Abstract
This study evaluated the tumorigenesis risk of induced pluripotent stem (iPS) cells after transplantation into the cochlea. One mouse embryonic stem (ES) cell line and three mouse iPS cell lines, one derived from adult mouse tail-tip fibroblasts (TTFs) and two from mouse embryonic fibroblasts (MEFs), were neurally induced by stromal cell-inducing activity. Before transplantation, the efficiency of neural induction and the proportion of residual undifferentiated cells were evaluated using immunocytochemistry, and no significant differences were observed in the ratios of colonies expressing βIII tubulin, nestin, or octamer (Oct)3/4. Four weeks after transplantation into the cochleae of neonatal mice, the number of surviving transplants of TTF-derived iPS cells generated by retroviral infection was significantly higher than those of MEF-derived iPS cells generated by plasmid transfection. Teratoma formation was identified in one of five cochleae transplanted with TTF-derived iPS cells. However, no significant differences were found in the cell proliferation activity or the extent of differentiation into mature neurons among the cell lines. These findings emphasize the necessity of selecting appropriate iPS cell lines and developing methods to eliminate undifferentiated cells after neural induction, in order to establish safe iPS cell-based therapy for the inner ear.
Introduction
Hearing loss affects millions of people worldwide and is one of the most common disabilities. Hair cells and their associated auditory primary neurons, the spiral ganglion neurons, are essential for hearing, and defects in these cells can result in profound hearing impairment or deafness. Recent studies have indicated the potential of stem cell-based approaches for the regeneration of hair cells and spiral ganglion neurons, with embryonic stem (ES) cells being identified as the most promising stem cell candidate. Previously, ES cells have been used to generate glutamatergic neurons (30), neurons making synaptic contacts with cochlear hair cells (17,18), and in vitro hair cell-like cells (15,27), as well as in the functional recovery of transplanted cochleae in an animal model (24).
However, the use of ES cells introduces problems involving immune rejection and ethics. In this context, the establishment of induced pluripotent stem (iPS) cells, which are generated from a patient's own cells (36,37,42), could be of great significance. Previous studies have investigated the potential of iPS cell-based therapy for regenerative medicine in a variety of fields, including the nervous system (16,33,39,43), which strongly suggests that they have an equivalent capacity to ES cells.
We previously reported on the potential of ES cell-derived neural progenitors, which had been neurally induced by stromal cell-inducing activity (SDIA) (13), as transplants for the replacement of spiral ganglion neurons (17,18,24). We compared the capability of iPS cell-derived neural progenitors and ES cell-derived cells to differentiate into spiral ganglion neurons (23), which suggested the possibility of substituting iPS cells for ES cells in this instance. However, iPS cells have considerable variation in their capacity for differentiation, and there is a risk of tumorigenesis among their clones (19,41), which is a critical issue for clinical applications. We previously used only one clone of iPS cells, and did not clarify the risk for tumorigenesis after cochlear transplantation. Since then, numerous iPS clones have been generated using different transcription factor combinations and sources (3,21,25,26). The current study compared the survival and neural differentiation capabilities, and the risk of tumorigenesis after cochlear transplantation, among mouse ES cells and three clones of mouse iPS cells, which were generated using different sources and methods.
Materials and Methods
Animals
Postnatal day 3 (P3) C57BL/6 mice (Japan SLC, Hamamatsu, Japan) were cared for in the Institute of Laboratory Animals at Kyoto University Graduate School of Medicine in Japan. The Animal Research Committee of the Kyoto University Graduate School of Medicine approved all of the experimental protocols, which were performed in accordance with the National Institute of Health Guidelines for the Care and Use of Laboratory Animals.
