Abstract
The therapeutic efficacy of cell-based therapy after stroke can be enhanced by making the host brain tissue more receptive to the administered cells, which thereby facilitates brain plasticity. We hypothesized that simvastatin increases human umbilical cord blood cell (HUCBC) migration into the ischemic brain and promotes brain plasticity and neurological functional outcome after stroke. Rats were subjected to 2-h middle cerebral artery occlusion (MCAo) and administered subtherapeutic doses of simvastatin (0.5 mg/kg, gavaged daily for 7 days), HUCBCs (1 × 106, one time injection via tail vein), or combination simvastatin with HUCBCs starting at 24 h after stroke. Combination treatment of stroke showed an interactive effect in improvement of neurological outcome compared with simvastatin or HUCBC monotherapy groups. In addition, combination treatment significantly increased brain-derived neurotrophic factor/TrkB expression and the number of engrafted HUCBCs in the ischemic brain compared with HUCBC monotherapy. The number of engrafted HUCBCs was significantly correlated with functional outcome (modified neurological severity score). Combination treatment significantly increased neurogenesis and synaptic plasticity in the ischemic brain, and promoted neuroblast migration in cultured subventricular zone explants. Using primary cultured neurons (PCNs), we found that combination treatment enhanced neurite outgrowth compared with nontreatment control, simvastatin or HUCBC supernatant monotherapy. Inhibition of TrkB significantly attenuated combination treatment-induced neurite outgrowth. Our data indicate that combination simvastatin and HUCBC treatment of stroke increases BDNF/TrkB expression, enhances HUCBC migration into the ischemic brain, amplifies endogenous neurogenesis, synaptic plasticity and axonal growth, and thereby improves functional outcome after stroke.
Introduction
Human umbilical cord blood cells (HUCBCs) are a rich source of hematopoietic and mesenchymal progenitor cells, and have potential as a clinical therapeutic agent, since they are easily isolated without ethical and technical problems (5,16,23,24). HUCBCs when administered to ischemic brain proliferate, differentiate, and secrete factors beneficial for the host brain tissue in vivo (29,30,34). However, the effect of HUCBC transplantation is dependent on the number of transplanted HUCBCs (36), and the success of a vascular route for cell therapy has been limited by the low migration efficiency and low survival rates of the transplanted progenitor cells in the lesioned area. Therefore, identifying conditions that enhance HUCBC migration into the ischemic brain and increase the transplanted HUCBC survival is of considerable clinical interest.
Recent data suggest that selected cell-based and pharmacological therapies that promote neurogenesis, synaptic plasticity, and axonal outgrowth provide opportunities to improve clinical outcomes and brain functional recovery (17,39,44). Statins, 3-hydroxy-3-methylglutaryl-coenzyme A (HMG-CoA) reductase inhibitors, are a class of drugs originally used to lower cholesterol. Epidemiological data in a large population of first-ever ischemic stroke patients showed that pretreatment with statins before first-ever ischemic stroke was associated with better early outcome with a reduced mortality during hospitalization and neurological disability at hospital discharge (1). Our previous studies have shown that treatment of stroke with an effective dose of simvastatin (1 mg/kg) amplifies angiogenesis and vascular stabilization, and promotes arteriogenesis in rats (3,41,42). A subtherapeutic dose of simvastatin (0.5 mg/kg) alone or a subtherapeutic dose of bone marrow stromal cells (BMSCs) alone did not improve functional outcome. However, a significant increase in the functional outcome was found with combination treatment using a subtherapeutic dose of simvastatin and BMSCs after stroke in rats (8). Whether simvastatin promotes HUCBC migration into the ischemic brain and whether combination treatment of stroke with subtherapeutic doses of simvastatin and HUCBCs promotes endogenous neurogenesis and synaptic plasticity, and thereby augments poststroke restorative therapy have not been investigated. In this study, we investigate the therapeutic benefit of a combination of simvastatin and HUCBCs to amplify the therapeutic effect of HUCBC-based therapy to improve the functional outcome after stroke in adult rats. We hypothesize that combination treatment promotes HUCBC migration into the ischemic brain by increasing brain-derived neurotrophic factor (BDNF) and its receptor tropomyosin-related kinase B (TrkB) expression within the compromised brain.
