Abstract
This study was designed to retrospectively compare the impact of crude Sigma V collagenase (Sigma V, n = 52) with high-purified Serva NB1 collagenase (Serva NB1, n = 42) on human islet isolation outcomes. A three-step filtration was applied to the crude Sigma V to remove endotoxin contamination and impurities; in addition, this process was used as a lot prescreening tool. Isolation outcomes were determined by digestion efficacy, islet yields, purity, viability, glucose-stimulated insulin release, and endotoxin content. The digestion efficacy between Sigma V and Serva NB1 was statistically significant (Sigma V: 64.71% vs. Serva NB1: 69.71%, p = 0.0014). However, the islet yields were similar (Sigma V: 23422.58 vs. Serva NB1: 271097 IEq, p = 0.23) between groups. There was no significant purity difference observed in fractions with purities greater than 75%. Viability (Sigma V: 93.3% vs. Serva NB1: 94.8%, p = 0.061) and stimulation indexes (Sigma V: 3.41 vs. Serva NB1: 2.74, p = 0.187) were also similar between the two groups. The impact of cold ischemia and age on the isolation outcome in the Sigma V group was comparable to the Serva NB1 group. The endotoxin content of the final products in the filtered Sigma V group was significantly less than that in the high-purified Serva NB1 group (0.022 vs. 0.052 EU/ml, p = 0.003). Additionally, in the Sigma V group there was minimal lot to lot variation and no significant loss of enzymatic activity after filtration. These findings indicate that the use of Sigma V or other crude enzyme blends for research pancreata is warranted to reduce isolation costs and increase the amount of islets available for critical islet research. These findings also validate the need for a systematic enzyme analysis to resolve these inconsistencies in overall enzyme quality once and for all.
Introduction
Human islet transplantation is an emerging therapy for type I diabetes (11,13,27). One of the main limiting factors for wide-spread clinical application is inconsistent islet isolation outcomes. The quality of the enzyme used to dissociate the pancreas is of great importance for islet manufacturing. Significant global efforts to purify the components of collagenase and protease enzyme blends and to characterize the in vitro enzyme composition and digestion efficacy have been made; however, wide batch-to-batch, and even vial-to-vial, variability remains (23). This variability in enzyme blends has hindered the standardization of collagenase digestion in human islet isolation across centers and is associated with unpredictable islet isolation results (7,9,16). In the past, Liberase HI and Serva NB1 collagenase (Serva NB1) have been the most widely investigated enzymes for human islet isolation. Prior to 2007, Liberase HI was considered the gold standard enzyme for more than 10 years (14,19,20,26). However, following disclosure that it potentially contained bovine neural tissue contaminants, Liberase HI was discontinued for use in clinical islet transplantations. To reduce the risk of transmission of bovine spongiform encephalopathy, the islet community began to use Serva NB1. However, this enzyme change significantly increased islet isolation cost and decreased human islet isolation outcomes worldwide (1,9).
Before the introduction of Liberase HI, Sigma Collagenase V (Sigma V) and other crude Sigma collagenase blends were used for both human and other animal pancreatic islet isolations (29,30). However, these enzyme blends did not gain extensive application due to low digestion efficacy related to enzyme impurity, imbalanced combination of key active components, significant batch-to-batch and vial-to-vial enzyme variation, high endotoxin levels, and pigment contamination (17,29,30). Yet, most of the results regarding the use of the collagenase enzyme blends in human islet isolation were obtained long before the optimization of current standard isolation techniques (29). In this study, we investigated whether simple filtration of the low-cost crude collagenase, Sigma V, combined with lot screening, could represent an alternative to expensive purified enzyme blends.
Materials and Methods
Pancreas Preservation and Islet Isolation
Human pancreata were obtained from organ procurement organizations following informed consent and IRB approval. The pancreata were preserved, using either University of Wisconsin solution (UW) or Histidine-Tryptophan-Ketoglutarate (HTK), and transported to the cell isolation facility at the University of Illinois at Chicago. No donor randomization was applied.
