Abstract
Hippocampal cholinergic neurostimulating peptide (HCNP) is known to promote differentiation of septohippocampal cholinergic neurons. The HCNP precursor protein (HCNP-pp) may play several roles, for example, as an ATP-binding protein, a Raf kinase inhibitor protein, and a phosphatidylethanolamine-binding protein, as well as a precursor for HCNP. This study therefore aimed to elucidate the involvement of HCNP-pp in specific neural lineages after stroke using a hypoxic–ischemic (HI) rat model of brain ischemia. The specific neural lineages in the hippocampus were investigated 14 days after ischemia. Some bromodeoxyuridine (BrdU)+ neural progenitor cells in the hippocampus of hypoxic, HI, or sham-operated rats expressed HCNP-pp. Almost half of the BrdU+/HCNP-pp+ cells also expressed the oligodendrocyte lineage marker 2′,3′-cyclic nucleotide 3′-phosphodiesterase, whereas only a few BrdU+/HCNP-pp+ cells in the hippocampus in HI brains expressed the neuronal lineage marker, doublecortin (DCX). Interestingly, no BrdU+/HCNP-pp+ progenitor cells in hypoxic, HI, or sham-operated brains expressed the astrocyte lineage marker, glial fibrillary acidic protein. Together with previous in vitro data, the results of this study suggest that the expression level of HCNP-pp regulates the differentiation of neural progenitor cells into specific neural lineages in the HI hippocampus, indicating that neural stem cell fate can be controlled via the HCNP-pp mediating pathway.
Keywords
Introduction
Recent studies have shown that the proliferation and differentiation of neural stem/progenitor cells from the subventricular zone (SVZ) and dentate gyrus (DG) in the hippocampus play an important role in the response to cerebral ischemia, representing a promising discovery in stroke research (10,18,22,31,48). Focal cerebral ischemia can stimulate a cytogenic response in these areas, with cells migrating into the ischemic area to give rise to heterotypic lineages (3,28,49). Both gliogenesis and angiogenesis may contribute to brain repair following stroke, although a small number of cells can also give rise to postmitotic neurons (3,28,49).
Hippocampal cholinergic neurostimulating peptide (HCNP), originally purified from young rat hippocampus, was first reported by Ojika et al. (37). The HCNP precursor protein (HCNP-pp) has previously been reported to possess multiple functions, for example, as an ATP-binding protein, a Raf kinase inhibitor protein, and a phosphatidylethanolamine-binding protein (PEBP), as well as the HCNP precursor (20,30,39,50). We recently reported that HCNP-pp expression in cultured adult hippocampal progenitor cells (AHPs) was closely associated with differentiation into microtubule-associated protein (MAP) 2ab+ neurons and receptor-interacting protein+ oligodendrocytes, but not into glial fibrillary acidic protein (GFAP)+ astrocytes (42). These results suggest that the expression level of HCNP-pp acts as a key regulator of the differentiation of cultured AHPs into specific neural lineages, indicating that the control of neural stem cell fate can be achieved via the HCNP-pp mediating pathway.
The aim of this study was to elucidate the role of HCNP-pp expression in the differentiation of hippocampal neural stem/progenitor cells into neurons, oligodendrocytes, and astrocytes in vivo and to confirm the results of previous in vitro studies. We utilized a hypoxic–ischemic (HI) model to stimulate neural stem cells in the hippocampus and analyzed the neural lineages of AHPs generated 14 days after ischemic stroke using immunohistochemistry.
