglycemia at 42 days following hAM-ISC transplantation were given glucose solution intraperitoneally, and their blood glucose level was monitored. N.S., no significant difference between the normal group and hAM-ISCs group based on one-way ANOVA test. Data are shown as mean ± SEM except (A).
To evaluate a physiological function of transplanted hAM-ISCs, IPGTT was performed on normal mice (n = 5), sham-operated mice (n = 2), and hAM-ISCs transplanted mice (n = 5), which exhibited normoglycemia at 42 days following transplantation (Fig. 5F). Blood glucose levels in normal mice and hAM-ISC-transplanted mice rose with peak level at 30 min, followed by a drop to initial range within 150 min. Glucose level of the hAM-ISCs transplanted mice appeared a little higher than that of the normal but was not significant. In contrast, sham mice showed absence of tolerance against glucose challenging. These results demonstrate that hAM-ISC transplantation could normalize the blood glucose level of hyperglycemia in type 1 diabetic mice.
hAM-ISCs Directly Regulate the Blood Glucose Level of Type 1 Diabetic Mice
We further investigated whether transplanted hAM-ISCs could directly regulate the blood glucose level of recipient mice via the insulin they produced, overcoming immune rejection. RT-PCR analyses showed that kidney tissues of the mice that normalized blood glucose level following transplantation with hAM-ISCs 8 weeks or 6 months earlier expressed human genes of INS, PDX1, GLUT1, glucagon-like peptide 1 receptor (GLP1R), NKX6-1, NEUROD1, NEUROG3, and hAlu (transposable elements of the human genome) (Fig. 6A). Normal mouse kidney tissue did not express any of these human genes. Immunohistochemical analyses showed the presence of a number of transplanted human cells residing within a mouse kidney capsule where they were injected (Fig. 6B). Coimmunofluorescence images of the tissue section demonstrate that most of these cells colocalized human insulin protein and human nuclei proteins (Fig. 6C). To clarify the presence and origin of insulin in the blood of the normalized mice, three randomly chosen mice whose blood glucose level ranges between 128 and 138 mg/dl following hAM-ISCs transplantation were sacrificed after 56 days. Immunohistochemical studies showed that both size and number of pancreatic islets were markedly decreased in the pancreas of these mice compared to those of the normal mice (Fig. 7A). Mouse insulin and c-peptide levels in the blood of hAM-ISCs transplant mice were negligible compared to 1.9 ± 0.1 ng/ml of mouse insulin and 3.1 ± 0.4 ng/ml of mouse c-peptide in normal mice (Fig. 7B). In contrast, human insulin and c-peptide levels in hAM-ISCs transplant mice were 0.4 ± 0.1 and 0.6 ± 0.0 ng/ml, respectively. Both proteins were hardly detected in the blood of normal mice (Fig. 7C). These results demonstrate that human insulin was solely responsible for the regulation of the blood glucose level of hAM-ISC-transplanted mice. The results also suggest that transplanted hAM-ISCs could survive and actively synthesize human insulin in the kidneys of immunocompetent mice without the aid of any immunosuppressive drugs. To investigate how hAM-ISCs could overcome immune rejection of xenogeneic grafts, we examined the expression profile of immunity-related genes (Table 2) in hAMs, hAM-ISCs, and kidney tissues of two mice that were allowed to live for 56 days or 6 months, maintaining normal blood glucose level. Immune-related genes of HLA-ABC, β2M, CD40, CD59, TAP1, and TAP2 were consistently expressed in hAMs and hAM-ISCs regardless of differentiation in vitro (Fig. 7D). However, genes of HLA-DR, CD80, and CD86 were not expressed in both types of cells. Similarly, kidney tissues of hAM-ISCs transplant mice also showed expression of β2M, TAP1, and TAP2 at 2 and 6 months after transplantation. Interestingly, CD40 transcript was not detected in both mice kidneys, whereas HLA-ABC and CD59 transcripts were seen at 2 months but disappeared after 6 months of transplantation.

