Abstract
Coexpression of CD140b (PDGFRβ) and CD146 has been used to isolate endometrial mesenchymal stem-like cells (eMSCs), which have a perivascular location. This study aims to evaluate a single marker for purifying eMSCs. Using an antibody panel with novel specificities, we screened human endometrial tissues and stromal cell suspensions by flow cytometry and immunohistochemistry to identify perivascular markers. Sorted subpopulations were examined for colony-forming unit (CFU), self-renewal, and differentiation assays for mesenchymal stem cell (MSC) function. We also transplanted sorted eMSCs under the kidney capsule of superimmunodeficient NSG mice. Magnetic bead selection was compared with flow cytometry sorting (flow sorting) using CFU assay. One novel marker (W5C5) was particularly effective in selecting eMSCs. W5C5+ cells comprise 4.2 ± 0.6% (n = 34) of endometrial stromal cells and reside predominantly in a perivascular location in both basal and functional layers of endometrium. The clonogenicity of W5C5+ cells is significantly greater than W5C5- and unselected cells. W5C5+ cells differentiated into adipocytes, osteocytes, chondrocytes, myocytes, and endothelial cells. W5C5+ cells produce endometrial stromal-like tissue in vivo. In terms of clonogenicity, magnetic bead-selected W5C5+ cells gave rise to significantly higher CFU numbers compared to flow-sorted W5C5+ cells. This study identified W5C5 as a single marker capable of purifying eMSCs possessing MSC properties and reconstituting endometrial stromal tissues in vivo. W5C5 enriches eMSCs to high purity and provides a simple protocol for their prospective isolation using magnetic bead selection rather than flow sorting. W5C5 selection may provide an alternate, readily available autologous source of MSC, obtainable with minimal morbidity using an office endometrial biopsy procedure for future cell-based therapies.
Keywords
Introduction
Mesenchymal stem cells (MSCs), also known as mesenchymal stromal cells, were originally isolated from bone marrow as an adherent nonhematopoietic population (13,32) by in vitro culturing. Mesenchymal stem-like cells have been identified in many human tissues including heart, liver, dental pulp, adipose tissue, synovial membrane, umbilical cord blood, amniotic fluid, and endometrium (1,8,9,17,36,40). Key features of MSCs are their self-renewal and multipotency. MSCs differentiate into mesoderm-derived lineages including adipocytes, chondrocytes, osteocytes in vitro (7,31,32,43). Recent studies have also demonstrated that MSCs differentiate into nonmesenchymal lineages: neurons (22,39), skeletal muscle (10), myocardium (26), and endothelium (27). In addition to this broad differentiation capacity, immunomodulatory properties of MSCs are an important aspect for therapeutic purposes. Substantial evidence shows that MSCs modulate T cell-mediated immunological responses and have low immunogenicity and MSCs home to sites of ischemia or injury when administered systemically (42). MSCs are now considered an available cell source for cell-based therapies and tissue engineering applications. Most knowledge on mesenchymal stem cell (MSC) properties has been generated from in vitro studies conducted on heterogeneous population of adherent cells. However, in terms of clinical application, this conventional in vitro method for MSC isolation has a major disadvantage due to contamination with cells other than MSCs. Because the properties and gene expression of MSCs are altered by culture conditions (2), considerable research effort is currently devoted to identifying specific MSC markers for their prospective isolation (8,27).
The human endometrium is the highly regenerative mucosal lining of the uterus, which grows 4–7 mm within 4–10 days every menstrual cycle (25). This dynamic regenerative capacity is physiologically regulated by cyclical changes of plasma sex steroid hormone levels (14,15,29). The endometrium comprises epithelial cell-lined glands surrounded by a substantial supportive vascularized stroma. We have previously identified a small population (1.25%) of colony-forming stromal cells in human endometrium (6,35). Those that formed large colony-forming units (CFU) (0.02%) underwent self-renewal, had high proliferative potential, and were multipotent, differentiating into adipocyte, chondrocyte, osteocyte, and smooth muscle cells (17). We also have demonstrated that these endometrial MSC-like cells (eMSCs) can be purified on the basis of their coexpression of two perivascular markers, cluster of differentiation 140b (CD140b; platelet-derived growth factor receptor β [PDGFRβ]) and CD146 (36). The use of this combination of markers was the first report on the prospective isolation of MSC-like cells from a human mucosal tissue, and this combination was confirmed by a recent study identifying human MSCs in multiple organs (8). These markers identified the perivascular location of MSCs in many tissues, and colocalization studies suggested that they are subpopulations of pericytes (8,36).
The use of two markers to identify MSCs necessitates flow cytometry-based cell sorting or two sequential magnetic bead selections for purification. Cell sorting is a complex procedure requiring sophisticated equipment and may have detrimental effects on cell viability. Two sequential magnetic bead selections may also affect the cell viability. In contrast, a single marker would provide a simple and safe isolation protocol for prospective purification of eMSCs. This major advantage for clinical application can be achieved with magnetic beads rather than flow cytometry sorting. The aim of the study was to evaluate whether a single marker would provide a purer population of eMSCs to markedly simplify their isolation and analysis and to provide an isolation protocol more suitable for future cell therapies based on magnetic bead selection.