Fifty-two isolations were performed using Sigma Collagenase V and 42 isolations were performed using Serva NB1 purified enzyme blend. In order to reduce the variability due to differences in isolation procedures, only the isolations conducted in the period between June 2007 and December 2009 were compared. The same islet isolation protocol was used for all isolations during this time. Serva NB1 (Premium and GMP grades, SERVA Electrophoresis GmbH, Heidelberg, Germany) was reconstituted with cold HBSS (Mediatech, Manassas, VA), supplemented with 10 U/ml heparin, and complemented with neutral protease (SERVA Electrophoresis GmbH). Variable units of collagenase (1600–2057 Wunsch units) and neutral protease (200–257 units) were used based on the pancreas weights. Sigma V, with an enzyme activity of FALGPA 1.0–3.0 mg/solid (Sigma, St. Louis, MO), was reconstituted with 350 ml of perfusion solution (Mediatech), which was supplemented with 20 mM of HEPES (Mediatech) and 10 mM of glutamine (Invitrogen, Carlsbad, CA), to a final concentration of 2.86 mg/ml. There is no direct conversion between Wunsch units used for the Serva enzyme to the FALGPA units used for the Sigma V enzyme. This concentration was chosen based on results from previous rodent and monkey studies (2,21). No additional neutral protease was used in the preparation of Sigma V. Each lot of Sigma V has an associated certificate of origin, tracking and certifying the source animals as disease free in antemortem and postmortem testing; no bovine neural material is used in the manufacturing of Sigma V. This enzyme is also heat-treated twice to inactivate potential pathogenic viral agents.
To reduce endotoxin and pigment levels, the reconstituted enzyme went through a three-step filtration process using decreasing pore size filters (0.8, 0.45, and 0.22 μm) at 2–10°C. The crude enzyme was filtrated with three cellulose nitrate-based filters (500 ml volume, Nalgene) using a standard “wall vacuum” at a pressure of 16–17 lb/inch2. To reduce filtration variance, the same types of filters were used for every filtration step. Throughout these isolations, multiple lots of cellulose nitrate-based filters were used with comparable results. The three-step filtration process was also used as a prescreening tool for enzyme lot selection. When testing a new lot of Sigma V, the enzyme was used only when at least 250–300 ml of enzyme passed through the first 0.8-μm filter and the rest passed through an additional 0.8-μm filter. In the case where the membrane became blocked before 200 ml of enzyme passed through the filter, the lot was discarded and not used for islet isolations. In our experience, if the enzyme passed through the initial 0.8-μm filters, there were no subsequent problems with the additional filters to complete the three-step filtration process. Approximately 40% of the enzymes tested did not make it through the filtration screening. However, when the enzyme passed through our easy filtration process, it was successfully used for human islet isolations every time. Therefore, we did not have to waste any pancreata or time testing these enzymes.
The isolation, purification, and culture procedures were performed as previously described (13,18,25,27). Briefly, the pancreata were trimmed and distended with either Serva NB1 or Sigma V enzymes and digested using a modified Ricordi semiautomatic method. The digestion phase was stopped between 10 and 20 min based on microscopic observation of islet cleavage (degree of islets released from exocrine tissue) and tissue volume by the same experienced personnel. Digested tissues were then collected and washed three times. The tissues were incubated with UW solution for 30 min prior to continuous density purification using the UIC-UB gradient (4) in a Cobe 2991 cell separator (Cobe 2991, Cobe, CO) and subsequently cultured in CMRL culture media (Mediatech) at 37°C supplemented with ITS (Invitrogen), sodium bicarbonate (Sigma), HEPES, human albumin (Grifols, Los Angeles, CA), and ciprofloxacin (Hospira Inc., Lake Forest, IL).
Islet Quality Score
Final quality of isolated human islets was scored using a standardized system based on size distribution, fragmentation, density, border sharpness, and shape (22). Each of these criteria was scored from 0 to 2. Islets of maximal quality scored 10; islets of poorest quality scored 0.
