Abstract
For the advancement of porcine xenotransplantation for clinical use in type 1 diabetes mellitus, the concerns of a sustainable and safe digestion enzyme blend must be overcome. Incorporating good manufacturing practices (GMP) can facilitate this through utilizing GMP-grade enzymes. In conjunction, still taking into account the cost-effectiveness, a wide concern. We evaluated how GMP-grade enzyme blends impact our piglet islets and their long-term effects. Preweaned porcine islets (PPIs) were isolated from 8- to 10-day-old pigs. Digestion enzyme blends, collagenase type V (Type V), collagenase AF-1 GMP-grade with collagenase NB 6 GMP-grade (AF-1 and NB 6), and collagenase AF-1 GMP-grade with collagenase neutral protease AF GMP-grade (AF-1 and NP AF) were compared. Islet quality control assessments, islet yield, viability, and function, were performed on days 3 and 7, and cell content was performed on day 7. GMP-grade AF-1 and NB 6 (17,209 ± 2,730 islet equivalent per gram of pancreatic tissue [IE/g] on day 3, 9,001 ± 1,034 IE/g on day 7) and AF-1 and NP AF (17,214 ± 3,901 IE/g on day 3, 8,833 ± 2,398 IE/g on day 7) showed a significant increase in islet yield compared to Type V (4,618 ± 1,240 IE/g on day 3, 1,923 ± 704 IE/g on day 7). Islet size, viability, and function showed comparable results in all enzyme blends. There was no significant difference in islet cellular content between enzyme blends. This study demonstrated a comparison of GMP-grade collagenase enzyme blends and a standard crude collagenase enzyme in preweaned-aged porcine, a novel topic in this age. GMP-grade enzyme blends of AF-1 and NB 6 and AF-1 and NP AF resulted in substantially higher yields and as effective PPIs compared to Type V. In the long run, considering costs, integrity, and sustainability, GMP-grade enzyme blends are more favorable for clinical application due to high reproducibility in comparison to undefined manufacturing processes of standard enzymes.
Introduction
Exogenous insulin is the most dependable, sought-after treatment for type 1 diabetes mellitus (T1DM) 1 , a T-cell-mediated autoimmune disorder characterized by a destruction of insulin-producing beta cells, resulting in ketoacidosis and hyperglycemia 2 . While insulin therapy has proven to be an overwhelmingly successful, life-sustaining method of treating T1DM patients, subcutaneous insulin injections display high variability; this defect gives rise to an unpredictable therapeutic response resulting in inadequate glycemic control and heightened risk of hypoglycemia 3 . To ameliorate the shortcomings of insulin therapy, researchers have established islet allotransplantation to restore euglycemia in patients disabled by intransigent hypoglycemia and insulin deficiency 4 . However, this method of T1DM therapy is challenged by the harsh scarcity of organ donors, resulting in the extensive search for a plausible, immunocompatible alternative for beta cell replacement: porcine islet xenotransplantation 5 .
Successful xenotransplantation from pig to humans could give rise to an unlimited supply of cost-effective organs, tissues, and cells, resolving the shortage of expensive human islet cells required for allotransplantation 6 . Preparation to produce highly functioning, viable and sustainable porcine islets involves a scrupulous isolation and digestion protocol, necessitating a proper enzyme blend 7,8 . Moreover, authorization of superior-quality, consistently pure islets for usage in clinical settings requires utilization of highly purified dissociation enzymes that abide by the good manufacturing practices (GMP) guidelines 9 . GMP guidelines serve as regulation for manufacturing, testing, and quality assurance for products to be used for human consumption; such guidelines include defined and controlled processes, a clean and hygienic manufacturing facility, and good documentation practices. Reagents subordinate to these guidelines have been banned from human islet isolation procedure for clinical purposes because of the risk of transmission of prions, a destructive disease 10 .
This endeavor for a consistent enzyme blend, however, has yet to desist. A standard enzyme used, crude collagenase from Clostridium histolyticum, type V (Type V; Sigma-Aldrich, St. Louis, MO, USA), has proven to generate isolation outcomes exhibiting endotoxin levels significantly lower than that of a highly purified enzyme blend produced under GMP guidelines, producing promising results at a much lower cost 11,12 . Studies have also demonstrated crude collagenase yields islet numbers highly comparable to those of purified enzymes, while demonstrating efficient release of islets from the acinar tissue of the pancreas as well as preservation of islet size 13 , inevitably making this cost-effective enzyme blend more appealing and widely used 14 . In spite of this, this crude collagenase fails to completely eliminate the predicament of lot-to-lot inconsistencies; this may be attributed to subsequent deterioration of crude enzyme activity over time through storage 15 , which, in turn, nullifies the cost-effectiveness of Type V in the long run, as the longevity of GMP-grade enzyme blends outweighs the immediate low cost of crude collagenase.
In this study, we aim to observe the outcome of utilizing GMP-grade enzyme blends, collagenase AF-1 with collagenase NB 6 (Nordmark Biochemicals, Uetersen, Germany) and collagenase AF-1 with neutral protease AF (Nordmark Biochemicals), using doses comparable to the enzyme activities of our standard enzyme Type V. Highly purified collagenase AF-1 mainly contributes collagenase activity, whereas collagenase NB 6, as crude collagenase, mainly contributes neutral protease and tryptic-like activities to the doses. Highly purified Neutral Protease AF contributes neutral protease activity. We hypothesize that GMP-grades AF-1 (26.1PZ-U/g, 0.2DMC-U/g, 0.7TLA-U/g) and NB 6 (3.5PZ-U/g, 3.5DMC-U/g, 8.8TLA-U/g) and AF-1 (29.6PZ-U/g, 0.2DMC-U/g, 0.8TLA-U/g) and NP AF (9.1DMC-U/g) will be as effective in producing islet yield, size, and function as the standard crude enzyme, Type V.