In this study, we adapted flow cytometry analysis and immunohistochemistry to screen 10 monoclonal antibodies with perivascular or stromal immunoreactivity in human endometrium using an antibody panel (3) and our in vitro CFU assay for identifying a potential single marker of eMSCs. MSC properties were confirmed using multilineage differentiation and self-renewal assays. To determine if eMSCs reconstitute stromal tissue, we investigated their in vivo activities. This study found that one novel single marker, W5C5, enabled the prospective isolation of multipotent, self-renewing endometrial MSCs that generated endometrial stroma in vivo. The endometrial MSC population selected with W5C5 beads had significantly higher cloning efficiency than the flow cytometry sorted (flow-sorted) W5C5+ population. Bead-selected (beaded) W5C5+ endometrial MSCs may offer an alternate, readily available autologous source of MSCs, obtainable with minimum morbidity using office endometrial biopsy procedure for future cell-based therapies.
Materials and Methods
Human Tissue Collection
Human endometrial tissue (n = 43), including underlying myometrium, was collected from ovulating women aged 36–53 (44.5 ± 0.8) years undergoing hysterectomy or endometrial biopsy for nonendometrial benign pathologies. Informed written consent was obtained from each patient, and ethics approval was obtained from the Southern Health Human Research and Ethics Committee B. Menstrual cycle stage was assessed according to well-established histological criteria for the normal menstrual cycle (28) by experienced histopathologists. This study used 19 proliferative and 19 secretory phase samples.
Novel Antibody Panel
Ten monoclonal antibodies against novel markers of bone marrow-derived MSCs (Fig. 1A) were obtained as culture supernatants from an antibody panel with known and novel specificities (3).
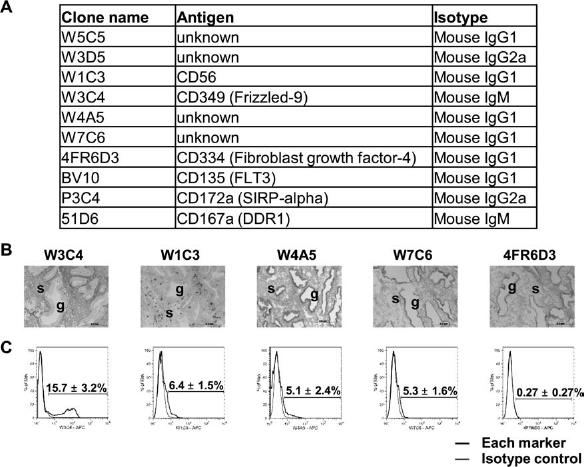
Novel marker expression in human endometrium. (A) Antibody panel of 10 novel markers for mesenchymal stem cells (MSCs). (B) Immunohistochemical staining with five marker antibodies on human endometrial tissue sections. (C) Representative flow cytometric histograms showing the expression of the five markers on human endometrial stromal cell fractions. s, stroma; g, gland. Data are mean ± SEM. FLT-3, fms-related tyrosine kinase 3; SIRP-α, signal regulatory protein-α; DDR1, discoidin domain receptor tyrosine kinase 1.
Preparation of Single-Cell Suspensions of Human Endometrial Stromal Cells
Endometrial tissue including the nonmenstruated basalis layer was scraped off the underlying myometrium to ensure all endometrial tissue was collected. Endometrial tissue was then dissociated using enzymatic and mechanical dissociation and separated into stromal single-cell suspensions and epithelial clumps as previously described (6,17,23) with several modifications. Endometrial tissue samples were washed in DMEM/F-12 medium containing 15 mM HEPES buffer (Invitrogen, Auckland, New Zealand), 5% new born calf serum (Invitrogen), and 1% antibiotic–antimycotic (Invitrogen) (Bench Medium), then weighed and cut into small pieces <1 mm3. The tissue fragments were digested with 0.5% (wt/vol) collagenase I (Worthington Biochemical Corporation, Lakewood, NJ, USA) and 40 μg/ml deoxyribonuclease type I (Worthington Biochemical Corporation) in DMEM/F-12 (Invitrogen) supplemented with 10% fetal calf serum (Invitrogen), 2 mM glutamine (Invitrogen), antibiotic–antimycotic (DMEM+) for 1.5 h at 37 °C on a shaker. Typically, 10 ml of the cell dissociation medium was used per 1 g tissue. The dissociated cells were filtered through a sterile 40-μm cell strainer (BD Biosciences, Durham, NC, USA). Most of the stromal cells and blood cells, present as a single-cell suspension, passed through the cell strainer into a sterile 50-ml polycarbonate tube, whereas the undigested fragments, mostly comprising glandular clumps, were retained on the strainer. Stromal single-cell suspensions were layered over Ficoll-Paque PLUS (GE Healthcare Bio-Sciences AB, Uppsala, Sweden) and centrifuged to remove red blood cells. The medium/Ficoll interface, mainly containing stromal cells and peripheral blood mononuclear cells, was carefully aspirated, washed with Bench Medium, and then subjected to flow cytometry.
Multicolour Flow Cytometry
Fresh isolated endometrial stromal cell suspensions (up to 1 × 107 cells/100 μl) were incubated with primary antibodies on ice for 45 min in Bench Medium as previously described (36). The antibodies used are listed in Table 1. Cells were then incubated with a directly conjugated secondary antibody on ice for 30 min, depending on first antibody isotype. Cells were pelleted and washed with Bench Medium after each incubation. Flow cytometric cell sorting and analysis was performed on a MoFlo (Beckman Coulter, Fullerton, CA, USA) using Summit software (version 5.2, Beckman Coulter). The initial selection for analysis was based on forward versus side scatter profile, dead cells were excluded by electronic gating on 7-aminoactinomysin D (Invitrogen) or SYTOX blue (Invitrogen) profile, and remaining cells were analyzed using multiple combinations of the antibodies. Isotype-matched controls were included for each antibody.