Materials and Methods
Animal Model
All animal experiments were approved by the Animal Care and Use Committees of Nagoya City University Graduate School of Medical Sciences and conformed to the guidelines for the use of laboratory animals published by the Japanese government (Law No. 105, October 1973). Male Sprague–Dawley rats (postnatal days 10–13) were obtained from Japan SLC, Inc. (Hamamatsu, Japan). All pups were housed with their mothers under a 12-h light/dark schedule (light on 08:00–20:00) and given free access to food and water. Pups were subjected to a modified Levine procedure to produce HI brain injury on postnatal day 14 (26). Briefly, pups were anesthetized with isoflurane (induction at 4% followed by maintenance at 2–2.5%), and the right common carotid artery was sectioned between double ligatures of 4–0 surgical silk. Pups were allowed to recover for 2 h and were then exposed to an additional 2 h of hypoxia (8% oxygen, 92% nitrogen) in a plastic container at 37°C. After HI insult, pups were returned to their mothers. Contralateral hemisphere of each HI model, left side, was examined as hypoxia brain control. Sham-operated control animals were treated identically, but without sectioning of the right carotid artery and subsequent hypoxia.
BrdU Labeling and Experimental Design
Proliferating cells were labeled using BrdU (Sigma-Aldrich, Tokyo, Japan), which is incorporated into newly synthesized DNA. At 5 days after HI insult, pups in the sham and HI groups received three doses of intraperitoneal (IP) BrdU (50 mg/kg), given once every 4 h over a period of 8 h (Fig. 1). Animals were killed 2 h or 9 days after the last BrdU injection (i.e., 5 or 14 days after HI insult). Day 5 after HI insult was chosen because this was within the maximal period of HI-induced cell proliferation identified in the previous study (47) and avoided labeling of proliferating glia (41). Each group contained five pups.
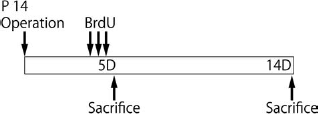
Experimental schedule. Each mouse received three doses of BrdU 50 mg/kg intraperitoneally, given every 4 h over a period of 8 h, at 5 days after hypoxic–ischemic surgery. Brains were dissected at 2 h or 9 days after BrdU administration and fixed for immunohistochemistry in 4% paraformaldehyde in 0.1 M phosphate buffer after transcardial perfusion with cold saline (0.9%).
Tissue Preparation
Pups were killed under anesthesia with pentobarbital (50 mg/kg IP) and were perfused transcardially with 0.9% saline followed by 4% paraformaldehyde in phosphate buffer. Brains were removed and postfixed in the same fixative at 4°C overnight, cut into coronal blocks containing the hippocampus (approximately 5 mm thickness), and postfixed in the same fixative at 4°C for an additional 2 nights. After fixation, each tissue block was equilibrated in 30% sucrose solution in phosphate-buffered saline (PBS) and cut using a cryostat (Leica Microsystems, Germany) into 10-μm-thick coronal sections. Sections were then stored in a cryoprotective solution at −20°C until used for immunohistochemistry.
Immunohistochemistry
Free-floating coronal sections (10 μm) were incubated in PBS containing 0.3% Triton X-100 at 4°C overnight, then washed three times (5 min each) in PBS at room temperature (RT). For BrdU immunohistochemistry, sections were incubated in 2N HCl at 37°C for 20 min to denature DNA, rinsed twice in 0.1 mol/L borate buffer (pH 8.5) at RT for 15 min each, and then three times (5 min each at RT) in PBS. Sections were double- or triple-stained for BrdU, HCNP, and various lineage markers (sex-determining region Y-box 2 (SOX2): neural progenitor cells; doublecortin (DCX): immature neurons; glial fibrillary acidic protein (GFAP): astrocytes; CNPase: oligodendrocytes). Sections were incubated with primary antibodies diluted with PBS containing 0.3% Triton X-100 and 1% bovine serum albumin at 4°C overnight. The following antibodies were used as primary antibodies; rat monoclonal anti-BrdU antibody (1:500, AbD Serotec, UK), mouse monoclonal anti-SOX2 antibody (1:200, R&D Systems, MN, USA), guinea pig polyclonal anti-DCX antibody (1:2,000, Millipore), mouse monoclonal anti-GFAP antibody (1:1,000, Millipore, MA, USA), and mouse monoclonal anti-CNPase antibody (1:500, Millipore). We also generated a rabbit polyclonal anti-HCNP antibody and incubated sections with this antibody after dilution (1:500), as described previously (33,35). After three more 5-min washes in PBS, the sections were labeled with secondary antibodies for 2 h at RT, using AlexaFluor 350, 488, 594 (Molecular Probes, OR, USA) or Pacific Blue-labeled goat anti-mouse, rat, guinea pig, or rabbit IgG antibodies (1:400, Invitrogen, CA, USA) as secondary antibodies. After several washes with PBS, the sections were mounted on glass slides and covered with coverslips using Fluoromount (Diagnostic BioSystems, CA, USA). Fluorescent signals were detected under an Axiovision fluorescent microscope (Zeiss, Germany) at excitation wavelengths of 365 nm (AlexaFluor 350), 470 nm (AlexaFluor 488), and 590 nm (AlexaFluor 594) and an LSM5 Pascal laser confocal microscope (Zeiss) at excitation wavelengths of 405 nm (Pacific Blue, Invitrogen), 488 nm (AlexaFluor 488), and 543 nm (AlexaFluor 594).
