Abstract
Cell therapy is expected to restore perfusion and improve function in the ischemic/infarcted myocardium; however, the biological mechanisms and local effects of transplanted cells remain unclear. To assess cell fate in vivo, hexadecyl-4-[18F]fluorobenzoate (18F-HFB) cell labeling was evaluated for tracking human circulating progenitor cells (CPCs) with positron emission tomography (PET) and was compared to the commonly used 2-[18F]fluoro-2-deoxy-d-glucose (18F-FDG) labeling method in a rat myocardial infarction model. CPCs were labeled with 18F-HFB or 18F-FDG ex vivo under the same conditions. 18F-HFB cell-labeling efficiency (23.4 ± 7.5%) and stability (4 h, 88.4 ± 6.0%) were superior to 18F-FDG (7.6 ± 4.1% and 26.6 ± 6.1%, respectively; p < 0.05). Neither labeling approach significantly altered cell viability, phenotype or migration potential up to 24 h postlabeling. Two weeks after left anterior descending coronary artery ligation, rats received echo-guided intramyocardial injection in the infarct border zone with 18F-HFB-CPCs, 18F-FDG-CPCs, 18F-HFB, or 18F-FDG. Dynamic PET imaging of both 18F-HFB-CPCs and 18F-FDG-CPCs demonstrated that only 16–37% of the initial injection dose (ID) was retained in the injection site at 10 min postdelivery, and remaining activity fell significantly over the first 4 h posttransplantation. The 18F-HFB-CPC signal in the target area at 2 h (23.7 ± 14.7% ID/g) and 4 h (17.6 ± 13.3% ID/g) postinjection was greater than that of 18F-FDG-CPCs (5.4 ± 2.3% ID/g and 2.6 ± 0.7% ID/g, respectively; p < 0.05). Tissue biodistribution confirmed the higher radioactivity in the border zone of 18F-HFB-CPC rats. Immunostaining of heart tissue sections revealed no significant difference in cell retention between two labeled cell transplantation groups. Good correlation with biodistribution results was observed in the 18F-HFB-CPC rats (r = 0.81, p < 0.05). Compared to 18F-FDG, labeling human CPCs with 18F-HFB provides a more efficient, stable, and accurate way to quantify the distribution of transplanted cells. 18F-HFB cell labeling with PET imaging offers a better modality to enhance our understanding of early retention, homing, and engraftment with cardiac cell therapy.
Introduction
Despite considerable advances in cardiovascular therapeutics over the past few decades, our ability to effectively treat myocardial infarction and subsequent congestive heart failure remains limited by the fact that necrotic or scarred myocardium cannot be restored. Cell-based regenerative therapy is a promising strategy for myocardial repair. Initial animal studies and clinical trials have demonstrated that intramyocardial injection of circulating progenitor cells (CPCs) can restore regional perfusion and improve function in the ischemic/infarcted myocardium (6,9). However, further understanding the biological mechanisms and local effects of transplanted cells remains mostly restricted to postmortem histological assessment (28), which is a single time point analysis and unpractical with human subjects. Little quantitative data on the biodistribution and in vivo kinetics of transplanted stem/ progenitor cells in the myocardium and the whole body have been reported. Therefore, the development of non-invasive imaging approaches for monitoring the fate and tissue distribution of transplanted cells would provide useful insights.
Positron emission tomography (PET) is a well-developed tool for studying myocardial blood flow, metabolism, and cardiac function and is proving to be useful for evaluating cell therapeutic responses. As an advanced nuclear imaging technology, PET combines high sensitivity, good spatial resolution, and whole-body imaging to track transplanted stem cells (29). Therefore, PET can be used as a noninvasive imaging technique to assess cell fate longitudinally. Moreover, PET provides the potential for quantification of cell numbers in specific tissues of interest. PET imaging techniques have been proposed and evaluated in several cell delivery models (1,2,7). To implement PET cell imaging, it is important to develop a stable radiolabeling method without altering cell viability or function. One common approach to assess the distribution pattern of transplanted cells with PET is 2-[18F]fluoro-2-deoxy-d-glucose (18F-FDG) cell labeling (7). Our group has used this technique to evaluate the ability of tissue-engineered matrices to improve the early retention of transplanted human CPCs in an ischemic hindlimb model (30). Although 18F-FDG-labeled cells could be detected by PET, we and others have also observed significant leakage of 18F-FDG from labeled cells (1,30).
Hexadecyl-4-[18F]fluorobenzoate (18F-HFB), a lipophilic long-chain ester derivative, is a novel radioligand that is absorbed into the cell membrane and thus might be a superior cell tracker for PET imaging. Recently, it has been used to image rat mesenchymal stem cells (MSCs) by micro-PET after intravenous injection to healthy animals (12). However, use of 18F-HFB to label human cells for the assessment of cardiac cell therapy has not been reported. In addition, it has not been compared to other commonly used cell tracking methods, such as 18F-FDG, nor has its cytotoxic effect on labeled cells been evaluated.
In this study, we evaluated 18F-HFB labeling properties for monitoring intramyocardially injected CPCs with PET imaging in a rat myocardial infarction (MI) model. Also, we assessed 18F-HFB cytotoxicity and the possibility of 18F-HFB labeling for the evaluation of human CPCs delivered in a collagen matrix. All in vitro and in vivo studies of 18F-HFB labeling were compared to an 18F-FDG labeling approach.
Materials and Methods
Radiotracer Synthesis
18F-HFB was synthesized by a three-step procedure according to previously described methods (12). Briefly, the triflate precursor was synthesized after esterification of 4-(N,N-dimethylamino)benzoyl chloride (Sigma-Aldrich, Oakville, Canada) with 1-hexadecanol (Sigma-Aldrich). 18F-HFB was produced by 18F substitution and purified by high-performance liquid chromatography with 20–40% yield (decay corrected) and high radiochemical purity (> 99%). Pure 18F-HFB was dissolved in 10% dimethyl sulfoxide (DMSO)/saline.
