Abstract
In the context of islet transplantation, experimental models show that induction of islet intrinsic NF-κB-dependent proinflammatory genes can contribute to islet graft rejection. Isolation of human islets triggers activation of the NF-κB and mitogen-activated kinase (MAPK) stress response pathways. However, the downstream NF-κB target genes induced in human islets during the isolation process are poorly described. Therefore, in this study, using microarray, bioinformatic, and RTqPCR approaches, we determined the pattern of genes expressed by a set of 14 human islet preparations. We found that isolated human islets express a panel of genes reminiscent of cells undergoing a marked NF-κB-dependent proinflammatory response. Expressed genes included matrix metallopeptidase 1 (MMP1) and fibronectin 1 (FN1), factors involved in tissue remodeling, adhesion, and cell migration; inflammatory cytokines IL-1β and IL-8; genes regulating cell survival including A20 and ATF3; and notably high expression of a set of chemokines that would favor neutrophil and monocyte recruitment including CXCL2, CCL2, CXCL12, CXCL1, CXCL6, and CCL28. Of note, the inflammatory profile of isolated human islets was maintained after transplantation into RAG-/- recipients. Thus, human islets can provide a reservoir of NF-κB-dependent inflammatory factors that have the potential to contribute to the anti-islet-graft immune response. To test this hypothesis, we extracted rodent islets under optimal conditions, forced activation of NF-κB, and transplanted them into allogenic recipients. These NF-κB activated islets not only expressed the same chemokine profile observed in human islets but also struggled to maintain normoglycemia posttransplantation. Further, NF-κB-activated islets were rejected with a faster tempo as compared to non-NF-κB-activated rodent islets. Thus, isolated human islets can make cell autonomous contributions to the ensuing allograft response by elaborating inflammatory factors that contribute to their own demise. These data highlight the potential importance of islet intrinsic proinflammatory responses as targets for therapeutic intervention.
Keywords
Introduction
The substantial loss of β-cell mass and function following transplantation into allogeneic recipients constitutes a major impediment impacting upon the clinical success of islet transplantation as a treatment for type I diabetes and the prevention of diabetic complications (17). Both islet extrinsic and intrinsic factors may contribute to the loss of β-cell mass and function. Extrinsic immune factors that contribute to islet cell loss include both innate (9,35) and graft-specific adaptive immune responses (23,37). The nuclear transcription factor NF-κB can induce inflammatory as well as survival and death pathways in a variety of cell types (22). Animal studies demonstrate that inflammatory-activated islets have the potential to elaborate NF-κB-dependent genes that dampen glucose regulation and promote antigraft immunity. Some of these islet intrinsic NF-κB-dependent factors have been identified, including inducible nitric oxide synthase (15,25) and cyclooxygenase 2 (47), which negatively impact glucose-stimulated insulin release and can induce islet apoptosis (15,25), whereas islet expression of tissue factor (TF) (35) would promote local coagulation (5). Elaboration of chemokines (45) by engrafted islets would aid the recruitment of inflammatory cells (26,32), whereas molecules such as Fas (10) and activated transcription factor 3 [ATF3] (18) would promote β-cell apoptosis. Significantly, blockade of the NF-κB pathway in islets, prior to transplantation by treatment with NF-κB inhibitors (11,16) or antioxidants (7,38), can improve islet graft function in experimental models. These animal data demonstrate that islet-intrinsic, NF-κB-dependent proinflammatory responses can make major contributions to the ensuing antigraft immune response.
In the context of clinical islet transplantation, induction of islet-intrinsic NF-κB-dependent proinflammatory genes may contribute to loss of human islet cell mass, function, and rejection. Indeed, isolated human islets exhibit evidence for activation of the NF-κB and mitogen-activated kinase (MAPK) stress response pathways (1,8). Despite evidence that the NF-κB pathway is activated by islet isolation (1,8) and that suppression of islet intrinsic proinflammatory genes improves islet transplantation in animal models (7,16,26,29,38), the downstream NF-κB target genes induced in human islets during the isolation process are poorly described. Therefore, in this study, we determined the pattern of genes expressed by isolated human islets. In doing so, we found that isolated human islets express an array of NF-κB-dependent proinflammatory genes prior to transplantation, suggesting an islet cell autonomous contribution to the ensuing allograft response. Indeed the islet graft may potentially drive inflammation and injury, which may act to exacerbate the anti-islet-graft immune response. These data highlight the potential importance of islet intrinsic proinflammatory responses as targets for therapeutic intervention.
