Abstract
The choice of enzyme blend is critical for successful islet isolation. Islet yield, viability, integrity, and function are important factors that influence the outcome of islet transplantation. Liberase HI has been used as a standard enzyme for pancreas digestion and has successfully produced islets that reversed diabetes. However, the replacement of Liberase HI with collagenase NB1 has significantly influenced the process outcome, both in quality and quantity of the isolated islets. The assessment of islet cells by Flow Cytometry (FC) has been reported to be useful for evaluating islet quality. The aim of this study was to assess the isolation outcomes and islet quality when comparing human islet cell processed with Liberase HI and NB1. A total of 66 islet isolations, 46 processed using Liberase HI and 20 using Serva NB1, were retrospectively analyzed. Islet yield, function in vitro, islet cell viability by FC, as well as isolation-related factors were compared. There was no significant difference in donor characteristics such as age and height; however, body mass index (BMI) in the Liberase HI group was significantly higher. There was also no significant difference in prepurification, postisolation, or postculture IEQ or percent recovery between the two groups. Flow data showed Liberase HI preparations had a significantly higher percent of live cells (DAPI-) and NG+/ TMRE+ when compared to NB1. Stimulation Indices (SI) for Liberase HI (n = 45) showed 3.17 and NB1 (n = 18) 2.71 (p = NS). The results of Annexin V/DAPI staining for live, apoptotic, and necrotic cells were 50.7 ± 2.24%, 14.4 ± 1.02%, and 27.8 ± 1.92% for Liberase HI versus 48.1 ± 1.93%, 12.3 ± 0.92%, and 33.9 ± 2.28% for NB1. Islets isolated using Liberase HI showed higher viable β cells by NG/TMRE staining and decreased necrosis by Annexin V/DAPI staining. FC assessment may be useful for determining the choice of digestion enzyme to maximize viable islets.
Introduction
Islet transplantation has recently been shown to have significant influence on the quality of life of selected patients with brittle type 1 diabetes by ameliorating diabetic complications particularly hypoglycemia unawareness and frequent hospitalizations (24,28,30). This therapeutic procedure has been demonstrated by several centers globally; however, multiple pancreata were used to obtain sufficient islets to reverse a single diabetic recipient (30,31). A key component that influences a successful islet isolation outcome is the choice of an appropriate enzyme type for pancreas digestion so that high quality clinical grade islets can be obtained (16, 19,29). Different types of enzymes are currently being used to digest the gland to optimize islet isolation for transplant application (4,8,16,17). It is not yet clear which of these enzymes is superior to the others in order to obtain high integrity quality viable islets. Since the development of Liberase HI (Roche, Indianapolis, Indiana) the number of patients receiving islets has increased dramatically and indeed the number of patients transplanted with islets were further increased when the Edmonton protocol was initiated more than a decade ago (3,30,31). Nevertheless, switching to a mammalian-free enzyme blend, collagenase NB1 (Serva, Heidelberg, Germany), influenced the isolation results and the number of patients transplanted with islets decreased significantly (3,6). A retrospective comparison between the two enzymes, Liberase HI and collagenase NB1, reported that the former is superior over the NB1 enzyme using digested tissue amount, islet yield, and cytokine gene expression as the main focus of their study results (7). However, a later study found that by adjusting the NB1 concentration, one could potentially acquire high-quality islets for clinical use (33). Assessment of cellular viability and cellular composition by flow Cytometry (FC) of pancreatic islets isolated from pancreata digested with collagenase and thermolysin/neutral protease from two different manufacturers—Roche Applied Science Liberase HI and Serva NB1—is the purpose of this study.
Materials and Methods
Human Pancreatic Islet Isolation and Enzyme Preparation
All islet isolations were performed using a modified automated method with the same protocol, as previously described (1,2,27). All tissue obtained for this study had verification of research consent and institutional review board approval from City of Hope National Beckman Research Institute. Enzyme digestion data was collected from pancreata (n = 66) perfused with either Liberase HI (Roche Diagnostics, Roche Applied Science, Indianapolis, IN, n = 46) or Serva premium grade Collagenase NB1 with neutral protease (SERVA Electrophoresis GmbH, Heidelberg, Germany, n = 20) via the pancreatic duct and then enzymatically/ mechanically dissociated using the modified semiautomatic digestion technique (27) with continuous shaking at 37°C. The Serva NB1 consisted of two components. The collagenase was reconstituted separately from the neutral protease (NP) and combined immediately prior to enzyme perfusion. The Liberase HI had both components in one vial and used thermolysin instead of NP. Digestion was stopped with a combination of protein and cold media dilution. All digested tissue was pooled and islets purified with Biocoll Separation Solution (Biochrom AG, Cedarlane Laboratories, Canada) using continuous density gradients 1.1 and 1.08 g/ml. Islet number, diameter, and purity were determined using dithizone (DTZ) staining and expressed as islet equivalents (IEQ) (18,26).