Cell Isolation and Culture
Human CPC procurement was approved by the Human Research Ethics Board of the University of Ottawa Heart Institute. After acquiring informed consent from healthy human donors, total peripheral blood mononuclear cells (PBMNCs) were freshly isolated from human peripheral blood by Histopaque 1077 (Sigma-Aldrich) density-gradient centrifugation. Subsequently, PBMNCs were cultured on fibronectin-coated plates in endothelial basal medium (EBM-2; Clonetics, Guelph, Canada) supplemented with endothelial growth medium (EGM)-2-MV-SingleQuots [5% fetal bovine serum, vascular endothelial growth factor (VEGF), insulin-like growth factor-1, and human epidermal growth factor]. After 4 days, the adherent population (CPCs) was collected as described previously (20).
Cell Radiolabeling
Harvested CPCs were incubated with 18F-HFB for 30 min in a sterile centrifuge tube under various conditions, including different dose ranges of 18F-HFB (0.4–0.6, 1.5–2.5, or 5.0–7.0 mCi), different concentrations of CPCs (2.0 × 106 cells in 1, 3, or 5 ml PBS), and different temperatures (4°C, 22°C, or 37°C). In parallel experiments, CPCs were labeled with 18F-FDG under these same conditions. At the end of incubation, cells were washed to remove unbound radioactivity. The radioactivity was measured both in cells and in the supernatant by using a dose calibrator (Capintec, Ramsey, NJ). With correction for radiolabel decay, cell-labeling efficiency was calculated as the activity in cells over the total activity used in the incubation.
Cell Labeling Stability
Radiolabeled cells were resuspended in 2 ml PBS and incubated at 37°C for 2 and 4 h. The retention of 18F-HFB or 18F-FDG within CPCs was calculated.
Viability of Labeled CPCs
The relative survival of labeled cells was evaluated by using the Vi-CELL analyzer (Beckman Coulter, Mississauga, Canada) with a Trypan Blue Dye Exclusion Method. Radiolabeled or unlabeled CPCs (1 × 106) suspended in 1.0 ml EBM were plated, and viability was assessed after an incubation of 2 h, 4 h, 24 h, or 5 days. Cell viability was also measured with CPCs incubated with nonradioactive HFB.
Phenotype of Labeled CPCs
The phenotype of 18F-HFB-CPCs and 18F-FDG-CPCs was assessed by flow cytometry analysis (FACSAria cell sorting system; BD Biosciences, San Jose, CA) using CD144, vascular endothelial growth factor receptor-2 (VEGFR-2), L-selectin (CD62L), and CD34 antibodies (Becton Dickinson, Mississauga, Canada) at 24 h postlabeling.
Migratory Potential of Labeled CPCs
In vitro cell migration assays were performed as described previously (31). Briefly, 18F-HFB-CPCs, 18F-FDG-CPCs, or unlabeled controls were incubated with 4′6-diamidino-2′-phenylindole (DAPI, Molecular Probes, Eugene, OR), and then 2 × 104 cells were placed separately in the upper chamber of a modified Boyden chamber with VEGF-free media. The lower chamber contained serum-free media with 50 ng/ml VEGF (Sigma-Aldrich). After 24 h of incubation, cells that migrated into the lower chamber were counted in six random high-powered fields.
Retention of 18F-HFB in Matrices
Collagen-based matrices (pH ~7.5) consisted of a mixture of blended neutralized type I rat tail tendon collagen (0.4%, wt/vol; Becton Dickinson, Mississauga, Canada) and chondroitin 6-sulfate (1:6, wt/wt; Sigma), crosslinked with 0.02% (vol/vol) glutaraldehyde (30). 18F-HFB-CPCs (1 × 106, in 50 μl PBS) or 18F-HFB alone was added to the matrix solution (450 μl) on ice. Matrix mixture was plated and incubated at 37°C for 60 min to allow complete gelation. Matrix gels were then transferred into 2 ml PBS and incubated at 37°C for 2 h. Before and after incubation, the radioactivity in matrices was measured.
Myocardial Infarction Animal Model
Procedures were performed with the approval of the University of Ottawa Animal Care Committee, in accordance with the Canadian Council on Animal Care's Guide to the Care and Use of Experimental Animals. Sprague–Dawley rats (8–9 weeks old, Charles River, Wilmington, MA) were anesthetized (2% isoflurane, inhaled), intubated, and ventilated, and then the heart was exposed via a sternotomy. Subsequently, a 7–0 suture was placed in the anterior myocardium at one third of the length of the heart, towards the apex, so that the left coronary anterior descending artery was completely ligated to induce MI (23). After 2 weeks, the survival for all animals was 77.3%.
Echo-Guided Cell Delivery
Two weeks after ligation, MI rats were anesthetized and anchored in a supine position. Echocardiography was performed with a Vevo770 system (VisualSonics, Toronto, Canada) in B mode with the use of a 716 series real-time microvisualization (RMV) scanhead probe. Rats randomly received one of the following treatments in 50 μl PBS (n = 8/group), administered by echo-guided intramyocardial injection into the infarct border zone with a 27-gauge needle: (1) 2 × 106 18F-HFB-CPCs, (2) 2 × 106 18F-FDG-CPCs, (3) 18F-HFB, or (4) 18F-FDG. To this end, the syringe was secured in a micromanipulator (VisualSonics), and both the needle and RMV scanhead probe were aligned before the injection procedure (18,27). The needle was retracted from the ultrasound field of view with the use of the micromanipulator until the needle tip was in the desired location within the heart, which was proximal to the infarct area (defined by poor wall motion). The cell mixture was then injected into the border zone of the anterior wall.