Antibodies Used for Flow Cytometry and Immunohistochmistry

To determine the viability of flow-sorted W5C5+ and W5C5- fractions, the flow-sorted cells were washed once with Bench Medium immediately after sorting. Washed cells were subjected to flow cytometry analysis after adding a new aliquot of SYTOX blue. The flow analysis was performed within 15 min of the original sorting.
Magnetic Bead Selection
Magnetic bead selection was performed according to the manufacturer's instruction (Miltenyi Biotec, Bergisch Gladbach, Germany). Briefly, freshly isolated endometrial stromal cell suspensions (up to 1 × 107 cells/100 μl) were incubated with phycoerythrin (PE)-conjugated W5C5 antibody (10 μl/107 cells) [a kind gift from Christoph Gaissmaier (TETEC AG, Reutlingen, Germany) and Katherin Pütsch (Miltenyi Biotec)] on ice for 30 min in 0.5% fetal calf serum in PBS (Bead Medium). Cells were then incubated with anti-PE magnetic-activated cell sorting (MACS) MicroBeads (Miltenyi Biotec) on ice for 30 min. Cell suspensions (up to 1 × 108 cells/500 μl) were applied onto Miltenyi columns in a magnetic field, followed by washing the column with 500 μl Bead Medium three times. While most W5C5- cells passed through the column, magnetically labeled W5C5+ cells were mostly retained on the column. The columns were removed out of the magnetic field, and W5C5+ cells were flushed out firmly with 1 ml of Bead Medium.
In Vitro Colony-Forming Assays
Flow-sorted subpopulations of endometrial stromal cells were seeded at a clonal density of 50–100 cells/cm2 onto fibronectin-coated 100-mm culture dishes and cultured in DMEM+ supplemented with 10 ng/ml human fibroblast growth factor 2 (Millipore, Billerica, MA, USA) (F-DMEM+). The first media change was after the first week, and half media changes were done twice weekly thereafter. Colonies were monitored microscopically to ensure they were derived from single cells. Cultures were terminated after 5–7 weeks and stained with hematoxylin. Clusters, ≥50 cells, were counted, and the colony-forming efficiency determined (6).
In Vitro Differentiation Assays
Flow-sorted subpopulations of endometrial stromal cells were cultured in F-DMEM+ at clonal density (50–100 cells/cm2) to obtain colony-forming W5C5+ cells. The resultant colonies were cultured with or without passaging to increase cell numbers and then incubated separately with adipogenic, osteogenic, myogenic. and angiogenic differentiation induction media for 4 weeks as described (27,31,36,43). To induce angiogenic differentiation, cells were seeded at 10,000 cells/cm2 and cultured with endothelial cell basal medium-2 (EBM-2), supplemented with endothelial cell growth medium-2 microvascular (EGM-2 MV) SingleQuots (Lonza) at 37°C with 5% CO2. The cultures were maintained with medium changes every 3 or 4 days for 4 weeks (27). For in vitro chondrogenic differentiation, 3–5 × 105 cells were cultured as a pelleted micromass in a centrifuge tube in chondrogenic differentiation medium for 4 weeks. Cells from the same original clones were also cultured concurrently for 4 weeks in DMEM+ as an undifferentiated control.
To assess each differentiation, cells were harvested for RNA or fixed with 4% paraformaldehyde (PFA) and stained with Oil Red O (adipogenic), an alkaline phosphatase kit (osteogenic) (Sigma-Aldrich, St. Louis, MO, US) or immunocytochemistry using antibodies (myogenic and angiogenic) as described in Table 1. Nuclear counterstaining was done with Mayer's hematoxylin. For immunocytochemistry, fixed cells were incubated with anti-α smooth muscle actin antibody, anti-smooth muscle myosin heavy chain antibody, or anti-CD31 antibody for 1 h at room temperature and diaminobenzidine (DAB, Sigma-Aldrich) staining was conducted with EnVision+ System-HRP (DAKO). To assess chondrogenic differentiation, RNA was extracted from micromass cultures and Alcian blue staining was performed on 4% PFA-fixed paraffin sections (4 μm) of micromasses, followed by nuclear Fast Red counterstain. Stained cells were examined under an Olympus BX41 microscope (Olympus, Tokyo, Japan), and images were captured using an Olympus DP25 digital camera (Olympus).
Differentiated cells were also assessed for expression of lineage-specific genes using RT-PCR. Total RNA was isolated, and genomic DNA contamination was removed using ISOLATE RNA Mini kit (BIOLINE, London, UK), and 1 μg RNA was reverse transcribed to complementary DNA using the SuperScript III First-Strand Synthesis System (Invitrogen). Subsequent PCR reactions were performed using GoTaq Green Master Mix (Promega) in a Gene Amp PCR System 2700 (Applied Biosystems, Foster City, CA, USA). Primer sequences are shown in Table 2 (17,36). The PCR products were separated by 1.5% agarose gel electrophoresis and visualized with GelRed (Biotium, Hayward, CA, USA) staining using ChemiDoc XRS system (Bio-Rad Laboratories, Hercules, CA, USA).