Materials and Methods
All experiments were conducted in accordance with the standards and procedures of the American Council on Animal Care and Institutional Animal Care and Use Committee of Henry Ford Health System.
Middle Cerebral Artery Occlusion (MCAo) Model and Animal Groups
Adult male Wistar rats (Jackson Laboratory) weighing 270—300 g were used for inducing a preclinical stroke model (MCAo). Transient right hemisphere MCAo was induced for 2 h by advancing a 4-0 surgical nylon suture (18.5—19.5 mm) with an expanded (heated) tip from the external carotid artery into the lumen of the internal carotid artery to block the origin of the MCA (4). Twenty-four hours after surgery, rats were divided randomly into four groups and treated with: a) phosphate-buffered solution (PBS, GIBCO), gavaged daily for 7 days; b) subtherapeutic dose of simvastatin (0.5 mg/kg, Sigma), gavaged daily for 7 days (8); c) subtherapeutic dose of HUCBCs (1 × 106, Saneron CCEL Therapeutics, Inc., Tampa, FL) in 1 ml of PBS intravenous injected via a tail vein, one time; d) combination simvastatin (0.5 mg/kg, daily for 7 days) and HUCBCs (1 × 106). Immunosuppression with cyclosporine was not performed in the present study, which is consistent with previous studies performed by us and others (5,30). Cyclosporine may also affect stroke outcome. Cyclosporine A increases the risk of convulsions in patients with cerebral infarction and/or at an early stage following focal cerebral ischemia (40). 5-Bromodeoxyuridine (BrdU, 100 mg/kg/day, Sigma-Aldrich, St. Louis, MO), as a marker for proliferating cells, was injected intraperitonealy 24 h after stroke daily for 14 days.
Neurological Functional Tests
A series of functional tests including a modified neurological severity score (mNSS), adhesive removal test, and foot-fault evaluation were performed before MCAo and 1, 7, and 14 days after MCAo by an investigator who was blinded to the experimental groups, as previously described (5,6).
mNSS is a composite of motor, sensory, balance, and reflex tests (5). Neurological function was graded on a scale of 0 to 12 (normal score 0; maximal deficit score 12) with one point awarded for the exhibition of specific abnormal behavior or for lack of a tested reflex. A greater impairment of normal function results in a higher score. The adhesive removal test requires the use of adhesive-backed paper dots of equal size (113.1 mm2) as bilateral tactile stimuli on the distal-radial region of each forelimb (4). Prior to surgery, rats were trained for 3 days to remove the adhesive. Once the rats are able to remove the adhesive dots within 10 s, they were subjected to MCAo. Post-MCAo, the time to remove each stimulus from both forelimbs was recorded on three trials per day. Individual trials were separated by at least 5 min. In the foot-fault test, rats are tested for placement dysfunctions of forelimb (5,6). Rats were placed on elevated hexagonal grids of different sizes. The number of steps used to cross the grid was counted up to 100 steps or 10 min, whichever came first, and the total number of foot-faults for each forelimb was recorded. Foot-fault values were presented as the number of missteps of the contralateral forelimb divided by the total number of steps counted. It is well established that in animals with MCAo, the impaired (contralateral) limbs faulted more often than nonimpaired limbs.
Lesion Volume Measurement
Rats were sacrificed 14 days after MCAo by transcardial perfusion with saline, followed by perfusion and immersion in 4% paraformaldehyde before being embedded in paraffin (n = 8/group). Seven coronal sections of tissue were processed and stained with hematoxylin and eosin for calculation of the lesion volume. The seven brain sections were traced with the use of the Global Laboratory image analysis system (Data Translation). To measure the lesion volume, the indirect lesion area was calculated, in which the intact area of the ipsilateral hemisphere was subtracted from the area of the contralateral hemisphere. Lesion volume is presented as a volume percentage of the lesion compared with the contralateral hemisphere (4,33). Lesion volume was measured by blinded manner.
