Abstract
In murine allogeneic transplantation models, ICOS gene-transduced bone marrow-derived mesenchymal stem cells (MSCsICOS-EGFP) were evaluated for their effects on GvHD severity and long-term survival. Lethally irradiated BALB/c or first filial generation of BALB/c and C57BL/6 (CB6F1) mice were transplanted with bone marrow cells and splenocytes from C57BL/6 mice to establish acute GvHD models. Recipient mice were injected with MSCsICOS-EGFP, MSCs, MSCsEGFP, ICOS-Ig fusion protein, MSCs + ICOS-Ig, or PBS (control group). Long-term survival, GvHD rates and severity, CD4+ T-cell apoptosis and proliferation, and Th1/Th2/ Th17 effecter cell polarization were evaluated. In the C57BL/6 → CB6F1 HSCT model, the long-term survival in the MSCICOS-EGFP group was higher than that in the GvHD group (74.29 ± 7.39% vs. 0, p < 0.01), and this survival rate was also higher than that in the MSC, ICOS-Ig, or MSC + ICOS-Ig groups (42.86 ± 8.36%, p = 0.004; 48.57 ± 8.45%, p = 0.03; or 50.43 ± 8.45% p = 0.04, respectively). The survival advantages of MSCICOS-EGFP-treated group were confirmed in the C57BL/6 → BALB/c HSCT model. In both HSCT models, the low mortality in the MSCICOS-EGFP group was associated with lower incidence and severity of acute GvHD. Treatment with MSCsICOS-EGFP induced more CD4+ T-cell apoptosis compared with that in the GvHD group. The effect on CD4+ T cells was shown as early as day 2 and maintained until day 14 (p < 0.05 on days 2, 3, 7, and 14). Furthermore, we demonstrated that MSCsICOS-EGFP were able to suppress Th1 and Th17 polarization and promote Th2 polarization on both protein expression and gene transcription levels. Higher serum levels of IL-4, IL-10, and lower levels of IFN-γ, IL-2, IL-12, and IL-17A were detected in the MSCICOS-EGFP group. The MSCsICOS-EGFP could also induce GATA-3, STAT6 expression and inhibit T-bet, STAT4, ROR-gt expression. Our results showed that injection of MSCsICOS-EGFP is a promising strategy for acute GvHD prevention and treatment. It provides synergistic benefits of MSC immune modulation and ICOS-B7h pathway blockage.
Keywords
Introduction
Despite recent advancement in preventing and treating graft-versus-host disease (GvHD) associated with allogenic hematopoietic stem cell transplantation (allo-HSCT), morbidity and mortality rates remain high (2). Currently, standard treatment of GvHD is still limited to steroids and other immunosuppressants in most transplantation centers. Even though these treatment approaches are effective to some extent, the likelihood of lethal infections may increase regardless of whether the GvHD has been controlled (36). In addition, there is no effective treatment available for severe steroid-refractory GvHD. Therefore, it is critical to explore new strategies for GvHD prevention and treatment.
Inducible costimulator (ICOS), a member of the cluster of differentiation 28 (CD28) family, plays unique roles in T-cell activation and differentiation (10). ICOS ligand [ICOSL, B7-related protein 1 (B7RP-1), or B7 homolog (B7h)] is constitutively expressed on antigen-presenting cells. The expression of B7h could be induced in some nonlymphoid tissues (including heart, lung, kidney, and testes) by tumor necrosis factor-α (TNF-α) or lipopolysaccharide, which were often produced during GvHD responses (9,37). Blockage of the ICOS-B7h pathway could reduce solid organ graft rejection, such as cardiac, liver, and pancreatic islet cells, and prolong allograft survival (7,16,30).
In our previous study, we have demonstrated that blocking the ICOS-B7h signal by ICOS-Ig fusion protein could inhibit the function of alloreactive T-lymphocytes and therefore decrease the incidence and severity of acute GvHD in a mouse model (3). Unfortunately, application of ICOS-Ig fusion protein is limited due to the amount of the fusion protein needed, in vivo variability of Ig fusion protein, and the potential immune interference that may be induced by Ig fusion protein.
Mesenchymal stem cells (MSCs) are pluripotent nonhematopoietic progenitor cells. Many studies indicated that MSCs are able to suppress the activation, proliferation, maturation, and effector functions of lymphocytes, including T cells, B cells, natural killer cells, and dendritic cells (DCs) (4,45). The beneficial effects of MSCs have been demonstrated in several studies showing that MSCs may enhance engraftment of hematopoietic stem cells, accelerate reconstitution of hematological and immune system after allo-hematopoietic stem cell transplantation (allo-HSCT), as well as migrate to inflamed tissues and participate in the tissue repair process during GvHD (5,23,25). However, even though injection of MSCs had been considered a promising treatment for GvHD, some studies showed that MSCs lost their immunosuppressive potential after allotransplantation and failed to prevent GvHD in some murine models and clinical studies (33,38). The conflicting results may be related to the timing and dosing of MSC infusion, origin of MSCs, or posttransplantation migration capability (48).
Gene-modified MSCs have been explored in some studies. They showed that interleukin (IL)-7, IL-10, and chemokine C-X-C motif receptor 4 (CXCR4) genetransduced MSCs have produced promising results in reducing severity of acute GvHD (6,24,28). In this study, ICOS-transduced MSCs were evaluated in the allotransplant murine models. We demonstrated that ICOS-transduced MSCs were able to reduce the severity of acute GvHD and increase long-term survival via combining immunomodulatory effects of MSCs and the blockage of the ICOS-B7h pathway.