Donor and Pancreas Characteristics
Pancreata utilized in the study were compared according to donor characteristics: age, height, donor weight, body mass index (BMI), cold ischemia time (CIT), and pancreas weight. Isolation outcome parameters were also compared between the two enzymes: switch time, IEQ pre-Cobe, IEQ postisolation, IEQ postculture, percent recovery postisolation, percent recovery postculture, IEQ per gram pancreas weight pre-Cobe, IEQ per gram pancreas weight postisolation, IEQ per gram pancreas weight postculture.
Assessment and Characterization of Dissociated Human Islet Cells
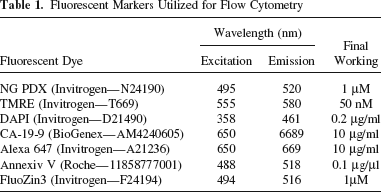
Isolated islets were initially cultured in serum-free CMRL 1066 medium containing 0.25% human serum albumin (HSA; pH 7.4) as previously described (14, 22,34), using T175 non-tissue culture-treated flasks (Sarstedt Inc.), and incubated (37°C, 5% CO2) for 24–72 h. Aliquots of 2500 IEQ with purities > 80% were utilized for flow cytometric analysis (n = 66). Single cells were prepared from whole human islets using a previously described dissociation method (12) using TrypLE enzyme, washed with PBS, and 1 × 106 cells/ condition were stained with fluorescent dyes (12,15): Newport Green PDX (NG), a zinc binding dye for β-cell identification, tetramethylrhodamine ethyl ester (TMRE), to assess mitochondrial membrane potential, 4′,6-diamidino-2-phenylindole dihydrochloride (DAPI), to identify live versus dead cells, Annexin V-FLUOS (Annexin V), to differentiate apoptotic and necrotic cells, and ductal surface marker anti-carcinoma antigen 19-9 (CA19-9; mouse monoclonal antibody) labeled with secondary antibody goat-anti-mouse IgG Alexa® 647 (10 ml and 10 mg/ml each) (15). Flow cytometry was performed using the CyAn™ ADP Flow Cytometer by Dako (UK). Table 1 shows detailed concentrations and excitation/emission wavelengths of fluorescent markers utilized in this study. Single stained NG, TMRE, and CA19-9-Alexa 647 were used for compensation. These dyes were also used in combination with DAPI for simultaneous detection of NG/TMRE/DAPI, Annexin V/DAPI, and NG/TMRE/CA-19-9-Alexa647/DAPI. A control “auto” consisting of cells and PBS only was used to determine background and autofluorescence. For the CA19-9 antibody an isotype control was initially used to detect any nonspecific binding.
Fluorescent Markers Utilized for Flow Cytometry
Whole Islet Viability and Islet Function
Viability of islets postculture was determined using fluorescence microscopy as described previously (21). Islets were stained with Fluorescein Diacetate (FDA) and Ethidium Bromide (EtBr) for visualization under fluorescence microscope to determine live percentage of whole islets.
Glucose-Stimulated Insulin Secretion
Glucose-stimulated insulin secretion (GSIS) of whole islets was performed using the dynamic perifusion assay. Briefly, 300–500 IEQ (n = 69) were incubated overnight (37°C) in RPMI-1640 containing 3 mM glucose and 10% fetal bovine serum. Islets were then perifused in a small column between Cytodex™ beads, (1 h, 37°C, equilibration), in Kreb's Ringer buffer solution containing 3 mM low glucose and 1% human serum albumin, then challenged with 16.7 mM high glucose. Insulin from eluted 1-min fractions was measured using the human insulin ELISA Kit (Mercodia). Stimulation indices were calculated by dividing average values of the initial response over the average of the baseline.
Statistical Analysis
Data was recorded as mean percentage ± SEM. ANOVA and MANOVA correlation and analysis of variance were performed using SAS 9.1 for Windows (SAS Institute Inc., Cary, NC) and Excel data analysis (Microsoft Office Excel 2003). Results were verified using the GraphPad Prism 5 program, version 5.01 for Windows, two sample t-test. A value of p < 0.05 was considered statistically significant while values of p > 0.05 were not significant (NS).