Immunohistochemical Assessment
For immunostaining, a standard paraffin block was obtained from the center of the lesion (bregma −1 to +1 mm). A series of 6-μm-thick sections was cut from the block. Every 10th coronal section for a total of five sections was used for immunohistochemical staining. Antibody against BrdU (a marker of proliferating cells, 1: 100; Boehringer Mannheim), doublecortin (DCX, a marker of migrating neuroblasts, 1:100; C-18, Santa Cruz Biotechnology), synaptophysin (Syn, a marker of presynaptic plasticity and synaptogenesis, 1:1000, Chemicon), and anti-human-specific monoclonal nuclear protein NuMa (1:15; Ab-2, 107-7, EMD Chemicals) (9,22), BDNF (1:300, Santa Cruz), and TrkB (1:500, Santa Cruz)) immunostaining was performed. In addition, Bielshowsky silver and Luxol fast blue (LFB) histohemical stainings were used to identify axons and myelin, respectively, as previously described (7). Control experiments consisted of staining brain coronal tissue sections as outlined earlier, but omitted the primary antibodies.
Immunostaining Quantitation
For quantification of NuMa, BrdU, DCX, BDNF, synaptogenesis, Bielshowsky silver, and LFB immunostaining, five slides from the standard reference coronal section of each brain, with each slide containing eight fields from the ischemic border zone (IBZ, adjacent to the ischemic core area), were digitized under a 40x objective (BX40; Olympus Optical) using a 3-CCD color video camera (DXC-970MD, Sony) interfaced with a Micro Computer Imaging Device (MCID) software (Imaging Research). For quantitative measurements, the total numbers of NuMa-positive human cells (HUCBCs) both in the ischemic ipsilateral and contralateral brain hemisphere, the percentage number of BrdU-positive cells in the subventricular zone (SVZ), and the percentage number of the DCX-positive neuroblasts both in the SVZ and in the ischemic ipsilateral striatum were measured. For quantitative synaptophysin, BDNF, and TrkB expression, and Bielshowsky silver and LFB, the percentage of synaptophysin-, BDNF-, and TrkB-positive area in the IBZ, and the percentage of Bielshowsky silver- and LFB-positive areas in the bundle of stratum in the IBZ were also measured in each section using the MCID imaging analysis system, respectively (7,8). Immunostaining and quantitation were performed by an investigator blinded to the experiment.
Enzyme-Linked Immunosorbent Assay (ELISA)
To test whether combination treatment regulates BDNF expression in the ischemic brain after stroke, MCAo, simvastatin, HUCBC, and combination treatment animals were killed 3 days after MCAo (n = 4/group). Brain extracts were obtained from the ischemic border identified visually (bregma −1 to +1 mm, border region encompassing the ischemic core) at 3 days after MCAo. Tissue blocks were dissected on ice, and wet weight was rapidly measured. The brain extracts were divided into 200-μl triplicate samples. Using ELISA kits (R&D Systems, Minneapolis, MN; Calbiochem), BDNF ELISA was performed.
Subventricular Zone (SVZ) Explant Cell Migration
To investigate whether combination treatment increases endogenous SVZ neural progenitor cell (NPC) migration, the SVZs derived from MCAo, simvastatin treatment, HUCBC treatment, or combination treatment animals 7 days after MCAo (n = 4/group) were dissociated. The SVZ explants were cut to 1-mm3 sections and plated in BD Matrigel™ Matrix (BD Biosciences, Bedford, MA) in 24 wells (6 wells each group) with 1 ml of Neuralbasal-A medium (Invitrogen) containing 2% of B27 supplement (Invitrogen). For specific measurement of SVZ-NPC migration, DCX (a marker of migrating neuroblast, 1:250, Santa Cruz) immunofluorescent conjugated with Cy3 staining was performed and the cell migration length was measured at 5 days after culture. The 4x objective with 1.5x electronic zoom of an Olympus IX71 microscope with a CCD camera (CoolSNAP, Proper Scientific Photometrics) and Meta View software (Universal Imaging, West Chester, PA) were used for acquiring images. The average distance of DCX-positive cell migration from the explant culture edge was measured by an investigator blinded to the explanted culture using the Meta View software.