Materials and Methods
Human Islet Preparations
Information on the eight-principle islet isolates used for the microarray study are presented in Table 1. Human pancreatic islets were separated by a closed loop method as described previously (39). Pancreases were removed surgically from heart beating deceased donors and preserved with cold storage solution (Viaspan® Belzer UW, Bristol-Myers Squibb, Princeton, NJ). The pancreas was disaggregated by infusing the ducts with cold collagenase and neutral protease (Serva, Germany), dissected and placed in a metal chamber. Dissociated islet and acinar tissue was washed, and islets were separated from the acinar tissue on a continuous Biocoll (Biochrom AG, Berlin, Germany) density gradient (Polysucrose 400 and amidotrizoeic acid) on a refrigerated apheresis system (model 2991, COBE Laboratories). Islets were washed and collected in M199 medium supplemented with Hepes buffer and 20% human albumin. Purified islets were counted in duplicate, and islet number and mass were expressed in terms of islet equivalents. Immediately after extraction, ~5,000 IEQ pure islets (fractions 3–8) were either snap frozen in liquid nitrogen or resuspended in mRNA isolation buffer (RLT lysis buffer, Qiagen). Samples were then stored as −80°C for further downstream manipulations. Of the nine islet isolates used for qPCR analysis, three were taken from the microarray analysis (Table 1), with an additional six new preparations tested, so three samples were common to both modalities.
Characteristics of the Eight-Principle Islet Preparations Analyzed by Microarray
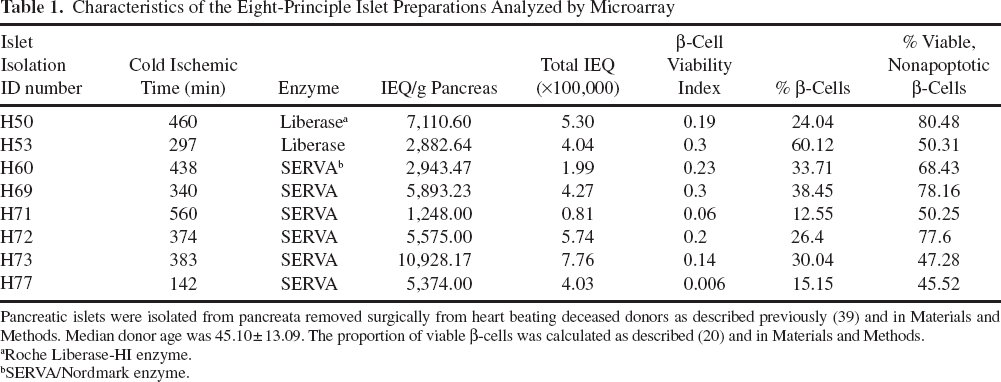
Pancreatic islets were isolated from pancreata removed surgically from heart beating deceased donors as described previously (39) and in Materials and Methods. Median donor age was 45.10 ± 13.09. The proportion of viable β-cells was calculated as described (20) and in Materials and Methods.
Roche Liberase-HI enzyme.
SERVA/Nordmark enzyme.
Real-Time Quantitative PCR
Total RNA was extracted using the RNeasy Plus Mini Kit (Qiagen) according to the manufacturer's instructions. Single-stranded cDNA was produced using the Superscript III reverse transcriptase kit (Invitrogen, Australia) according to manufacturer's instructions, to a maximum of 25 cycles. Primers (Tables 1 and 2) were designed using Primer3 with sequences obtained from GenBank and synthesized by Sigma Aldrich (Australia). PCR reactions were performed on the RG300 Real-Time PCR System (Rotorgene) using FastStart SYBR Green Master Mix (Roche Diagnostics). Human glyceraldehyde 3-phosphate dehydrogenase (GAPDH) or mouse peptidylprolyl isomerase A (or cyclophilin A; CPH2) were used as housekeeping genes, and data were analyzed using the ΔΔCt method (28); GAPDH typically appeared after nine cycles. Amplification was run to a maximum of 40 cycles.
Primers for Human Genes

Microarray Analysis