PET Imaging for Localization of Labeled CPCs In Vivo
For some animals (n = 5/group), 18F-PET images were acquired using the Inveon small animal PET scanner (Siemens, Knoxville, TN) for cell localization at 10 min, 2 h, and 4 h postinjection. 13N-NH3 PET imaging was also performed for anatomic delineation of the heart and liver. Image reconstruction resulted in an image resolution of approximately 1.3 mm. 18F-microPET image data were analyzed in three cardiac axes (axial, coronal, and sagittal) with the Inveon Research Workplace and fused with 13N-NH3 images. Regions of interest (ROIs) were manually drawn around the injection site, normal myocardium, lung, and liver by visual inspection. The same ROIs were used at the three scan time points. The radioactivity of each ROI was expressed as percentage of the injected dose per gram (% ID/g) for all tissues, and the percentage of the injected dose (% ID) was also calculated in the MI border zone as % ID/g × ROI volume (cm3, assuming a tissue density of 1 g/cm3) to determine the relative retention of injected cells in the target site.
Biodistribution of Labeled CPCs
After PET scan completion (at 5 h postinjection), rats were sacrificed and tissues were harvested. Biodistribution of the radioactivity accumulation in different tissues was determined by a gamma counter (PerkinElmer Life and Analytical Sciences, Waltham, MA), and the tissues were weighed. Data were expressed as % ID/g.
Immunofluorescence Assessment of Transplanted Human CPCs
Hearts were sliced into three sections: the infarct center, the border zone, and the normal myocardium. Tissue samples were fixed with 4% paraformaldehyde and embedded in paraffin, and slides were prepared in 5-μm serial sections. Transplanted human CPCs were localized by immunofluorescence staining with anti-human mitochondria antibodies (1:40; Chemicon, Temecula, CA). The sections were mounted with mounting medium containing DAPI (Vector Laboratories, Burlingame, CA). The percentage of transplanted human cells [(number of human mitochondria+DAPI+ cells / number of DAPI+ cells) × 100] per field of view (/FOV) was calculated from four random slides of each section as described previously (30).
Statistical Analysis
Data are expressed as mean ± standard deviation (SD). Statistical analyses between groups were performed with a one-way analysis of variance, with Bonferroni corrections as appropriate. Correlation analyses were performed by linear regression. Differences with p < 0.05 were considered statistically significant.
Results
Ex Vivo CPC-Labeling Efficiency
The factors influencing cell-labeling efficiency were studied to determine the optimal labeling conditions for in vivo tracking studies. There was no correlation between the cell uptake and the amount or the concentration of radiotracer in the incubation (total volume: 1 ml; cell number: 2 × 106) for both 18F-HFB (p = 0.78, n ≥ 5/dose range) and 18F-FDG (p = 0.46, n ≥ 4/dose range) groups (Fig. 1A).
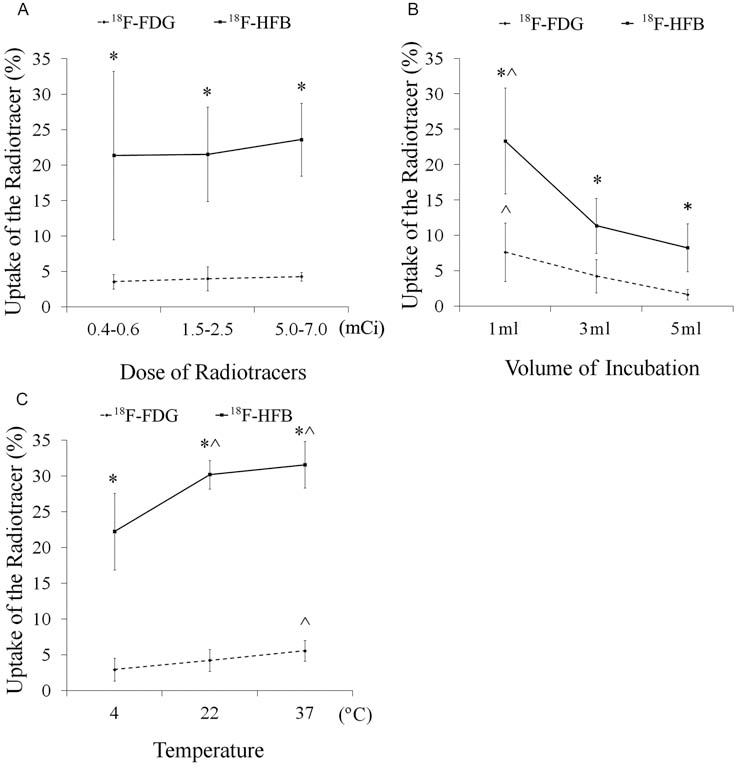
Cell-labeling efficiency of 18F-HFB and 18F-FDG. (A) The effect of radiotracer dose on labeling efficiency. (B) The effect of cell concentration on labeling efficiency. (C) The effect of temperature on labeling efficiency (*p < 0.05 vs. 18F-FDG labeling; ^p < 0.05 vs. other conditions within group; n ≥ 4/group). FDG, fluoro-2-deoxy-d-glucose; HFB, hexadecyl-4-fluorobenzoate.
CPCs (2 × 106) were incubated with 18F-HFB or 18F-FDG in three different volumes (1, 3, and 5 ml) resulting in different cell concentrations (2 × 106, 0.7 × 106, and 0.4 × 106 CPCs/ml). Labeling efficiency in 5 ml was 8.3 ± 3.4% (18F-HFB) and 1.6 ± 0.7% (18F-FDG), and in 3 ml was 11.4 ± 3.9% (18F-HFB) and 4.2 ± 2.3% (18F-FDG). As shown in Figure 1B, labeling efficiency was significantly increased for both 18F-HFB and 18F-FDG labeling when incubation was performed in 1 ml (23.4 ± 7.5% and 7.6 ± 4.1%, respectively; p < 0.05). Due to no effect of radiotracer concentration on cell labeling, a cell concentration-dependent incorporation of 18F-HFB labeling (r = 0.81, p < 0.05) was confirmed with an overall labeling efficiency of 4.2–48.4%.