Primary Cultured Neuron (PCN) and Neurite Outgrowth Measurement
To test whether combination treatment increases dendrite outgrowth and whether the mechanisms underlying the enhanced neurite outgrowth are mediated by the BDNF/TrkB pathway, PCN culture was employed and oxygen-glucose deprivation (OGD) was induced in vitro, as previously described (7). Briefly, embryonic day 17 (E17) cortical cells were isolated from embryonic brain of Wistar rats and cultured in four-chamber slides with Neuralbasal-A medium (GIBCO) containing 2% B27 medium supplement (GIBCO) and antibiotics for 7 days. To mimic the ischemic condition in vivo, OGD was induced within an anaerobic chamber (model 1025, Forma Scientific) by 85% N2, 10% H2, and 5% CO2, at 37°C for 1 h. The PCN cultures were removed from the anaerobic chamber and rinsed with PBS. The PCN cultures were divided into six groups, six chambers each group with 0.5 ml medium: 1) nontreatment for control; 2) + simvastatin 1 μM; 3) + HUCBC supernatant (0.5 ml); 4) + simvastatin + HUCBC supernatant; 5) BDNF 50 ng/ml; 6) simvastatin + HUCBC supernatant + TrkB inhibitor (K252a, 200 nM, Calbiochem, Cat# 480354), for 24 h. Then the PCN cultures were performed for neuron-specific class III β-tubulin (TUJ1, a phenotypic marker of neural cells) immunofluorescent staining using a monoclonal anti-TUJ1 antibody (Covance, 1:1000) with Cy3 for PCN number counting and neurite outgrowth measurement (7,14).
To trace the axonal arbors of fluorescently labeled neurons, the fluorescent photomicrographs were captured at 40x magnification with a digital camera, the TUJ1-positive PCN numbers and the length of TUJ1-positive dendrites were measured using MCID analysis system (27). The average number of TUJ1-positive PCNs per 40x field and the average length of total 20 neuronal dendrite outgrowths were presented. Neurite outgrowth was measured by blinded manner.
Statistical Analysis
Two-way ANOVA (univariate or multivariate analysis of variance) with +/- Simvastatin and +/- HUCBC as two of the factors was used for analyzing the data of functional evaluation, lesion volume, BrdU, DCX, synaptophysin, Bielshowsky silver, LFB, BDNF/TrkB immunostaining, and neuroblast migration in SVZ, and one-way ANOVA was used for neurite outgrowth in PCN. Post hoc test (Tukey) was used for multiple comparison if an overall treatment group effect was detected at p < 0.05. Independent two-sample t-test was used for testing the number of NuMa-positive HUCBCs that migrate into the ischemic ipsilateral and contralateral brain between two groups. Pearson partial correlations after bivariate correlation were used to analyze the correlation of the engrafted HUCBC number in the ischemic brain with the neurological functional outcome (i.e., mNSS, adhesive removal, and foot-fault test, respectively). All data are presented as mean + SE.