Cell Lines
The cell lines used in this study are shown in Table 1. These included three iPS cell lines, one of which was generated by the retroviral transduction of adult mouse tail-tip fibroblasts (TTFs) to produce 256H13 cells (21) carrying the Discosoma coral red fluorescent protein (DsRed) gene driven by the cytomegalovirus early enhancer/chicken β-actin (CAG) promoter. The other two iPS cell lines were generated by the retroviral transduction of mouse embryonic fibroblasts (MEFs) to produce 20D17 cells (25), or by the plasmid transfection of MEFs to produce 440A3 cells (26). We also used the mouse ES cell line G4-2 (generously donated by Dr. Hitoshi Niwa, Riken CDB, Kobe, Japan), which was derived from the E14tg2a ESC line (7) and carried the enhanced green fluorescent protein (EGFP) gene driven by the CAG promoter (see Acknowledgments for source of cells).
Summary of Cell Lines Used in This Study
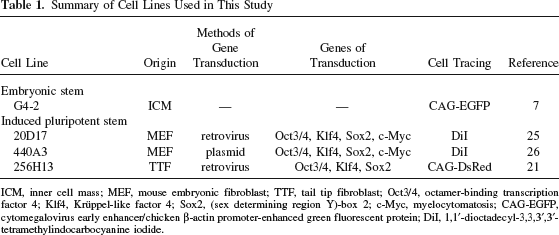
ICM, inner cell mass; MEF, mouse embryonic fibroblast; TTF, tail tip fibroblast; Oct3/4, octamer-binding transcription factor 4; Klf4, Krüppel-like factor 4; Sox2, (sex determining region Y)-box 2; c-Myc, myelocytomatosis; CAG-EGFP, cytomegalovirus early enhancer/chicken β-actin promoter-enhanced green fluorescent protein; DiI, 1,1′-dioctadecyl-3,3,3′,3′-tetramethylindocarbocyanine iodide.
Antibodies
The primary antibodies used were as follows: anti-βIII tubulin mouse monoclonal antibody (1:500; Covance Research Products, Berkeley, CA); anti-nestin mouse monoclonal antibody (1:500; BD Pharmingen, Franklin Lakes, NJ); anti-octamer-binding transcription factor 3/4 (Oct3/4) mouse monoclonal antibody (1:500; Santa Cruz Biotechnology, Santa Cruz, CA); and anti-Ki67 rabbit monoclonal antibody (1:500; Thermo Fisher Scientific Anatomical Pathology, Runcorn, UK). The secondary antibodies used were Alexa-488-conjugated anti-mouse or rabbit antibody (1:1000; Molecular Probes, Eugene, OR) and Alexa-568-conjugated anti-mouse or rabbit antibody (1:1000; Molecular Probes).
Neural Induction
SDIA was used for the neural induction of pluripotent stem cells (13), as reported previously (17,18,23, 24). Undifferentiated iPS or ES cells were cultured to form differentiated colonies on a feeder layer of PA6 stromal cells (RCB1127; Riken Cell Bank, Kobe, Japan). Colonies that formed on the PA6 monolayer after 7 days of culture were isolated. Following fixation with 4% paraformaldehyde in 0.1 M phosphate buffer for 15 min, immunostaining for βIII tubulin, nestin, and octamer (Oct)3/4 against undifferentiated and differentiated ES or iPS colonies was performed to test the efficacy of neural induction in each cell line. Specimens were viewed with a BX50 microscope (Olympus, Tokyo, Japan), and the number of βIII tubulin-, nestin-, or Oct3/4-positive colonies, as well as the total number of colonies, were counted. The ratio of βIII tubulin, nestin, or Oct3/4 expression was then calculated. Four independent cultures were performed.