Materials and Methods
Mice
Male C57BL/6 (B6; H-2b, CD45.2+) and female BALB/c (H-2d, CD45.2+) mice were purchased from Silaike company (Shanghai, China). Female CB6F1 (C57BL/6 × BALB/c, H-2b×d, CD45.2+) mice were purchased from the Animal Production Area of the Chinese Scientific Academy (Shanghai, China). Mice were 6 to 8 weeks old at the time of HSCT. All mice were housed in a specific pathogen-free condition at Animal Facilities of Second Military Medical University (SMMU, Shanghai, China). For the first 3 weeks after transplantation, all mice had access to filtered water or autoclaved hyperchlorinated drinking water. Experiments were conducted in compliance with institution and government guidelines. All animals were handled in accordance with institutional and governmental directives, and all animal experimental protocols were approved by the local authorities.
Construction of Recombinant Adenovirus
The recombinant adenovirus Ad5/F11b-ICOS-EGFP and control adenovirus Ad5/F11b-EGFP were constructed in Qian lab at the Institute of Oriental Hepatobiliary Surgery Hospital, SMMU. Basically, a murine ICOS gene (Shanghai Yiyuan Bio-Gene Technology Development Co., Ltd., ShangHai, China) was inserted into the PDC318 vector (Shanghai GeneCore BioTechnologies Co., Ltd., Shanghai, China), then cotransfected with plasmid pPE3-F11B-EGFP (Microbix Biosystems, Mississauga, ON, Canada) into 293 packaging cells (Microbix Biosystems) to construct Ad5/F11b-ICOS-EGFP. Ad5/F11b-ICOS-EGFP was then propagated in 293 cells and purified by cesium chloride (Jiangxi DongPeng New Materials Co., Ltd., XinYu, JiangXi, China) gradient centrifugation. The viral titer was 1.185 × 1010 PFU/ml by 50% tissue culture infectious dose assay. Ad5/F11b-EGFP (viral titer 3 × 109 PFU/ml) were constructed and used as a vector control.
Preparation and Transfection of Bone Marrow (BM)-Derived MSCs
BM cells of donor mice (B6) were collected by flushing femurs and tibias with Dulbecco's modified Eagle's medium (Invitrogen, Carlsbad, CA, USA) containing 10% fetal bovine serum (FBS; Invitrogen). All cells were cultured in 75-cm2 culture flasks (BD Pharmingen, San Diego, CA, USA) at a concentration of 1 × 106 cells/ml at 37°C in a humidified atmosphere with 5% CO2. After 72 h, the nonadherent cells were removed. Before further expansion and experimental use, the characteristics of MSCs, such as morphology, cell growth pattern, phenotype, and karyotype were confirmed. In addition, their differentiation potentials into adipocytes, osteoblasts, or chondrocytes were verified using standard in vitro culture conditions as described previously (4,27,31). All experiments were performed using MSCs that had no more than six passages, during which high levels of MSC markers [stem cell antigen 1-positive (Sca-1+), CD29+, CD44+, CD90.2+, fetal liver kinase-positive (Flk-1+), and CD117-, CD34-] were expressed as determined by flow cytometry (Beckton Dickson Company) analysis. MSCs were reanimated and maintained in culture for no more than 10 in vitro passages.
MSCs were cultured at a density of 2 × 104 cells/cm2. Once MSCs were completely adhered, Ad5/F11b-ICOS-EGFP was added into the medium at 600 multiplicity of infection. The medium was discarded 4 h later, and fresh medium was added. MSCs transfected with Ad5/F11b-ICOS-EGFP were called MSCsICOS-EGFP, while MSCs transfected with control vector Ad5/F11b-EGFP were named as MSCsEGFP. Expression of EGFP was confirmed in both MSCsICOS-EGFP and MSCsEGFP by inverted fluorescence microscope (Olympus, Tokyo, Japan) as early as 6 h after transfection and maintained for about 14 days in in vitro culture (Fig. 1a). ICOS and/or EGFP transfection efficiency was confirmed by flow cytometry (Fig. 1b). Transfected MSCsICOS-EGFP were highly positive for Sca-1, CD29, and CD44; moderately positive for Flk-1 and CD90.2; and negative for CD117 and CD34. Similar phenotypes were observed in MSCsEGFP and MSCs. In addition, growth pattern and differentiation potentials of MSCsICOS-EGFP and MSCsEGFP were confirmed.

MSCsICOS-EGFP attenuate the severity of GvHD and induce prolonged survival. (a) Transfection of MSCsICOS-EGFP. This is the photo observed by inverted fluorescence microscope 12 h after transfection (left: 100×; right: 200×). (b) Flow cytometry analysis shows a representative flow cytometric graph on transfection. (c) In B6 → CB6F1 HSCT, Kaplan–Meier estimates of survival rates over 120 days; four replicated experiments were combined. (d) GvHD scores in B6 → CB6F1 HSCT model. (e) In the B6 ® BALB/c HSCT model, Kaplan–Meier estimates of survival rates over 60 days; three replicated experiments were combined. (f) GvHD scores in B6 → BALB/c HSCT model. (g) Histologic analysis of tissue samples, MSCICOS-EGFP group (upper panel) and GvHD group (lower panel).