Results
Combination Treatment of Stroke Significantly Improves Neurological Outcome
Two-way ANOVA analysis did not reveal a significant interaction between simvastatin and HUCBC monotherapy on adhesive removal and foot-fault test. Instead, simvastatin monotherapy showed a significant effect on foot-fault test 14 days after MCAo (F = 8.5, p < 0.01); HUCBC monotherapy showed significant functional benefits both on foot-fault test (7 days: F = 39.1, p < 0.01; 14 days: F = 44.0, p < 0.01) and adhesive-removal test (7 days: F = 22.5, p < 0.01; 14 days: F = 20.3, p < 0.01) after stroke. However, statistically significant effects of simvastatin monotherapy (7 days, F = 15.6, p < 0.01; 14 days: F = 21.8, p < 0.01) and HUCBC monotherapy (7 days, F = 27.4, p < 0.01; 14 days: F = 36.9, p < 0.01) and a positive interactive effect of them (14 days: F = 7.3, p < 0.05) on mNSS after stroke were confirmed by two-way ANOVA (Fig. 1A). These data indicate that combination treatment of stroke significantly improves functional outcome after stroke.

Combination simvastatin and human umbilical cord blood cell (HUCBC) treatment of stroke improves functional outcome, decreases lesion volume, and enhances HUCBC migration in the ischemic brain in rats. (A) Modified neurological severity score (mNSS) test. (B) Lesion volume. (C) NuMa-HUCBC migration in the ischemic brain. (D) Correlation of NuMa-HUCBCs with mNSS. n = 8/group. MCAo, middle cerebral artery occlusion. Scale bar in C: 50 μm.
Lesion Volume in the Ischemic Brain
Figure 1B shows a significant decrease in the ischemic lesion volumes 14 days after stroke in HUCBC treatment alone and combination groups without interaction with simvastatin treatment compared with the MCAo control group (p < 0.05, n = 8/group).
Combination Treatment of Stroke Significantly Increases the Number of Engrafted HUCBCs in the Ischemic Brain
Figure 1C shows that there is a significant increase in the number of NuMu-positive HUCBCs in the ipsilateral hemisphere in the combination treatment rats compared to HUCBC monotherapy rats (p < 0.05, n = 8/group). In addition, the immunostaining images showed that NuMu-HUCBCs primarily aggregated around the vascular vessels in the ischemic area of the brain. Figure 1D shows that the increased number of HUCBCs in the ischemic brain is significantly correlated with the improvement of mNSS test (r = −0.751, p < 0.05). These data indicate that combination treatment of stroke significantly increases the number of engrafted HUCBCs in the ischemic brain as well as promotes functional outcome after stroke. The numbers of HUCBCs present in the ischemic brain may contribute to improved functional recovery after stroke.
Combination Treatment of Stroke Significantly Increases NPC Proliferation and Migration in the Ischemic Brain
Neurogenesis is related to functional recovery after stroke (18). The rostral SVZ is one of neuroproliferative regions in adult animals, and the NPCs generated in the SVZ migrate to the ipsilateral striatum after injury (20). Figure 2A—E shows that the percentage of BrdU-positive cells in the SVZ significantly increased in simvastatin treatment alone and combination treatment groups compared with MCAo control group (p < 0.05, n = 8/group). Figure 2F—J shows that simvastatin, HUCBC, and combination treatment groups significantly increased DCX neuroblast cell numbers in the SVZ compared with MCAo control group (p < 0.05, n = 8/group). There was no interaction for simvastatin and HUCBC monotherapy on BrdU or DCX cells in the SVZ. However, a significant main effect for simvastatin (F = 5.5, p = 0.028) or HUCBC monotherapy (F = 26.3, p < 0.01) and a significant interaction effect (F = 5.2, p = 0.032) in DCX neuroblast numbers in the ischemic striatum were found. These data indicated that combination treatment of stroke significantly enhanced endogenous NPC generation in the ipsilateral SVZ and increased SVZ neuroblast migration into the ischemic ipsilateral striatum.