RNA quality was assessed using an Agilent 2100 Bio-analyzer (Agilent Technologies, Palo Alto, CA), and all samples had RNA integrity number (RIN) of >8. Labeled cRNA generation and array hybridization were performed at the Ramaciotti Centre for Gene Function analysis (UNSW, Sydney, Australia) according to the manufacturer's instructions. Eight RNA samples were hybridized, each to a single Affymetrix HGU133+2 microarray, in three batches. Arrays were normalized using RMA in Partek® software, version 6.4 (Partek, Inc., St. Louis, MO, USA). Quality control plots identified a batch effect relating to the date of hybridization, which, in addition to a potential gender effect, was removed using the Batch Remover tool in Partek, resulting in normalized, log2 microarray data. Probeset detection calls of Present/Marginal/Absent were calculated using the
Most Heterogeneously Expressed Genes
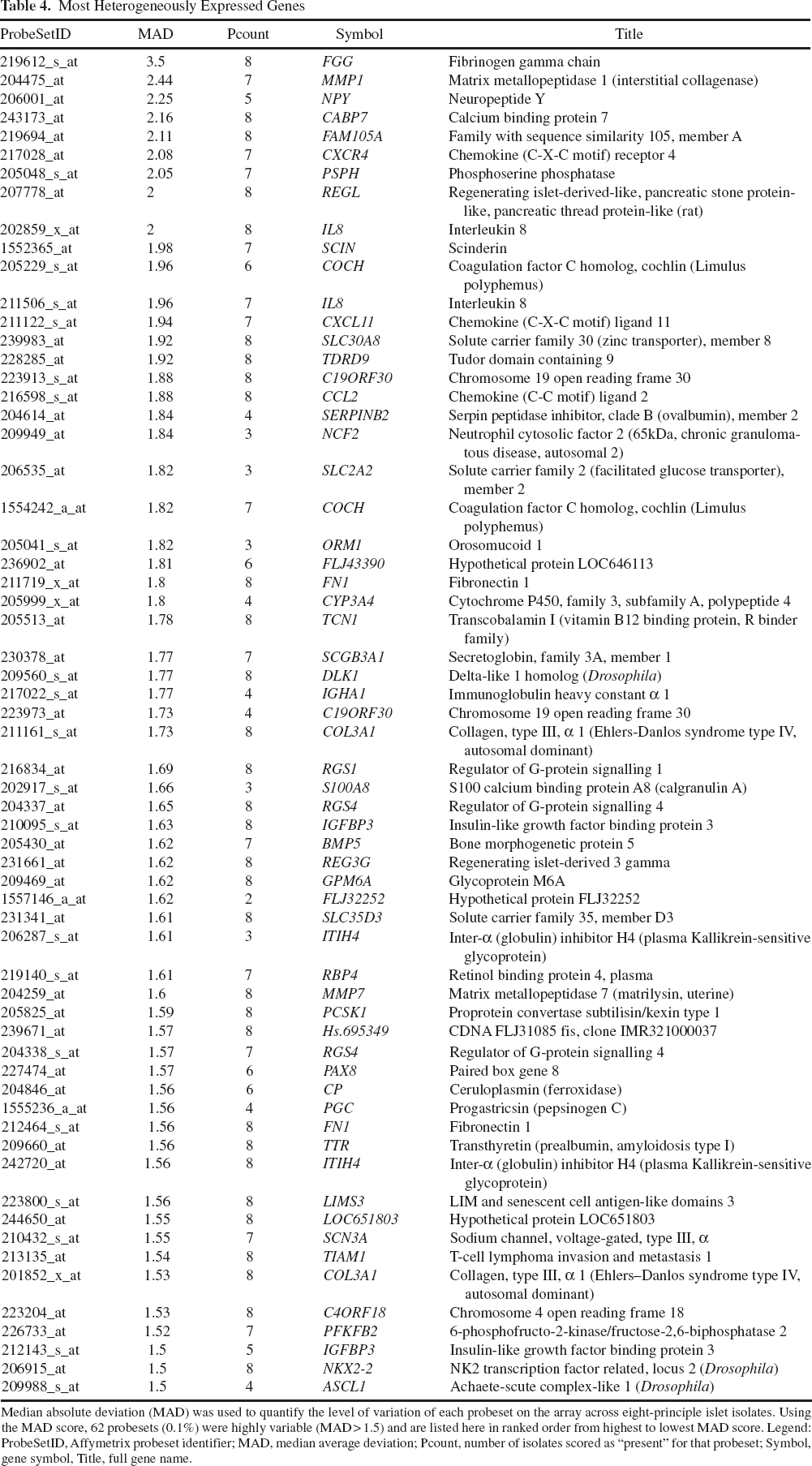
Median absolute deviation (MAD) was used to quantify the level of variation of each probeset on the array across eight-principle islet isolates. Using the MAD score, 62 probesets (0.1%) were highly variable (MAD > 1.5) and are listed here in ranked order from highest to lowest MAD score. Legend: ProbeSetID, Affymetrix probeset identifier; MAD, median average deviation; Pcount, number of isolates scored as “present” for that probeset; Symbol, gene symbol, Title, full gene name.
Comparison to Laser Capture Microdissected (LCM) Islets
Cross-platform meta-analysis of gene expression patterns between pancreatic samples obtained via laser capture microscopy (LCM) (31) and our isolated islets was conducted. The LCM data consisted of three samples, hybridized to the Affymetrix HG-U133 A and B arrays (
Mice
Female recombination activating gene knockout (RAG-/-), C57BL/6, and BALB/c mice (6–8 weeks old) were purchased from the Animal Resource Center (ARC, Perth, Australia). All procedures and experiments conducted complied with institutional AEC guidelines.
Human Islet Transplantation
Human islets (~2,000 IEQ) were transplanted under the renal capsule of normoglycemic RAG-/- mice using protocols we have described (16). For gene analysis of human islet grafts, at 4 weeks posttransplantation, the renal capsule was peeled back from the kidney to reveal the intact islet graft, which was immediately prepared for mRNA isolation.
Rodent Islet Isolation, NF-κB Gene Expression, and Transplantation
Rodent islets were isolated from BALB/c mice following protocols we have described (15,16). For cytokine treatment, rodent islets were cocultured in media containing 200 U each of interleukin (IL)-1β, tumor necrosis factor (TNF)-α and interferon (IFN)-γ (R&D Systems) for 4 h either alone or in combination. For transplantation, BALB/c H-2d islets were isolated and stimulated with or without IL-1β, TNF-α and IFN-γ for 4 h, and subsequently transplanted under the renal capsule of streptozotocin-induced diabetic C57BL/6 H-2b mice, a full allogeneic strain combination, as we have described (51). For gene expression analysis, mRNA was isolated (mRNA isolation kit; Qiagen) and assessed by qRT-PCR following protocols we have described (27).