Establishment of Experimental Allogeneic Transplantation Murine Models (C57BL/6 → BALB/c and C57BL/6 → CB6F1) and Administration of MSCsICOS-EGFP
In the C57BL/6 → BALB/c experimental HSCT model, recipient mice (BALB/c) were exposed to a single dose of 600 cGy total body irradiation [TBI; cesium Ce 137 (137Ce) source], while in the C57BL/6 → CB6F1 model, recipient mice (CB6F1) were exposed to a single dose of 800 cGy TBI. BM cells were prepared from the femurs and tibiae of C57BL/6 donor mice. The cells were washed and resuspended in Dulbecco's modified Eagle's medium before injection. Donor splenocytes (SCs) were prepared with red blood cells removed by hypotonic lysis using red blood cell buffer (BD Pharmingen). BM cells and SCs from the B6 donors were resuspended and injected intravenously into the recipient animals on day 0 within 4 to 6 h after TBI. CB6F1 recipients received 6 × 107 SCs and 5 × 106 BM cells per mouse to establish a major histocompatibility complex (MHC)-haploidentical murine model (n = 35/ group). Followed by haploidentical HSCT, BABL/c recipients were transplanted with 2 × 107/mouse SCs and 5 × 106/ mouse BM cells to establish a MHC-mismatched murine model (n = 20/group) to further validate the results. In the BM groups, mice were injected intravenously only with BM cells. Mice in the control groups were injected intravenously with the sterile phosphate-buffered saline (PBS; Sigma-Aldrich, St. Louis, MO, USA) only after TBI.
GvHD mice were treated in the following groups. On day 0, mice in the MSCICOS-EGFP or MSCEGFP group were injected intravenously with MSCsICOS-EGFP or MSCsEGFP, respectively (2 × 105/mouse). Mice in the MSCs + ICOS-Ig and MSCs groups were injected intravenously with MSCs (2 × 105/mouse). On day 0, 2, 4, and 8, mice in the ICOS-Ig group and MSCs + ICOS-Ig group were injected with ICOS-Ig fusion protein intraperitoneally (100 μg/ day/mouse; R&D Systems, Minneapolis, MN, USA). Mice in the GvHD group received BM cells and SCs only after TBI.
The recipient mice were monitored daily for survival for up to 120 days in an MHC-haploidentical murine model. Body weight, GvHD incidence according to the GvHD clinical scoring system, and clinical scores of GvHD were measured weekly for up to 7 weeks. The survival advantages (up to 60 days) of MSCICOS-EGFP-treated mice and severity of acute GvHD were confirmed in the MHC-mismatched murine model. GvHD clinical scoring system sums changes in five clinical parameters: weight loss, posture (hunching), activity, fur texture, and skin integrity. A severity scale from 0 to 2 was used for each parameter with maximum index score of 10 (8).
Blockage of ICOS-B7h Pathway
DCs were prepared from CB6F1 recipient mice as described previously (40). Specifically, bone marrow cells were obtained, adjusted to 2 × 105/ml and plated on six-well plates (BD Pharmingen). Cells were cultured for up to 5 days in the presence of 10 ng/ml of mouse granulocyte macrophage colony-stimulating factor and 1 ng/ml mIL-4 (both bought from R&D Systems) at 37°C, 5% CO2. The loosely adherent clusters were used on day 6 as immature DCs. To further induce maturation, 10 ng/ml of lipopolysaccharide (Sigma-Aldrich) was added during the last 16 h. A mature DC (mDC) phenotype, that is, CD80, CD86, human leukocyte antigen-DR (Biolegend Company, San Diego, CA, USA), was used to confirm DC maturation.
In vitro MSCICOS-EGFP binding with B7h was evaluated. MSCsICOS-EGFP and mDCs were harvested and resuspensed at a concentration of 1 × 107 cells/ml in PBS. After MSCsICOS-EGFP (100 μl) were cultured with mDCs (100 μl) for 24 h at 37°C, expressions of ICOS on the MSCICOS-EGFP surface and B7h on the mDC surface were measured to confirm in vitro binding. Expression of ICOS on the MSCICOS-EGFP surface and expression of B7h on the mDC surface were evaluated by flow cytometry and used as negative controls. After MSCsICOS-EGFP (100 μl) were incubated with 5 μg mouse B7RP-1-IgG fusion protein (R&D systems) for 45 min at 37°C, expression of ICOS on the MSCICOS-EGFP surface was used as ICOS-positive control. After mDCs (100 μl) were incubated with 5 μg mouse ICOS-IgG fusion protein (R&D systems, USA) for 45 min at 37°C, expression of B7h on the mDC surface was used as B7h positive control. Rat IgG control was purchased from eBioscience (San Diego, CA, USA). MSCEGFP binding with B7h in vitro was also evaluated and used as a control.
Flow Cytometry
A total of 1 × 106 cells were resuspended in a final volume of 200 μl PBS for flow cytometric analysis. Commercial monoclonal antibodies CD4, CD8, CD25, CD29, CD34, CD44, Scal-1, CD90.2, Flk-1, CD117, ICOS, and B7RP-1 (eBioscience) were applied for analysis. Two- or three-color flow cytometry (Aria; BD Company, Franklin Lakes, NJ, USA) was performed to measure the expression of surface molecules according to standard techniques. Background staining for antibodies was performed in negative cell lines and with matched flurochrome-conjugated isotype controls. The stained cells were incubated with mAb for 20 min at 4°C, washed twice with PBS, resuspended in PBS, and analyzed on a flow cytometer with Cell Quest software (BD company).
CD4+ T-Cell Monitoring
Recipient mice were humanely exsanguinated on days 1, 2, 3, 7, and 14 after transplantation. Total numbers of spleen cells and CD4+ cells were evaluated. Apoptosis of CD4+ T cells at various time points were evaluated by flow cytometry with Fluorescein isothiocyanate-conjugated Annexin V (eBioscience) and propidium iodide (PI) staining according to manufacturer's instructions.