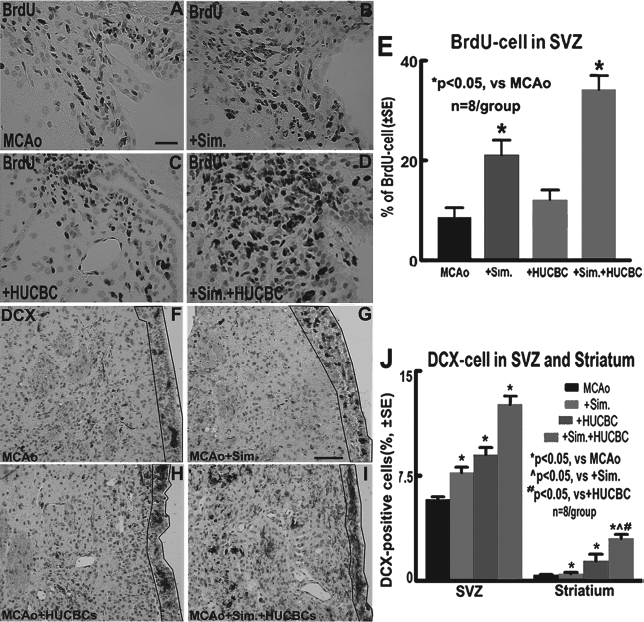
Combination simvastatin and HUCBCs treatment of stroke enhances neurogenesis in the ischemic brain. (A—E) 5-Bromodeoxyuridine (BrdU) cells in the subventricular zone (SVZ) and quantitative data. (F—J) Doublecortin (DCX) neuroblasts in the SVZ (frame) and striatum and quantitative data. n = 8/group. Scale bar in A: 50 μm, scale bar in G: 100 μm.
Combination Treatment of Stroke Increases Synaptic Plasticity and Axon Growth in the Ischemic Brain
Synaptic plasticity and axonal growth promote functional outcome after stroke. To test whether combination treatment of stroke induces synaptic plasticity and axon growth, synaptophysin, Bielschowsky silver, and LFB immunostaining were performed. Synaptophysin is a marker for presynaptic plasticity and synaptogenesis (35). Bielschowsky silver is a marker for axons (21,31). LFB is a marker of myelin. Figure 3A—E displays that HUCBC monotherapy did not increase synaptophysin. Simvastatin monotherapy significantly increased synaptophysin expression in the bundles of the IBZ (F = 16.9, p < 0.01). Moreover, there was a positive interaction between simvastatin and HUCBC monotherapy (F = 10.0, p < 0.01). Figure 3F—J shows that simvastatin (F = 64.8, p < 0.01) and HUCBC monotherapy (F = 32.6, p < 0.01) significantly increased Bielshowsky silver axon density in the IBZ compared with MCAo control. However, combination treatment displayed a significant interaction between the two monotherapy groups (F = 8.3, p = 0.007). In addition, the correlation analysis showed there was a significant correlation of synaptophysin staining in the ischemic brain with mNSS (r = −0.77, p < 0.05). Figure 3K—O shows that simvastatin treatment alone significantly increased LFB myelin in the bundles of the IBZ compared with MCAo control (p < 0.05, n = 8/group). HUCBC treatment alone did not increase LFB myelin; however, there was a significant increase when combined with simvastatin (F = 6.6, p = 0.016) measured by two-way ANOVA. These data indicated that combination treatment of stroke significantly increased synaptic plasticity and axonal growth after stroke, which may also contribute to the functional benefit after stroke.

Combination simvastatin and HUCBCs treatment of stroke enhances synaptic plasticity and axon growth in the ischemic brain. (A—E) Synaptophysin immunostaining and quantitative data. (F—J) Bielschowsky silver staining and quantitative data. (K—O) Luxol fast blue (LFB) immunostaining and quantitative data. n = 8/group. IBZ, ischemic border zone. Scale bar in A: 50 μm, scale bar in F and K: 40 μm.