Materials and Methods
Islet Isolation: Procurement and Digestion
Donor pancreata were obtained from 8- to 10-day-old, preweaned Yorkshire pigs (S&S Farms, Ramona, CA, USA) of either sex. All animal procedures were performed under protocol approved by the University of California, Irvine Institutional Animal Care and Use Committee (IACUC# AUP-17-129). Nonsurvival surgery was performed in <4 to 5 min on porcine to obtain pancreatic tissue, as previously described 16 . The procured pancreata were preserved in Hank’s Balanced Salt Solution (HBSS; Gibco-Thermo Fisher Scientific, Waltham, MA, USA) on ice until mechanical digestion; cold ischemic time did not exceed 1 h. Pancreatic tissue was mechanically minced and washed until 1 mm3 fragments were obtained, and were separated into ≤2 g of tissue per 50 ml conical tube 17,18 . Ischemia time was carried out through enzymatic digestion of the following collagenases: Type V (29.6PZ-U/g of pancreas, 9.3DMC-U/g, 9.4TLA-U/g), collagenase AF-1 (26.1PZ-U/g, 0.2DMC-U/g, 0.7TLA-U/g) with collagenase NB 6 (3.5PZ-U/g, 3.5DMC-U/g, 8.8TLA-U/g), and collagenase AF-1 (29.6PZ-U/g, 0.2DMC-U/g, 0.8TLA-U/g) with Neutral Protease AF (9.1DMC-U/g) (Table 1). For the collagenase and neutral protease activities of Type V, which were originally not in PZ-Units and DMC-Units, conversion rates as found in the literature or as evaluated by analyses of Type V batches were applied 19 . Enzymes were prepared at least 30 min before use to ensure they were kept cold until use; enzymes were dissolved in HBSS and sterile-filtered through a 0.22-µm filter. The appropriate dose of cold enzyme was added and digestion volume totaled to 25 ml. Pancreatic tissue was digested at 37°C, shaking at 100 rpm for 15 min. Digested tissue was efficiently neutralized with HBSS supplemented with 10% porcine serum (Gibco-Thermo Fisher Scientific) and filtered through a 500-µm mesh.
Enzyme Dosing Details.

GMP: good manufacturing practices; NP AF: neutral protease AF
Islet Culture
Preweaned porcine islets (PPIs) were plated in T-225 untreated suspension flasks (Corning Inc., Corning, NY, USA) and cultured in islet maturation media (IMM) at 37°C and 5% CO2 (Thermo Forma Series II 3210 Water Jacketed CO2 Incubators; Thermo Fisher Scientific) for 3 to 7 days 18 . One hundred percent media change on day 1 with IMM. One hundred percent media change on day 3 with IMM supplemented with 100 μM Necrostatin-1 (Abcam, Cambridge, UK). Fifty percent media change on day 5 with IMM supplemented with Necrostatin-1. One hundred percent media change on day 7 with IMM supplemented with Necrostatin-1. Islet assessment was performed on days 3 and 7 except cellular content, which was only performed on day 7.
Islet Yield and Islet Size
To determine islet count (IC) and islet equivalent (IE) values, a 100-µl aliquot was dithizone-stained (MP Biomedicals, Irvine, CA, USA) for 2 to 3 min. Dithizone-stained PPIs were counted in duplicate with a standard stereomicroscope (Max Erb, Santa Ynez, CA, USA) including a 10× eyepiece graticule 20,21,22 . Pictures of dithizone-stained PPIs were taken with an EVOS XL digital inverted microscope (Thermo Fisher Scientific). PPI yields were quantified as islet count per gram of pancreatic tissue (IC/g) and islet equivalent per gram of pancreatic tissue (IE/g). IE/IC index ratio was calculated as IE yield divided by IC yield to indicate islet size 23 .
Islet Viability
Duplicate samples of 100 IE aliquot were stained with calcein AM (Invitrogen, Carlsbad, CA, USA) and propidium iodide (Invitrogen) for 15 min 17 . Stained islets’ fluorescence was examined through a microplate reader (Tecan Infinite F200; Tecan, Mannedorf, Switzerland). Percent viability was calculated through the equation: (calcein AM-positive cells/[calcein AM-positive cells + propidium iodide-positive cells]) × 100. Islet cellular viability is described below, proceeding islet cellular content procedures.
Islet Function: Glucose Stimulated Insulin Release Assay
A triplicate sample of 100 IE was incubated at 37°C and 5% CO2 for 1 h in each media in the corresponding order: low glucose (2.8 mM; L1), high glucose (28 mM; H), back to low glucose (2.8 mM; L2), and high glucose plus 3-isobutyl-1-methylxanthine (28 mM + 0.1 mM IBMX; H+) 16 . Supernatant was collected in labeled tubes and stored at −20°C until analysis. Analysis insulin secretion levels were quantified using a porcine insulin enzyme-linked immunosorbent assay (Porcine Insulin ELISA; Mercodia, Uppsala, Sweden) and further analyzed through a microplate reader (Tecan; and Magellan V7, Phoenix, AZ, USA). Insulin levels were normalized with DNA content, as described below, and indicated as pg of insulin/ng of DNA/hour (pg/ng DNA/h). The stimulation index (SI) is the insulin concentration released from the H divided by the insulin concentration released from the L1.
Islets, after completion of GSIR assay, were collected and lysed with TE buffer and sonicated (Sonics VibraCell Ultrasonic Processor Model VC70 T; Sonics & Materials, Inc., Newton, CT, USA) for 30 s on ice 18 . Lysed cells were centrifuged at 4°C at 1,400 × g for 15 min. Supernatant was collected, fluorescence-stained (Quant-iT PicoGreen dsDNA kit; Molecular Probes, Eugene, OR, USA), and examined through a microplate reader (Tecan).