Results
Liberase HI and Serva Collagenase NB1/neutral protease were used for islet isolation. Liberase HI had an average thermolysin activity of 71278 U. The average neutral protease concentration for the Serva was 123.55 DMC. Serva uses a DMC assay to detect NP activity. Postculture islets were analyzed by flow cytometry. Table 2 shows the characterization of the donor and islet isolation from human pancreata (n = 66) used for this comparison. There was no significant difference in terms of age, height, CIT, pancreas weight, percent recovery, IEQ/g, and total IEQ between the two groups. The average weight and BMI of the donor pancreas in the Liberase HI group was significantly higher than those obtained with NB1; however, there was no significant difference in the islet number or isolation and culture recovery percentages between the two groups. There was a significant difference in switch time, with the Serva enzyme taking longer to digest the pancreas. It is worth noting that the endotoxin value as specified in the manufacturer Certificate of Analysis was significantly higher for Liberase than Serva NB1 (25.65EU vs. 0.765EU). Flow cytometry results for cells stained with NG, TMRE, CA19-Alexa647, Annexin-V, and DAPI (Fig. 1) suggest that, of the 10 staining combinations, four showed significant differences between Liberase HI and Serva NB1 enzymes: NG+, NG+/TMRE+, and DAPI-.

Cell viability and composition percentages for islets prepared with Liberase HI or Serva NB1 digestion enzymes. Dissociated human islets were stained for Newport Green (NG), tetramethylrhodamine ethyl ester (TMRE), Annexin-V, and 4′6-diamidino-2-phenylindole dihydrochloride (DAPI). A significant difference in the percentage of viable β-cells (stained positive for NG and TMRE) was seen for islets that were isolated using Liberase HI compared to Serva NB1. Live cell percentage as determined by DAPI exclusion was higher for the Liberase HI isolated islets (p < 0.0001). The percent of necrotic cells was also significantly higher in the Serva group versus the Liberase HI (p = 0.0482)
Donor, Pancreas, and Islet Characteristics
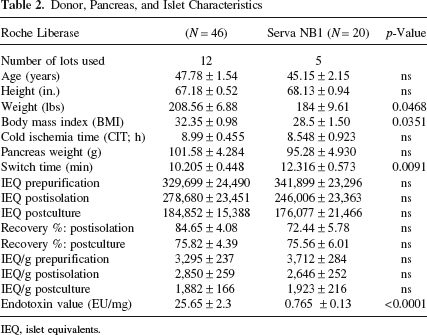
IEQ, islet equivalents.
Staining for NG+ only cells was higher in the NB1 group (71.5 ± 4.05% vs. Liberase HI 52.8 ± 3.58%, p < 0.005). Staining for TMRE+ and NG+ cells (subpopulation within the NG+ group) was significantly higher for preparations in the Liberase HI group than the NB1 group (57.8 ± 2.51% vs. 40.6 ± 3.13%; p < 0.0005). DAPI data showed the Liberase HI group had a significantly higher percent of live cells than NB1 (73.4 ± 1.34% vs. 60.7 ± 1.67%; p < 0.0001). Further breakdown of DAPI negative staining using Annexin-V showed no statistical difference in terms of live or apoptotic cells between the two groups; however, the NB1 group had a higher percentage of necrotic cells than Liberase HI (33.9 ± 2.28% vs. 27.8 ± 1.93%; p < 0.05, respectively).
The percentage of CA19+ cells was not significantly different between the two groups (n = 18, Liberase HI 26.9 ± 4.99% vs. NB1 22.0 ± 3.38%). Stimulation index for Liberase was 3.17 ± 0.25, while Serva SI was 2.71 ± 0.33; however, there was no statistically significant difference between the two groups.
Discussion
Clinical islet transplantation has been shown to be an effective treatment for a select group of patients with type 1 diabetes mellitus and for patients with severe chronic pancreatitis (11,28,30,31,35). Transplanted islets are better able to ameliorate metabolic control of blood glucose compared to insulin injections alone, preventing complications associated with diabetes (28). However, despite many improvements achieved in islet isolation, the islet yield may be insufficient and, consequently, islets from multiple organs are needed to achieve euglycemia (28,30,31).