Combination Treatment of Stroke Increases BDNF and TrkB Expression in the Ischemic Brain
BDNF/TrkB play an important role in neuronal survival, differentiation, synaptic plasticity, and neurogenesis, and enhance functional recovery after brain injury (10). To test the molecular mechanism of combination-induced brain plasticity, immunostaining for BDNF/ TrkB was performed. Fig. 4A—K shows that simvastatin or HUCBC treatment alone significantly increased BDNF and TrkB expression in the ipsilateral IBZ compared with MCAo control (p < 0.05, n = 8/group). A positive interactive effect of combination simvastatin and HUCBC treatment on BDNF (F = 9.2, p < 0.01) and TrKB (F = 6.31, p = 0.018) was confirmed by two-way ANOVA. To further verify whether single or combination treatment upregulates BDNF, ELISA assay of the ipsilateral brain tissue was also employed. Figure 4F shows that HUCBC treatment significantly increased BDNF protein level measured by ELISA assay compared with MCAo control group (p < 0.05, n = 4/group).

Combination simvastatin and HUCBC treatment of stroke increases brain-derived neurotrophic factor/tropomyosin-related kinase B (BDNF/TrkB) expression in the ischemic brain. (A—E) BDNF immunostaining and quantitative data. (F) Brain tissue ELISA assay. (G—K) TrkB immunostaining and quantitative data. n = 8/group. Scale bar in A and G: 50 μm.
Combination Treatment of Stroke Increases Ipsilateral SVZ Neuroblast Migration In Vitro
To further investigate whether combination treatment increased neurogenesis, the ipsilateral SVZ explant cultures derived from animals were employed. Figure 5A and B shows that simvastatin treatment and HUCBC treatment significantly enhanced DCX neuroblast (simvastatin: F = 68.3, p < 0.01; HUCBC: F = 85.6, p < 0.01) migration in the SVZ explant cultures compared with MCAo alone group. However, combination treatment displayed a significantly interaction effect on DCX neuroblast migration (F = 14.1, p < 0.01) compared with simvastatin or HUCBC treatment alone, respectively.

Combination simvastatin and HUCBCs treatment of stroke enhances neural progenitor cell (NPC) migration in the ipsilateral SVZ explant cultures derived from the ischemic brain, and increases neurite outgrowth in primary cultured neurons (PCNs) after oxygen-glucose deprivation (OGD). (A, B) DCX neuroblasts and quantitative data. (C—I) Neurite outgrowth and quantitative data in the PCNs. (C) Control, (D) + simvastatin 1 μM; (E) + HUCBC supernatant; (F) + simvastatin + HUCBC supernatant; (G) + BDNF 50 ng/ml; (H) + simvastatin + HUCBC supernatant + TrkB inhibitor (k252a); (I) quantitative data. n = 6/group. TUJ1, antibody for β III tubulin. Scale bar in A: 10 μm, scale bar in C: 50 μm.
Combination Treatment and BDNF Increase Neurite Outgrowth, Inhibition of TrkB Significantly Attenuates Combination Treatment-Induced Neurite Outgrowth in PCN Cultures
To further investigate the molecular mechanism of the combination treatment increase of axonal growth after stroke, PCN neurite outgrowth measurement was performed. Figure 5C—I shows that simvastatin (1 μM), HUCBC supernatant, and BDNF (50 ng/ml) significantly increased neurite outgrowth (TUJ1 staining of βIII tubulin) compared with nontreatment control. Moreover, combination simvastatin and HUCBC supernatant significantly increased neurite outgrowth compared with simvastatin or HUCBC supernatant monotreatment groups (p < 0.05, n = 6/group). However, inhibition of TrkB by using K252 significantly attenuated neurite outgrowth in the combination treatment group (p < 0.05, n = 6/group).
Discussion
HUCBCs can be used for autologous or allogeneic transplantation (32,37). Compared with BMSCs, HUCBCs are less mature than BMSCs and can be successfully used even when there is only a half-match of acceptors. Transplantation of a human umbilical cord blood-derived neural-like stem cell line (HUCB-NSCs) in a rat model of cortical infarct shows HUCB-NSC survival and extensive migration into damaged brain and some differentiation to neurons and astrocytes (24). HUCBCs when administered intravenously selectively migrate to the ischemic area in the brain and enhance functional recovery after stroke (5). Therefore, the intravenous treatment with HUCBCs may be a convenient strategy for clinical treatment of stroke patients. In the present study, we show that combination treatment with HUCBC and simvastatin significantly increased the number of engrafted HUCBCs in the ischemic brain, and improved functional outcome after stroke.