Reporter Assays
Reporter assays were carried out essentially as we have described (15,27). Briefly, MIN6 cells (34) were transfected with 0.6 μg of the NF-κB luciferase reporter NF-κB.Luc, plus 0.2 μg CMV.β-galactosidase and stimulated with 200 U each of IL-1β, TNF-α and IFN-γ either alone or in combination. Luciferase activity was assayed in cell lysates harvested 8 h poststimulation using luciferase assay kit (Promega). Results were normalized to β-galactosidase activity (GalactoStar; EBioscience) to give relative luciferase activity.
Statistical Analysis
Statistical significance for comparisons between isolated and LCM islets was determined using
Results
Whole Genome-Wide Analysis of the Human Islet Transcriptome
Eight independent human islet isolates prepared within our clinical transplant center were subjected to whole genome transcript profiling on Affymetrix microarrays. Microarray data were RMA normalized, and both the batch effect and the potential gender effect were removed (see Methods). These normalized transcript profiles are visualized as line plot (Fig. 1A), which represents a global view of the genetic architecture of human islets, prepared for clinical transplantation. Of significance for this discussion, the level of interisolate heterogeneity is clearly evident as fluctuations in expression levels for specific genes between individual islet preparations. We used the median absolute deviation (MAD) to quantify the level of variation of each probeset on the array, where a MAD score of 0.5 or 1 indicates that there is at least a 1.41- or 2-fold variation, respectively, in 50% of the isolates. We compared the average expression level of each probeset to its MAD (Fig. 1B), revealing that those probesets with the highest variability tended to be moderately expressed on average. It is clear that the majority of probesets (94%) are homogeneously expressed between isolates (MAD < 0.5), yet this leaves 3,254 (6%) probesets with moderate to high variability across isolates (MAD > 0.5). Using the MAD score to identify probesets with extremely variable patterns, we could identify 381 (0.7%) with MAD > 1.0, 62 with MAD > 1.5, and 7 with MAD > 2.0 (Fig. 1C and Table 4). Furthermore, combining MAD score with the number of times each probeset was declared as detected above background (i.e., Pcount, see Methods) revealed that variance arose from modulation of gene expression levels for probesets scored as present in every sample (i.e., Pcount 8, black dots in Fig. 1B) as well as absolute changes, as indicated by probesets scored as present in at least one, but not all, samples (i.e., Pcounts 1–7, gray dots, Fig. 1B). These data indicate that isolated human islets exhibit significant heterogeneity in a relatively small subset of genes.

A global analysis of genes expressed by isolated human islets. (A) Gene expression levels for isolated human islets were assessed by microarray (
Gene Set Enrichment Analysis (GSEA) of Isolated Human Islets
A classical approach to gene expression profiling might interrogate the most highly expressed genes in human islets. But we questioned whether interrogating heterogeneously expressed genes might provide insights into the underlying molecular status of human islets prepared within a clinical transplant context. Accordingly, gene set enrichment analysis (GSEA) (48) of the heterogeneously expressed genes was used to identify the perturbed molecular processes. GSEA evaluates micro array data at the level of gene sets, avoiding the bias of a candidate gene approach (48). First, all genes on the U133 + 2 microarray were ranked according to their MAD score, from most to least variable. Using GSEA, this preranked score was then interrogated against the collection of all “curated” gene sets from the Molecular Signatures Database, version 2.5 (48), which contains gene sets from various expert curated pathway databases as well as manually curated published microarray studies.
Dramatically, GSEA analysis identified enrichment within the set of genes with a high MAD score for gene sets associated with activation of the NF-κB stress/ inflammation pathway; namely, the HINATA_NFKB_UP gene set (FDR < 0.001), which represents the upregulated genes identified by treating keratinocytes or fibroblasts with NF-κB by Hinata et al. (19). The enrichment plot for member genes in the HINATA_NFKB_UP gene set are presented in Figure 2A, whereas the expression data and MAD score for each gene are presented in heat map format in Figure 2B. These data provide a map of the key genes triggered by organ preservation and or islet isolation (8).

Molecular evidence for NF-κB activation in isolated human islets. (A) Genes ranked by decreasing MAD and by the GSEA method were found to be enriched for various NF-κB gene sets, including the HINATA_NFKB_UP gene set (
Identification of the Chemokine CCL2 as Being Expressed by Isolated Human Islets
One striking feature of the gene expression profile of human islets, illustrated by GSEA analysis in the heat map data in Figure 2B, was the high prevalence of chemokines and cytokines within the heterogeneously expressed gene set. Indeed, chemokines and cytokines represented ~20% of all expressed genes identified by GSEA as being associated with the HINATA_NFKB_UP gene set. Of note, the chemokine CCL2 was fourth in this gene set and has been previously identified as a NF-κB-dependent gene expressed by isolated human islets (32,40), validating this approach.