Cytokine Profiling
On days 7 and 14 after transplantation, blood samples from recipient mice were collected and centrifuged to obtain serum. Interferon (IFN)-γ, IL-4, IL-2, IL-10, IL-12, and IL-17A levels in serum were measured by enzyme-linked immunosorbent assay kits (eBioscience) according to manufacturer's instructions. Samples and controls were run in duplicate. The absorbance at 450 nm was measured using a Thermomax microplate reader (Bio-Rad, Hercules, CA, USA).
Histologic Analysis
On day 14 after transplantation, GvHD target organs, including liver, spleen, lung, and intestines, were obtained and formalin (The Science Company, Denver, CO, USA) preserved, paraffin embedded, sectioned, and hematoxylin and eosin stained (Nanjing Jiancheng Bioengineering Institute, NanJiang, China). Pathologic assessment of degree of inflammation was made by a pathologist who was unaware of the origin of the sections.
Quantitative Analysis of Transcription Factor Gene Expression by Real-Time Polymerase Chain Reaction (PCR)
SCs of the recipient mice were harvested at day 7 and day 14 after transplantation. Total RNA was extracted using TRIzol (Invitrogen) according to the manufacturer's instructions. The extracted RNAs with a purity range from 1.6 to 2.0 were used for cDNA transcription, which then were used as template for real-time quantitative PCR (7300 Fast Real-Time PCR system; Applied Biosystems, Foster City, CA, USA). Primers were obtained from Sangon Biotech Co. Ltd. (Shanghai, China): T-box expressed in T cells (T-bet)-For: TGGTTCCGGCCCATGCGAAC, T-bet-Rev: CCCAGCGGGAGAGGAGCTGT; signal transducer and activator of transcription 4 (STAT4)-For: GTG GCCCGCTCCACAATGGG, STAT4-Rev: GGCCTCTG AGGGTGCGTTGG; STAT6-For: GTGGCTGAGCGA GTGCCCTG, STAT6-Rev: GTGAAGCCTCGGCCCA GCAG; guanine–adenine–thymine-adenine-binding protein 3 (GATA3)-For: CCTGAGCAGCCACCACACCG, GATA3-Rev: CAGCGACGGGTCTGGGGAGA; retinoic acid receptor-related orphan receptor γ t isoform (ROR-γt)-For, TGCAAGACTCATCGACAAGG, ROR-γt-Rev: AG GGGATTCAACATCAGTGC; β-actin-For: CCACCCCG GGGAAGGTGACA, β-actin-Rev: CCAGCCCTGGCTG CCTCAAC. PCR conditions were: activation 10 min at 95°C, annealing 5 s at 54°C, elongation 15 s at 72°C, 40 cycles. The expression levels were calculated based on the real-time PCR method. The copy number for each target gene was normalized to the copy number for β-actin using the 2-ΔΔCT method (21).
Mixed Lymphocyte Reaction (MLR)
CD4+ and CD8+ T cells from recipient mice on day 7 were purified and stimulated with concanavalin A (ConA; Sigma-Aldrich). Mouse SCs were prepared as previously described. They were then resuspended in 5 ml of Roswell Park Memorial Institute (RPMI) 1640 media (Sigma-Aldrich), counted, and adjusted to a concentration of 2.5 × 106 cells/ml. Freshly thawed ConA was added to the responder cells at a concentration of 2.5 mg/ml. Cells were cultured for 48 h before testing.
For MLR cultures, CD4+ and CD8+ T cells (2 × 104 cells) obtained from recipient mice on day 7 were used as responder T cells using CD4+ T- or CD8+ T-positive selection kit (Stem Cell, Vancouver, BC, Canada). Responder cells were incubated with MSCsICOS-EGFP (1 × 104 cells), MSCs (1 × 104 cells), ICOS-Ig (5 μg), or B7h-Ig (5 μg), respectively, in 96-well round-bottom plates (BD Pharmingen). Irradiated mDCs from recipient mice (4 × 104) [γ-irradiation, 30 Gy cyanocobalamin cobalt Co 60 (60Co) source; the Second Military Medical University, Shanghai, China] were used as stimulator cells. All cells were cocultured in 96-well round-bottom plates at a final volume of 300 μl/ well in the presence of RPMI-1640 containing 10% FBS. Responder CD4+ or CD8+ T cells only were used as controls. After 3 days of coculturing, T-cell proliferation was evaluated by a cell counting kit-8 (Dojindo, Shanghai, China) assay. The value of optical density (OD) in each well was measured with a Thermomax microplate reader at 450 nm. Stimulation indices were calculated as follows: (OD value of cocultures - OD value of control medium)/ (OD value of control T - OD value of control medium). Supernatants were collected and tested for cytokine profiles. The experiments were repeated three times.
Statistical Analysis
Survivals in different groups were evaluated using Kaplan–Meier estimated event rates (log rank test) with SPSS 11.5 software (IBM, Armonk, NY, USA). The significance of observed differences was determined by ANOVA, and Bonferroni's correction was employed for multiple comparisons. Data were shown as mean ± standard deviation for separate experiments. A value of p < 0.05 was considered statistically significant.