Transplantation Procedure
G4-2 ES cells and 256H13 iPS cells were genetically labeled with GFP and DsRed, respectively. Colonies that formed on the PA6 monolayer after 6 days of culture were isolated by incubation with collagenase B (Roche Diagnostics, Tokyo, Japan) for 5 min at room temperature. After dissociation of those isolated colonies into single cells by treatment with 0.25% trypsin-ethylenediamine tetraacetic acid (EDTA) (Invitrogen) for 5 min at 37°C, 20D17 and 440A3 iPS cells were labeled with 5 μg/ml 1,1′-dioctadecyl-3,3,3′,3′-tetramethylindocarbocyanine perchlorate (DiI; Invitrogen) for 5 min at room temperature, and then for an additional 25 min at 4°C. After labeling, cells were washed with phosphate-buffered saline (PBS) three times and resuspended at the density of 1 × 104 cells/μl in fresh Dulbecco/Vogt modified Eagle's minimal essential medium. SDIA-treated cells were transplanted into C57BL/6 P3 mouse cochleae (n = 20). Animals were anesthetized using a combination of general anesthesia with sevoflurane and local anesthesia with 0.5% lidocaine. The left otic bulla of each animal was opened to expose the round window, and suspensions of each cell line (4 × 104 cells in 4 μl Dulbecco/Vogt modified Eagle's minimal essential medium) were injected into the scala tympani through the round window membrane, which was then covered with fibrin glue.
Fates of Transplanted Cells
SDIA-treated cells that had been transplanted into neonate mouse cochleae were examined 4 weeks after transplantation using immunohistochemistry. Under general anesthesia with sevoflurane, cochleae were dissected out and fixed with 4% paraformaldehyde for 4 h. After decalcification with 10% EDTA for 5 days, 10-μm-thick cryostat sections were made. Midmodiolus sections were provided for immunostaining for βIII tubulin and Ki67. At the end of the staining procedures, nuclear staining was performed with 4′,6-diamino-2-phenyl-indole (DAPI). Surviving transplants were defined as labeled cells with normal nuclear morphology, and the numbers of surviving transplants and βIII tubulin- or Ki67-positive transplants were counted in two midmodiolus sections of each cochlea. The average value for each animal was used in the analysis. The ratio for the expression of βIII tubulin or Ki67 in surviving transplants was calculated. In addition, hematoxylin and eosin (H&E) staining was performed in four randomly selected sections to examine tumor generation in the cochleae.
Statistical Analysis
Differences in the ratio of expression for βIII tubulin, nestin, or Oct3/4 in the colonies before and after neural induction (n = 4 for each group) were statistically examined with two-factor factorial analysis of variance (ANOVA), followed by Bonferroni test for multiple comparisons. Differences in the numbers of surviving transplants in one midmodiolus section, and in the ratio of expression of βIII tubulin or Ki67 in surviving transplants (G4-2, n = 2; 20D17, n = 5; 440A3, n = 4; 256H13, n = 4) were statistically examined with one-factor factorial ANOVA, followed by Tukey-Kramer test for multiple comparisons. Values of p < 0.05 were considered to be statistically significant. All data are presented as the mean ± SE.
Results
Efficacy of Neural Induction Before Transplantation
The expression of nestin or βIII tubulin was observed in colonies derived from each cell line following SDIA treatment (Fig. 1a–h), with typical filamentous structures of the nestin at higher magnifications (Fig. 1a–d, insets). The expression of Oct3/4 was found in colonies derived from each cell line (Fig. 1i–l). The ratios of βIII tubulin, nestin, or Oct3/4 expression before and after SDIA treatment are shown in Figure 2, with significantly higher values of βIII tubulin and nestin expression after SDIA treatment, indicating efficient neural induction. There were no significant differences in the ratio of nestin or βIII tubulin expression among cell lines. To identify undifferentiated cells in colonies after neural induction by SDIA, Oct3/4 expression was examined (Fig. 1i–l) and the ratios of expression were unexpectedly high (Fig. 2c) after SDIA treatment, implying the presence of undifferentiated cells in colonies before cell transplantation. No statistical differences in the ratio of Oct3/4 expression were found among cell lines. In short, transplants derived from each cell line contained neural-induced and undifferentiated cells in approximately similar ratios.