Chemokine Expression Pattern Expressed by Human Islets
Given the role of NF-κB in controlling chemokine expression and the immunological relevance of chemokines to transplantation and the success of islet engraftment (40,45,53), we examined chemokine transcripts in isolated human islets. To do this, we first utilized a classical bioinformatics approach on our microarray data set by ranking transcripts for all known chemokines by their expression level. These data, presented in heat map format in Figure 3A, demonstrate that a clear subset of chemokine transcripts were highly expressed by isolated human islets. Namely, isolated human islets expressed relatively high levels of transcripts for chemokine ligand (C-X-C motif) 12 [CXCL2], chemokine ligand (C-C motif) 2 [CCL2], CXCL12, CXCL1, CXCL6, and CCL28. Concurring with our GSEA results, RTqPCR analysis showed CCL2 to be robustly expressed in all samples (avg = 9.5, Pcount = 8), but also highly variable (MAD=1.8) (Fig. 3B). Indeed, CCL2 was expressed at levels comparable to that of the islet genes pancreatic and duodenal homeobox 1 (PDX-1) and islet amyloid polypeptide (IAPP) (Fig. 3B).
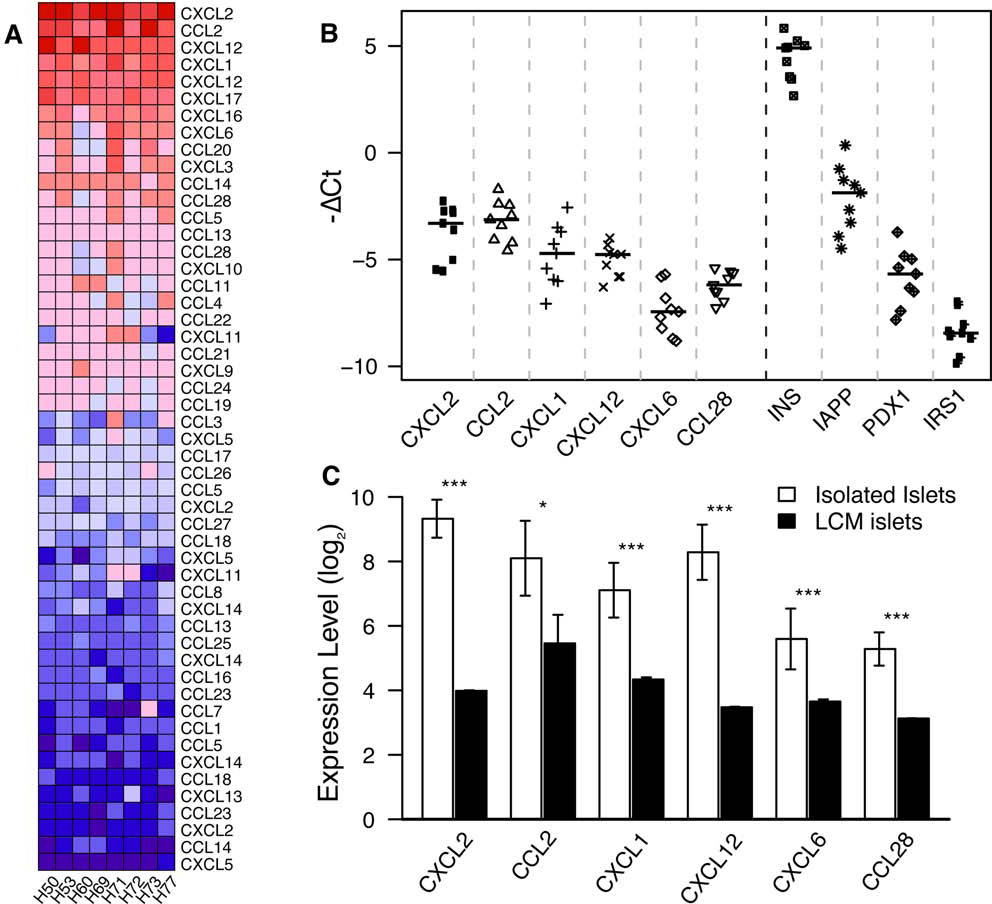
Chemokine expression is elevated in isolated human islets. (A) Heat map analysis of median normalized chemokine gene expression in isolated human islets. Each column represents an individual islet isolate. Gene names are indicated. (B) Using RTqPCR, expression levels of chemokines were assessed in isolated human islets (ΔCt,
Of further interest, with the exception of CCL2, the chemokines CXCL2, CXCL12, CXCL1, CXCL6, and CCL28 showed a homogeneous expression pattern across all samples as indicated by the low median absolute deviation (e.g., MAD≤0.8) and high Pcount (e.g., Pcount≥7). RTqPCR analysis in nine islet isolates demonstrated the high expression of CXCL2, CXCL1, CXCL12, CXCL6, and CCL28 in all islet isolates examined; indeed gene expression levels for these chemokines were comparable to that of the islet expressed genes PDX-1 and IAPP but higher than that of insulin receptor substrate-1 (IRS-1) (Fig. 3B). Thus, human islets express a pattern of NF-κB activation typified by variable expression of CCL2, but consistently high expression of CXCL2, CXCL1, CXCL12, CXCL6, and CCL28.