RT-PCR Primers Used for Differentiation Assays

In Vivo Tissue Reconstitution Assay
Nonobese diabetic severe combined immunodeficient interluekin 2 receptor γ-deficient (NOD/SCID/IL2 receptor γ chainnull; NSG) mice (38) were used for xenotransplantation experiments. Ethics approval for the use of animals was granted by the Monash Medical Centre Animal Ethics Committee A. Single-cell suspensions of cultured W5C5+ cells (5 × 105) in 5–10 μl of DMEM+ was injected underneath the kidney capsule as described (23,24). Some NSG mice were ovariectomized at the time of transplantation, and 1 μg of estradiol valerate (E2) (Sigma-Aldrich) in 100 μl of peanut oil was subcutaneously injected once a week with or without daily subcutaneous injection of 1 mg of progesterone (P4) (Sigma-Aldrich) for the last 2 weeks to mimic human menstrual cycles. These xenotransplanted mice were nephrectomized 10 weeks after transplantation for histological analysis.
Graft-bearing kidneys excised from NSG mice were directly embedded in Tissue-Tek OCT compound (Sakura Finetech, Torrance, CA, USA), frozen, and serially sectioned at 6 μm by using a Leica cryostat (Leica Microsystems, Inc.). Serial cryosections were air-dried, washed with PBS, and fixed in 4% PFA. Histological analysis was performed by hematoxylin and eosin staining. For immunohistochemistry, sections were permeabilized with 0.1% Triton X-100, blocked with protein block serum-free (DAKO), and then incubated with antihuman vimentin antibody (Table 1) for 60 min at room temperature. The primary antibody was visualized by incubation with Alexa Fluor 568-conjugated secondary antibody (Invitrogen) for 30 min at room temperature. Nuclei were stained with 1 μg/ml Hoechst 33258 (Sigma-Aldrich) for 5 min. Images were collected using a Leica DMR fluorescence microscope (Leica Microsystems, Wetzlar, Germany) equipped with a Leica DC200 digital camera.
Statistical Analysis
Data were analyzed using GraphPad PRISM software (version 5.03; GraphPad Software, Inc., San Diego, CA, USA). Gaussian distribution was examined using Kolmogorov–Smirnov test. Student's t test or Mann–Whitney test was performed to determine statistical significance between two groups according to distribution. For comparing three groups, the data were analyzed using the Kruskal–Wallis test, followed by the Mann–Whitney test with Bonferroni's correction to identify individual significances. Data are presented as mean ± SEM for parametric data or median and range for nonparametric data. Results were considered statistically significant when p < 0.05.
Results
Localization and Clonogenicity of Novel Marker-Positive Cells
Since eMSCs reside in a perivascular location (36), we first investigated the localization of the 10 novel marker antibodies (Fig. 1A) in human endometrium by immunohistochemistry. CD135 (fms-related tyrosine kinase 3; FLT3), CD172a [signal regulatory protein (SIRP)-α] and CD167a [discoidin domain receptor tyrosine kinase 1 (DDR1)] epitopes were not expressed on human endometrial stroma. Therefore, antibodies reactive with these molecules were used as isotype controls as they were produced as hybridoma supernatants similar to the other antibodies in the panel. W1C3 (CD56), W3C4 (CD349), W4A5, W7C6, and 4FR6D3 (CD334) immunoreacted with human endometrium (Fig. 1B, C), but only W5C5 and W3D5 were identified as uniquely perivascular markers throughout whole endometrium in not only basalis but also functionalis (Fig. 2A). These two candidate markers were next screened for expression on freshly isolated endometrial stromal cells using flow cytometry. Whereas 4.2 ± 0.6% (n = 34) of endometrial stromal cells expressed W5C5 antigen, only 0.4 ± 0.2% (n = 7) expressed W3D5, which was apparently lower than expected from immunohistochemistry data (Fig. 2B), indicating that the W3D5 epitope is lost during enzymatic dissociation. Therefore, we focused on W5C5 as a potential novel single marker for prospective isolation of eMSCs, as this epitope appeared to be an enzyme-insensitive marker.

Localization and clonogenicity of human endometrial W5C5+ cells. (A) Immunohistochemical staining with W5C5 and W3D5 antibodies on human endometrial tissue sections. Arrows demonstrate that W5C5+ and W3D5+ cells are perivascular in location. g, gland. Scale bar: 200 μm. (B) Representative flow cytometric histograms of W5C5 (n = 31) and W3D5 (n = 7) expression on human endometrial cells. (C) Cloning efficiency of unselected, W5C5- and W5C5+ cells. Bars are medians. (D) Microscopic images of colonies derived from W5C5+ cells (left) and W5C5- cells (right). Scale bar: 500 μm (top) and 200 μm (lower). ∗p < 0.05.
We next assessed MSC activity of flow-sorted W5C5+ cells. We first evaluated the clonogenicity using colony-forming unit (CFU) assay. The cloning efficiency of W5C5+ (median, 3.6; range, 0.7–6.9; n = 6) cells was significantly higher than unselected cells (median, 0.6; range, 0.1–3.8; n = 7) (p < 0.05) and W5C5- cells (median, 0.2; range, 0.0–1.2; n = 14) (p < 0.05) (Fig. 2C), although the cloning efficiency of unselected cells was not significantly higher than W5C5- cells (p = 0.15) (Fig. 2C). These results collectively suggest that the W5C5+ population includes most clonogenic cells in human endometrial stroma and that a single marker, W5C5, can be used for isolating clonogenic endometrial stromal cells. Microscopic analysis revealed that clonogenic W5C5+ cells were typically smaller, densely packed with a high nuclear/cytoplasmic ratio and immature cell features, compared with W5C5- cell clones (Fig. 2D).