Glucose-Stimulated Insulin Secretion and Viability Assays
Static glucose incubations (GSI) were performed, as previously described, to evaluate islet physiology and potency (3). Briefly, 10 purified islets were hand-picked and incubated with Krebs ringer buffer containing 1.67 mM (low) glucose and 20 mM HEPES for 1 h. The islets were then transferred into new Krebs buffer containing 16.7 mM (high) glucose for 1 h and insulin concentration was determined using a conventional enzyme-linked immunosorbent assay (ELISA, Mercodia, Sweden). The stimulation index (SI) was calculated by dividing insulin release during high glucose (16.7 mM) by insulin release during basal glucose (1.67 mM). The postisolation islet viability was determined using fluorescent staining with Syto-Green (Invitrogen) and ethidium bromide (Sigma), as previously described (5,24,31).
Endotoxin Measurement
Endotoxin content was measured in the Sigma V enzyme after filtration and in the final islet preparation using the Endosafe Portal Test System (PTS™, Charles River Laboratory). Similar testing was done with the Serva enzyme in order to compare endotoxin levels before use and in the final islet product. In brief, 1.0 ml of enzyme or final islet product was spun down for 10 s, using a bench centrifuge at 1000 rpm. Supernatants (25 μl) were injected into cartridges provided by Charles River in triplicates. Readouts were expressed as EU/ml.
Statistical Analysis
All results were expressed as either mean ± SD or SE. Differences between Sigma V and Serva NB1 were analyzed by paired or unpaired Student's t-tests and chi-square tests. Statistical analysis for multiple comparisons between Sigma V lots were analyzed by one-way ANOVA. Level of statistical significance for most analyses was set at p < 0.05; multiple comparisons using ANOVA were considered significant at p < 0.01.
Results
Human Pancreata Characteristics
Pancreas characteristics are shown in Table 1. Donor age, gender, weight, and body mass index did not show any significant differences between the two groups. The percentage of organs preserved in UW or HTK solutions, which are used for organ flush and cold storage, was also similar between groups. The only significant difference observed was cold ischemia time, which was significantly higher for the Sigma V group (10.63 ± 2.0 h vs. Serva NB1 9.32 ± 2.95 h group, p = 0.023).
Donor Characteristics of Human Pancreata
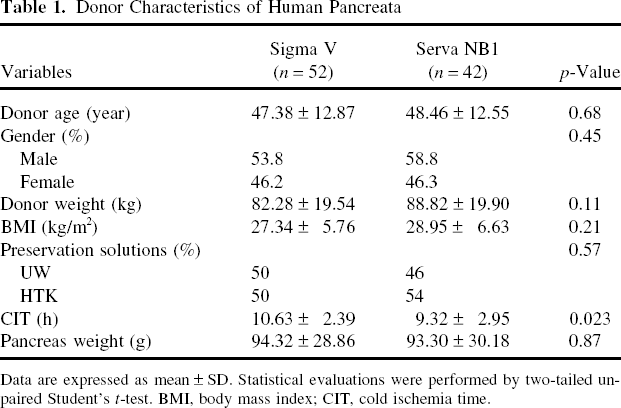
Data are expressed as mean ± SD. Statistical evaluations were performed by two-tailed unpaired Student's t-test. BMI, body mass index; CIT, cold ischemia time.
Islet Isolation Outcomes
A comparison of outcome variables from human islet isolations using either the Sigma V or the Serva NB1 enzymes is summarized in Table 2. The time required to free the majority of the islets from the surrounding exocrine tissue was not different between the Sigma V and the Serva NB1 groups. Enzyme digestion efficacy was calculated by dividing the weight of the digested tissue by total pancreas weight. The difference in digestion efficacy between Serva NB1 and Sigma V was statistically significant (69.71 ± 23.74% vs. 64.71 ± 19.12%, p < 0.05). However, when we compared pre- and postpurification yields, the mean islet equivalents (IEq) of the Sigma V group was very similar to the yields of the Serva NB1 group. Also pre- and postpurification islet yield per gram of pancreas were comparable among the groups.