Islet Viability and Cellular Content: Flow Cytometry Assay
Duplicate samples of 3,000 IE aliquot were dissociated with Accutase (Innovative Cell Technologies, San Diego, CA, USA) for 15 min in a 37°C, 100 rpm shaking water bath to obtain single cells 24 . Dissociated cells were filtered through a 40-µm filter (Corning Inc.) and stained with 7-aminoactinomycin D dye (7-AAD; Invitrogen) for 30 min on ice to detect live and dead cells. Stained cells were both fixed with 4% paraformaldehyde for 10 min and permeabilized with intracellular staining permeabilization wash buffer (BioLegend, San Diego, CA, USA) for 15 min on ice. Cells were blocked and incubated with Protein Block (Abcam) for 30 min on ice to minimize nonspecific binding, followed by staining for intracellular markers with fluorescently conjugated antibodies through intracellular staining permeabilization wash buffer (BioLegend) with 0.5% bovine serum albumin (Equitech-Bio, Inc., Kerrville, TX, USA) for 30 min on ice. Phycoerythrin (PE)-conjugated anti-insulin (CST, Beverly, MA, USA), APC-conjugated anti-glucagon (Novus Biologicals, Littleton, CO, USA), and PE-conjugated anti-somatostatin (Novus Biologicals) antibodies were used as a marker for beta, alpha, and delta cells, respectively. Fluorescein isothiocyanate-conjugated anti-GLUT2 (Novus Biologicals) antibody was used with PE-conjugated anti-insulin for double staining to identify GLUT2-positive beta cells. Stained cells were quantified and analyzed using the NovoCyte 3000VYB Flow Cytometer (ACEA Biosciences, Inc., San Diego, CA, USA) and FlowJo software (FlowJo, Ashland, OR, USA). For gating controls, unstained, single-stained, fluorescence minus one, and matching isotype controls were used.
Immunohistochemical Evaluations of Islets
Islets collected on day 7 were fixed in neutral buffer formalin (Cat # 22-026-354, Fischer Scientific) for at least 48 h. The fixed samples were prepared for paraffin processing through serial dehydration in increasing concentrations of ethanol solutions using a Leica TP1020 tissue processor (Leica Microsystems, Buffalo Grove, IL, USA). After preparation, the tissues were embedded in paraffin wax (Sigma Aldrich) using a Leica EG 1150C tissue embedder (Leica Microsystems). The tissue blocks were sectioned into 5-µm sections using a Leica RM 2255 microtome (Leica Microsystems) and incubated at 65°C for 1 h before staining. Mouse and Rabbit Specific horseradish peroxidase/3, 3-diaminobenzidine (ABC) Detection Kit (cat#ab64264, Abcam) was utilized to detect insulin-positive cells according to the manufacturer protocol and slides were counterstained with hematoxylin.
Statistical Analysis
All data are represented as mean ± standard error of mean (SEM). To determine whether IC/g, IE/g, IE/IC index ratio, islet viability, SI, islet function, and islet cellular composition results were statistically significant, a one-way analysis of variance followed by a post hoc Tukey’s honestly significant difference test was carried out. The disparity of the values compared were considered statistically significant if P-values <0.05.
Results
Yield and Islet Size of PPIs
Preweaned Yorkshire porcine at 8 to 10 days ranged from 3 to 6 g of pancreatic tissue collected during surgery. To compare islet yield between enzyme groups, IC and IE counts were done on days 3 and 7 of culture. IC and IE were then normalized and represented as per gram of pancreas of tissue digested. On day 3, PPI tissue digested with Type V produced 8,087 ± 812 IC/g (n = 6) (mean ± SEM) and 4,618 ± 1,240 IE/g (n = 6) (Table 2), theoretically yielding 24,260 to 48,520 IC per pancreas and 13,853 to 27,705 IE per pancreas. PPI tissue digested with AF-1 and NB 6 produced 16,993 ± 23,60 IC/g (n = 11) and 17,209 ± 2,730 IE/g (n = 11) (Table 2), theoretically yielding 50,979 to 101,958 IC per pancreas and 51,626 to 103,252 IE per pancreas. AF-1 and NB 6 materialized a twofold (P < 0.05) increase in IC/g yield and a fourfold (P < 0.01) increase in IE/g yield compared to Type V. PPI tissue digested with AF-1 and NP AF produced 20,794 ± 38,93 IC/g (n =5) and 17,214 ± 3,901 IE/g (n = 5) (Table 2), theoretically yielding 62,381 to 124,763 IC per pancreas and 51,641 to 103,282 IE per pancreas. AF-1 and NP AF materialized a threefold (P < 0.01) increase in IC/g and a fourfold (P < 0.05) increase in IE/g compared to Type V. At day 7, PPI tissue digested with Type V produced 1,626 ± 136 IC/g (n = 5) and 1,923 ± 704 IE/g (n = 6) (Table 2), theoretically yielding 4,879 to 9,758 IC per pancreas and 5,770 to 11,540 IE per pancreas. PPI tissue digested with AF-1 and NB 6 produced 8,496 ± 675 IC/g (n = 10) and 9,001 ± 1,034 IE/g (n = 10) (Table 2), theoretically yielding 25,488 to 50,976 IC per pancreas and 27,004 to 54,007 IE per pancreas. AF-1 and NB 6 materialized both a twofold decrease in IC/g (P < 0.01) and IE/g (P < 0.05) from day 3 of culture to day 7. PPI tissue digested with AF-1 and NP AF 7,229 ± 510 IC/g (n = 5) and 8,833 ± 2,398 IE/g (n = 5) (Table 2), theoretically yielding 21,686 to 43,373 IC per pancreas and 26,500 to 52,999 IE per pancreas. AF-1 and NP AF materialized a threefold (P < 0.01) decrease in IC/g and a twofold decrease in IE/g on day 3 of culture compared to day 7. Type V resulted in an eightfold decrease in IC/g and a fourfold decrease in IE/g from day 3 to day 7 of culture, despite its insignificance. There was no notable difference in IE/IC ratio between the different collagenases; however, both AF-1 and NB 6 and AF-1 and NP AF had bigger islet sizes. All collagenase groups maintained their islet size ratio from day 3 to day 7 of culture.