Image Analysis and Quantification
The percentage of HCNP+ newborn cells was assessed by observing brain sections at 5 and 14 days after HI insult. Five coronal sections from each of five rats were obtained every 100 μm, located −3.14 to −3.64 mm from the bregma. The area including the dentate granular cell layer (GCL), the subgranular zone (SGZ), and the hilus was considered to be the region of interest for quantification. BrdU+ cells were detected, and the expression of HCNP-related protein signals in these cells was determined using fluorescence and confocal microscopy. The percentage of BrdU+/HCNP-pp+ cells was calculated relative to the total number of BrdU+ cells.
The phenotype of BrdU+/HCNP-pp+ cells at 14 days after ischemia was assessed using a triple-immunostaining technique. BrdU+/HCNP-pp+ cells in the GCL, SGZ, and hilus were detected, and their expression of DCX, GFAP, or CNPase was subsequently determined using Axiovision. The triple-positive percentages were calculated as BrdU+/HCNP-pp+/DCX+, BrdU+/HCNP-pp+/GFAP+, and BrdU+/HCNP-pp+/CNPase+ cells relative to total BrdU+/HCNP-pp+ cells. Mean values were obtained from five sections from each of five rats.
Statistical Analysis
Each experiment was repeated independently at least three times. One-way analysis of variance (ANOVA) and Tukey–Kramer tests were used to compare means among groups. The results are reported as means ± one standard deviation (SD). Statistical significance is represented by ∗p < 0.05.
Results
HCNP-pp Expression in Neural Progenitors in Hippocampus After HI
We previously reported that HCNP-pp was expressed at low levels in neural stem cells during the neonatal period (39,52). HCNP-pp is expressed in neurons and oligodendrocytes in the developing brain and may control lineage differentiation via mitogen-activated protein kinase (MAPK) signaling (32). We also recently reported that downregulation of HCNP-pp expression in AHPs in vitro may encourage the differentiation of GFAP+ astrocytes (42). However, little is known about HCNP-pp expression in neural progenitor cells in the adult brain after ischemia. To determine the validity of the HI model, we initially confirmed the presence of ischemia-induced progenitor cells in the hippocampus. Bromodeoxyuridine (BrdU)+ progenitor cells expressing sex-determining region Y-box 2 (SOX2) were increased at the inner edge of the dentate granule cells in the hippocampus 5 days after HI, compared with the contralateral hippocampus as hypoxia control or with sham-operated brains (Fig. 2A). This suggested that the ischemic model produced results consistent with those from previous reports (34).