Comparison of Chemokine Expression Levels Between Islets In Situ and Postisolation
To determine whether the expression levels for the expressed chemokines represented a change relative to expression in islets in the native pancreas, gene expression levels for isolated islets were compared by meta-analysis to that found in islet cells enriched by laser capture dissection microscopy from pancreas tissue (31). We combined the array data from Marselli et al. (31) with ours using careful normalization to account for the differences in microarray design and experimenter (see Methods); this ensured that data from both studies are directly comparable. Significantly, this analysis demonstrated that chemokine mRNA expression levels were markedly increased postisolation overexpression levels seen for islets in situ (
Expression of the NF-κB-Dependent Stress Response Genes A20 and ATF3 in Isolated Human Islets
NF-κB governs the expression of proinflammatory factors, but also genes involved in regulating cellular apoptosis and survival (4,49,52); in islets two well-described NF-κB-regulated genes are the stress response genes ATF3 and A20 [also known as tumor necrosis factor α-induced protein 3 (TNFAIP3)] (18,27). Microarray analysis showed that human islet preparations expressed significantly high mRNA levels for ATF3 and A20 (Fig. 4A), increased by 2.51- and 7.31-fold, respectively, as compared to islets within the native pancreas in situ. High expression of ATF3 and A20 mRNA was also detected in islet preparations by RTqPCR analysis, confirming the microarray result (Fig. 4B). Thus, isolated human islets express ATF3 and A20, both are NF-κB-regulated genes controlling islet survival that are considered to only be present in inflammatory activated islets (15,18,27). High expression of NF-κB-regulated ATF3 and A20 is consistent with the observation that organ preservation and or islet isolation results in activation of NF-κB (1), which subsequently induces the gene expression patterns we identify here.
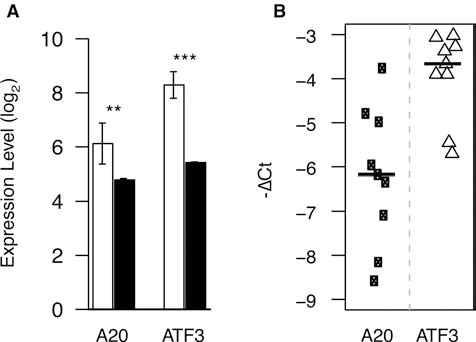
Expression of pro- and antiapoptotic factors are elevated in isolated human islets. (A) Meta-analysis for expression levels of A20 (tumor necrosis factor α-induced protein 3) and activated transcription factor 3 (ATF3) in isolated human islets (
Islet Gene Expression Patterns Following Transplantation
We next questioned whether the proinflammatory profile of human islets would reverse or subside after transplantation. To determine the stability of the inflammatory gene expression pattern after transplantation, human islets were grafted under the renal capsule of euglyceamic immunodeficient RAG-/- mice. Using RTqPCR, we compared gene expression levels for inflammatory genes between freshly isolated human islets with that of human islets recovered after 4 weeks of transplantation (Fig. 5). Examination of the inflammatory stress response genes ATF3 and A20 revealed a 13.7-fold decrease in ATF3 expression (

Change in gene expression 4 weeks after transplantation of human islets into mouse recipients. Human islets were transplanted under the kidney capsule in recombination activating gene knockout (RAG-/-) mice, and expression levels were assessed 4 weeks later using RTqPCR for key genes regulating apoptosis (A) or chemokines (B) (-ΔΔCt relative to pretransplantation,
Induction of CXCL1, CXCL2, CXCL28, and CCL2 in Islets Requires NF-κB Activation
To further understand the molecular mechanism by which human islets acquired an inflammatory chemokine gene expression profile, and the role of NF-κB in driving this gene signature, we turned to a rodent islet model. In the case of rodent (BALB/c) islets isolated under optimal conditions that is extracted from a living anesthetized donor, the chemokines CXCL1, CXCL2, CXCL28, and CCL2 were only weakly expressed. In contrast, when NF-κB was activated by stimulating BALB/c islets with inflammatory cytokines, high expression of CXCL1, CXCL2, CXCL28, and CCL2 mRNA was induced (Fig. 6A). Analysis of NF-κB reporter activity in MIN6 β-cells that demonstrated NF-κB activation was achieved upon treating islet cells with these same cytokines (Fig. 6B). To demonstrate NF-κB activation was necessary for expression of CXCL1, CXCL2, CXCL28, and CCL2, isolated BALB/c islets were transduced by recombinant adenovirus expressing the physiologic NF-κB inhibitor IκBα. As shown in MIN6 cells, overexpression of IκBα suppressed cytokine-induced NF-κB reporter activity (Fig. 6B). In the case of primary islets overexpressing IκBα, cytokine induction of CXCL1, CXCL2, CXCL28, and CCL2 was suppressed (Fig. 6C). Thus, islets isolated under optimal conditions do not express significant mRNA levels for CXCL1, CXCL2, CXCL28, and CCL2; however, activation of NF-κB is both sufficient and necessary to drive the expression of these chemokines in islets.
