Abstract
Repair of segmental bone defects remains a major challenge for orthopedic surgeons. This study aimed to investigate whether recombinant human bone morphogenetic protein-2 (rhBMP-2)-loaded calcium sulfate (CS) combined with mesenchymal stem cell (MSC) sheets could accelerate bone regeneration in ulnar segmental defects of rabbits. In vitro, the osteogenic differentiation of MSCs cultured on rhBMP-2-loaded CS was investigated. Forty complete 1.2-cm bone defects were treated with CS (group A), rhBMP-2-loaded CS (group B), MSC sheet-wrapped CS (group C), and MSC sheet-wrapped rhBMP-2-loaded CS (group D). At 4 and 8 weeks after implantation, the samples were treated by X-ray, microcomputed tomography, and histological observation. The rhBMP-2 could be released from the rhBMP-2-loaded CS scaffolds and maintain its bioactivity. The alkaline phosphatase (ALP) of MSCs cultured on rhBMP-2-loaded CS was significantly higher than that of CS at both 7 and 14 days (p < 0.05). The defects treated with MSC sheet-wrapped rhBMP-2-loaded CS showed significantly higher scores by X-ray analysis and more bone formation determined by both histology and microcomputed tomography than the other three groups at both 4 and 8 weeks after implantation (p < 0.05). No significant difference in X-ray score and bone formation was found between groups B and C, both significantly higher than group A (p < 0.05). The results suggested that MSC sheet-wrapped rhBMP-2-loaded CS may be an effective approach to promote the repair of segmental bone defects and has great potential for repairing large segmental bone defects in clinic.
Keywords
Introduction
Repair of large segmental bone defects, resulting from trauma, inflammation, or tumors, remains a major challenge for orthopedic surgeons. Autografts and allografts are widely used for bone defect repair in clinical situations, and autografts are considered the gold standard for filling bone defects; however, it is penalized by shortcomings such as donor site morbidity and donor shortage (9). The immunological response and endemic risk cannot be ignored with allografts.
These difficulties have resulted in the search for alternative bone graft substitutes. In recent years, regenerative medicine and tissue engineering have provided the possibility of successful repair and restoration of function in damaged or diseased tissues (19,23), which is based on scaffolds, cells, and growth factors.
Among the numerous materials used for scaffolds, calcium sulfate (CS) is a safe, absorbable, and moldable material, of which biocompatible properties have been well documented in clinical trials (4,35) and animal studies (10,13). However, scaffolds can be combined with mesenchymal stem cells (MSCs) and/or growth factors to improve the quality and speed of bone repair.
MSCs are multipotent cells that can be induced to differentiate into several mesodermal cell types such as osteoblasts, chondrocytes, adipocytes, tenocytes, and myoblasts (27). In previous studies, MSCs seeded onto different materials were shown to regenerate critical size bone defects (11,12,17). However, many MSCs fail to attach or their adhesion rate is poor due to the low surface to volume ratio of scaffolds (42). To address this issue, we adopted a cell transplantation method in which MSCs are cultured and lifted as a cell sheet structure (24). Thus, the scaffolds are wrapped by cell sheets.
Besides the scaffold material and cells, other important components in bone regeneration are osteoinductive factors like bone morphogenetic proteins (BMPs). Osteoblast differentiation is regulated by BMPs, and recombinant forms of human BMP-2 and BMP-7 (also known as osteogenic protein-1) have been synthesized for clinical use (38). Recombinant human (rh) BMPs require an efficient delivery system or carrier material to provide sustained release and prolong their osteoinductivity. CS is a good antibiotic carrier in both clinical (22) and animal studies (8), so we considered that whether CS could be a good rhBMP-2 carrier.
In this study, we hypothesized that rhBMP-2-loaded CS combined with MSC sheets would synergistically improve new bone formation. We investigated the osteogenic differentiation of MSCs on rhBMP-2-loaded CS scaffolds in vitro and the ability of rhBMP-2-loaded CS scaffolds, combined with MSC sheets, to form new bone in ulnar segmental bone defects.