Islet Yield and Islet Size of PPIs on Day 3 and Day 7 of Culture.

IC/g: islet count per gram of pancreatic tissue; IE/g: islet equivalent per gram of pancreatic tissue; IE/IC: islet equivalent to islet count ratio; NP AF: neutral protease AF; Type V: collagenase type V.
Values represented as mean ± SEM. *P < 0.05 compared to control media on the same day of culture. **P < 0.01 compared to control media on the same day of culture. †P < 0.05 compared to the same media on day 7 of culture. ††P < 0.01 compared to the same media on day 7 of culture.
In terms of morphology, Type V displayed a majority of fairly spherical, rounded islets with good integrity and some fragmentation on day 3 (Fig. 1A). By day 7, Type V islets remained to be fairly spherical with fragmentation, however, more jagged and largely reduced in integrity (Fig. 1B). AF-1 and NB 6 displayed to be a majority of spherical, mix of rounded and irregular islets with sufficient integrity and very little fragmentation on day 3 (Fig. 1C). By day 7, AF-1 and NB 6 islets remained to be spherical islets with very little fragmentation, however, more rounded, slightly reduced in integrity, and presence of little clusters (Fig. 1D). AF-1 and NP AF displayed to be a majority of spherical, well-rounded islets with great integrity and little fragmentation on day 3 (Fig. 1E). By day 7, AF-1 and NP AF islets remained to be spherical, well-rounded islets, however, with reduced integrity and more fragmentation (Fig. 1F).
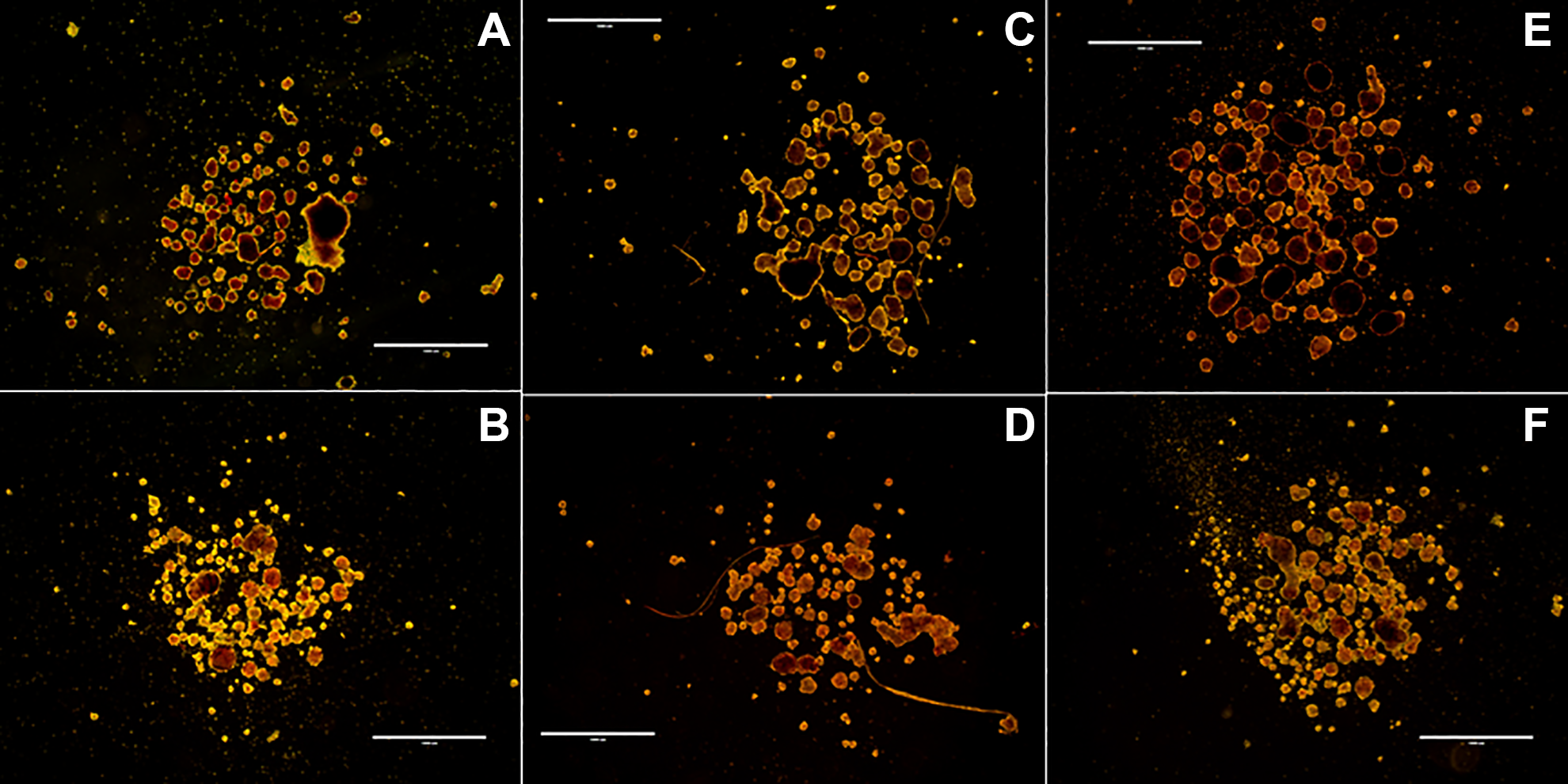
Dithizone-stained PPIs, 20/40 ph, scale: 1,000 µm. (A) Type V on day 3. (B) Type V on day 7. (C) AF-1 and NB 6 on day 3. (D) AF-1 and NB 6 on day 7. (E) AF-1 and NP AF on day 3. (F) AF-1 and NP AF on day 7.