Results
MSCsICOS-EGFP Attenuate GvHD Severity and Improve Long-Term Survival
In our MHC-haploidentical murine model (B6 → CB6F1), long-term survival and GvHD were monitored in recipient mice for up to 120 days. Our results showed that administration of MSCs or ICOS-Ig could significantly improve the long-term survival rates com pared to the GvHD group (42.86 ± 8.36% vs. 0, p < 0.01, 48.57 ± 8.45% vs. 0, p < 0.01, respectively). Administration of MSCs and ICOS-Ig simultaneously could improve the long-term survival slightly more to 50.43 ± 8.45%, p < 0.01. The highest survival benefit was achieved when MSCsICOS-EGFP were administered (74.29 ± 7.39% vs. 0, p < 0.01). The long-term survival in the MSCICOS-EGFP mice was significantly higher than that in the GvHD mice, but there were also significant improvements compared with the MSC group (42.86 ± 8.36%, p = 0.0079), the ICOS-Ig group (48.57 ± 8.45%), and the MSC + ICOS-Ig group (50.43 ± 8.45%) (Fig. 1c). Mice in the BM groups had the highest survival rate in all groups (95.62 ± 6.41%). All mice in the control groups died within 14 days after transplantation.
In terms of GvHD severity, our results showed that MSCICOS-EGFP treatment was associated with the lowest incidence of acute GvHD (20.31%) (Table 1). Similarly, clinical GvHD score in the MSCICOS-EGFP group was significantly lower than that in the GvHD group (p < 0.01) (Table 1). The differences between MSCICOS-EGFP and MSC, ICOS-Ig, or MSC + ICOS-Ig groups were also statistically significant (p = 0.04, p = 0.03, or p = 0.03, respectively) (Fig. 1d).
Acute GvHD Rate and Clinical Scores in MHC-Haploidentical Model

Versus GvHD group (n = 35).
aGvHD: acute graft-versus-host disease; MHC: major histocompatibility complex; MSCs: mesenchmyal stem cells; ICOS: inducible costimulator; EGFP: enhanced green fluorescent protein; Ig: immunoglobulin.
The survival data of MSCICOS-EGFP administration in our MHC-haploidentical model were promising. However, chances of model-specific findings cannot be ruled out. Therefore, we validated the study results in another BM transplant (BMT) model. Our previous studies have shown that MSCs (1 or 5 × 105/mouse) or ICOS-Ig fusion protein could reduce the severity of acute GvHD after MHC class I- and II-disparate allo-HSCT (B6 → BABL/c) (4). Here we examined whether MSCsICOS-EGFP can attenuate the severity of acute GvHD in the same model (B6 → BALB/c) and whether the benefits from MSCsICOS-EGFP were superior to that from MSCs or ICOS-Ig fusion protein alone. Figure 1e shows survival curves of mice in the MHC-mismatched model. As we expected, survival rates in the MSCICOS-EGFP, MSC, MSCEGFP, and ICOS-Ig groups were significantly higher than that in the GvHD group (50.00 ± 11.18% vs. 0, p < 0.01; 20.00 ± 8.94% vs. 0, p = 0.0002; 15.00 ± 7.98% vs. 0, p = 0.0042; 25.00 ± 9.68% vs. 0, p = 0.0004; n = 20). Meanwhile, the survival rate in the MSCICOS-EGFP group was also higher versus the MSC, MSCEGFP, or ICOS-Ig group (p = 0.0364, 0.0204, or 0.046, respectively, n = 20). Similarly, clinical GvHD score in the MSCICOS-EGFP group was significantly lower than that in the GvHD, MSC, MSCEGFP, or ICOS-Ig group (p = 0.01, 0.04, 0.04, and 0.04, respectively, n = 20) (Table 2). Lower mortality in the MSCICOS-EGFP group was associated with lower clinical GvHD score (Fig. 1f), implying the survival benefit was related to the reduction of GvHD severity.
Acute GvHD Rate and Clinical Scores in MHC-Mismatched Model

Versus GvHD group (n = 20).
Since most induced acute GvHD (aGvHD) occurs within the first 14 days after HSCT, target organ samples, including liver, spleen, lung, and intestines, of recipient mice were obtained on day 14 for histologic analysis (11). Our results showed that in the GvHD group, severe inflammation and tissue injury were present in multiple organs on day 14. However, there was little evidence showing lymphocyte infiltration and tissue injury in the GvHD target organs in the MSCICOS-EGFP group (Fig. 1g).
These results suggested that an early injection of MSCs, ICOS-Ig, or MSCsICOS-EGFP after allo-transplant could provide protective effects in both MHC-mismatched and haploidentical murine GvHD models. The best protective benefit was achieved when MSCsICOS-EGFP were administered.
MSCsICOS-EGFP Competitively Bind to B7h Molecules on mDCs
The ICOS-B7h signaling pathway plays an important role in T-cell activation. To confirm that ICOS expressed on MSCsICOS-EGFP is able to bind to B7h molecules on mDCs and therefore block the signal transduction, in vitro binding was analyzed. In our study, while MSCsICOS-EGFP and mDCs were cultured separately, the expression of ICOS on MSCsICOS-EGFP and B7h on mDCs was 91.64% and 69.73%, respectively (Fig. 2a). Once B7h-IgG fusion protein occupied binding site, the expression of ICOS on the MSCsICOS-EGFP was reduced to 5.79%. The same observation was found with the mDCs. After mDCs were incubated with ICOS fusion protein, surface expression of B7h was reduced to 4.88% (Fig. 2b). After MSCsICOS-EGFP and mDCs were cocultured for 24 h, expression of ICOS on the MSCsICOS-EGFP and B7h on the mDCs was 6.42% and 3.91%, respectively (Fig. 2c). This demonstrated that ICOS on the MSCsICOS-EGFP was able to competitively bind to B7h on the mDC and therefore block signal transduction through ICOS-B7h pathway. Similar observation was not found in the MSCsEGFP using the same assay procedure (Fig. 2).