Materials and Methods
Fabrication of rhBMP-2-Releasing CS–rhBMP-2 Scaffolds
Osteoset (Wright Medical, Arlington, TN, USA), a medical-grade CS powder, was used. rhBMP-2 (Gene Script, Piscataway, NJ, USA) was dissolved in a buffered solution at 1 mg/ml. To prepare the rhBMP-2-loaded CS scaffolds, 0.48 g of CS powder, 0.135 ml of mix solution (sterile distilled water, provided by Wright Medical), and 15 μl of rhBMP-2 were aseptically mixed in a dish. The mixture was transferred to a 4-mm diameter x 12-mm thick circular mold to produce implantation cylinders. A putative marrow cavity (diameter = 1 mm) was drilled along the major axis of the CS constructs (Fig. 1A).

Experimental procedures for the assembly of scaffold–cell constructs with cell sheet construction. (A) A putative marrow cavity (diameter = 1 mm) was drilled along the major axis of the calcium sulfate (CS) constructs. (B) Rabbit bone marrow-derived mesenchymal stem cell (BM-MSC) sheets were harvested using a cell scraper. (C) The harvested BM-MSC sheets spread in cell culture plate. (D) The recombinant human bone morphogenetic protein-2 (rhBMP-2)-loaded CS scaffold was wrapped by MSC sheets.
Also, 0.08 g of CS powder, 0.025 ml of mix solution, and 2.5 μg of rhBMP-2 were mixed, and the mixture was placed in a 6-mm diameter x 2-mm thick circular mold to make disks for cell studies. Control scaffolds were prepared without addition of rhBMP-2. All procedures were performed under sterile conditions.
MSC Isolation and Culture
Bone marrow (BM)-derived MSCs were isolated from rabbits and cultured as reported previously (6). Briefly, BM-MSCs were aspirated from bone marrow, gradient centrifuged, and plated in flasks containing Dulbecco's modified Eagle's medium (DMEM) low glucose (Gibco, Grand Island, NY, USA) containing 10% fetal bovine serum (Gibco) and 1% antibiotics (100 U/ml penicillin and 100 U/ml streptomycin). After a 48-h culture, the medium was removed, and fresh medium was added to each flask. Cells were maintained at 37°C in a humidified atmosphere with 5% CO2, and the medium was changed every 2 days. When adherent cells reached 80–90% confluence, they were detached with 0.25% trypsin-EDTA (Gibco) and replated at 1:3 in regular growth medium to allow for continued passaging. Cells performed a maximum of nine doublings during expansion and culturing procedures.
Osteogenic Differentiation of MSCs
The osteogenic differentiation of MSCs cultured on rhBMP-2-loaded CS scaffolds (6-mm diameter, 2-mm thickness) was examined by measuring alkaline phosphatase (ALP) activity expressed by the cells. Scaffolds were first placed in a 96-well plate, and MSCs were seeded onto samples at a density of 5 × 104 cells per sample and cultured for 7 or 14 days in control medium. The medium was changed every 2 days. At 7 and 14 days, the medium was removed and the samples were transferred to a new plate, washed with PBS, followed by adding a cell lysis solution. The samples were then processed through two freeze–thaw cycles (-70°C and room temperature, 45 min each) to rupture the cell membrane and extract the proteins. ALP activity of the cell lysate was determined with a pNPP Phosphatase Assay kit (Nanjing Jiancheng Bio-engineering Co., Ltd. Nanjing, China). Total protein content of the cell lysate was measured using a BCA Protein Assay kit (Nanjing Jiancheng Bio-engineering), according to the manufacturer's protocol. Dividing the quantity of ALP by the amount of total protein normalized the specific amount of ALP. ALP activities of MSCs cultured on CS scaffolds were used as controls, and ALP activities of MSCs cultured on cell culture plates served as blanks.
Fabrication of Cell Sheet–Scaffold Constructs
Confluent cells in flasks at densities of 50,000 cells/cm2 were cultured in DMEM for 2 weeks, until BM-MSC sheets formed and could be detached intact from the substratum using a cell scraper (Fig. 1B, C). The sheets were then wrapped around the rhBMP-2-loaded CS cylinders and single CS cylinders (Fig. 1D).
Animals and Surgical Procedure
Twenty New Zealand white rabbits (provided by the Laboratory Animal Center of Zhejiang University, Hang-zhou China), weighing 2.5–3.0 kg, were used. The Institutional Animal Care and Use Committee approved all animal experimental protocols. The animals were anesthetized with an intravenous injection of 3% sodium pentobarbital (30 mg/kg). Under sterile conditions a posterior lateral longitudinal incision of 3 cm was made bilaterally over the distal ulna. Soft tissues were dissected, and the bone was exposed by gentle retraction of muscles. Soft tissues were dissected, and the bone was exposed by gentle muscle retraction. Forty complete 1.2-cm bone defects were created with a highspeed saw under irrigation with physiological saline, and the periosteum was removed at the bone defect. The radius was left intact for mechanical stability.