Viability of PPIs
Viability was examined through Calcein AM and propidium iodide staining on day 3 and day 7 of culture. At day 3 (92% ± 1%) and day 7 (93% ± 1%) of culture, Type V showed no significant difference (Fig. 2A). AF-1 and NB 6 significantly (P < 0.05) decreased over culture time from day 3 (94% ± 1%) to day 7 (88% ± 2%) (Fig. 2A). At day 3 (95% ± 1%) and day 7 (92% ± 3%) of culture, AF-1 and NP AF showed no significant difference. In addition, single-cell viability was examined through 7-AAD staining on day 3 and day 7 of culture paired with cellular composition analysis through flow cytometry. Findings from 7-AAD-stained viability validate the insignificant difference in collagenases (90% ± 1%, 87% ± 3%, 92% ± 1%, 86% ± 3%, 90% ± 1%, 87% ± 2%, respectively) (Fig. 2B). Overall, viability remained at reasonable levels of >80% throughout culture for all collagenases.

Islet viability on day 3 and/or day 7 of culture. (A) Calcein AM/PI-stained PPI viability. PPIs digested from Type V (n = 6), AF-1 and NB 6 (n = 11, 6), and AF-1 and NP AF (n = 5, 4) were stained using fluorescent dyes CalAM and PI, and analyzed with a microplate reader. Islet viability was calculated by the equation: [CalAM-positive cells/(CalAM-positive cells + PI)] × 100. (B) 7-AAD-stained PPI viability. PPIs digested from Type V (n = 3), AF-1 and NB 6 (n = 5), and AF-1 and NP AF (n = 5) were dissociated, stained with 7-AAD dye, and analyzed by flow cytometry. Values represented as mean ± SEM. *P < 0.05.
Functionality of Islets In Vitro
GSIR assay was used to establish the functionality of the islets in vitro on days 3 and 7 of culture, through determining the amount of insulin released when islets are stimulated by varying amounts of glucose. Insulin (mU/l) and DNA concentrations (ng/ml) were obtained through ELISA and DNA assays, respectively, and insulin release was normalized, and data are represented as pg/ng DNA/h. There was no significant difference between Type V (L1: 3.71 ± 2.35 pg/ng DNA/h, H: 2.26 ± 0.42 pg/ng DNA/h, L2: 1.71 ± 0.45 pg/ng DNA/h, H+: 2.98 ± 1.06 pg/ng DNA/h), AF-1 and NB 6 (L1: 2.68 ± 0.40 pg/ng DNA/h, H: 3.35 ± 0.32 pg/ng DNA/h, L2: 1.58 ± 0.28 pg/ng DNA/h, H+: 2.24 ± 0.29 pg/ng DNA/h), and AF-1 and NP AF (L1: 1.58 ± 0.41 pg/ng DNA/h, H: 2.41 ± 0.62 pg/ng DNA/h, L2: 1.33 ± 0.22 pg/ng DNA/h, H+: 2.00 ± 0.28 pg/ng DNA/h) on day 3 of culture (Fig. 3A). Incongruously, Type V shows an abnormal response to 2.8 mM glucose concentration at L1. There was no significant difference between Type V (L1: 0.57 ± 0.12 pg/ng DNA/h, H: 0.87 ± 0.18 pg/ng DNA/h, L2: 0.71 ± 0.15 pg/ng DNA/h, H+: 1.89 ± 0.63 pg/ng DNA/h), AF-1 and NB 6 (L1: 1.75 ± 0.36 pg/ng DNA/h, H: 3.85 ± 1.60 pg/ng DNA/h, L2: 1.63 ± 0.21 pg/ng DNA/h, H+: 3.86 ± 0.74 pg/ng DNA/h), and AF-1 and NP AF (L1: 0.86 ± 0.26 pg/ng DNA/h, H: 1.32 ± 0.46 pg/ng DNA/h, L2: 0.91 ± 0.19 pg/ng DNA/h, H+: 2.08 ± 0.23 pg/ng DNA/h) on day 7 of culture (Fig. 3A). Nevertheless, AF-1 and NB 6 shows an overall higher response in insulin release. More importantly, from day 3 to day 7 AF-1 and NB 6 displays maintenance of insulin release throughout culture, in contrast to the other enzyme groups that display a decrease in response.