Three different temperatures (4°C, 22°C, and 37°C) were also tested. The highest retention of 18F-HFB (31.6 ± 3.2%) and 18F-FDG (5.6 ± 1.4%) in CPCs was obtained when labeling occurred at 37°C (Fig. 1C).
On the basis of these results, in subsequent in vitro and in vivo experiments, 2 × 106 CPCs were incubated with 2 mCi of radiotracer (18F-HFB or 18F-FDG) in 1 ml at 37°C. Overall, with any given condition, 18F-HFB-labeling efficiency was better than that of 18F-FDG (all p < 0.05).
Labeling Stability
To evaluate the efflux rate of 18F-HFB and 18F-FDG from the cells, the retention of radioactivity in labeled CPCs was measured. After a 2-h or 4-h incubation, the retention of 18F-HFB in labeled CPCs (91.6 ± 5.1% and 88.4 ± 6.0%, respectively) was significantly higher compared to 18F-FDG-CPCs (42.5 ± 4.3% and 26.6 ± 6.1%; p < 0.05), demonstrating greater labeling stability of 18F-HFB-CPCs (Fig. 2).

Stability of radiolabeling in 18F-HFB-CPCs and 18F-FDG-CPCs. After a 2-h or 4-h incubation, the retention of 18F-HFB in labeled CPCs was significantly higher than 18F-FDG-CPCs (*p < 0.05 vs. 18F-FDG-CPCs; n = 5/group). CPCs, circulating progenitor cells.
Cytotoxicity of Labeling in CPCs
To investigate the effect of radiolabeling on cells, CPC viability at 0 h, 2 h, 4 h, 24 h, and 5 days postlabeling was measured (Fig. 3A). There were no significant differences among 18F-HFB-CPCs, 18F-FDG-CPCs, and the unlabeled controls in the first 4 h. At 24 h postlabeling, however, a reduction in 18F-HFB-CPC viability (65.8 ± 13.3%) was observed compared to the controls (81.8 ± 6.9%, p < 0.05). After 5 days, there was a loss in viability for both 18F-HFB-CPCs (62.5 ± 10.4%) and 18F-FDG-CPCs (72.1 ± 14.7%) compared to the unlabeled cells (84.0 ± 13.3%, p < 0.05). To further investigate whether the increased radioactivity in 18F-HFB-CPCs or the HFB compound itself causes cell death, we also incubated CPCs with nonradioactive HFB. There was no significant difference between HFB-CPCs (68.7 ± 7.0%) and 18F-HFB-CPCs in 24-h cell viability. However, viability of HFB-CPCs (69.7 ± 14.7%) at 5 days was better than 18F-HFB-CPCs, but less than the controls, suggesting that both increased radioactivity and HFB itself contributed to the death of 18F-HFB-CPCs.

The effect of radiolabeling on cell viability and phenotype. (A) Labeled cell viability. (*p < 0.05 vs. untreated CPCs; ^p < 0.05 vs. 18F-FDG-CPCs; #p < 0.05 vs. HFB-CPCs; n = 6 per group). (B) Representative flow cytometric analysis of cell phenotype at 24 h postlabeling. The expression of cluster of differentiation 144 (CD144), vascular endothelial growth factor receptor 2 (VEGFR-2), CD62L, and CD34 on the unlabeled CPCs, 18F-FDG-CPCs, and 18F-HFB-CPCs.
The phenotype of labeled CPCs was confirmed at 24 h postlabeling by cell surface marker expression analysis of CD144+, VEGF-R2+, L-selectin (CD62L)+, and CD34+ (Fig. 3B and Table 1). There was no significant difference among these three groups for any of the markers (all p ≥ 0.30), suggesting that neither radiolabeling method affected the phenotype of CPCs.
Cell Phenotypes and Function at 24 h Postlabeling

n = 6 per group. CD144, cluster of differentiation 144; VEGF-R2, vascular endothelial growth factor receptor 2; CPCs, circulating progenitor cells; FDG, fluoro-2-deoxy-d-glucose; HFB, hexadecyl-4-fluorobenzoate.
To define whether radiolabeling induces the cytotoxicity in cell function, in vitro migration potential of labeled cells was tested at 24 h and 5 days postlabeling (Fig. 4). The number of migrating cells/FOV was not significantly different between 18F-HFB-CPCs, 18F-FDG-CPCs, and unlabeled controls after 24 h (p = 0.90) and 5 days (p = 0.81), indicating the functional activity of human CPCs to migrate in response to VEGF in an in vitro assay was not affected by 18F-HFB or 18F-FDG radiolabeling.

The effect of radiolabeling on cell migration potential. There was no significant difference in the migrated cell number per view between 18F-HFB-CPCs, 18F-FDG-CPCs, and unlabeled controls at 24 h (p = 0.90) and 5 days (p = 0.81) after labeling.
In Vitro Retention of 18F-HFB or 18F-HFB-CPCs in the Collagen Matrix
To identify whether collagen matrices retain 18F-HFB, the retention of 18F-HFB in matrices was measured in vitro. When 18F-HFB was added directly to the matrix, 65.9 ± 5.4% 18F-HFB was retained in the matrices after its gelling. After a 2-h incubation, 93.4 ± 1.4% of this retained activity was still in the matrix, while the radioactivity of 18F-HFB-CPCs within the matrix was 90.5 ± 6.0% and the radioactivity of 18F-HFB-CPCs alone was 86.6 ± 3.5%, with no significant difference among these three groups (p = 0.14) (Fig. 5). The retained radioactivity of 18F-HFB alone without cells in the collagen matrix was significantly higher than that of 18F-FDG alone in the matrix (4.1 ± 1.0%, p < 0.005).

18F-HFB retention in matrices. There was no difference between 18F-HFB + matrix, 18F-HFB-CPCs, and 18F-HFB-CPCs + matrix. However, retention of 18F-HFB in the matrix was significantly higher than that of 18F-FDG (*p < 0.05 vs. all 18F-FDG groups; ^p < 0.05 vs. 18F-FDG + matrix; n ≥ 4/group).