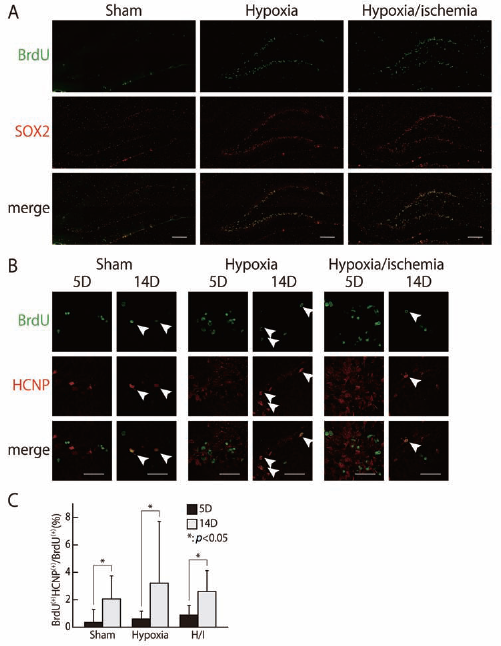
Changes in HCNP precursor expression in hippocampus after hypoxia–ischemia. Most BrdU+ cells in the dentate granular layer expressed the neural progenitor marker SOX2 at 5 days after hypoxic–ischemic surgery (A; green BrdU, red SOX2). The number of double-positive cells in the hippocampus tended to be higher in hypoxic and hypoxic–ischemic brains, compared with sham-operated brains (A, C). Some immature BrdU-positive progenitor cells showed HCNP precursor expression (B; green BrdU, red HCNP precursor). The number of immature BrdU+ progenitor cells gradually increased at 14 days in all groups, compared with the numbers at 5 days, although all numbers were small (B, C). Animals: n = 5 per each group. Scale bar: 20 μm. Arrowheads indicate positive cells. ∗p < 0.05.
We then examined the involvement of HCNP-pp during neurogenesis in HI brains by monitoring its expression in BrdU+ AHPs. BrdU positivity was considered to identify neural progenitor cells, and HCNP-pp expression was detected in BrdU+ cells by immunohistochemistry (Fig. 2B). At 5 days after ischemia, the incidences of BrdU+/HCNP-pp+ progenitor cells were higher in the hippocampus of HI and hypoxia brains, compared with sham-operated brains, although the difference was not significant. At 14 days after HI surgery, the proportion of BrdU+/HCNP-pp+ progenitor cells had increased in all groups, compared with the numbers at 5 days after surgery (sham 5D: 0.35 ± 0.94%, 14D: 2.06 ± 1.67%; hypoxia 5D: 0.60 ± 0.57%, 14D: 3.21 ± 4.50%; hypoxia–ischemia 5D: 0.88 ± 0.70%, 14D: 2.60 ± 1.52%) (p < 0.05) (Fig. 2B, C). These results suggest that some progenitor cells express HCNP-pp at some stage during neural lineage development, whereas this expression of HCNP-pp during neural lineage development was not specific in hippocampus of HI or hypoxia.
Different Lineages of Neural Progenitor Cells Expressing HCNP-pp
To examine changes in the expression of HCNP-pp during progenitor cell differentiation, BrdU+ progenitor cells in the hippocampus of HI, hypoxic, or sham-operated rats were coimmunostained for HCNP-pp and neural lineage markers, including doublecortin (DCX) for neurons, 2′,3′-cyclic nucleotide 3′-phosphodiesterase (CNPase) for oligodendrocytes, and GFAP for astrocytes (Figs. 3–5). BrdU+ progenitors were immunostained with each lineage marker over the 14 days following surgery. DCX+ neuroblasts in the hippocampus of HI rats were faintly observed among the BrdU+/HCNP-pp+ progenitors, although no DCX+ cells were seen among the BrdU+/ HCNP-pp+ progenitors in the hippocampus of hypoxic or sham-operated brains (HI: 16.0 ± 36.2%; hypoxia: 0%; sham: 0%) (Figs. 3 and 6). This suggests that the characteristics of BrdU+ neuroblasts induced by HI might differ from those in hypoxic or sham-operated brains.