Stroke results in increases in neurogenesis in the SVZ in adult brain, which might be an endogenous attempt to self-repair (43). Increasing neurogenesis decreases cognitive and behavioral deficits following stroke (25). Neurogenesis consists of two processes: NPC proliferation in the neuroproliferative regions and migration into lesioned area (2). In the present study, we found that combination treatment of stroke significantly increased the number of BrdU-proliferating cells in the SVZ and also significantly increased the number of migration neuroblasts in the neighboring striatum, ipsilateral to the lesion. The increased neurogenesis is consistent with the benefit of functional outcome seen in the combination treatment animals. Our data also showed that combination treatment of stroke with subtherapeutic doses of simvastatin and HUCBCs failed to induce a significant interactive effect in lesion volume, foot-fault, and adhesive removal tests. However, combination treatment evoked significant interactive effects in the neuroblast migration, synaptogenesis, axon, and myelin growth in the ischemic brain after stroke. In vitro study also showed that combination treatment significantly enhanced SVZ NPC migration as well as PCN neurite outgrowth. Therefore, combination treatment of stroke with subtherapeutic doses of simvastatin and HUCBCs significantly enhanced neurogenesis, synaptic plasticity, and axon growth, which may, in concert, mediate functional improvements.
BDNF/TrkB plays an important role in neurogenesis and synaptic plasticity. Intraventricular infusion of TrkB-Fc fusion protein promotes ischemia-induced neurogenesis in the dentate gyrus of the adult rat (13). Cortically expressed BDNF supports the maintenance of cortical neuron size and dendrite structure (11). BDNF mutation influences synaptic plasticity (19). BDNF modifies neuronal glutamate sensitivity, [Ca2+] homeostasis, and promotes neuronal and synaptic plasticity after stroke (28). Voluntary exercise leads to an upregulation of BDNF and improves synaptic function (12). Simvastatin upregulats BDNF and increases neurogenesis, which is associated with therapeutic improvement after traumatic brain injury (26,38). Simvastatin treatment improves functional recovery after experimental spinal cord injury by upregulating the expression of BDNF (15). In the present study, combination treatment of stroke showed a significant upregulation of BDNF/ TrkB and synaptophysin expression as well as an increase of axonal growth in the ischemic brain. In vitro study also showed that combination treatment significantly increased SVZ explant NPC migration and PCN neurite outgrowth. However inhibition of TrkB attenuated neurite outgrowth induced by combination treatment. Our previous study also found that BDNF promotes SVZ explant cell migration, and inhibition of BDNF by using anti-BDNF neutralizing antibody significantly inhibited SVZ explant cell migration (6). These data support our hypothesis that the BDNF/TrkB pathway promotes neurogenesis and synaptic plasticity and axon growth after stroke.
In summary, our data demonstrate that combination simvastatin and HUCBC treatment of stroke significantly upregulates BDNF/TrkB, enhances HUCBC intracerebral transplantation, and increases neurogenesis, synaptic plasticity, and axon growth in the ischemic brain, which improve neurological functional recovery after stroke in rats. These studies have important implications for the restorative use of cell-based therapy, and provide a basis for priming brain with pharmacological agents to amplify the therapeutic effect of cell-based treatment of neurological disease.
Footnotes
Acknowledgments
The authors thank Qinge Lu and Supata Santra for technical assistance. This work was supported by National Institute of Neurological Disorder and Stroke (NINDS) 1R41NS064708 (J.C.), National Institute on Aging RO1 AG031811 (J.C.), NINDS PO1 NS23393 (M.C.), and American Heart Association grant 09GRNT2300151 (J.C.). J. Chen is a consultant for Saneron CCEL Therapeutics, Inc.