The 40 defects were randomized into four groups (n = 5/group). In group A, only CS scaffolds were implanted into the bone defects. In group B, rhBMP-2-loaded CS scaffolds were implanted. In group C, MSC sheet-wrapped CS scaffolds were implanted. In group D, MSC sheet-wrapped rhBMP-2-loaded CS scaffolds were implanted. After implantation, muscles, fascia, and skin were closed separately over the defects, and no internal and external fixations were used. Each rabbit was administered 400,000 U of penicillin intraoperatively and on the first postoperative day to prevent infection. All rabbits were sacrificed at 4 or 8 weeks after surgery.
Gross Observation
Postoperative activities, food intake, and wound healing were observed in the animals. The status of bone repair and growth of callus were studied in samples taken out through the original incision after the animals were sacrificed.
Radiological Examination
Anterior–posterior radiographs of the bilateral forearm were obtained to assess bone healing at 4 and 8 weeks after implantation. Bone formation was assessed in each group under a triple-blinded protocol, based on the Lane and Sandhu X-ray score (18) (Table 1).
Lane and Sandhu X-Ray Scores

Microcomputed Tomography
Each sample was examined with a microcomputed tomography (μ-CT) system (Explore Locus, GE Healthcare, London, Ontario, Canada). Specimens were placed in a sample holder filled with water and oriented so that the long axis of the block was parallel to the sample holder axis. A high-resolution protocol (45.0 × 45.0 × 45.0 μm) was applied. Finally, μ-CT slices were compared with corresponding histological slides to verify the reliability of the discrimination criteria. The gray-level threshold was set to distinguish newly formed bone tissue from the CS scaffold ghost. The CS scaffold ghost showed a mean gray level of <520 ± 45, whereas that of the newly formed bone was >1,000 ± 125. Furthermore, our objective was to compare the regenerated bone tissue, so the gray-level threshold was set at 1,000 to highlight newly formed bone tissue. Digitized data were analyzed with Micro View 2.2 software (GE Healthcare) and the volume of the newly formed bone/the total volume of the defect was evaluated.
Histological Observations
Specimens from bone defect sites were fixed in 10% paraformaldehyde, decalcified with formate-sodium formate, and embedded in paraffin. Sagittal plane sections (7 μm) from each implant were prepared from the interface region, stained with hematoxylin and eosin, and examined under a light microscope (Olympus, Tokyo, Japan).
Statistical Analysis
Results are expressed as means and SDs. Lane and Sandhu X-ray scores and newly formed bone tissues were examined by one-way analysis of variance (ANOVA). Data analyses were performed using the SPSS software (ver. 15.0; SPSS, Inc., Chicago, IL, USA). Tukey's test was used for multiple comparisons, and the level of significance was set at p < 0.05.
Results
ALP Measurement
Specific ALP expression increased on rhBMP-2-loaded CS scaffolds from 7 to 14 days (Fig. 2). At 7 days, the ALP (mean ± SD; n = 5) level was 0.51 ± 0.08 nmol/s/mg protein for MSCs on rhBMP-2-loaded CS scaffolds, which was higher than 0.30 ± 0.03 nmol/s/mg protein on CS scaffolds (p < 0.05). At 14 days, ALP increased to 1.22 ± 0.21 nmol/s/mg protein in cells on the rhBMP-2-loaded CS scaffolds, which was significantly higher than 0.42 ± 0.04 nmol/s/mg protein for cells on CS scaffolds (p < 0.05). Moreover, the ALP levels of MSCs on the rhBMP-2-loaded CS and CS scaffolds were much higher than those of baseline ALP for MSCs on cell culture plates at both 7 and 14 days (p < 0.05).

Expressions of alkaline phosphatase (ALP) on rhBMP-2-loaded CS scaffolds, CS scaffolds and cell culture plates after coculturing for 7 and 14 days. The ALP of MSCs cultured on hBMP-2-loaded CS scaffolds was significantly higher than that on CS scaffolds and cell culture plates at both 7 and 14 days (p < 0.05).