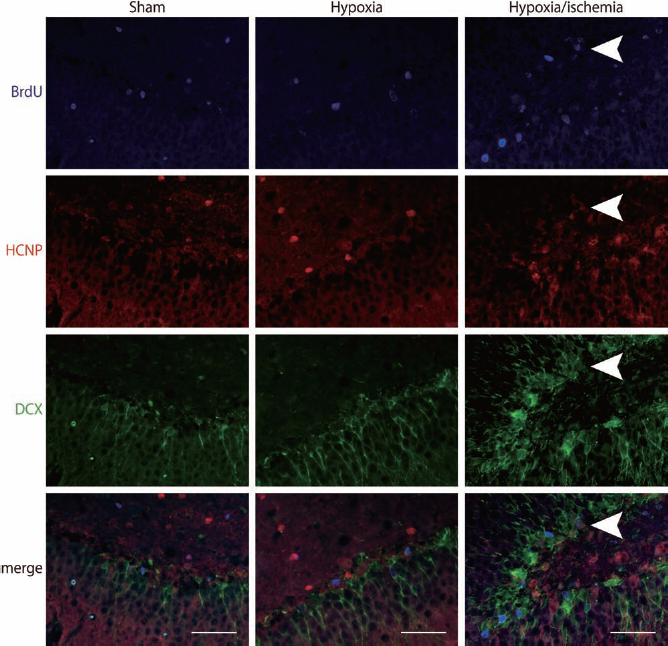
Expression levels of HCNP precursor determine phenotypic expression of the neural lineage marker DCX. Colocalization of the neural lineage maker, DCX, CNPase, was evaluated in BrdU+/HCNP-pp+ at 14 days after surgery in sham-operated, hypoxic, and hypoxic–ischemic rats (blue BrdU, red HCNP precursor, green DCX). Less than 20% of BrdU+ progenitor cells in the hippocampus expressed DCX in hypoxic–ischemic rats, and no DCX+ cells were seen in the hippocampus in sham-operated or hypoxic rat brains. Scale bars: 20 μm.

Expression levels of HCNP precursor determine phenotypic expression of the neural lineage marker CNPase. Colocalization of the neural lineage maker, CNPase, was evaluated in BrdU+/HCNP-pp+ at 14 days after surgery in sham-operated, hypoxic, and hypoxic–ischemic rats (blue BrdU, red HCNP precursor, green CNPase). Almost half of the BrdU+/HCNP-pp+ cells also expressed the oligodendritic marker CNPase at 14 days after surgery, with no significant differences among the three groups. Scale bars: 20 μm.

Expression levels of HCNP precursor determine phenotypic expression of the neural lineage marker GFAP. Colocalization of the neural lineage maker, GFAP, was evaluated in BrdU+/HCNP-pp+ at 14 days after surgery in sham-operated, hypoxic, and hypoxic–ischemic rats (blue BrdU, red HCNP precursor, green GFAP). No GFAP+/BrdU+/HCNP-pp+ cells were detected in the hippocampus in any of the groups, although GFAP+ cells were increased in the hippocampus in hypoxic–ischemic brains, compared with hypoxic and sham-operated brains.
We assessed CNPase positivity in this subpopulation to determine the relative contribution of BrdU+/HCNP-pp+ progenitor cells to the oligodendrocyte lineage. CNPase+ cells comprised 63.3 ± 44.2%, 47.8 ± 48.3%, and 46.7 ± 48.0% of BrdU+/HCNP-pp+ cells in the hippocampus of HI, hypoxic, or sham-operated brains, respectively, at 14 days after surgery (Figs. 4 and 6). This suggests that almost half of the BrdU+/HCNP-pp+ cells in all groups at this stage may represent the oligodendrocyte lineage, although there were no significant differences among the three groups. In contrast, no GFAP+ cells were detected among the BrdU+/HCNP-pp+ cells in the hippocampus in any of the groups, whereas GFAP+ cells were present among the BrdU+/HCNP-pp- cells (Figs. 5 and 6). These results are consistent with those of our previous reports on the differentiation of adult progenitor cells into specific neural lineages (42) and on the expression pattern of HCNP-pp in the adult rat brain (35,52).
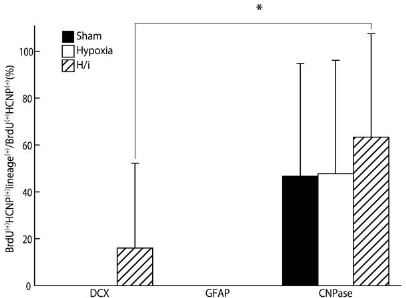
Summary of cell lineages observed under experimental conditions as demonstrated by BrdU/HCNP staining colocalized with the different neural lineage markers, DCX, GFAP, and CNPase. Data are expressed as means ± SD (n = 5). ∗p < 0.05.