Dynamic PET Imaging and Cell Kinetics of Transplanted CPCs in Myocardium
The range of radioactivity of injected 18F-HFB-CPCs was 0.6–10.9 MBq (0.02–0.29 mCi) and cell viability was 86.6–94.3%, while the range of activity of 18F-FDG-CPCs was 0.5–3.5 MBq (0.01–0.10 mCi) and cell viability was 88.1–93.5%.
After cell delivery, PET images of rats were acquired (Fig. 6A). Dynamic PET imaging (Fig. 6B) delineated the myocardial infarct area (ammonia images) and demonstrated the kinetics of transplanted CPCs (fusion images) in the myocardium and other organs (such as liver and lungs). Only a small portion of transplanted cells was retained in the injection site of both 18F-HFB-CPC (11.8 ± 7.7% ID) and 18F-FDG-CPC (11.0 ± 8.9% ID) rats at 10 min posttransplantation. In images of the 18F-HFB-CPC rats, radioactivity in the infarct border zone progressively declined to 6.9 ± 3.5% ID after 2 h and 4.7±2.7% ID after 4 h. However, this was greater than that observed for 18F-FDG-CPCs (2.5 ± 1.2% ID and 1.4 ± 0.5% ID at 2 and 4 h, respectively; p < 0.05) (Fig. 6C).
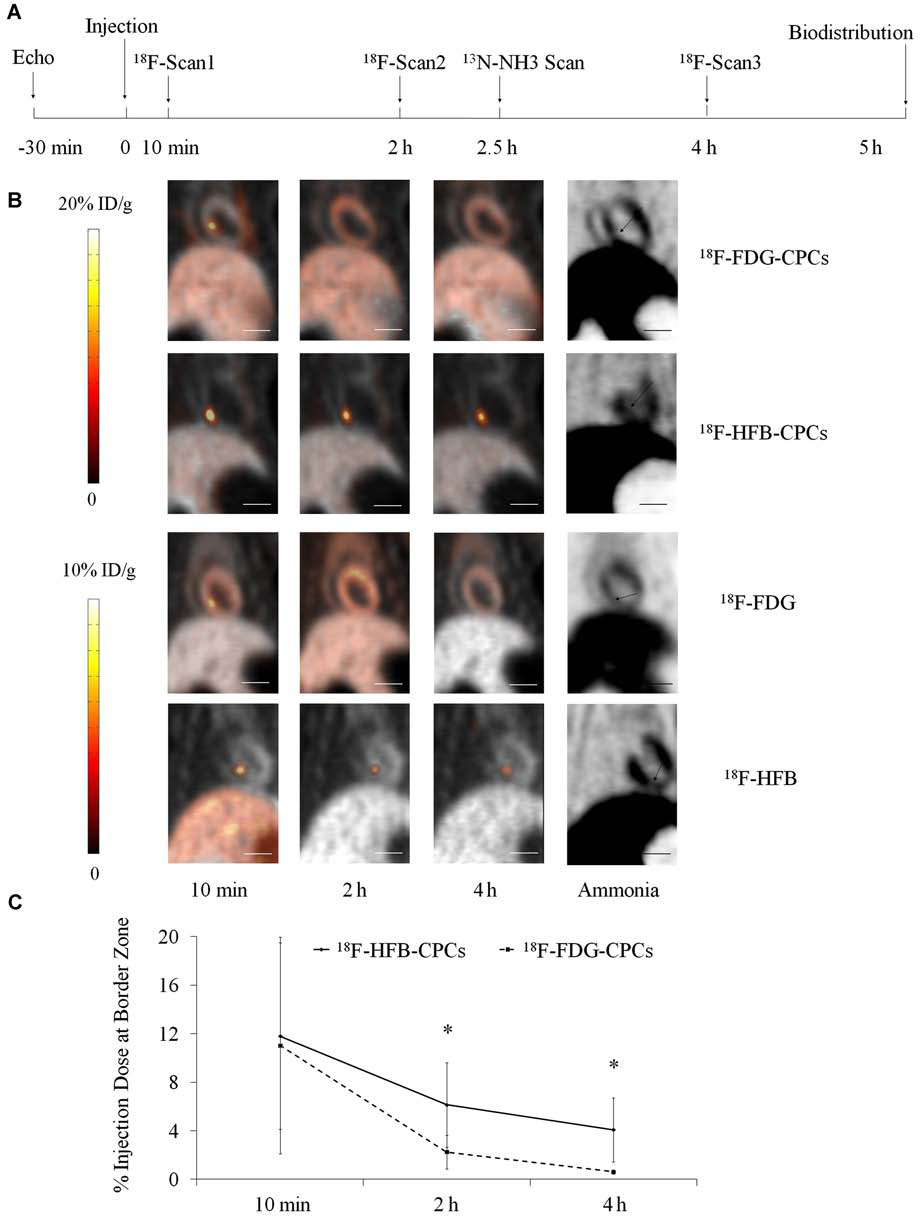
Dynamic PET imaging of labeled-CPCs after intramyocardial injection. (A) Flow diagram of in vivo study. (B) PET fusion image (coronal view) of 13N-NH3 (gray) and 18F (color) indicated the distribution of transplanted cells or radiotracers in the MI rat at 10 min, 2 h, and 4 h postinjection; 13N-NH3 image showing reduced myocardial perfusion in the infarct area, (arrows). Scale bar: 10 mm. (C) Kinetics of transplanted CPCs at the border zone of infarcted myocardium (*p < 0.05 vs. 18F-FDG-CPCs, n = 5/group). PET, positron emission tomography; ID, injection dose.