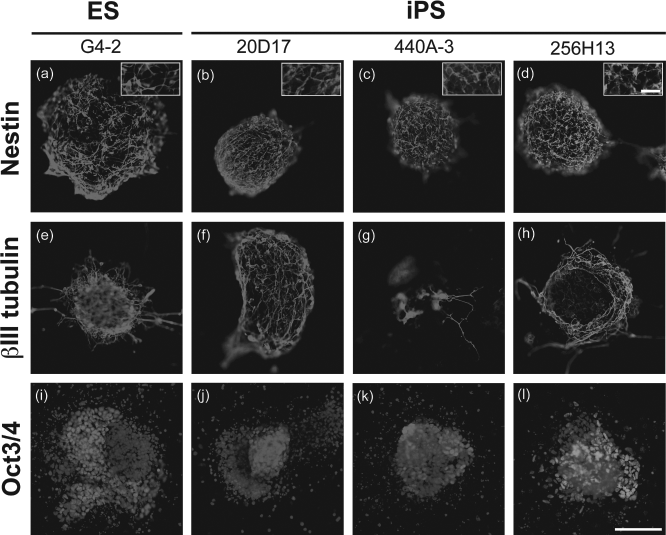
In vitro neural differentiation of embryonic stem (ES) cells (ES) and induced pluripotent stem (iPS) cells by stromal cell-derived inducing activity (SDIA). Immunocytochemical analysis of colonies following SDIA treatment for nestin (a–d), for βIII tubulin (e–h), and for octamer-binding transcription factor 3/4 (Oct3/4) with 4′,6-diamino-2-phenyl-indole (DAPI) counterstaining of nuclei (i–l). Insets (a–d) demonstrate the filamentous staining pattern of the nestin antibody. Scale bar: 100 μm. Inset scale bar: 20 μm. See text and tables for definition of cell types.

Efficacy of neural induction by SDIA. Percentages of βIII tubulin-positive (a), nestin-positive (b), or Oct3/4-positive (c) ES and iPS cell colonies before and after SDIA treatment. Significantly higher values of βIII tubulin (a) and nestin (b) expression were observed after SDIA treatment, with no significant differences among cell lines. Percentages of Oct3/4-positive colonies decreased significantly after SDIA treatment, with no significant differences among cell lines (c). *p < 0.05, **p < 0.01, ***p < 0.001.
Fates of Transplants
Four weeks after transplantation, surviving transplant-derived cells were found in two of the three cochleae transplanted with ES cell-derived cells, five of the six transplanted with 20D17 iPS cell-derived cells, four of the six transplanted with 440A3 iPS cell-derived cells, and four of the five transplanted with 256H13 iPS cell-derived cells (Fig. 3a–d). The high settlement rates of transplant-derived cells in each cell line indicate the stability of our transplantation procedure. No significant differences in the presence of transplant-derived cells were found among the cell lines, indicating no significant difference in the capability for survival. Cochlear specimens that showed the settlement of transplant-derived cells were used for further histological analyses.

In vivo neural differentiation and proliferation of ES and iPS cell-derived neural progenitors. Enhanced green fluorescence protein (EGFP) expressing ES cell-derived transplants were found in the scala tympani (arrows in a). DiI-positive iPSC-derived cells were found in the scala tympani (arrows in b, c). Discosoma red fluorescent protein (DsRed)-expressing iPSC-derived transplants were found in the scala tympani (arrow in d), in the scala media (arrowheads in d), and in the scala vestibuli (winding arrows in d). Blue fluorescence shows nuclear labeling with 4′,6-diamino-2-phenyl-indole (DAPI). Immunohistochemical analysis of markers for neurons (βIII tubulin) (e–h) and a marker for cell proliferation (Ki67) (i–l) in the C57BL/6 mouse cochlea 4 weeks after transplantation. Arrows (e–h) and arrowheads (i–l) indicate βIII tubulin- or Ki67-positive transplants, respectively. Scale bars: 500 μm (a–d), 10 μm (e–l). st, scala tympani; sm, scala media; sv, scala vestibuli; mo, modiolus.