Discussion
The results of this study demonstrated that the proportion of BrdU+/HCNP-pp+ progenitors in the hippocampus gradually increased during the first 14 days after ischemia in HI, hypoxic, or sham-operated rat brains, although the proportions remained small in all groups. Foremost, we showed that the HCNP-pp expression was associated with the phenotypic expression of differentiated lineage markers, such as DCX, GFAP, and CNPase. Almost half of the BrdU+/HCNP-pp+ progenitor cells in the hippocampus may differentiate along the oligodendrocyte lineage in HI, hypoxic, or sham-operated brains. In contrast, a small number of double-positive-stained cells in the hippocampus expressed the neuronal lineage marker, DCX, but only in HI brains. No BrdU+/HCNP-pp+ progenitors expressed the astrocyte lineage marker GFAP in any of the groups of rats. These results suggest that HCNP-pp contributes to the differentiation of progenitor cells into specific neural lineages in the ischemic brain.
The SVZ provides a well-characterized source of newly generated neurons during development. In the first few weeks of postnatal life, the SVZ also generates astrocytes and oligodendrocytes that migrate radially towards overlying structures to complete a sequence of glial development (27,46). The hippocampal DG, along with the SVZ, provides a source of newly generated neurons throughout the life of rodents (29). Under normal conditions, the DG and SVZ generate immature neurons that migrate towards the CA1 pyramidal layer and the olfactory bulb, respectively, where they can differentiate into mature neurons (12). Several recent studies have demonstrated significant postnatal neurogenesis in rodent models of ischemic brain injury (25). Following neonatal HI, immature neurons may emerge from either the SVZ or DG and migrate towards the site of injury, followed by differentiation into neurons or glia (18,25,28,36,55). However, few neuronal progenitors originating from the cells of the lateral ventricle, including the SVZ, differentiate into region-appropriate neurons (21,45). Furthermore, neonatal stroke may disrupt gliogenesis, reducing the overall number of new glial cells and shifting the population toward astrocytes (28,45). This acceleration of gliosis might be one of the factors responsible for interrupting functional neuronal recovery after ischemic injury. Overall, these results suggest that the induction of differentiation of newly generated neural progenitor cells into mature neurons may have potential applications in the treatment of ischemic brains.
We previously purified a peptide, HCNP, from the soluble fraction of hippocampi from young adult rats, which induced the synthesis of acetylcholine in cholinergic neurons from the medial septal nucleus (37). HCNP and nerve growth factor (NGF) play synergistic roles in the induction of acetylcholine synthesis in an in vitro tissue culture system (38,39). HCNP-pp has been reported as a modulator of cell signaling that functions as an endogenous inhibitor of multiple kinases (20,30,50,53). We also reported that HCNP-pp may inhibit the MAPK cascade during brain development (32). Furthermore, HCNP-pp expression was reported in all cultured AHPs, although its expression has only been confirmed in vivo in neurons and oligodendrocytes of postnatal rat brain during development (11,35,52). We recently showed that elevated HCNP-pp expression accompanied the differentiation of AHPs into neurons and oligodendrocytes, whereas its downregulation resulted in AHP fate commitment into astrocytes (42). These distinct patterns of HCNP-pp expression in neural progenitor cells are likely to contribute to the appropriate development of the brain, especially the hippocampus. In the current study, BrdU+/HCNP-pp+ neuronal progenitors induced in HI, as well as in sham-operated brains, did not differentiate into GFAP+ astrocytes as shown in vitro; instead these HCNP+ progenitors mainly developed along the CNPase-positive oligodendrocyte lineage. Furthermore, a few BrdU+/HCNP-pp+ progenitors induced by HI were able to develop along the DCX+ neuronal lineage. It is possible that continuous HCNP-pp expression may inhibit the differentiation of neural progenitor cells into astrocytes and that HCNP-pp+ progenitors induced by HI may contain clusters of cells with different characteristics from those in the nonischemic hippocampus.