Islet isolation involves multiple steps: pancreas preservation, pancreas digestion with various enzymes, mechanical dissociation to free the islets, purification, and islet culture. During these processes, there are several factors that may be harmful and negatively influence the quality of isolated islets. Therefore, understanding the impact of these parameters on the outcome of islet preparation may help predict islet quality prior to clinical islet transplantation (29). The focus of this study was to assess the effects of different variables such as cellular composition and islet viability, on islet cells processed using Liberase HI or NB1 by FC.
Although past studies have reported that human islet cellular composition can be measured by FC using dissociated islet cells, these studies were limited not only by the sample size (number of isolations) but also by the specific indicators and markers used to assess the islets (10,12,20,25). There are many different indicators that have been utilized to measure cell viability, such as TMRE, DAPI, 7-aminoactinomycin D (7-AAD), and propidium iodide (PI) (9,10,12,13,25,32). Previous studies have used 7-AAD in combination with TMRE; however, these two dyes overlap. We opted for DAPI and a laser filter, which is more sensitive, has a shorter wavelength, and does not compete with additional markers used in this study (12,32). However, viability of dispersed islets using DAPI alone does not make a distinction between live and early apoptotic cell populations, thereby giving an elevated live percentage. Islets in early stage apoptosis were indistinguishable by DAPI staining alone; however, with the additional use of Annexin-V/DAPI, 14.4% of DAPI-negative cells stained apoptotic and 50.7% were live for Liberase HI. Necrotic cells (27.8 + 1.93%), as determined by Annexin-V/DAPI, and dead cells (26.5+1.32%), by DAPI staining alone yielded similar results for Liberase HI. This study showed that there is a marked difference for cells positive for active mitochondrial membrane potential as determined by TMRE staining and those negative for DAPI and positive for Annexin-V, indicating apoptosis. TMRE attaches to the negatively charged mitochondrial membrane of healthy cells and therefore Annexin-V would be excluded from live cells. Therefore, using two different dyes, TMRE and Annexin-V, to detect early and late apoptosis respectively would be useful markers to predict islet quality. There were 57.8 + 2.52% NG+/TMRE+ cells and 50.7 + 2.24% Annexin-V/DAPI- cells for Liberase HI while 40.6 + 3.13% NG+/TMRE+ cells and 48.1 + 1.93% Annexin-V/DAPI- cells for NB1.
It has been reported that digestion enzyme lot plays an important role in determining the isolation outcome (4,5,16,17). This study focused on cell viability and composition, and variability among two enzymes (Liberase HI and Serva Collagenase NB1) from different manufacturers was observed. Despite the fact that the Serva group had higher staining for NG, the islets from pancreata digested with Liberase HI had significantly higher cell viability, as determined by NG, TMRE, and DAPI, and lower necrosis as determined by Annexin-V/DAPI than those pancreata digested with Serva enzyme. Using Liberase HI resulted in significantly better islet yield; however, in 2007 it was mandatory to switch to mammalian-free enzyme due to the possibility of prion contamination from bovine protein utilized in the production process (7,8,23,33). The enzyme change to Serva collagenase NB 1 resulted in a marked variability in reproducible islet results among centers (6,17). Even with lot-to-lot variability (the use of 12 different lots), the Liberase on average had higher cell viability as determined by FC, lower switch time, and although higher endotoxin, this did not affect the viability or yield. In addition, the effect of enzyme types has shown no significant difference in stimulation indices for the isolated islets. However, a trend of higher SI for islets isolated from organ digested with Liberase HI than Serva enzyme was observed.
In conclusion, this study determined that FC assessment of cellular composition and viability of the islet preparation—in association with other potency assays—may provide an excellent means to improve current islet preparation and culture condition. FC results can help substantiate the suitability of the digestion enzyme used for the pancreas storage and digestion.
Footnotes
Acknowledgments
This work was supported by a grant from the National Institute of Health (NIH), the Islet Cell Resources (ICR) 5U42RR016607, and City of Hope National Medical Center (COH). The authors wish to thank the Islet Cell Resource Consortium (ICR) of the City of Hope National Medical Center for providing islet cells used to carry out the study. We particularly wish to thank the islet isolation team of the Southern California Islet Cell Resources Center (SCICRC). Special words of appreciation to: Lucy Brown and her team at the Analytical Cytometry Core Lab for their help with the flow cytometry and to the SCICRC Quality Control department for all QC SI and FDA/EtBr testing results. We would like to thank Dr. Silvia da Costa for editing and constructive input. The authors declare no conflicts of interest.