We observed the expression of βIII tubulin in transplant-derived cells in each cell line (Fig. 3e–h), and the ratios of expression are shown in Table 2. There were no significant differences in these ratios among the cell lines, suggesting that they all have a similar ability to replace neurons in the cochlea. Ki67 expression, which is a marker of proliferating cells, was also observed in transplant-derived cells of each cell line (Fig. 3i–l), indicating that the proliferative capacity of SDIA-treated cells was maintained 4 weeks after transplantation. The ratios of Ki67 expression are shown in Table 2, and did not differ significantly between the cell lines, suggesting no significant difference in proliferative activity. However, there was a significant difference among the cell lines in the numbers of transplant-derived cells in the cochleae: the number of transplant-derived 256H13 iPS cells was significantly higher than that of 440A3 iPS cells.
Characterization of Transplants After Transplantation

Data were compared with one-factor factorial analysis of variance (ANOVA), followed by Tukey-Kramer test for multiple comparisons. The number of transplant-derived 256H13 iPS cells was significantly higher than that of 440A3 iPS cells. *p < 0.05.
Teratoma formation was identified only in one cochlea that had been transplanted with 256H13 iPS cell line-derived cells (Fig. 4). This tumor contained both undifferentiated cells and differentiated cells such as duct-forming epithelial cells, cartilage, neural cells, and mesenchymal cells (Fig. 4). The remaining four cochleae transplanted with 256H13 iPS cell line-derived cells exhibited no tumor formation, similar to the cochleae transplanted with other cell line-derived cells.

Teratoma formation in one cochlea transplanted with tail tip fibroblast (TTF)-iPSCs (256H13). The specimen was fixed with 4% paraformaldehyde (PFA) in PBS, embedded with OCT compound, sectioned, and stained with hematoxylin and eosin. (a) A midmodiolar section of the cochlea. (b) A section 100 μm medial to (a). (c, d) Higher magnification of (a) and (b), respectively. Arrow (c) indicates undifferentiated cells. Arrow (d) and arrowhead (d) indicate cartilage and keratinized epithelium, respectively. Arrows (e, f, g) indicate neural cells, mesenchymal cells, and ducts, respectively. Scale bar: 500 μm (a, b), 100 μm (c, d), 50 μm (e–g).
Discussion
Here we compared the neural differentiation capability and the risk of tumorigenesis after cochlear transplantation among three clones of iPS cells and ES cells. Before transplantation, the efficacy of neural induction and the proportion of residual undifferentiated cells after SDIA treatment were evaluated using marker proteins such as nestin, βIII tubulin, and Oct3/4. SDIA was shown to induce the neural differentiation of MEF- and TTF-derived iPS cells, as well as ES cells. Unexpectedly, colonies derived from each cell line contained residual undifferentiated cells even after neural induction, indicating that transplants derived from such cell lines also contained undifferentiated cells. Presumably, some of the SDIA-treated cells may express both pluripotent and neuronal markers, in accordance with a recent study that neural precursor cells derived from human embryonic and fetal central nervous system expressed mRNA for pluripotent markers (35). No significant differences in the ratios of marker protein expression were identified among the cell types, suggesting no apparent difference in the quality of transplants before cochlear transplantation.
Four weeks after transplantation, the majority of cochleae that had been transplanted exhibited the settlement of iPS or ES cell-derived neurons, similar to previous studies (23,24). The ratio of βIII tubulin expression in transplant-derived cells did not differ among cell lines, suggesting that all three of the iPS cell lines studied had a similar capability to ES cells for the settlement of stem cell-derived neurons in cochleae, as we observed in a previous study (23).
We investigated the proliferative activity after cochlear transplantation by immunostaining for Ki67. Interestingly, 4 weeks after transplantation, transplant-derived cells with proliferation activity were present in cochleae that had undergone transplantation with all cell types, including ES cells. Moreover, no difference in the ratio of Ki67-positive cells in the transplant-derived cells was identified among the cell lines. Previous reports describing the transplantation of ES cell-derived cells into cochleae found no tumor formation (1,2,4–6,8,9,14,24,28–30,32), suggesting that the observed proliferative activity of transplant-derived cells might not be directly associated with the risk of tumorigenesis.