Gross Observation
All rabbits ate a normal diet and behaved appropriately after surgery, and all survived until the scheduled date of sacrifice with no apparent complication, such as incision infection or skin necrosis.
At 4 weeks, most part of defects in group A was filled with fibrous tissue and could be recognized from the sample surface. New bone formed in the extremities of defects and the areas next to the radius in groups B and C; however, the outside of the defects was filled with fibrous tissue. Group D showed much more bone and less soft tissue than that of the other groups (Fig. 3D).

Gross observation of ulnar defects at postoperative 4 weeks (A, B, C, D) and 8 weeks (E, F, G, H). (A, E) CS group; (B, F) rhBMP-2-loaded CS group; (C, G) MSC sheet-wrapped CS group; (D, H) MSC sheet-wrapped rhBMP-2-loaded CS group. Scale bar: 300 μm.
At 8 weeks, the ulna repaired with the MSC sheet-wrapped rhBMP-2-loaded CS scaffolds showed good bony union (Fig. 3H), and the defects were not filled with fibrous tissue. The ulna achieved bony union in groups B and C; however, some of the defects were filled with fibrous tissue. Bony union only appeared at the extremities and areas next to the radius in group A.
X-Ray Examinations
X-ray images were obtained at 4 and 8 weeks after surgery to evaluate bone healing and bone union conditions within defects (Fig. 4). The bone defects had not been repaired at 4 weeks in group A. At 8 weeks, osteoid tissue had formed in the areas next to the radius, and the CS was degraded completely. In groups B and C, the CS was largely degraded at 4 weeks, and some callus and osteoid tissue had formed at the extremities and areas next to the radius. At 8 weeks, the CS was degraded completely, and a bony connection between the CS and bone tissue was observed. The medullary cavity achieved partial or slight recanalization, and the bone defects were partially repaired. The CS was mostly degraded at 4 weeks in group D. More callus and osteoid tissue had formed in the defects. At 8 weeks, the CS was degraded completely. The medullary cavity almost achieved recanalization, and the bone defects were mostly repaired.

Radiographic analysis of ulnar defects at postoperative 4 weeks (A, B, C, D) and 8 weeks (E, F, G, H) and Lane and Sandhu X-ray scores. (I) 4 weeks; (J) 8 weeks in each group. (A, E) CS group; (B, F) rhBMP-2-loaded CS group; (C, G) MSC sheet-wrapped CS group; (D, H) MSC sheet-wrapped rhBMP-2-loaded CS group. Results are expressed as mean ± SD. #Compared with group CS, p < 0.05; *compared with other groups, p < 0.05. Scale bar: 5 mm.
Bone formation scores were evaluated according to the Lane and Sandhu score standard, and average scores were used. The X-ray scores in each group increased with time and group D > group C and group B > group A at postoperative 4 and 8 weeks (p < 0.05). No significant difference was observed between groups B and C at any time point (Fig. 4I, J).
Bone Volume Measured on μ-CT
Newly formed bone was woven at postoperative 4 weeks, whereas the newly formed bone was much denser, resembling cortical bone at postoperative 8 weeks. The volume of newly formed bone in group D was significantly greater than that in groups A, B, and C at each time point (p < 0.05) (Figs. 5–7). No difference in the volume of the newly formed bone was observed between groups B and C at any time point. The volume of newly formed bone in groups B and C was significantly higher than that of group A at 4 and 8 weeks after implantation (p < 0.05).

The set of microcomputed tomography (μ-CT) images shows a representative transverse section and sagittal section of the forearm at a standardized position in the middle of the bony defect at 45 μm resolution at 4 weeks after implantation. (A, E) CS group; (B, F) rhBMP-2-loaded CS group; (C, G) MSC sheet-wrapped CS group; (D, H) MSC sheet-wrapped rhBMP-2-loaded CS group. Scale bar: 1 mm.

The set of μ-CT images shows a representative transverse section and sagittal section of the forearm at a standardized position in the middle of the bony defect at 45 μm resolution at 8 weeks after implantation (A, E) CS group; (B, F) rhBMP-2-loaded CS group; (C, G) MSC sheet-wrapped CS group; (D, H) MSC sheet-wrapped rhBMP-2-loaded CS group. Scale bar: 1 mm.