Outside the injection site, some activity was present in the normal myocardium, lungs, liver, and other tissues (Fig. 7). At 10 min postinjection (Fig. 7A), a transient high lung uptake was observed in 18F-HFB-CPC (2.3 ± 0.9% ID/g) and 18F-FDG-CPC (2.3 ± 1.9% ID/g) rats. In animals treated with 18F-HFB alone, a different tracer distribution was observed, with an obvious high liver activity (5.1 ± 2.4% ID/g), whereas high uptake was visible in the normal myocardium of 18F-FDG rats (1.3 ± 0.3% ID/g). At 2 h postinjection (Fig. 7B), the most notable change was the relocation of activity from the injection site to the normal myocardium of 18F-FDG-CPC rats. At 4 h postinjection (Fig. 7C), retention of 18F-HFB-CPCs in the border zone (17.6 ± 13.3% ID/g) was still observed. In contrast, there was no significant difference between the activity of injection site and normal myocardium of 18F-FDG-CPC rats (2.6 ± 0.7% ID/g vs. 2.4 ± 0.4% ID/g). In both 18F-HFB-CPC and 18F-FDG-CPC groups (1.6 ± 0.6% ID/g and 1.9 ± 1.1% ID/g, respectively), a slightly higher uptake was detected in the liver compared to 18F-HFB and 18F-FDG rats.
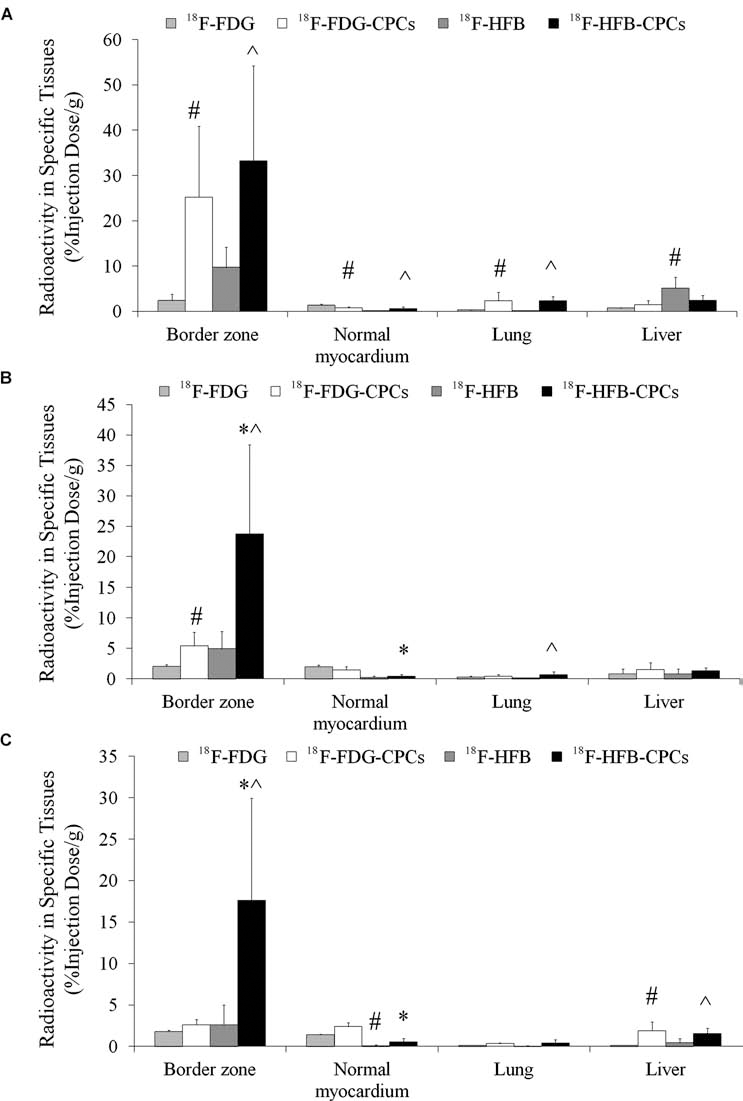
The dynamic distribution of transplanted CPCs. PET images indicated radioactivity measured at the border zone, normal myocardium, lung, and liver at (A) 10 min postinjection, (B) 2 h postinjection, (C) 4 h postinjection (*p < 0.05 vs. 18F-FDG-CPCs; ^p < 0.05 vs. 18F-HFB; #p < 0.05 vs. 18F-FDG; n = 5/group).
Radionuclide Biodistribution of CPCs in Rats
Consistent with the in vivo 18F-HFB-CPC images, the majority of radioactivity was detected in the border zone at 5 h after intramyocardial injection of CPCs. Tissue radionuclide biodistribution (Fig. 8) indicated that 17.2 ± 8.9% ID/g was detected in the border zone of 18F-HFB-CPC rats, compared to only 3.1 ± 1.5% ID/g in 18F-FDG-CPC rats (p < 0.05). No significant radioactivity was detectable in the normal myocardium of 18F-HFB-CPC (0.7 ± 0.6% ID/g) and 18F-HFB (0.5 ± 0.4% ID/g) rats, whereas 18F-FDG-CPC (3.0 ± 1.6% ID/g) and 18F-FDG (3.7 ± 1.7% ID/g) rats showed relative high uptake in this region. Outside the border zone, liver, spleen, lung, and bone marrow showed cell accumulation in cell transplantation rats.

Biodistribution of radioactivity in different organs at 5 h postinjection. The activity of 18F-HFB-CPCs in the border zone of infarcted myocardium was greater than that of 18F-FDG-CPCs (*p < 0.05 vs. 18F-FDG-CPCs; ^p < 0.05 vs. 18F-HFB; #p < 0.05 vs. 18F-FDG).