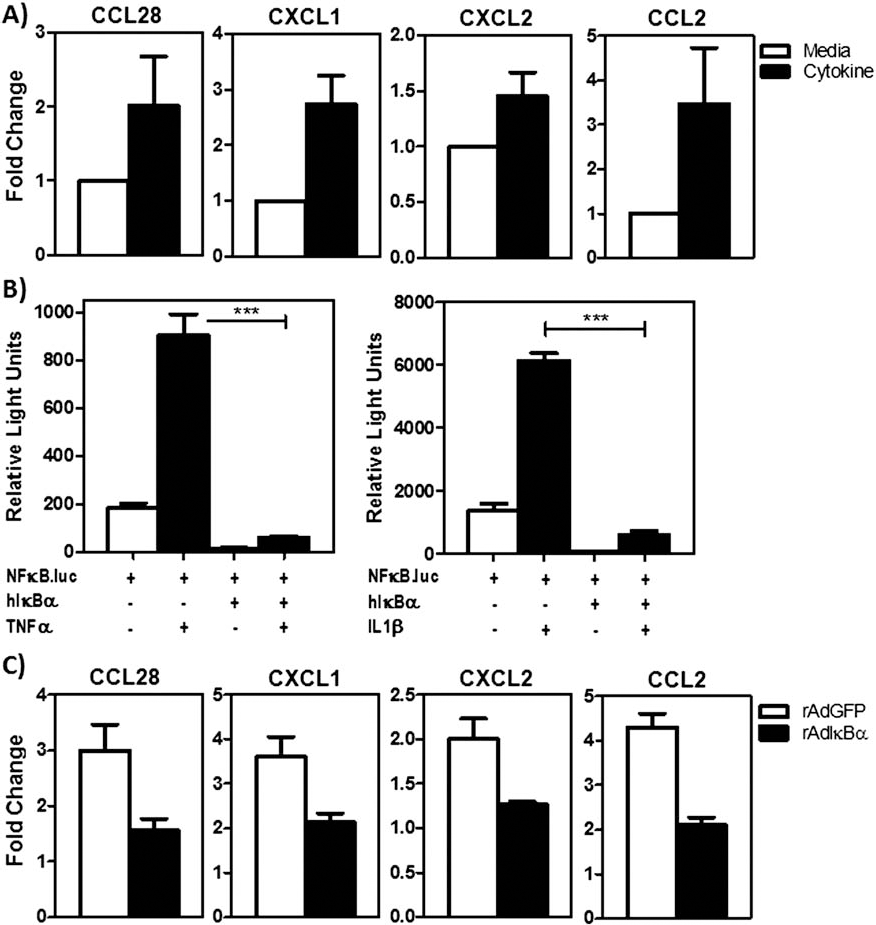
NF-κB control of chemokine genes in islet cells. (A) RTqPCR analysis of chemokine mRNA levels in isolated rodent islets cultured with (black bars) or without (open bars) cytokines for 4 h (
Effect of an NF-κB Gene Signature on Graft Outcomes
To test how a preexistent NF-κB gene signature would affect transplant outcomes, major histocompatibility complex 2-expressing (H2d) BALB/c mouse islets, exposed or not to cytokines, were transplanted under the renal capsule of diabetic and allogeneic H2b C57BL/6 mice (Fig. 7). As expected, control islets isolated under optimal conditions and cultured in media were able to rapidly restore normal metabolic control (Fig. 6B). Subsequently, these islet grafts were rejected with a mean survival time of ~22 days (

Impact of a preexistent NF-κB gene signature upon islet graft outcomes. Blood glucose levels (BGL) for diabetic allogeneic transplant recipients of islet grafts cultured prior to transplantation (A) without (NT;
Discussion
This study identifies that isolated human islets express a molecular signature consistent with that of cells undergoing a marked proinflammatory response prior to the event of transplantation. This proinflammatory phenotype is not shared by islets in situ nor by rodent islets isolated under optimal conditions from living anesthetized donors, suggesting that this proinflammatory phenotype is a response to the isolation process. Activation of the NF-κB pathway by stress associated with organ procurement and islet extraction from the pancreas represents one candidate driving this response. The NF-κB pathway is a major sensor for a broad range of physiological stresses including inflammatory mediators, but also UV damage and osmotic stress. Isolated islets prepared for transplantation undergo significant stress that is not seen by islets in situ. Firstly, brain death results in a “sympathetic/autonomic storm” (41). This leads to a cascade of events including inflammatory cytokine and catecholamine release, disturbed metabolism, tissue ischemia, cellular injury, and death (3,43). After organ procurement, there is a period of cold ischemia, and during the isolation process, islets are exposed to collagenase, a bacterial enzyme that may further exacerbate the inflammatory process. Following separation from the acinar tissue islets lose their vascular supply and are exposed to osmotic stresses during purification on density gradients. Finally, they undergo an additional period of cell culture outside of their in situ environment resulting in further stress. Therefore activation of the NF-κB pathway in isolated human islets has been demonstrated in previous studies (1,8). The NF-κB pathway induces a broad array of cellular responses including those related to inflammation, survival, and cell death (22). Thus, islet intrinsic activation of the NF-κB pathway (1,8) and subsequent elaboration of proinflammatory genes (35,40) during organ preservation and islet isolation (44) could be expected to have significant consequences for islet function following transplantation. A molecular overview of the transcriptional response of islets to isolation stress would aid our understanding of the molecular processes effecting islet function and islet graft survival after transplantation.