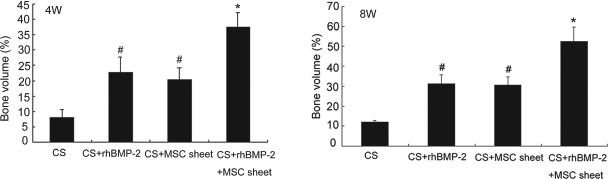
Bone formation measured in μ-CT (error bars represent means ± SD for n = 5). #Compared with CS group, p < 0.05; *compared with other groups, p < 0.05.
Histological Analysis
The CS was largely degraded at 4 weeks after implantation. In group A, woven bone formed at the extremities of the defects, whereas other parts of the defects were filled with fibrous tissue (Fig. 8A). In groups B and C, newly formed woven bone was observed in the extremities of the defects and areas next to the radius and also in the nearby marrow cavities (Fig. 8B, C). The outside of the defects was filled with fibrous tissue. In group D, some woven bone formed in the defects, even in the outside of the defects, and a bony bridge across the defects was observed (Fig. 8D).

Histological examination of the repaired bone tissues in the bone defects at 4 (A, B, C, D) and 8 weeks after implantation (E, F, G, H). (A, E) CS group; (B, F) rhBMP-2-loaded CS group; (C, G) MSC sheet-wrapped CS group; (D, H) MSC sheet-wrapped rhBMP-2-loaded CS group. w, woven bone tissue; c, cortical bone tissue; f, fibrous tissue; m, bone marrow; black arrow, osteoblasts. H&E x20. Scale bar: 200 μm.
At 8 weeks after implantation, both ends of the original ulna were united with the regenerated new bone, particularly in group D, and the CS disappeared completely. Compared with that at 4 weeks, the newly formed bone was much denser, resembling normal cortical bone, particularly in group D. In group A, bone formed in the extremities and inside the defects, blocking the marrow cavity (Fig. 8E). Cortical bone regenerated at the inside of defects in groups B and C, and the outside of the defects was filled abundantly with denser woven bone and little fibrous tissue, whereas the medullary cavity achieved partial or slight recanalization (Fig. 8F, G). In group D, cortical bone regenerated on both sides of the defects, and the medullary cavity almost achieved recanalization (Fig. 8H). Four of five group D-treated bone defects were fully repaired at 8 weeks after implantation, based on macroscopic observation, histology, and μ-CT.
Discussion
The results of this study demonstrated that the MSC sheet-wrapped rhBMP-2-releasing CS scaffolds had a synergistic effect on bone formation, as shown by X-ray, μ-CT, and histology. Moreover, we revealed that the rhBMP-2-loaded CS scaffolds had a significant positive effect on bone formation, and the effect was similar to that of MSC sheet-wrapped CS scaffolds.
In vitro cell culture experiments are a useful way to evaluate material biocompatibility. CS has been widely used in clinical situations and shows good biocompatibility, by supporting MSCs attachment and proliferation. The differentiation ability of cells on scaffolds indicates viability and indirectly demonstrates material biocompatibility (33). ALP is a well-defined marker for osteogenic differentiation of MSCs and can be regulated by many growth factors. In this study, MSCs differentiated on the rhBMP-2-loaded CS scaffolds, with a clear increase in ALP activity from 7 to 14 days. Additionally, the ALP level of MSCs cultured on rhBMP-2-loaded CS scaffolds was significantly higher than that on CS and cell culture plates at both 7 and 14 days. This confirmed that rhBMP-2 released from the rhBMP-2-loaded CS scaffolds retained its bioactivity, thus improving osteogenic differentiation of MSCs. In a previous study, rhBMP-2 released from scaffolds retained its osteoinductivity over a 28-day period in an in vitro culture (16), which promoted bone regeneration in vivo.
CS does not show intrinsic osteoinductivity, but surely displays osteoconductivity (13). In this study, rhBMP-2 was incorporated into the CS scaffolds and sustained localized release occurred, inducing bone formation. Bone tissue formation was regulated not only by the growth factor conditions, but also by the degradation rate of the scaffold. Minimal bone formation occurred in the slowly degrading scaffolds. Consistent with previous studies (2), the extent of tissue formation increased significantly in the more rapidly degrading gels, demonstrating the importance of tuning the scaffold degradation rate with the rate of new tissue formation. The scaffold degradation rate also likely influences the rate at which growth factors diffuse from a scaffold. In this study, CS displayed a more rapid resorption at 4 weeks after implantation, consistent with a previous report (36). Osteoset can be completely reabsorbed in human tissue within 4–6 weeks without an immune response (36). In this study, the CS scaffold was mostly degraded at 4 weeks and completely disappeared at 8 weeks after implantation. When CS was absorbed, rhBMP-2 was released and thus improved bone regeneration. Moreover, CS scaffolds serve as a concentrated source of calcium, which is required during active bone remodeling and mineralization. Group B showed higher X-ray scores, more newly formed bone, and better quality new bone than those of group A (Figs. 7, 8). The release of rhBMP-2 from the rhBMP-2-loaded CS scaffolds enhanced the regenerative capacity of bone, consistent with previous studies (3,30).