Detection of Transplanted CPCs by Immunofluorescence
To confirm the results obtained by PET imaging and radiotracer biodistribution and to gain further insight into transplanted CPC incorporation in the myocardium, immunofluorescence was performed on heart tissue sections. In the heart of cell-transplanted rats, injected CPCs (human mitochondria+ cells) were found predominantly in the border zone (Fig. 9A, B). In contrast, transplanted cells were rarely detectable in the infarct scar and normal myocardium. There was no significant difference in the percentage of transplanted cells/FOV at the border zone between 18F-HFB-CPC rats (3.0 ± 0.7%) and 18F-FDG-CPC rats (2.6 ± 0.6%, p = 0.21). To further evaluate the accuracy of 18F-HFB and 18F-FDG labeling, a correlation between immunostaining and biodistribution results was analyzed. Better correlation was observed for the 18F-HFB-CPC group (r = 0.81, p < 0.05) (Fig. 9C) compared to that of 18F-FDG-CPCs (r = 0.51, p < 0.05) (Fig. 9D).

Immunofluorescence identification of transplanted cells in the border zone of infarcted myocardium. (A, B) Transplanted CPCs (arrows) in the border zone of (A) 18F-HFB-CPC and (B) 18F-FDG-CPC rats identified by specific anti-human mitochondria (red, cytoplasm) and DAPI (blue, nucleus). Scale bar: 12.5 μm. (C, D) Correlation between immunofluorescence and radionuclide biodistribution of (C) 18F-HFB-CPCs and (D) 18F-FDG-CPCs in rat hearts. A better correlation was observed for the 18F-HFB-CPC group.
Discussion
The results of our study demonstrate that labeling with 18F-HFB is a feasible method to assess the homing and tissue distribution of transplanted human CPCs in vivo with PET. Labeling CPCs with 18F-HFB provided a more efficient, stable, and accurate way to quantify cell distribution compared to the commonly used 18F-FDG- labeling method. PET imaging of 18F-HFB-CPCs revealed a significant cell washout from the myocardium immediately after intramyocardial injection, with only a small proportion (2–7%) of transplanted CPCs remaining in the border zone of infarcted myocardium 4 h after delivery.
Consistent with previous use in rat MSCs (12), 18F-HFB uptake into human CPCs was modest (12–48%), and 18F-HFB is efficiently retained in CPCs over 4 h (77–95%) in vitro. Optimal labeling efficiency was obtained at 37°C and high cell concentration conditions. In the comparison study, 18F-FDG, the widely available radiotracer, showed a low labeling efficiency of less than 10%, with a high efflux (only 22–33% retained in CPCs) over the same time period. Overall, the labeling efficiency and stability for 18F-HFB was significantly higher than 18F-FDG. This may be due in part to the uptake and retention mechanism of 18F-FDG, which depends on cell glucose transporters. 18F-FDG remains trapped within the cells as the fructose-6 phosphate analogue (18F-FDG-6P), except when it is dephosphorylated to 18F-FDG by glucose-6-phosphatase (15). Consequently, the 18F-FDG-labeling method is likely better suited for cells with low levels of glucose-6-phosphatase; and this may partly explain the difference in labeling efficiency of 18F-FDG in various cell types (7,16). In contrast, 18F-HFB, a lipophilic molecule, incorporates within the cell membrane and is independent of any receptor-mediated effect. The calculated logP value for 18F-HFB is 10.189 (for 18F-FDG, it is −2.0085); since a high logP value is indicative of hydrophobic molecules that cannot be dissolved directly in an aqueous buffer, it is easily understandable that 18F-HFB sticks and is retained in the lipophilic cell membrane. The distribution and metabolism of 18F-HFB within the labeled cells will help us to better understand this novel labeling method and might be one of our future research directions.
PET imaging demonstrated a greater signal of 18F-HFB-CPCs in the injection site compared with 18F-FDG-CPCs a few hours after cell transplantation. There was no significant difference in the distribution of 18F-HFB-CPCs and 18F-FDG-CPCs at 10 min postinjection. However, after 2 and 4 h, the signal intensity of 18F-HFB-CPCs was much greater than that of 18F-FDG-CPCs. In contrast, the radioactivity was high in the normal myocardium of 18F-FDG-CPC rats at this time period. However, immunofluorescence examination revealed that transplanted human CPCs were rare in the normal myocardium. We consider the well-known efflux of 18F-FDG from labeled cells as the main reason for the high uptake in this area. Consistent with the PET images of free 18F-FDG, the normal myocardium exhibited a high portion of activity that was unbound 18F-FDG and not transplanted 18F-FDG-CPCs. We also found that 18F-HFB radioactivity was rarely taken up by infarcted and normal myocardium. Therefore, 18F-HFB cell labeling may be a more reliable method for assessing the distribution of transplanted human CPCs, especially in the myocardium.
PET imaging indicated an 18F-HFB-CPC-associated distribution of radioactivity, which is markedly different from the distribution of the free 18F-HFB after intramyocardial injection. Up to 4 h after injection, radioactivity was predominantly located in the border zone (injection site) of 18F-HFB-CPC rats. Immnunostaining revealed many intact CPCs in this region and good correlation between the histological examination and tissue radionuclide biodistribution results. Recently, many advanced molecular techniques have been developed, such as real-time PCR, which is more accurate and sensitive to quantify cells (17) and might be a perfect complement to traditional confirmatory approaches in the future studies. Although few transplanted cells were found in the infarct scar area, cytokines and growth factors might recruit transplanted cells to the injured territory (21,26). It is known that acute ischemia enhances the expression of chemoattractants and cytokines mediating homing of CPCs. However, the infarct center may lack living cells and thus may not furnish the necessary instructive signals and/or blood flow for the delivery of oxygen and nutrients needed for transplanted cell survival and differentiation (5). Therefore, the border zone might be the better region for stem cell delivery, homing, and engraftment.