By contrast, the number of transplant-derived cells in the cochleae differed among the cell lines. Although there was no significant difference between the two MEF-derived clones of iPS cells, iPS cells derived from TTFs using retrovirus gave rise to significantly higher cell numbers in transplants than iPS cells derived from MEFs using plasmids. However, there is a possibility that the difference in cell tracing methods (TTFs with CAG-DsRed expression vs. MEFs with DiI tracing) affected the number of transplants after transplantation. In addition, in 1 out of 20 transplantation experiments, teratoma formation was observed only in one cochlea that had been transplanted with retrovirally induced TTF-derived iPS cells. Retroviral integration of the exogenous transcription factors may activate or inactivate host genes, resulting in tumorigenicity in the cochlea, as was the case in some patients who underwent gene therapy (22). Another possibility is that TTF-derived iPS cells have a higher risk for tumorigenesis than MEF-derived iPS cells, which is consistent with previous observations of transplantation into nonobese diabetic/severe combined immune deficiency (NOD/SCID) mouse brains (19).
To our knowledge, this is the first documentation of teratoma formation in cochleae after cell transplantation. Previously, the transplantation of undifferentiated ES cells (6,28,32) and fetal or adult neural stem cells (10–12,38,40) into cochleae was not found to result in tumor formation. The number of cells that can be transplanted into cochleae is limited because of their tiny size, and the numbers of settled transplant-derived cells in cochleae are reportedly low (4,20,34). Therefore, the risk for settlement of undifferentiated cells after transplantation appears to be small. Notably, teratoma formation in the cochlea was identified after transplantation of TTF-derived iPS cells, suggesting that their use involves a relatively high risk of tumorigenesis. Miura et al. validated the risk for tumorigenesis of iPS cell lines following neural induction, and indicated that their origin is a key element for tumorigenesis, in association with the persistence of undifferentiated cells (19,41). The present finding supports this hypothesis.
Recent studies have demonstrated that iPS cells have a similar capability to ES cells for the generation of inner ear cells (23,27). These previous findings support the development of iPS cell-based therapy for inner ears, which might avoid the problems associated with the use of ES cells. However, the present findings demonstrate that adult TTF-derived iPS cells can be associated with a high risk of tumor formation in the cochlea, and so the source of the iPS cells is a critical issue for the safety of iPS cell-based therapy, although further systematic investigation is necessary to determine the risk for tumorigenesis in the cochlea. The elimination of undifferentiated cells from transplants and/or the selection of pure progenitors must also be considered. This could be achieved by combining fluorescence-activated cell sorting with a recently reported novel method to track differentiating neural progenitors in pluripotent cultures using microRNA-regulated lentiviral vectors (31).
Footnotes
Acknowledgments
The authors thank Keisuke Okita and Shinya Yamanaka [Center for iPS Cell Research and Application (CiRA), Kyoto University, Kyoto, Japan] for providing mouse iPS cell lines, 20D17, 440A3 and 256H13; Jun Takahashi (Department of Biological Repair, Field of Clinical Application, Institute for Frontier Medical Sciences, Kyoto University, Japan) for providing mouse PA6 stromal cells; Hitoshi Niwa [RIKEN Center for Developmental Biology (CDB), Japan] for providing mouse ES cell line G4-2; and Yoshinobu Toda (Center for Anatomical Studies, Kyoto University Graduate School of Medicine, Kyoto) for histological analysis. This study was supported by a Grant-in-Aid for Regenerative Medicine Realization, a Grant-in-Aid for Scientific Research, and a grant from the 21st Century Centers of Excellence (COE) program from the Ministry of Education, Science, Sports, Culture and Technology of Japan. The authors declare no conflicts of interest.