Surface Phenotype of Human Endometrial W5C5+ Cells
W5C5+ cells were then examined for expression of lineage markers and typical MSC phenotypic markers using flow cytometry. Freshly isolated W5C5+ cells expressed MSC markers, CD29 (11.6 ± 3.3%, n = 3), CD44 (77.3 ± 10.6%, n = 3), CD73 (99.3 ± 0.4%, n = 3), CD90 (70.5 ± 6.5%, n = 13), CD105 (99.1 ± 0.4%, n = 3), CD117 (86.0 ± 12.5%, n = 3), CD140b (73.1 ± 11.5%, n = 9), CD146 (28.3 ± 4.3%, n = 30), and STRO-1 (60.3 ± 14.4%, n = 4) (Fig. 3A, B). A small number of W5C5+ cells expressed several lineage markers, CD24 (11.6 ± 2.2%, n = 3), CD31 (5.3 ± 0.8%, n = 16), CD45 (4.7 ± 0.6%, n = 11), and epithelial cell adhesion molecule [EpCAM] (27.5 ± 4.7%, n = 14) (Fig. 3A, B).
We further explored the relationship between the W5C5 marker and existing eMSCs markers, CD140b and CD146 (36). Most W5C5+ cells expressed CD140b, while all the W5C5+CD146+ cells were positive for CD140b (Fig. 3C). Accordingly, we next focused on W5C5+CD146+ cells.

Surface phenotype of human endometrial W5C5+ cells identified by multicolor flow cytometry. (A) Representative flow cytometric traces of lineage markers coexpressed on freshly isolated W5C5+ cells. (B) Percentages of marker expression. (C) Schematic showing relationship between W5C5+ (n = 31), CD140b+ (n = 15), and CD146+ (n = 31) cells. PDGFRβ, platelet-derived growth factor receptor β; STRO-1, marker for nonhematopoietic stromal cells; EpCAM, epithelial cell adhesion molecule.
Comparison Between W5C5+CD146+ Cells and W5C5hi Endometrial Stromal Cell Populations
The CFU assay was used to compare cloning efficiencies between CD140b+CD146+ eMSCs and W5C5+ CD146+ cells. The cloning efficiency of W5C5+CD146+ cells (6.2 ± 2.3%; n = 5) was significantly higher than CD140b+CD146+ cells (4.4 ± 1.8%; n = 5) (p < 0.05) (Fig. 4A). Furthermore, the proportion of W5C5+CD146+ cells in CD146+ cells was significantly higher in the proliferative phase (median, 35.0; range, 9.1–66.2; n = 16) compared to secretory phase (median, 18.4; range, 3.2–51.8; n = 15) (p < 0.05) of the menstrual cycle (Fig. 4B), suggesting that W5C5+CD146+ cells may be involved in endometrial proliferation. We have also demonstrated W5C5+ cells in menstrual endometrium (unpublished observations). However, the isolation of this highly clonogenic population still requires two markers. We found that most W5C5+CD146+ cells had a higher intensity of W5C5 expression than W5C5+CD146- cells (Fig. 4C). Consequently, we next flow-sorted W5C5 high positive (W5C5hi) cells using the sorting gate drawn with a dotted line in Figure 4C and compared CFU activity with W5C5+CD146+ cells sorted through the gate drawn with a dashed line (Fig. 4C). W5C5hi cells had a high clonal capacity (4.0 ± 1.0%; n = 5) equivalent to W5C5+CD146+ cells (3.2 ± 1.1%; n = 5) (p = 0.20) (Fig. 4D).

Comparison between W5C5+CD146+ and W5C5hi human endometrial cells. (A) The cloning efficiency of W5C5+CD146+ cells and CD140b+CD146+ cells (n = 5). (B) The proportion of W5C5+CD146+ cells in CD146+ population. Bars are medians. (n = 31) (C) Sorting gates for W5C5+CD146+ cells (dashed line) and W5C5hi cells (dotted line). (D) Cloning efficiency of W5C5hi cells and W5C5+CD146+ cells. ∗p < 0.05.
Multilineage Potential of W5C5+ Cells
Colonies of W5C5+ cells were expanded in DMEM+ and then cultured in osteogenic, adipogenic, myogenic, and chondrogenic induction media. The majority of W5C5+ cells differentiated into adipocytes after incubation in adipogenic induction medium as visualized by Oil Red O staining of lipid droplets (Fig. 5A). An early stage adipocyte lineage marker, peroxisome proliferatoractivated receptor γ2 (PPARγ2), was also expressed in the differentiated cells, but not when cultured in control medium (Fig. 5A).