Several recent studies have revealed that some signal cascades may be involved in the differentiation of neural progenitors in the ischemic brain and the spinal cord injury (9,14,23,43,54). Notch signaling via Jagged1 (Jag1) and Delta-like ligand 4 (DLL4) may represent one important cascade in the differentiation of neural progenitors towards neuronal cells; this signal may be enhanced by growth factors such as epidermal growth factor and fibroblast growth factor-2 (FGF-2) (2,15,40). The activation of glial genes, such as GFAP via the JAK-STAT pathway, may be one of the key mechanisms in the astrocytic differentiation of neural progenitors (6). Some environmental factors, including transforming growth factor β, may also play a crucial role in astrocytic specification of neural progenitors in neonatal HI brain (4). Regarding oligodendrocyte differentiation, MAPK activation may represent an important specific intracellular signaling factor, promoted by platelet-derived growth factor, FGF-2, NGF, neurotrophin 3, and brain-derived neurotrophic factor (1,5,7,13,16,24,51). The levels of HCNP-pp have been found to be lowered in the brain cortex in chronic hypoxic rats, suggesting elevated levels of phospho-extracellular signal-regulated kinase (pErk) (8). pErk in the hippocampus was elevated in response to ischemic stimuli, showing a biphasic pattern at 15 min and 24 h after ischemic reperfusion. N-methyl-d-aspartate receptors and Src family tyrosine kinases were essential for the upregulation of Erk activation. Furthermore, HCNP-pp was involved in this biphasic phosphorylation of Erk via blockade of the Src-Raf cascade in the acute ischemic hippocampus, while Src family tyrosine kinases were critical for modulation of the Ras/Raf/Mek/Erk cascade (19). These reports suggest that HCNP-pp may play a crucial role in changes in Erk activation after brain ischemia, with implications for the regulation of cell survival or death.
HCNP-pp has recently been shown to regulate activity of the Erk pathway in myoblasts via its role as a scaffold protein (17). Overexpression of HCNP-pp may result in the conversion of epithelial cells to a highly migratory fibroblast-like phenotype, indicating a role for HCNP-pp as a key regulator of cell motility and in switching the cells from proliferation to differentiation via modulation of the Erk pathway (44,56). However, the involvement of pErk downregulation in the differentiation of neural progenitor cells into astrocytes remains unknown. Our in vivo and in vitro data and the observations that HCNP-pp overexpression inhibits differentiation into GFAP+ astrocytes, while reduction in HCNP-pp induces astrocyte differentiation, suggest a role for HCNP/HCNP-pp in neurogenesis and/ or gliogenesis in AHPs via novel mechanisms, other than its function in inhibiting the Erk pathway.
The main limitation of the current study was that the cell lineages were only followed for 14 days after ischemia. Further studies are needed to confirm the involvement of HCNP/HCNP-pp in the differentiation of the neural lineage, including examining the effects of aging. Additionally, laboratory investigations may reveal cellular and molecular links between neurogenesis (i.e., BrdU) and HCNP-pp expression under pathological conditions, such as ischemic brain injury.
In conclusion, this study demonstrated that the downregulation of HCNP/HCNP-pp expression might be involved in the regulation of gliogenesis in neural progenitor cells during brain ischemia, through inducing the differentiation of astrocytes. Future studies to elucidate the exact mechanisms underlying the effects of alterations in HCNP/HCNP-pp on specific neural lineages might lead to the discovery of new target molecules to suppress gliosis induced by brain ischemia. A better understanding of regulatory pathways associated with neural cell differentiation may guide the development of therapeutic strategies targeting HCNP-pp to enhance neurogenesis following ischemia.
Footnotes
Acknowledgments
N.M. is supported by grants from Mitsubishi Pharma Research Foundation, Ichihara International Scholarship Foundation, and General Assembly of Japanese Association of Medical Sciences. The authors declare no conflicts of interest.