Human gene and protein expression in graft-bearing kidneys. (A) Human β-cell-related gene expression in graft-bearing kidney tissues removed from mice described in Figure. 5B. GMK1, GMK2, mouse kidneys transplanted with hAM-ISCs 8 weeks or 6 months earlier, respectively; NMK, normal mouse kidney; NC, negative control. See Table 1 for gene definitions. (B) Hematoxylin/eosin-stained, graft-bearing kidney section. Arrowheads indicate boundary between transplanted hAM-ISCs residing in mouse kidney capsule and the region of mouse kidney tissue. Scale bar: 100 μm. (C) Immunofluorescence staining of the upper region of transplanted hAM-ISCs in graft-bearing kidney shown in (B). Immunostaining was done against anti-human insulin (hINS) and anti-human nuclei antibodies (hNUCLEI). Arrows indicate colocalization of both proteins. Scale bar: 50 μm. Boxes a and b are enlarged view. Scale bar: 10 μm.

Regulation of mouse blood glucose level by human insulin from transplanted hAM-ISCs. (A) Pancreata section of normal mice and mice whose blood glucose level was normalized by human insulin secreted from transplanted hAM-ISCs. (B) Mouse insulin and c-peptide in sera of normal mice and mice transplanted with hAM-ISCs. (C) Human insulin and c-peptide in sera of normal mice and mice transplanted with hAM-ISCs (n = 3). a, b are significantly different from each other, p < 0.05. (D) Expression of human immune-related genes by hAMs and hAM-ISCs in vitro and by kidneys of GMK1 and GMK2 as in Figure 6A. Pancreata section was stained by anti-mouse insulin antibodies. Scale bar: 100 μm. See Table 1 for gene definitions.
Function of Immune-Related Molecules

Discussion
Throughout this study, we demonstrate that hAMs can effectively differentiate into insulin-secreting cells in vitro and that, following transplantation into immunocompetent mice with type 1 diabetes, these cells are capable of directly normalizing blood glucose levels of the mice through secretion of their own human insulin. Moreover, the therapeutic effect has been maintained for 210 days without any immunosuppressants.
We observed that hAM-ISCs engrafted in the kidney capsule of immunocompetent mice were viable and physiologically functional even as long as 210 days overcoming immune rejection. Many studies have also reported successful engraftment of human placental membrane-derived stem cells in animal recipients without any immunosuppression. These xenogeneic cells engrafted various tissues exhibiting microchimerism after 4 months following transplantation into neonatal swine and rats (3) or engrafted both fetal and placental tissues at least 12 weeks after delivery when transplanted in utero into pregnant rats (10). In adults, they survived for 15 days when intraperitoneally infused to bleomycin-treated mice (6) or for more than 4 weeks after injection into the infarcted myocardium of rats (39). Immune-tolerating properties of human placenta-derived cells have also been demonstrated by in vitro studies. Undifferentiated hAMs have been shown to suppress allogeneic (4,24) and xenogeneic (3) immune responses of peripheral blood mononuclear cells in mixed lymphocyte reactions and suppress umbilical cord blood lymphocytes proliferation induced by allogeneic cells in vitro (25). Particularly, cells negative for HLA-DR did not induce allogeneic T-cell proliferation response while cells positive for HLA-DR induced proliferation (28). In the present study, FACS analyses showed that most hAMs were HLA-DR negative and majority of them failed to differentiate into insulin-secreting cells after differentiation culture in vitro. These undifferentiated cells, which had been transplanted along with differentiated cells, might contribute to attenuate xenogeneic immune rejection against differentiated cells and/or differentiated cells themselves could have overcome the rejection. We have observed that hAMs either before or after differentiation expressed none of the genes including HLA-DR, HLA-DM, and costimulatory genes, such as CD80 and CD86. hAM-ISCs-bearing kidneys after 210 days of transplantation maintained similar expression pattern along with discontinued expression of CD40 gene, another costimulatory protein found on antigen-presenting cells. While these properties are advantageous to protect hAMs from allogeneic rejection, it remains in question whether they also play a role in the tolerance of human xenogeneic graft.