Multilineage potential of W5C5+ human endometrial cells. Representative histological staining (left) and RT-PCR data (right) after 4 weeks culture of W5C5+ human endometrial cells in (A) adipogenic, (B) osteogenic, (C) chondrogenic, (D) myogenic, and (E) angiogenic induction media. A single experiment representative of three separate experiments on three different patient samples is shown. Left, cultured cells in each induction medium. Middle, high power view. Right, cultured cells in control medium. Scale bar: 100 μm. PPARγ2, peroxisome proliferator-activated receptor γ2; CBFA1, core binding factor α1; ALP, alkaline phosphatase; αSMA, α smooth muscle actin.
W5C5+ cells underwent osteogenic differentiation as shown by positive staining for alkaline phosphatase (ALP) and expressed the critical early stage osteogenic transcription factor, core-binding factor α1 (CBFA1) (Fig. 5B).
W5C5+ cells were cultured as a micromass pellet to assess chondrogenic differentiation, and cells reminiscent of chondrocytes surrounded by Alcian blue-positive cartilaginous matrix were observed (Fig. 5C). Cells cultured in control medium failed to maintain a pellet structure due to lack of extracellular matrix production. RT-PCR analysis showed expression of the early-stage chondrogenic marker, collagen type X in the differentiated, but not undifferentiated control cells (Fig. 5C).
W5C5+ cells subjected to myogenic differentiation media immunostained strongly for the early-stage smooth muscle cell (SMC) marker, α smooth muscle actin (αSMA), and later stage marker myosin heavy chain (MHC) (Fig. 5D), compared with cells cultured in control medium.
After angiogenic differentiation, W5C5+ cells expressed CD31, an endothelial marker. W5C5+ cells in control medium were partly and slightly stained for CD31, suggesting that W5C5+ endometrial endothelial cells survived and proliferated in control medium (Fig. 5E). Thus, W5C5+ cells demonstrated multipotentiality in vitro, similar to bone marrow-derived MSCs, clonogenic (17), and CD140b+CD146+ endometrial cells (36).
In Vivo Tissue Reconstitution of W5C5+ Cells
To investigate the in vivo regenerative capability of W5C5+ cells, we transplanted W5C5+ cells under the kidney capsule of NSG mice. Some of these mice were ovariectomized (OVX) at the time of transplantation, and estradiol valerate (E2) was administrated weekly with daily injections of progesterone (P4) for the last 2 weeks. White growths (small masses) were identified macroscopically on two transplanted kidneys (one kidney of six OVX mice kidneys and one kidney of four non-OVX mice kidneys) (Fig. 6A). No macroscopic masses were generated in the remaining mice; however, histological and microscopic analyses of the transplantation sites revealed the existence of stromal-like connective tissues under all the kidney capsules of transplanted mice (Fig. 6B). Migratory human cells in murine kidney parenchyma were also observed in two of six OVX mouse kidneys and one of four non-OVX mouse kidneys (Fig. 6C), similar to the “chimeric vessel” (23) and “migrating endothelial type” observed in a kidney transplanted with endometrial side population cells (24). As the antibody against human vimentin (clone V9) only recognizes human vimentin, red-stained cells on immunofluorescent images were derived from human transplanted cells, but not from murine cells.
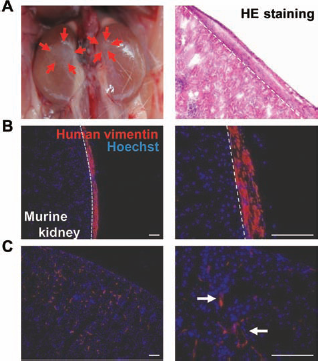
Human endometrial W5C5+ cells reconstitute stromal tissue in vivo. (A) Macroscopic view of kidneys 10 weeks after transplantation with 5 × 105 W5C5+ cells (left). Red arrows highlight the small white lesion produced by transplanted W5C5+ cells. HE staining of the transplanted site (right). (B, C) Immunofluorescence staining with human vimentin (red) and Hoechst 33258 (blue) of the transplants. (B) Stromal type. (C) Invasive type (white arrows). Scale bar: 100 μm.
Magnetic Bead Selection Versus Flow Cytometry Sorting of Human Endometrial W5C5+ Cells
Because of our interest in the future clinical application of eMSCs, we next compared magnetic bead selection with flow cytometry sorting of W5C5+ cells (Fig.7). Cloning efficiency of beaded W5C5+ population (median, 13.5; range, 12.3–14.3; n = 5) was significantly higher than not only W5C5- population (median, 3.8; range, 0.4–6.5; n = 5) (p < 0.05) but also the flow-sorted W5C5+ population (median, 6.5; range, 3.5–8.0; n = 4) (p < 0.05) (Fig. 7A). Flow cytometry analysis of the beaded W5C5+ population revealed that W5C5 purity was 20.6 ± 4.8% (n = 9). However, this is likely an underestimation of the percentage of W5C5+ cells. Comparison of the fluorescence intensities of beaded W5C5+ cells (Fig. 7F) with beaded W5C5- cells (Fig. 7G) showed that most of the W5C5+ fraction was shifted upward, although only 20.6 ± 4.8% were sufficiently bright to register in the positive gates (G1 and G2 in Fig. 7D), suggesting an underestimate of the true number of W5C5+ cells in the bead-sorted fraction due to the large population of W5C5lo cells. On the other hand, although the flow-sorted W5C5+ cells were sorted using the same gates (G1 and G2 in Fig. 7D), the W5C5 fluorescence intensity was slightly reduced as a small percentage of cells remained in the gate G4 (Fig. 7H), possibly because the fluorescent intensity was slightly decreased by the lasers during the initial sorting. Both beaded and sorted W5C5- cells (Fig. 7G, I) were clearly negative for W5C5. Accordingly, if the W5C5lo cells are in gate G4 of Figure 7F, the percentage of total W5C5+ cells in the bead-sorted W5C5+ is likely greater than 20.6 ± 4.8% (Fig. 7C), although unlikely higher than the flow-sorted purity (95.0 ± 1.4%; n = 5). Therefore, we speculated that the observed difference in cloning efficiencies arose from differences in their respective cell viabilities rather than contaminating W5C5- cells supporting growth of W5C5+ cells. Next, we have analyzed five samples to compare viabilities between flow-sorted and beaded (nonflow-sorted) W5C5+ fractions. The viability of the beaded W5C5+ fraction was 88.4 ± 2.7% after gating out debris and doublet cells, while the viability of the flow-sorted W5C5+ fraction was 92.3 ± 1.9% of W5C5+ counts (includes some W5C5+ dead cells). Although the flow-sorted W5C5+ cells were selected as live cells, their viability in relation to total count (includes cellular debris) was 63.4 ± 7.3%, indicating some of the flow-sorted W5C5+ cells had died and contributed to the debris. However, this figure may be an underestimate as a dead cell may contribute several events as debris. Consequently, we speculate that the real viability of flow-sorted W5C5+ fraction is less than 92.3 ± 1.9% and more than 63.4 ± 7.3%.