Isolation Outcomes of Human Pancreata

Data are expressed as mean ± SD. Statistical evaluations were performed by two-tailed unpaired Student's t-test. IEq, islet equivalent.
Successful separation of islets from exocrine tissue using a continuous density gradient depends on several factors, such as the percentage of free islets and the difference in cell density between islets and exocrine cells. The percentage of free islets in the Sigma V group after digestion was similar to the Serva NB1 group. In addition, we further compared the purification outcomes by evaluating the distribution of islet purity and tissue volume. Each collection fraction showed a similar distribution pattern and volume between the two groups (Fig. 1A, B). With regard to differences in purity of the fractions, only the purest fraction displayed a lower value in the Sigma V group compared to the Serva NB1 group (82.1 ± 13.6% vs. 87.3 ± 7.58%, p < 0.05), while all the remaining fractions presented similar purity. After purification, islets were divided into three groups accordingly to purity. The “high purity group” had purity greater or equal to 75%, the “middle purity group” had purity between 74% and 40%, and the “low purity group” had purity less than 40%. No significant difference was observed for fractions with purities greater than 75% (Fig. 1C). Overall, these data indicated that the purity differences did not affect the islet recovery rate during the purification process. The islet recovery rate, which was calculated by dividing the prepurification by the postpurification yield, was 82.01 ± 41.70% in the Sigma V group and 78.65 ± 36.73% in the Serva NB1 group (p > 0.05).
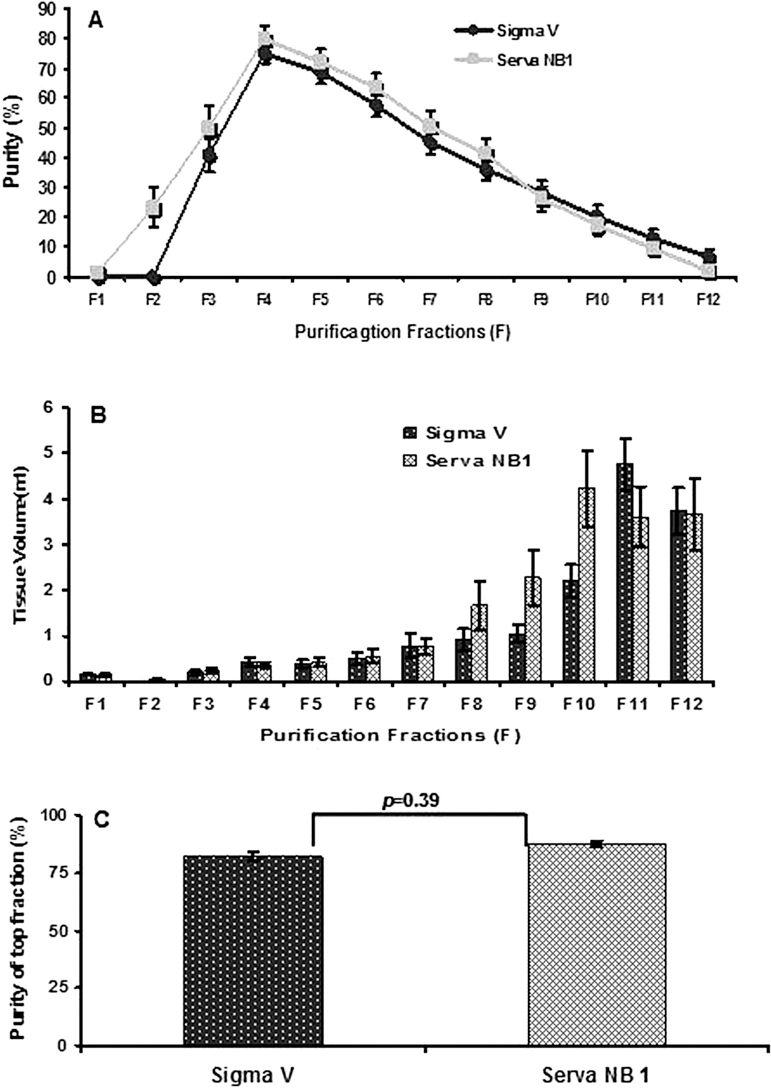
Purification outcomes of human pancreata. (A) Purified islet distribution over a density gradient of 1.066–1.078 g/cm3. (B) Tissue volume collected on continuous gradient. (C) Purity of top fraction. Data are expressed as mean ± SE and analyzed by unpaired Student's t-test by two-tail distribution (n = 52 for Sigma V and n = 42 for Serva NB1).