MSCs are multipotent cells that play roles in bone regeneration (40,44), and adding bone marrow-derived MSCs to scaffolds improves overall osteoconductivity (21,31,34). Compared with traditional cell suspension systems, the cell sheet technique achieves a highly efficient cell delivery system (26,41). The cell sheet technique has been demonstrated to be effective in soft tissues engineering, including skin (7), blood vessels, corneas (25), myocardium (32), and hard tissues (24,43), because the cell sheets effectively preserve cell–cell contact and the extracellular matrix. Furthermore, the layered cell sheets may mimic the in vivo deposition of bone matrix in which osteoblasts attach to a mineralized sheet (14). Moreover, the MSC sheets may act as periosteum to promote bone regeneration. Previous studies, using osteoconductive implants, demonstrated that intact periosteum is essential for bone regeneration (15) and that CS alone had no significant effect on bone healing without periosteum (20), consistent with the present results. Much less formed bone was observed in group A and most the defects were filled with fibrous tissue. However, group C exhibited more extensive osteogenesis and a better quantity, extent, and quality of newly formed bone tissue than those of group A at each time point.
Growth factors cannot efficiently repair large critical sized defects. Similarly, scaffolds containing only encapsulated growth factors provide a definite effect on bone regeneration for segmented bone defects. Scaffolds, cells, and growth factors constitute the three primary tissue engineering and regenerative medicine factors, and the combination of these three factors improves bone repair greatly, as confirmed by our results. The combination of rhBMP-2-loaded CS scaffolds with MSC sheets induced the strongest bone regenerative response at each time point compared with rhBMP-2-loaded CS and MSC sheet-wrapped CS scaffolds. However, no significant difference in bone formation was observed between groups B and C at 4 or 8 weeks after implantation. Additionally, μ-CT showed that the medullary cavity almost completed recanalization in group D. However, the outside of the defects in groups B and C was filled with fibrous tissue, and the medullary cavity achieved only partial or slight recanalization. Interestingly, our histological observations showed that the four groups developed regions and differing degrees of endochondral bone formation in defects with increased osteoclastic activity, suggesting that intramembranous bone formation predominates in small or mechanically stable defects, such as drill holes. When a fracture is not mechanically stable, endochondral ossification predominates, consistent with our results. Large bone defects had no additional fixation in our study. Moreover, typical chondrocytes could be seen in the bone defects, and newly formed bone tissues were in contact with chondrocytes (Fig. 9).

Histological examination of endochondral bone formation in the bone defects at 4 weeks after implantation. (A) CS group; (B) rhBMP-2-loaded CS group; (C) MSC sheet-wrapped CS group; (D) MSC sheet-wrapped rhBMP-2-loaded CS group. w, woven bone tissue; thick arrow, chondrocytes; thin arrow, osteoblasts. H&E x100. Scale bar: 200 μm.
MSC sheet-encapsulated rhBMP-2-loaded CS scaffolds were initially delivered together to regenerate new bone. MSCs can be harvested from a patient's bone marrow, expanded in culture, and formed in cell sheets for clinical application. Additionally, CS and rhBMP-2 are widely used in clinical situations. MSC sheet-wrapped rhBMP-2-loaded CS scaffolds may have potential for repairing large segmental bone defects.