Our findings also demonstrated a negative effect of 18F-HFB on cell viability after 24 h and 5 days, although no significant alteration was observed in cell phenotype and migration ability at these time points. Further investigation indicated free HFB is partly responsible for the observed cell death. We also found that labeling with 18F-HFB using the same concentration of CPCs (2 × 106 cell/ ml), no reduction of cell viability occurred in the lower radioactivity group (0.4–0.6 mCi), but a higher radioactivity (5–7 mCi) resulted in significant cell death (data not shown). Although the mean labeling efficiency was not changed, the radioactivity in single cells was different. In accordance with previous studies (3,4), the increased radioactivity could be another cause of cell death. Due to its high labeling efficiency, the low dose of 18F-HFB might be used in the future labeling case to reduce the cell death. This is the first study investigating the cytotoxicity of 18F-HFB labeled cells. Although cultivated CPCs showed a low sensitivity to irradiation-induced cell dysfunction, we would still recommend the use of a combination of radiolabeled cells with unlabeled cells in clinical trials (7). Also the dosimetry of 18F-HFB need be done before it is used for human application. For future studies in cell imaging with radiolabeling, it will be necessary to carefully check for radiation-induced impairment of cell viability, phenotype, and function at different time points.
Another limitation of 18F-HFB is the short half-life (110 min) of 18F, so that cell homing, retention, and survival can be assessed only in the first few hours. Other PET isotopes with longer half-lives may be worth pursuing. For example, 64Cu-pyruvaldehyde-bis-(N4-methylthiosemicarbazone) (half-life of 12.7 h) has been used for tracking C6 rat glioma cells up to 24–36 h (1). Another commonly used imaging technique considered for cell tracking is magnetic resonance imaging (MRI), in which cells are labeled with iron oxide (14) or paramagnetic particles (13). However, MRI signals cannot reliably indicate whether cells are dead or alive. While short-term monitoring of cells does not give insight into the long-term engraftment and function of the therapeutic cells, without the initial homing event, the cells cannot exert either their paracrine or regenerating effects. In addition, the synthesis of 18F-HFB is a multistep procedure and might not be available in some institutions. Every cell labeling approach has its own advantages and disadvantages. Therefore, the different cell labeling methods should be used for different subjects of investigation at different stages of cell transplantation.
In this study, we used an echo-guided intramyocardial injection technique to deliver cells into the target site of rat myocardium in a closed-chest fashion with a high success rate. By using this novel approach, we can remove the animals with unsuccessful injections from the study at the time of the injection to assure uniformity of the study groups. Successful injection was corroborated by PET images. Although the fusion (injection sites) and 13N-NH3 (infarct areas) PET images are shown at different slices, the maximum distance between the injection site and infarct scar was less than 3 mm, confirming cell delivery to the border zone.
The use of circulatory stem cells for therapy is an attractive approach considering the noninvasive nature of cell procurement (25,31). Experimentally, the administration of CPCs to ischemic or infarcted myocardium has resulted in increased blood vessel formation, decreased infarct size, and improved ventricular function (9). Reports have also shown that CPCs can prevent cardiomyocyte apoptosis, reduce remodeling, and improve cardiac function in areas of neovascularized ischemic myocardium (10). Intramyocardial delivery of CPCs has recently been shown to enhance left ventricular ejection fraction recovery in patients after acute myocardial infarction (AMI) (11). Previous work using 111In-oxine-labeled human PBMNCs has demonstrated that the intramyocardial injection technique displayed greater delivery efficiency compared with intracoronary and interstitial retrograde coronary venous delivery in an ischemic swine model (8). In our study, intramyocardial injection resulted in about 20% of transplanted CPC retention at 10 min postinjection, but subsequently, some injected cells moved to the pulmonary circulation, liver, spleen, and bone marrow. Therefore, the absolute number of CPCs engrafted in the heart is still rather low, which is in accordance with previous studies (8). A low rate of cell persistence is a potential limitation for cell therapy. One may speculate that the use of tissue engineered scaffolds will enhance the local retention and therapeutic effects of cells in the injured myocardium (22,24). We have previously used 18F-FDG labeling to successfully assess the ability of tissue- engineered matrices to improve the early retention of transplanted progenitor cells (30). The current study suggests that 18F-HFB may also be suitable for such an application. Unlike 18F-FDG, for which only 10–20% was retained in the matrix, the collagen matrices can bind 60–75% of 18F-HFB and about 90% of this activity will remain in the collagen matrix after a 2-h incubation. However, the leakage of 18F-HFB from labeled CPCs is minimal (only 8–15% after 4 h). Although this small amount of activity may be trapped in the matrix, it will be insignificant compared to the activity retained by 18F-HFB-CPCs in the matrix. Therefore, it is possible that 18F-HFB may be usable as a tracer to assess the location and fate of injected biomatrices. However, a more detailed in vivo evaluation needs to be done in future studies. 18F-HFB with PET imaging could be used to screen various bioengineering, pharmacological or genetic techniques that may enhance myocardial homing and therapeutic efficacy of transplanted stem cells (19). The prospects of 18F-HFB make it deserving of continue investigation.
Conclusions
In summary, the results of this study demonstrate that 18F-HFB is a usable tracer for in vivo monitoring of transplanted human CPCs in a rat model of myocardial infarction. 18F-HFB labeling with PET imaging can help evaluate the efficacy of stem cell delivery and retention in the target tissue quantitatively. Compared to 18F-FDG, the potential of 18F-HFB labeling to detect differences in cell homing between the border zone and normal myocardium suggests that this method may provide a better modality to enhance our understanding of early retention, homing, and engraftment with cardiac cell therapy.
Footnotes
Acknowledgments
This work was supported by the Heart and Stroke Foundation of Ontario (program grant PRG 6242; grant NA5905 to Drs. Ruel and Beanlands; grant T6793 to Dr. Suuronen), the Canadian Institutes of Health Research (grant MOP-77536 to Drs. Ruel and Suuronen), and the Canadian Foundation for Innovation (award 7346 to Dr. Ruel). Dr. Zhang and S. Thorn are recipients of Doctoral Research Awards from the Heart and Stroke Foundation of Canada. D. Kuraitis was supported by a Canadian Institutes of Health Research Canadian Graduate Scholarship. The authors declare no conflicts of interest.