Successful engraftment of human cells into immunocompetent animals overcoming immune rejection is more widely known in experiments using other types of adult stem cells. Transplantation of BM-MSCs into fetal sheep in early gestation could survive for 13 months, differentiating into diverse tissue cells (27). Intramuscular injection of BM-MSCs engrafted the musculature and contributed to both preexisting and new muscle fibers in hamsters 1 month later (36). Adipose-derived MSCs engrafted the injected site after 6 months of transplantation when injected into the tibialis muscle of the Duchennne muscular dystrophy (mdx) mouse (34) or engrafted the host muscles after 6 months when injected into the tail vein of mutated dysferlin (SJL) mice. which exhibit spontaneous myopathy (40). Umbilical cord-derived MSCs also could engraft and attenuate hyperoxia-induced lung injury in newborn rats (8). In this context, survival of hAM-ISCs in the kidneys of immunocompetent mice might reflect general characteristics of various MSCs whose immune-tolerating properties are common properties.
Under the present differentiation culture condition, we obtained 8.7% of insulin-positive cells in hAM-ISC population. The efficiency is a little higher than in previous reports using human embryonic stem cells (11,17). Different cell types and protocols used in both studies may account for the difference in results. We previously observed that hAMs expressed not only stem cell-specific genes, such as OCT4, REX1, and SCF, but also marker genes of primitive gut endoderm, GATA4 and hepatocyte nuclear factor 4α (HNF4A) (22). In the present study, we further found that undifferentiated hAMs expressed genes of NEUROD1, GLUT1, GCK, and PC2. NEUROD1, which are well known to play a critical role during the development of pancreatic endocrine cells from the gut endoderm, and its deletion in mice results in the severe reduction of endocrine cell formation and the eventual development of diabetes (29). GLUT1 and GCK genes play a key role in glucose sensing by pancreatic β-cells, particularly in human (12), and PC2 gene might possibly be involved in the processing of proinsulin into mature insulin. By contrast, human embryonic stem cells do not express genes of NEUROD1 (21), GCK, and PC2 (2) or barely express HNF4A (32). Therefore, hAMs appear to differentiate more efficiently into insulin-secreting cells than human embryonic stem cells in vitro.
Interestingly, immunohistochemical analyses have shown that most human cells residing in kidneys of hAM-ISC-transplanted mice were also positive for anti-human insulin antibody. Since merely 8.7% cells of in vitro differentiated hAMs before transplantation were insulin positive by FACS, a discrepancy exists between the two observations. It is possible that undifferentiated hAMs might have been removed from the injection site by an unknown mechanism or the stimulated yet not fully differentiated hAMs might have differentiated into insulin-positive cells in vivo. The in vivo differentiation capacity of human MSCs into insulin-secreting cells has been demonstrated in mice models (1,24).
We observed that sera of the normalized mice contained 0.4 ng/ml insulin and 0.6 ng/ml c-peptide, which were only about 20%, respectively, of the levels in control mice. In animals, diabetes becomes clinically apparent when more than 80% of β-cells are destroyed (30) and even very low level of insulin is able to restore normoglycemia in diabetic mice (31). In this context, the insulin level of the normalized mice might be a minimum amount to restore normoglycemia in diabetic mice.
In summary, we demonstrate that hAMs can readily differentiate into insulin-secreting cells in vitro, and the differentiated cells transplanted into the kidneys of type 1 diabetic mice can normalize hyperglycemia by releasing human insulin without inducing regeneration of mouse pancreatic β-cells. Furthermore, human insulin-secreting cells can successfully engraft in immunocompetent mice without immunosuppression, which lasts for 210 days. These results suggest that in vitro differentiated hAM-ISCs may be clinically useful for the treatment of patients with insulin-dependent diabetes. Further studies using nonobese diabetic (NOD) mice will help in the understanding of the precise mechanism of immune-privileged property of hAM-ISCs.
Footnotes
Acknowledgments
This work was supported by the Korea Science and Engineering Foundation (KOSEF) grant funded by the Korea government (MOST) (No. 2010-0011979) and a grant (11172 KFDA268) from Korea Food and Drug Administration in 2011. The authors declare no conflicts of interest.