However, cell layers and scaffold seeding (static or dynamic) both have advantages and uses. Scaffold seeding has been widely used for porous scaffolds and provides a significant number of cells. A cell layer can be used for scaffolds with low or no porosity, such as CS of this study. During scaffold seeding, cell distribution was uneven and the edge of the scaffolds often did not contain cells, whereas a layer was not provided for cells inside the scaffold. Thus, scaffold seeding in combination with a cell layer may be more effective for tissue repair with porous scaffolds. However, CS has no porosity and a cell layer was used in this study. An inner cavity was fabricated in the CS scaffolds to stimulate the roles of autologous MSCs from bone marrow in bone repair. Thus, MSCs from bone marrow participate in bone repair from the inner part of the construct. A larger inner scaffold cavity may be more convenient and efficient for MSCs participating in bone repair. However, a larger inner cavity means that scaffold thickness decreases, which may affect the mechanical strength of the scaffold. Using CS makes it difficult to fabricate a large inner cavity because it is very crisp. If the mechanical strength of a scaffold is not affected by a large inner cavity, then a thin scaffold with a large inner cavity combined with a layer of MSCs may be more effective in segmental bone repair.
Interestingly, we found that bone formation was greater in areas that were adjacent to bone resection areas and the intact radius in all groups, emphasizing that intact cells from surrounding tissues are crucial for a biological response to MSCs and/or growth factors. Growth factors and MSCs attract other progenitor cells or osteoclasts to initiate bone formation and remodeling (5,29). It may be that the initial bone formation sites existed during the early period after implantation. However, an early time point was not used to evaluate the initial sites of bone formation in this study. Nevertheless, new bone formed in the CS group at the extremities of defects where the endogenous periosteum was not removed at 4 weeks postoperation, indicating that the stumps may have initiated new bone formation during the early period, as occurred in the other three groups. However, a large quantity of bone filled into all bone defect areas in group D, particularly at the middle of the defect, indicating that other defect sites might also be initial sites for bone formation during the early period, due to the presence of the MSC sheets and rhBMP-2. Additionally, the length of regenerated bone was different. The length of the regenerated bone in group A was much shorter than in groups B and C, and the length of the regenerated bone in group D was the longest. These findings provide hints on clinical segmental bone repair; CS alone can be implanted for small segmental bone defects, whereas rhBMP-2-loaded CS or MSC sheet-wrapped CS can be implanted for larger segmental bone defects. We implant MSC sheet-wrapped rhBMP-2-loaded CS for much larger segmental bone defects. However, the contribution of MSCs, their role as artificial periosteum, and their destination in defects are still controversial. MSCs play positive roles in bone repair, and MSCs combined with rhBMP-2 improved the repair effect.
A limitation of this study was the lack of a negative control (empty defect alone). Large bone defects had very limited self-repair capacity, although the periosteum from the bony stumps was not removed. The empty defect group in a previous study showed that no bone formed, the extremities of bone defects were sclerotic, and that the medullary cavities were blocked (42). Strictly, the periosteum from the bony stumps should have been removed, at least from one of the two cut bone stumps. However, the effect of periosteum remnants around the stumps on bone formation was minimal, which could be seen in the CS group. However, the purpose of this study was to investigate whether rhBMP-2-loaded CS scaffolds combined with MSC sheets could synergistically repair large bone defects. Thus, the results were unaffected by not including an empty defect group and by not removing the periosteum from the bony stumps.
Another limitation is that we used allogeneic MSCs. If we had used human or ovine MSCs, we could have observed and better analyzed the source of newly formed tissue in vivo. However, if heterogeneous MSCs, such as human or ovine MSCs, were implanted into the bone defects of rabbits, most of the implanted cells would be rejected by the immune system, even if the rabbits had received an immunosuppressant. Our application of allogeneic MSCs did not cause an immunological reaction, consistent with previous reports that MSCs affect immunological modulation (1,28,39). However, autologous MSCs could have been released from the marrow in the bone defects and initiated repair, but the number of autologous MSCs was small, so the regenerative stimulus provided by the damaged bone tissue is very limited (42). Thus, the overlapping effects of allogeneic MSCs and/or BMP-2 were unaffected by using the same animal source. Additionally, allogeneic MSCs can be detected for 12 weeks after implantation in vivo (37). However, the survival, differentiation, and destination of implanted MSCs must be further investigated.
Conclusions
In summary, our results demonstrated the ability to deliver growth factors locally from biodegradable CS scaffolds and that MSC sheet- wrapped rhBMP-2-loaded CS scaffolds enhanced the repair of large segmental bone defects. Such cell and tissue engineering strategies offer therapeutic opportunities for large segmental defects in clinical situations.
Footnotes
Acknowledgments
The project was supported by the Medicine Health Sciences Grants of Zhejiang (2008A095) and the Natural Science Grants of Zhejiang (Y2100111). The authors declare no conflicts of interest.