Sigma V and Serva NB1 groups represented a similar size distribution (Fig. 2). The ratio of actual islet number (AIN) to islet equivalent number (IEN), an indicator of islet fragmentation, was also assessed to compare islet preparations digested with Sigma V or Serva NB1. This analysis revealed a similar ratio between the two groups (0.91 ± 0.03 of Sigma V vs. 0.89 ± 0.04 of Serva NB 1, p > 0.05).

The size characteristics of isolated human islets. Size distribution (n = 52 for Sigma V and n = 42 for Serva NB1) Data are expressed as mean ± SE and analyzed by unpaired Student's t-test by two-tail distribution. IEq, islet equivalent.
Tissue specificity and age difference are also important factors in determining the efficacy of collagenase digestion dissociation. Comparison of postpurification yields of three age groups revealed no significant difference between Sigma V and Serva NB1 (Fig. 3A). The association of cold ischemia and isolation outcomes was compared between the Sigma V and the Serva NB1 groups and no significant difference was observed in either short or prolonged ischemia groups (Fig. 3B).

Impact of age and cold ischemia on the isolation outcomes of Sigma V. (A) Age impact on isolation outcomes of Sigma V. (B) CIT impact on isolation outcomes of Sigma V. Data are expressed as mean ± SE and analyzed by unpaired Student's t-test by two-tail distribution. CIT, cold ischemia time. IEq, islet equivalent (n = 52 for Sigma V and n = 42 for Serva NB1).
Islet Quality
A comparison of islet quality among the Sigma V and Serva NB1 groups is summarized in Table 3. These results show similar values for stimulation indexes, as measured by static glucose incubations, and viability assessments, as measured with inclusion and exclusion dyes. In our FDA application to use Sigma V in clinical isolations, we demonstrated in a small number of samples (n = 3) that, although preisolation endotoxin levels of the filtered Sigma V enzyme were significantly higher compared to Serva enzymes (49.50 ± 13.03 vs. 17.10 ± 3.84 EU/ml, p < 0.014). Postisolation endotoxin levels were not significantly different (0.044 ± 0.042 EU/ml Sigma vs. 0.031 ± 0.016 EU/ml Serva). Overall, comparison of the 94 isolations in this study demonstrated that the Sigma V endotoxin levels were significantly lower than the Serva NB1 group (0.022 ± 0.026 vs.0.052 ± 0.006 EU/ml, p < 0.003) (Table 3).
Islet Quality-Related Characteristics

Data are expressed as mean ± SD. Statistical evaluations were performed by two-tailed unpaired Student's t-test. SI, stimulation index.
Lot-to-Lot Variation of Sigma V Performance on Isolation Outcomes
Table 4 summarizes the isolation outcomes of four different lots of Sigma V. After our filtration process and lot screening, we only observed minimal lot-to-lot differences.
Lot-to-Lot Variation of Sigma V

Data are expressed as mean ± SD. Statistical evaluations were performed by one-way ANOVA. IEq, islet equivalent. SI, stimulation index. Multiple comparisons using ANOVA were considered significant at p < 0.01.