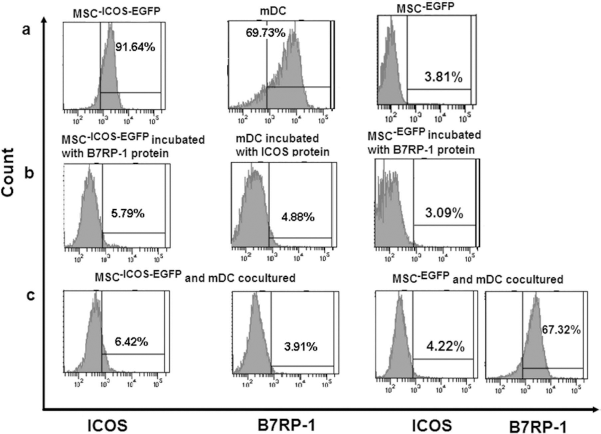
MSCsICOS-EGFP or MSCsEGFP competitively bind to B7RP-1 molecules in vitro. (a) Expression of ICOS on MSCsICOS-EGFP or MSCsEGFP and B7RP-1 on mDCs when cultured separately. (b) Expression of ICOS on MSCsICOS-EGFP or MSCsEGFP when incubated with B7RP-1 fusion protein and the expression of B7RP-1 on mDCs when incubated with ICOS fusion protein. (c) Expression of ICOS on MSCsICOS-EGFP or MSCsEGFP and B7RP-1 on mDCs after coculture for 24 h.
MSC-Mediated ICOS Pathway Blockage Induces CD4+ T-Cell Apoptosis in the Early Development of GvHD
In our study, we monitored how administration of MSCs as well as ICOS-B7h pathway blockage would affect the CD4+ T cells in the early development of GvHD.
Administration of MSCs was not able to induce CD4+ cell apoptosis compared to the GvHD group on day 3 (4.58 × 105 vs. 3.55 × 105, p = 0.07) or on day 7 (4.8 × 105 vs. 3.86 × 105, p = 0.05). It was, however, able to induce CD4+ cell apoptosis on day 14 (5.47 × 105 vs. 3.89 × 105, p = 0.046). On the other hand, administration of ICOS-Ig could reduce CD4+ T-cell counts as early as day 2 compared to the GvHD group (4.46 × 106 vs. 2.78 × 106, p = 0.041). The effectiveness of ICOS-Ig on CD4+ cells was maintained until day 7 (4.91 × 106 vs. 3.86 × 106, p = 0.045). However, the differences between the ICOS-Ig and the GvHD groups were not statistically significant at day 14. We also found that administration of MSCsICOS-EGFP was able to induce CD4+ cell apoptosis compared to the GvHD group from day 2 to day 14 (p < 0.05). In addition, it was able to induce more CD4+ cell apoptosis versus the MSC group on day 3 (6.17 × 105 vs. 4.58 × 105, p = 0.03) and day 7 (5.92 × 105 vs. 4.8 × 105, p = 0.04), and versus the ICOS-Ig group on day 14 (5.33 × 105 vs. 3.96 × 105, p = 0.03) (Fig. 3a).

MSCICOS-EGFP-induced CD4+ T-cell apoptosis in the early development of GvHD. (a, b) On days 2, 3, 7, and 14 post-BMT, spleen cells were collected from recipients in each group, reacted with anti-CD4 antibodies and PI, and were analyzed by flow cytometry. (a) Apoptosis of CD4+ T cells (n = 5/group). (b) The absolute counts of CD4+ T cells (n = 5/group). *p < 0.05 versus GvHD group; †p < 0.05 versus MSCICOS-EGFP group. Data are shown as mean ± standard deviation from three independent experiments performed in triplicate.
When MSCsICOS-EGFP were administered, the absolute counts of CD4+ T cells were significantly lower than that in the GvHD group from day 2 to 14 (p < 0.01). In addition, absolute CD4+ T-cell counts in the MSCICOS-EGFP-treated mice were significantly lower versus the MSCs group on days 3 and day 7 (3.19 × 106 vs. 4.21 × 106 on day 3, p = 0.01; 2.49 × 106 vs. 3.77 × 106 on day 7, p = 0.01) and versus the ICOS-Ig group on day 14 (2.51 × 106 vs. 3.26 × 106,p = 0.02) (Fig. 3b).
There was no significant difference in absolute counts or apoptosis of CD8+ T cells between treatment groups up to day 28 (data not shown, p > 0.05).
MSCsICOS-EGFP Modulate T-Helper Cell Subsets
Since both immunosuppressive MSC injection and ICOS-B7h pathway blockage could affect helper T-cell polarization, it is interesting to learn how effector T-helper cells were modulated after MSCICOS-EGFP injection and how they drive the evolving types of immune responses.
In our study, Th1 (IFN-γ, IL-2, and IL-12), Th2 (IL-4 and IL-10), as well as Th17 (IL-17A) cytokines from the recipients' serum were monitored on days 7 and 14. In addition, the expression of Th1 (T-bet and STAT4), Th2 (GATA-3 and STAT6), and Th17 (ROR-γt), specific transcription factors in recipients' SCs, was evaluated. The results showed that in the MSCICOS-EGFP, MSC, and ICOS-Ig groups, production of Th1 cytokines (IFN-γ, IL-2, IL-12) and Th17 cytokine (IL-17A) was significantly suppressed, while production of Th2 cytokines (IL-4 and IL-10) was significantly increased compared to the GvHD group on day 7 (p < 0.05). In addition, injection of MSCsICOS-EGFP could further increase the production of Th2 cytokines (IL-4 and IL-10) compared to the MSC or ICOS-Ig group (p < 0.05) (Fig. 4a). Similar results were found on day 14 as well (data not shown).