Magnetic bead versus flow cytometry sorting of human endometrial W5C5+ cells. Cloning efficiency of beaded W5C5+, beaded W5C5-, and flow-sorted W5C5+ population. Bars are medians. (B) Representative flow cytometric phenotyping data of beaded W5C5+ population. (C) Percentages of phenotypic marker expression. (D–I) Representative dot plot charts of flow data before (D, E) or after beading (F, G) or flow sorting (H, I). ∗p < 0.05. Note that (D) is the negative control for setting both W5C5 and CD146 gates.
The beaded W5C5+ population expressed several lineage markers, CD31 (3.8 ± 3.1%, n = 3), CD45 (3.6 ± 1.6%; n = 6), CD90 (93.3 ± 1.3%, n = 4) but not EpCAM (0.7 ± 0.7%; n = 6) (Fig. 7B, C). These data showed that the magnetic beading method yielded a W5C5+ population comprising more than 90% of stromal cells and was superior at excluding epithelial cells and leukocytes, although the other contaminating cells are also present. The expression of CD146 on the beaded W5C5+ (7.2 ± 3.4%; n = 3) (Fig. 7B, C) tends to be higher than nonbeaded cells (3.1 ± 0.4%; n = 34) (Fig. 3C) (p = 0.13).
Discussion
This study identified W5C5 as a novel single marker for purifying eMSCs, which self-renew; differentiate into adipogenic, osteogenic, chondrogenic, and myogenic cell lineages; and reconstitute mesodermal tissue in vivo. This is the first report to show that a single marker for the prospective isolation of eMSCs is comparable with the conventional method using coexpression of CD146 and CD140b (36). We have also demonstrated for the first time that transplanted W5C5+ eMSCs gave rise to endometrial stromal-like tissue in vivo (Fig. 6). Many in vitro experiments have proven that eMSCs exist in human endometrium (11,17,36,41). This study provides the first data showing that eMSCs regenerate endometrial stroma in vivo. Furthermore, we demonstrated that the W5C5+ population can be selected for purification using magnetic beads as well as flow cytometry sorting. A major advantage of bead selection is the rapid and simple isolation protocol obviating the need for prolonged flow cytometry preparation steps and access to sophisticated machines and software, saving time and reducing costs. Further advantages are the greater yields and quality of viable cells that are obtained with magnetic beads compared to flow cytometry, which adversely affects cell viability. Indeed, the beaded W5C5+ population has significantly higher clonal capacity than flow-sorted W5C5+ population, although the W5C5+ purity is greater by cytometry sorting. Thus, in terms of future clinical applications, bead selection is significantly more attractive than flow cytometry sorting, particularly for those requiring cell-based therapies.
W5C5 substantially enriched for eMSCs, resulting in a colony-forming capacity of 3.8% for W5C5+ stromal cells. This is a 3.0-fold increase over unselected endometrial stromal cells and a 14.7-fold increase over W5C5-cells. Almost all clonogenic stromal cells in human endometrial cell suspension were sorted into W5C5+ fraction. There is considerable variation between patient samples, particularly with endometrium as it is a dynamically remodeling tissue, and the viability is often affected by menstrual phase, age, and the underlying clinical condition at the time of surgery. This sample variation often masks differences in clonal capacities (36). Because of the intersubject variation associated with human endometrial samples, we compared the cloning efficiency of CD140b+CD146+ cells versus W5C5+CD146+ cells and W5C5+CD146+ cells versus W5C5hi cells (Fig. 4) using the same samples. W5C5+CD146+ cells had significantly higher clonal capacity than CD140b+CD146+ cells, and although there was no significant difference between W5C5hi cells and W5C5+CD146+ cells, the cloning efficiency of W5C5hi is 1.3-fold higher than CD140b+CD146+ cells, which also have the multilineage potential (36).