We outline the islet intrinsic genes expressed subsequent to the stress of organ procurement and islet isolation. We find that isolated islets can provide a major source of proinflammatory factors, which would be predicted to exacerbate the ensuing antigraft immune response after transplantation. As indicated by the GSEA analysis, these factors include known NF-κB target genes such as the macrophage and T-cell recruiting factor CCL2, neutrophil recruiting factor IL-8, and the β-cell toxic cytokine IL-1β(2). Isolated islets also expressed matrix metallopeptidase 1 (MMP1) and fibronectin 1 (FN1), factors involved in tissue remodeling, adhesion, and cell migration. In addition, human islets also express high levels of the transcription factor ATF3. ATF3 is expressed by damaged or dying tissues and in islets; ATF3 plays a critical role in the activation of pathways triggering β-cell apoptosis (18). Indeed, ATF3 knockout islets exhibit improved survival in syngeneic recipients (54). Collectively, the molecular profile of isolated human islets indicates that islets could make significant and cell autonomous contributions to the generation of a local, inflammatory milieu, posttransplantation.
That isolated human islets can act as a reservoir of potent immune factors posttransplantation is highlighted by our findings that human islets express very high levels of a specific set of chemokines. Specifically, islets express transcripts for CXCL2, CCL2, CXCL12, CXCL1, CXCL6, and CCL28. Of interest, CXCL1, CXCL2, and CXCL6 are chemoattractants for polymorphonuclear cells, whereas CCL2 is a chemoattractant for monocytes. The expression of these chemokines by isolated islets may impact on islet function and survival after transplantation.
Indeed, when rodent islets exemplifying the NF-κB gene signature of human islets were transplanted into allogeneic recipients they exhibited poor function and were rejected with a faster tempo when compared to noninflammatory islets isolated under optimal conditions. By extrapolation, these present data would predict that the NF-κB gene signature of human islets could contribute to a deterioration of islet graft function in the posttransplant milieu.
It is of interest to consider that, following intraportal infusion, transplanted islets are engulfed in a deleterious immediate blood-mediated immune reaction (IBMIR) involving localized coagulation, platelet aggregation, and complement activation (5,35). This response, triggered by expression of TF (35), itself under the control of NF-κB (30), is rapidly followed by infiltration of the engrafted islets by neutrophils, granulocytes, and monocytes (36). The mechanisms driving the cellular recruitment of polymorphs and monocytes to the graft site are not clear; however, our data indicate that islet intrinsic production of key neutrophil and monocyte chemokines, namely CXCL1, CXCL2, CXCL6, and CCL2, would contribute to the recruitment and activation of these early mediators of islet graft failure.
Some clinical studies support our finding that isolated human islets elaborate CCL2 (32,40); further islet CCL2 production is associated with increased recipient biochemical parameters of coagulation, liver damage, and inflammation posttransplant (32) as well as poor clinical islet transplant outcomes (6,40). Further to this, inhibiting the action of the NF-κB-dependent gene CCL2 (24) in experimental models improves early islet function in syngeneic transplantation (32) as well as promoting islet allograft survival (26). Collectively these examples, together with our present findings, demonstrate that human islets are capable of elaborating a broad repertoire of chemokines that potentially negatively impact islet engraftment and posttransplant survival.
This study reveals that isolated human islets also express certain stress response genes that may be beneficial to islet grafts. Isolated human islets express high levels of the chemokine CXCL12, a factor which could contribute to islet neovascularization through the recruitment of endothelial cell progenitors (21). Transplanted rodent islets stabilize hypoxia inducible factor (HIF)-1α protein (33) and elaborate vascular endothelial growth factor A [VEGFA] (50), as do isolated human islets (AW and STG unpublished observation), demonstrating that transplanted islets can respond to the physiological lack of oxygen by releasing factors including VEGFA and CXCL12 that encourage local neogenesis. High expression of A20 by human islets may also represent a beneficial response. A20 is regulated at the level of transcription by NF-κB (27), and in islets A20 plays a protective role by inhibiting apoptosis (15,27) and dampening NF-κB activation (14,15). These protective functions could improve islet graft function as overexpression of A20 improves syngeneic islet transplant outcomes (16). Thus, in the context of clinical islet transplantation, the induction of certain NF-κB genes, including CXCL12 and A20, as well as others (42), may be considered to be beneficial.
This present study demonstrates that isolated human islets exhibit a proinflammatory phenotype, which has the potential to contribute to poor graft outcomes. The expression pattern of induced inflammatory genes provides a list of candidates for possible therapeutic intervention, as well as a molecular gene set of potential predictive factors for increased graft immunogenicity. Of further note, the induced chemokine expression pattern remains stable posttransplantation; the permanence of this proinflammatory molecular signature suggests islet grafts can function as a stable reservoir of immune factors, which may be counter balancing immunosuppressive drug actions or acting as a barrier to generating immune tolerance. Further, these data would suggest that early interventions reversing the inflammatory changes to either quell islet intrinsic NF-κB activation (7,11,16) or target-specific factors elaborated by isolated islets (26,35) could be of benefit in the context of human islet transplantation.
Footnotes
Acknowledgments