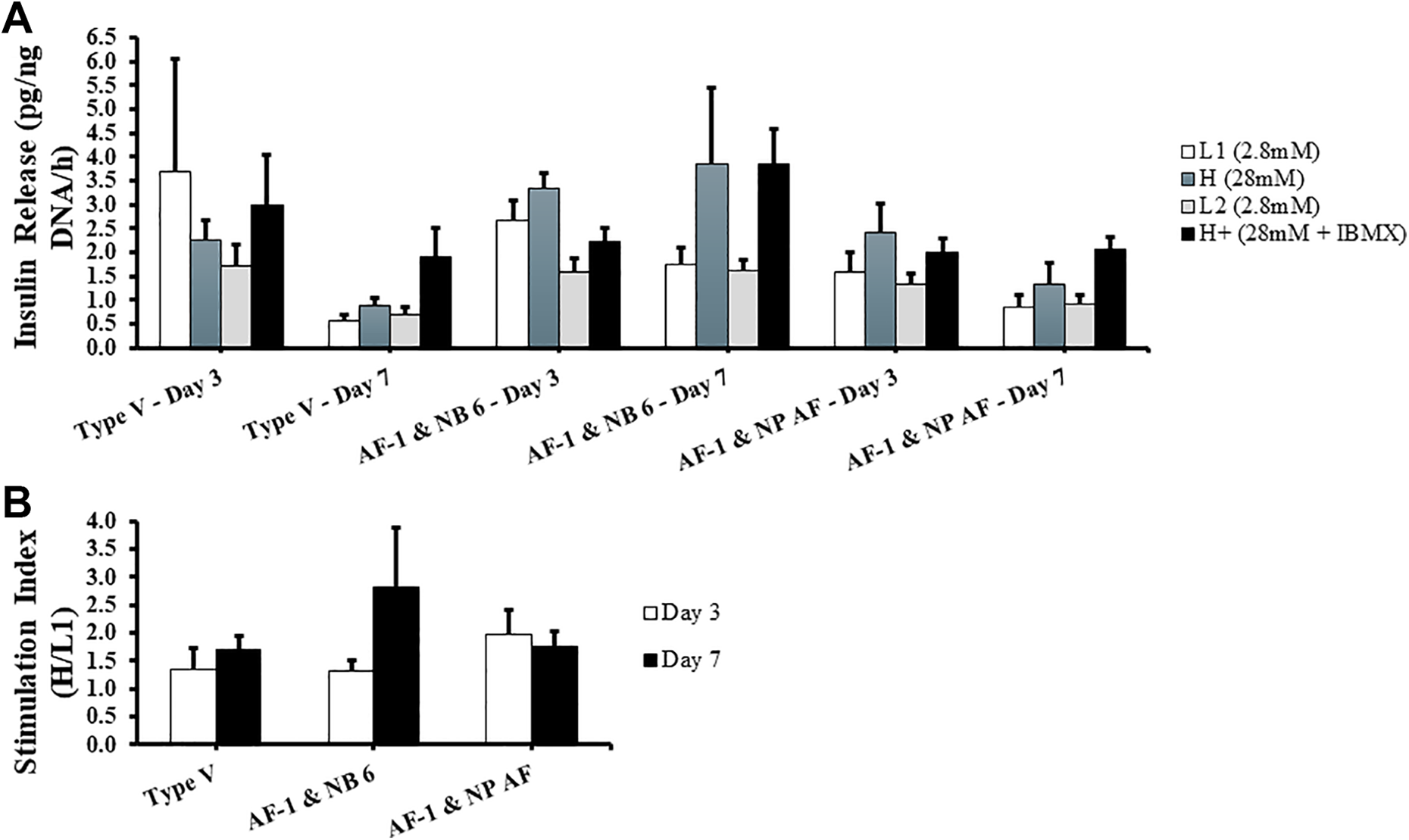
Islet function in vitro on day 3 and day 7 of culture. PPIs were ran under the glucose-stimulated insulin release assay. Islets were incubated at 37°C for 1 h in the following concentration and order of glucose medias: 2.8 mM (L1), 28 mM (H), 2.8 mM (L2), and 28 mM + IBMX (H+). Insulin released at each concentration was quantified using porcine insulin enzyme-linked immunosorbent assay and normalized to their DNA content. n = 5 for each group. Values are represented as mean ± standard error of the mean. (A) Insulin release in pg/ng DNA/h in their respective glucose concentrations. (B) Stimulation index—calculated as the amount of insulin secreted in H divided by L1.
SI was computed by dividing H by L1 and represented as a ratio. Day 3 SIs of Type V (1.35 ± 0.37), AF-1 and NB 6 (1.33 ± 0.18), and AF-1 and NP AF (1.98 ± 0.44) resulted in no notable difference; along with their day 7 SIs (1.70 ± 0.24, 2.83 ± 1.04, 1.76 ± 0.28, respectively) (Fig. 3B). AF-1 and NB 6 shows a higher SI compared to the other enzyme blends at day 7. Type V shows a slight increase in SI at day 7 compared to day 3; moreover, AF-1 and NB 6 shows a higher increase in SI at day 7 compared to day 3.
Cellular Composition of PPIs
Flow cytometry was run on dissociated single cells of the PPIs on days 3 and 7 to evaluate their cellular composition. Through this analysis we were able to quantify the major cellular composition of the islets: beta, alpha, delta, and GLUT2-positive beta cells. The percentage of beta cells in Type V (7.1% ± 2.6%), AF-1 and NB 6 (10.4% ± 1.7%), and AF-1 and NP AF (9.4% ± 1.2%) display no significant difference. However, both GMP-grade enzymes show higher levels of beta cell content (Fig. 4A). The percentage of alpha cells in Type V (9.2% ± 0.5%), AF-1 and NB 6 (6.4% ± 1.9%), and AF-1 and NP AF (8.2% ± 1.6%) display no significant difference (Fig. 4B). Both GMP-grade enzymes show lower levels of alpha cell content. The percentage of delta cells in Type V (3.2% ± 0.6%), AF-1 and NB 6 (2.9% ± 0.7%), and AF-1 and NP AF (2.9% ± 0.5%) display no significant difference (Fig. 4C). The percentage of GLUT2-positive beta cells in Type V (38.1% ± 12.8%), AF-1 and NB 6 (24.8% ± 2.6%), and AF-1 and NP AF (22.2% ± 4.5%) display no significant difference (Fig. 4D). Both GMP-grade enzymes show lower levels of GLUT2-positive beta cell content.