When looking at gene transcription factor level, compared to the GvHD group, expression of Th1 and Th17 transcription factors (T-bet, STAT4, and ROR-γt) was significantly suppressed in the MSC-, ICOS-Ig-, and MSCICOS-EGFP-treated mice on day 7 (p < 0.05). On the other hand, the expression of Th2 transcription factors (GATA-3 and STAT6) was significantly increased in the MSC, ICOS-Ig, and MSCICOS-EGFP groups on day 7 (p < 0.05). Furthermore, the expression of Th2 transcription factors (GATA-3 and STAT6) was statistically significant between the MSCsICOS-EGFP and MSCs or ICOS-Ig (p < 0.05) (Fig. 4b). Similar results were found on day 14 (data not shown). These results indicated that starting 7 days after transplantation, MSCsICOS-EGFP were able to suppress Th1 and Th17 differentiation and promote Th2 differentiation in both protein expression and gene transcription levels.
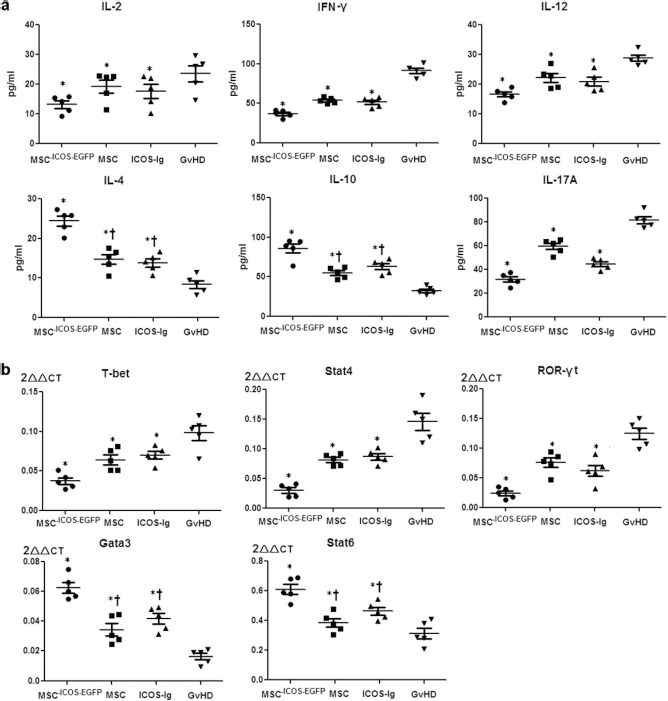
MSCsICOS-EGFP modulate T-helper cell subsets. (a) Th1 cytokine (IFN-γ, IL-2, IL-12), Th17 cytokine (IL-17A), and Th2 cytokine (IL-4 and IL-10) expression on day 7 in recipient mice in different treatment groups (n = 5/group). (b) Th1/Th17 (T-bet, STAT4, and ROR-γt), and Th2 (GATA-3 and STAT6) transcription factor expression in recipient mice on day 7 (n = 5/group). Shown is gene expression (2-ΔΔCT) of transcription factors. *p < 0.05 versus GvHD group; †p < 0.05 versus MSCICOS-EGFP group.
Immunosuppressive Function of MSCsICOS-EGFP
In our study, administration of MSCs alone was not able to suppress ConA-induced CD4+ and CD8+ T-cell proliferation. Similar results were observed when ICOS-Ig was added. However, administration of MSCsICOS-EGFP could significantly suppress CD4+ and CD8+ T-cell proliferation compared to the GvHD group (CD4+ SI: 26.45 vs. 46.33, p = 0.02; CD8+ SI: 24.86 vs. 48.72, p = 0.02). Furthermore, the differences between the MSCICOS-EGFP and MSC or ICOS-IG groups were statistically significant in CD4+ suppression (p = 0.03 and p = 0.02, respectively) (Fig. 5a). The difference of CD8+ suppression between the MSCICOS-EGFP and ICOS-Ig groups was statistically significant (p = 0.04), while there was no difference between the MSCICOS-EGFP and MSC groups (p = 0.08) (Fig. 5b).

Immunosuppressive function of MSCsICOS-EGFP in MLRs. (a) The ConA-induced CD4+ T-cell proliferation. (b) The ConA-induced CD8+ T-cell proliferation. (c) Responder CD4+ T-cell proliferation in the MLR. (d) Responder CD4+ T-cell proliferation with MSCsICOS-EGFP, anti-ICOS-mAb, anti B7h-mAb, and MSCsICOS-EGFP + anti-B7h-mAb. (e) IFN-γ, IL-2, IL-4, and IL-10 expression in the MLR supernatant. *p < 0.05 versus CD4+/CD8+/CD4+ + DC group (control group); †p < 0.05 versus MSCICOS-EGFP group; ‡p < 0.05 versus anti-ICOS-mAb. Data are shown as mean ± standard deviation from five independent experiments performed in triplicate.