Currently, the epitope identified by the W5C5 antibody is unknown despite several attempts to immunoprecipitate the cognate antigen. Most likely, the recognized epitope is a protein domain, as verified by the unaltered binding of the antibody on tunicamycin-treated W5C5+ retinoblastoma (WERI-Rb-1) cells or after incubation of untreated cells with neuraminidase, N-glycanase, and O-glycosidase. In addition, W5C5 antigen is not a phosphoinositol (PI)-linked molecule, because binding of the antibody was not influenced after treating the cells with PI-specific phospholipase C (PI-PLC). To identify the W5C5 epitope, we are currently preparing an expression library of WERI-Rb-1 cells. After transfection of hamster cells, we aim to select the W5C5+ clone and isolate and sequence the respective clone.
An increasing body of evidence suggests the existence of eMSCs in human endometrium (16). The present study adds to and extends this body of evidence by demonstrating the capacity of eMSCs to reconstruct human stromal tissue in vivo. In addition to their clonal capacity and multilineage potential, W5C5+ cells express typical MSC markers and are found in a perivascular location of not only the basalis but also the functionalis layer of endometrium, which is shed during menstruation and regenerates in the following menstrual cycle. Indeed, mesenchymal stem-like cells can be harvested from human menstrual blood (21,30). These findings are consistent with eMSCs identified by coexpression of CD140b and CD146 (36), which were also located perivascularly throughout the endometrium, confirming the reliability of this novel single marker for eMSCs. Similarly, MSCs in the other tissues also appear to exist in perivascular regions (8,9,27,37). Interestingly, in terms of their location, some BrdU label-retaining cells (LRCs), putative endometrial stem/progenitor cells, have also been identified in perivascular regions in mouse endometrium (5), although some LRCs were also found in other locations. Recently, it was also shown that human endometrial side population cells (ESPs) most likely reside in capillaries of human endometrium in both basalis and functionalis layers (24), and these cells also had MSC properties (4), although they were heterogeneous in composition.
The in vivo reconstitution data in the present study revealed that W5C5+ cells gave rise to migratory cells in the kidney parenchyma as well as stromal-like connective tissues (Fig. 6B, C). The migratory cells were morphologically similar to “migrating endothelial type” of endometrial side population cells (24), and the “chimeric vessel” has the potential of invading into the host tissue and incorporating into the vascular system of the host blood circulation (23). This, together with their in vitro angiogenic differentiation ability, indicates that W5C5+ cells are also capable of differentiating into endothelial cells. Thus, two major components of human endometrial stroma, stromal fibrobasts and blood vessels, arise from W5C5+ cells. It has previously been reported that bone marrow-derived MSCs with angiogenic potential differentiated into hematopoietic niche cells (27). Given that human endometrial epithelial stem/progenitor cells may need an appropriate niche (16), W5C5+ cells may also provide a stem cell niche for these progenitor cells. From morphological studies, it has been reported that endometrial epithelial regeneration occurs via cellular differentiation from stromal cells (18,19). However, it still remains unclear whether or not all endometrial components are derived from only one type of stem cell. Further study is expected to clarify the relationship and position of eMSCs in the endometrial stem/progenitor cell hierarchy (6).
The therapeutic potential of MSCs is currently being investigated in a number of phase I/II and III clinical trials, and many trials have been completed and achieved important results (12,33). The advantage of MSCs lies in their multilineage potential, their anti-inflammatory properties and their immunosuppressive effects (42), and their avoidance of ethical issues as encountered with embryonic stem cells. Additionally, easy and safe protocols are imperative for clinical applications. In spite of a growing need for readily available cell sources, relatively few cell sources have been considered suitable for cell-based therapies, and there is currently no consensus on the best method to prospectively isolate MSCs from the various source tissues due to phenotypic peculiarities in each tissue (20). Even established key MSC markers, such as CD271, require another marker to obtain higher purity (20). The human endometrium is a unique adult tissue that contains substantial stroma that regenerates cyclically under normal physiological conditions. Each month, the upper two thirds that is known as the functionalis layer is shed at menstruation and regenerates from the remaining basalis layer in the subsequent cycle. Even postmenopausal women can regenerate endometrium that supports pregnancy by treatment with hormone replacement therapy (34). Thus, human endometrium is one of the most attractive but overlooked sources of MSCs for cell-based therapies that is obtainable with less morbidity than bone marrow or adipose tissues, increasing the possibilities of autologous cell-based therapies for women. Moreover, W5C5 bead selection offers rapid easy protocols in purifying eMSCs without harsh steps, providing a safer protocol, amenable to clinical good manufacturing practice (GMP) compliance.
This is the first report on the prospective isolation of eMSCs using a novel single marker, W5C5. Given that W5C5+ cells reside throughout endometrium, they offer an alternate readily available autologous source of MSC obtainable with minimum morbidity using an office-based endometrial biopsy procedure for future cell-based therapies in tissue engineering applications. Also, this study confirms eMSCs reside in a perivascular location in the stroma and play an important role in cyclical regeneration of this highly regenerative tissue.
Footnotes
Acknowledgments
The authors acknowledge Frances Walker and Pamela Mamers for collection of the tissue and Dr. Mark Lawrence and Dr. Tony Lawrence for the provision of hysterectomy tissue. This study was supported by the National Health and Medical Research Council (NHMRC) of Australia grant 545992, NHMRC RD Wright Career Development Award 465121 (both to C.E.G), and Victorian Government's Operational Infrastructure Support Program. The authors declare no conflicts of interest.