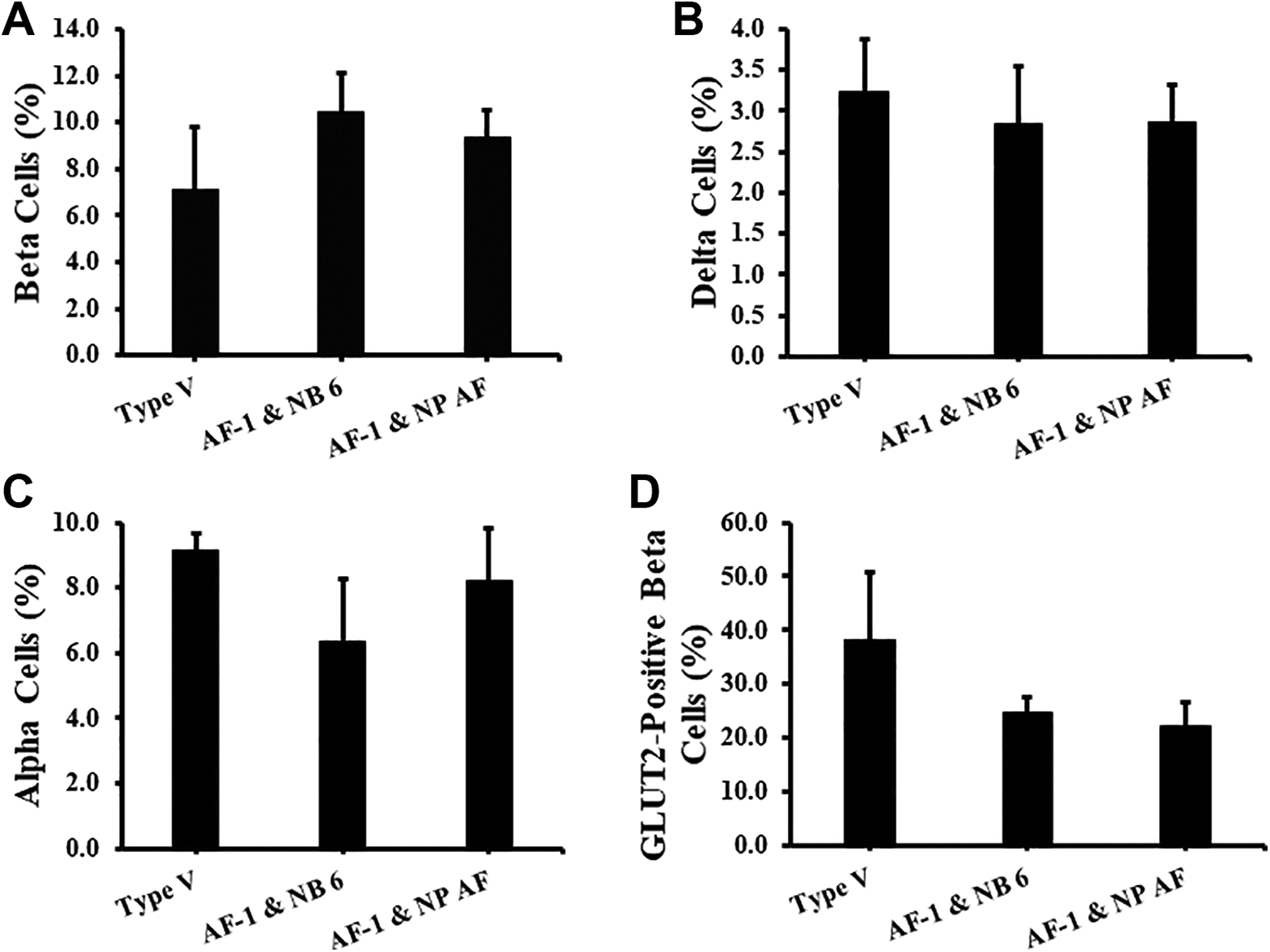
Islet cellular content and GLUT2 expression in beta cells on days 3 and 7 of culture. PPIs digested from Type V (n = 3), AF-1 and NB 6 (n = 5), and AF-1 and NP AF (n = 5) were dissociated, stained with 7-AAD viability dye, insulin, glucagon, somatostatin, and GLUT2 antibodies, and analyzed through flow cytometry. All gating of the percentage of endocrine cells and GLUT2-positive beta cells was done on live cells. Values represented as mean ± standard error of the mean. (A) The percentage of beta cells. (B) The percentage of alpha cells. (C) The percentage of delta cells. (D) The percentage of GLUT2-positive beta cells.
Immunohistochemical Analysis of Islets
In concurrence with our flow cytometry data, results from immunohistochemical analysis revealed a higher amount of insulin-positive cells per islet in AF-1 and NB 6 and AF-1 and NP AF compared to Type V (Fig. 5).

Immunohistochemical analysis of insulin-positive cells (brown) in islets on day 7. Scale: 10×. (A) Type V; (B) AF-1 and NB 6; (C) AF-1 and NP AF.
Discussion
Porcine xenotransplantations have been extensively studied to abate the limitations that human allotransplantation possess in treating T1DM. Many parameters in porcine islet isolations affect the production of successful islets in terms of yield, viability, and function 8 . Of the many parameters, pig donor age plays a role in obtaining an adequate supply of high-quality, viable islets; neonatal and adult aged pigs have been more prevalent and even shown to reverse diabetes 25,26 . Our study conveys that a scarcely investigated donor, preweaned-aged porcine, can be proficient enough for application as well, yielding a considerable amount of islets per pancreas tissue (Table 2). High yields of islets were analyzed as the only variable that correlates significantly with islet graft function in type 1 diabetic patients, proving its importance 27 . Another prominent parameter is the enzyme blend, taking a crucial role in producing large numbers of functional islets 7,28 . In this case, integrity tends to be the limiting factor. Although present in human, rat, and even adult pig isolations, analysis of optimization of enzyme blend is lacking in preweaned-aged porcine isolations 29,30,31 . The results presented reveal that consistent GMP-grade enzyme blends are capable of being as effective as crude enzymes in producing optimal PPIs.