Discussion
Each year there are approximately 6,000 organ donors available in the US, although less than 1,500 are used for either whole pancreas transplantation or islet transplantation (28). Thousands of pancreata are not used because of poor donor characteristics and/or economic concern for isolation cost. Some studies indicate that Liberase HI and Serva NB1 are superior over crude enzyme blends (17); however, the monetary cost of these enzymes are very high. Since cost burden is one of the primary factors contributing to the low number of isolations performed annually in the US, it is important to maximize the utilization of pancreatic donors, not only for clinical transplantation but also for research and drug development. Therefore, if a crude and primitive enzyme blend can perform at least equally as well as a highly purified enzyme, then using this crude blend for research pancreata is justified.
In this study, we evaluated the use of crude collagenase as an alternative to expensive, highly purified enzyme blends for human islet isolations. With a simple three-step filtration–purification approach, and a lot screening based on the ease of filtration, we were able to achieve comparable results, in terms of islet yields and quality, with Sigma V and Serva NB1.
The herein presented study was not randomized, but the donor and organ characteristics between the two tested groups were comparable for all the variables known to impact human islet isolation outcomes. Cold ischemia time was found to be significantly longer for the organs tested with the Sigma V enzyme; however, it is unlikely that one hour of ischemia time is of clinical relevance. Additionally, a similar distribution and purity of islet size was observed in the final product, indicating that neither cellular edema nor fragmentation was increased by the use of the crude collagenase Sigma V.
The availability of reliable enzymes for pancreas digestion is the limiting factor in islet manufacturing. The lack of lot-to-lot consistency and unpredictable in-process enzyme activity continues to be the Achilles heel of clinical islet transplantation. Enzyme lot testing is extremely expensive because it can only be completed on human pancreata during islet isolations. This inherently limits enzyme development for human islet isolation. We developed a prescreening method using our three-step filtration, in which only those lots that pass easily through the three-step filtration process are tested in human islet isolations. As a result, Sigma V lot-to-lot variation was not as large in our experience, compared to other studies (30).
Despite the use of costly manufacturing methods and a cGMP-conformed manufacturing environment, the endotoxin levels in islet preparations processed with the Serva NB1 were higher than in the Sigma V group. This is remarkable, since only a simple three-step filtration process was used to purify the Sigma V. Recent studies have demonstrated that endotoxin contamination of enzymes and the materials used during the islet isolation procedure play an important role in inflammation-induced functional stunning, destruction of islets, and amplification of the autoimmune and alloimmune reactions (6,8,12). One recent study demonstrated that endotoxin level in the lyophilized Sigma V blend was as high as 6.9 ng/mg (15) without filtration. However, our data demonstrates that when Sigma V is filtered, using our simple filtration process, a final product can be obtained that is comparable to the highly purified Serva enzymes. Although it is probable that enzyme activity is altered during filtration, this does not appear to significantly alter digestion efficiency, since the filtered Sigma V postpurification islet yield is comparable to that of Serva NB1.
This study reveals that use of the low-cost, filtered Sigma Collagenase V (approximately $600), in human islet isolations yields outcomes comparable to those obtained with the expensive, highly purified Serva NB1 ($3,000–$6,000 based on Serva Premium or GMP grade). This is particularly significant in the case of research pancreata, where the implementation of a low-cost collagenase would increase the amount of isolations performed for the purpose of research. Additionally, we have recently been approved by the FDA in our current IND to use Sigma V for human islet transplantation. To date we have performed two islet transplantations from Sigma V isolations (average yield 344,297 ± 33,351) and both patients have achieved insulin independence.
Importantly, these results highlight the critical, continuing need to optimize the current methods for enzyme purification and blending. A systematic analysis of essential enzyme components for successful human pancreas dissociation is necessary to ultimately deliver a well-defined enzyme blend that will provide reliable, high quality human islet preparations as other studied indicated (7,10).
Footnotes
Acknowledgments
The authors thank the member of the UIC isolation team for islet preparations, quality assessment, and data collection. This study was partially supported by NIH Center for Research Resources (Islet Cell Resources) NIH Clinical Islet Consortium (CIT). The authors declare no conflicts of interest.