Standard crude collagenase is a more prevailing enzyme in porcine islet isolations due to its inexpensive nature. It is characterized as a mixture of class I and class II collagenase with small amounts of clostripain, neutral protease, and tryptic activities, as is in collagenase from Clostridium histolyticum, Type V, used in this study 14 . Type V has shown to produce optimal islets with considerable yield, viability, and function despite its indistinctness 16,18,32 . On the other hand, Type V has also shown to possess wide intra-lot variation and inter-lot variation due to its undefined nature 33 . Moreover, Type V is produced under standard conditions and not under GMP compliance, which basically ensures consistent high quality due to a strictly controlled and rigorous monitored manufacturing process. With these complications, Type V is likely to be unpredictable in isolation outcomes, which can be seen in our standard error mean in both islet yield and islet function results (Table 2 and Fig. 3). Inconsistency in isolations is costly and risky, making other established alternatives more suitable 34 . It can lead to unnecessary expenses for materials or opposingly result in low yields, ultimately questioning the sustainability of the isolation process. For instance, an unstable enzyme allows for overdigestion to become more prominent, as seen in Type V’s approximately 25% to 50% decrease in IE/IC ratio (Table 2) compared to the other enzymes and its unresponsiveness in the static GSIR assay (Fig. 3), rendering futile islets. Consequently, the varying digestion efficacy associated with standard crude collagenase hinders its capabilities of gaining clinical application. An attempt to use simple filtration to reduce the inconsistency in standard Type V showed adequate and comparable results to highly purified enzyme blends 11 . Still, this filtration method needs to be more investigated; the composition of the enzyme blends itself may still be unavoidable in expediting a clinical protocol for porcine xenotransplantations.
Unlike the standard Type V, the GMP-grade enzymes are produced under controlled GMP-compliant conditions, ensuring safe and reliable products that yield high-quality islets while simultaneously providing high lot-to-lot consistency. In addition, AF-1 and NP AF are produced completely free from any animal-based components. Enzymes free from animal components have benefits of less degradation and endotoxin content 35 . Previously, highly purified enzymes have displayed high endotoxin levels, despite that, AF-1 and NB 6 and AF-1 and NP AF exhibit low endotoxin levels 11 . Endotoxin contamination of enzymes during digestion plays an important role in destruction of islets and amplification of the autoimmune response 11 . Let alone, the introduction of any potential animal-derived pathogen is excluded in AF-1, in turn increasing the likelihood of better isolation outcomes.
AF-1 is highly purified and nearly free from enzymatic activities of the following: clostripain, tryptic-like activities, and neutral protease. Having said that, a successful islet isolation requires both collagenolytic and proteolytic activity; proteolytic activity should be limited due to its opposing role in islet degradation and islet disintegration 14 . The supplementation of NB 6 and NP AF, respectively, contributes to this matter for the enzyme blends in this study. NB 6, being a crude collagenase, also contains a balanced ratio of proteolytic side activities and is made under GMP conditions. In the past, collagenase NB 1, a purified derivative of NB 6 and also a GMP-grade enzyme, has been studied and deemed successful in islet yields, purity, and function in human islet isolations 29,36 . AF-1 with NB 6 shows to be an uninvestigated, even in human isolations, novel option with potential. NP AF from Clostridium histolyticum is a highly purified proteolytic enzyme. The current standard enzyme blend for human islet isolations is collagenase from Clostridium histolyticum with a neutral protease because of its low cost, stable production, and strong digestion efficacy 37 . Previous studies in human islet isolations show that enzyme blend AF-1 supplemented with NP AF is successful and efficient, which is also seen in our results with porcine isolations 10 . However, other studies have determined that neutral protease, and even more with high concentrations of neutral protease, can become detrimental to islet survival and functional integrity during culture 38,39,40 . This can be indicative of why AF-1 and NP AF is slightly decreased in its insulin response and SI from day 3 to day 7 (Fig. 3). But, it was shown that supplementation with clostripain is an effective means to substantially reduce neutral protease activity in human isolations, providing a possibility in enhancing these enzyme blends in the future 38 . Further investigation on a definitive neutral protease and clostripain dose in combination with AF-1 needs to be accomplished.
Purified enzyme blends were known to be very costly before, deterring its application for use indirectly and directly in clinical application; however, affordable purified enzyme blends are now accessible. If cost is still concerning, manufacturing companies are able to provide research-grade enzymes that are enzymatically equivalent to their GMP grade, for initial protocol and application establishment to lower down research costs. This enzyme study provides significant results in low intra-lot variation and the basis for further analysis of low inter-lot variation. In the long run, GMP-grade enzyme blends exhibit great potential to be more cost-effective and promising for clinical application of porcine xenotransplantations.
Supplemental Material
Supplemental Material, sj-tif-1-cll-10.1177_0963689720977835 - Comparison of Islet Characterization from Use of Standard Crude Collagenase to GMP-Grade Collagenase Enzyme Blends in Preweaned Porcine Islet Isolations
Supplemental Material, sj-tif-1-cll-10.1177_0963689720977835 for Comparison of Islet Characterization from Use of Standard Crude Collagenase to GMP-Grade Collagenase Enzyme Blends in Preweaned Porcine Islet Isolations by Nicole Corrales, Soomin Park, Hien Lau, Ivana Xu, Colleen Luong, Samuel Rodriguez, Johanna Mönch, Michael Alexander and Jonathan RT Lakey in Cell Transplantation
Footnotes
Acknowledgments
The authors also acknowledge and appreciate the support from the Sue and Bill Gross Hall Stem Cell Center, a California Institute of Regenerative Medicine (CIRM)-supported facility at University of California, Irvine, the Brownstein Family Foundation, and the PADRE Foundation.
Ethical Approval
Our institution does not require ethical approval for reporting individual cases or case series.
Statement of Human and Animal Rights
All procedures in this study were conducted in accordance with the University of California Irvine, Institutional Animal Care and Use Committee, approved protocol number AUP-17-129.
Statement of Informed Consent
There are no human subjects in this article and informed consent is not applicable.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by the Juvenile Diabetes Research Foundation (JDRF Grant 2-SRA-2018-523-S-B), and the Department of Surgery, University of California, Irvine, CA, USA.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