The immunosuppression function of MSCsICOS-EGFP was further confirmed in the MLR assay. We found that administration of MSCs alone did not affect CD4+ T-cell proliferation (SI: 58.93 vs. 69.12, p = 0.06). However, coculturing with MSCsICOS-EGFP, ICOS-Ig, or B7h-Ig could significantly suppress responder CD4+ T-cell proliferation (SI: 32.16, 43.81, or 44.76 vs. 69.12, p = 0.0039, 0.0103, 0.0128, respectively). Furthermore, administration of MSCsICOS-EGFP could provide a significantly higher suppressive effect than administration of MSCs or B7h-Ig (p = 0.002 and p = 0.01, respectively) (Fig. 5c).
Since inhibition of the ICOS-B7h pathway could lead to suppression of CD4+ cell proliferation, we explored how blocking either ICOS or B7h would contribute to the immunosuppressive effect. Interestingly, we found that immunosuppression function was more significant with anti-B7h mAb than with anti-ICOS mAb (SI: 23.47 vs. 38.16, p = 0.04). More interestingly, when we added MSCsICOS-EGFP to anti-B7h-mAb in the MLRs, the suppression of CD4+ T-cell proliferation was even higher than that with MSCsICOS-EGFP alone (SI: 18.26 vs. 30.11, p = 0.02) (Fig. 5d).
We did not detect any significant inhibition of T-cell receptor-independent CD8+ T-cell proliferation by adding MSCsICOS-EGFP, MSCs, or ICOS/B7h-Ig fusion protein (data not shown). In addition, by measuring cytokines from the MLR supernatant, we further confirmed that Th1 cytokine (IFN-γ, IL-2) production was suppressed, and Th2 cytokine (IL-4, IL-10) production was induced with administration of MSCsICOS-EGFP (Fig. 5e).
Discussion
Immune modulatory effects of MSCs on cellular immunity have been shown in various studies on different T-cell effector subpopulations (4,5,23,25,33,38). In addition, blocking ICOS-B7h may provide a similar immunemodulatory function (45). Theoretically, combination of MSC administration and ICOS blockade could enhance the immunosuppressive effects. In addition, MSCs can be used as a vehicle to deliver ICOS into GvHD target organs and therefore enhance the expression of ICOS in local tissues. In this study, we explored whether combining ICOS-B7h pathway blockage and MSC injection could provide synergistic immunosuppressive results in GvHD models and, if so, what mechanisms are associated with said immune-modulatory function.
We demonstrated for the first time that MSCICOS-EGFP administration could attenuate GvHD severity and improve long-term survival in both MHC-haploidentical and MHC-mismatched murine models. Administration of MSCsICOS-EGFP could induce CD4+ T-cell apoptosis, inhibit CD4+ T-cell proliferation, suppress Th1/Th17 polarization, and promote Th2 polarization.
In acute GvHD, CD4+ T-cell activation and differentiation into effector cells is one of the most essential processes (35). In our study, MSC treatment alone was not able to induce CD4+ apoptosis on days 3 and 7. It was able to induce CD4+ T-cell apoptosis on day 14. On the other hand, administration of ICOS-Ig could reduce CD4+ T-cell count as early as day 2 yet lost the inhibition capability on day 14. When MSC immune modulation and ICOS pathway blockage were combined, the decrease in CD4+ T cells in recipient mice was detected as early as day 2, and the reduction was maintained during our monitoring period. It is known that the early phase, especially the first 3 days after transplantation, is very important for acute GvHD control. We believe early CD4+ T-cell suppression by MSCsICOS-EGFP may be essential for GvHD prevention.
Recently, a dominant role of B7h over ICOS was demonstrated in a mouse model using anti-ICOS mAb or anti-B7h mAb (39). Similarly, in our study, B7h blockage was more potent than ICOS blockage on inhibition of CD4+ T-cell proliferation. When MSCsICOS-EGFP were added to anti-B7h in the MLRs, the suppression of CD4+ T cells was further enhanced.
Acute GvHD is considered to be mainly mediated by the Th1 immune response (22). However, there are studies showing that adding Th2 cells to donor SCs may mitigate acute GvHD with significant attenuation of GvHD disorder in the GI tract and liver (12,13). Many studies (1,17,18,26,34,46) have explored roles of costimulatory signal played in the polarization of helper T-cell subsets. Again, the results are not conclusive. Polarization of helper T-cell function may be related to different expression levels of costimulatory receptors.
B7h plays an important role in controlling specific aspects of T-cell activation, differentiation, and function. It can enhance T-cell secretion of IFN-γ and IL-10 but not IL-2 (14,29). Specifically, Hubbard et al. demonstrated that deficiency of ICOS signal by gene knockout led to decreased serum IFN-γ levels and increased IL-4 and IL-10 levels in two GvHD models, suggesting that alloreactive ICOS-/- T cells were skewed toward Th2 differentiation (18). Furthermore, blockage of ICOS/ B7h interaction by anti-ICOS mAb could significantly decrease IFN-γ, IL-17, and TNF-α production (42). Similar T-effecter cell polarization was reported with administration of MSCs. Aggarwal et al. demonstrated that human MSCs could decrease IFN-γ production in Th1 cells and increase secretion of IL-4 in Th2 cells (1).
In our study, MSCsICOS-EGFP regulated the balance of Th1/Th2/Th17 manifested by elevated levels of IL-4 and IL-10, and decreased levels of IL-2, IFN-γ, and IL-12 after transplantation. Similar results were found in MLR supernatants. For transcription factors related to Th differentiation, the results showed decreased expression levels of T-bet and STAT4, which are related to Th1 differentiation, and increased expression levels of GATA3 and STAT6, which are related to Th2 differentiation. These results suggested that MSCsICOS-EGFP induced Th2 deviation by modulating expression of Th1/Th2-associated cytokines and transcription factors.
Th17 cells are a newly identified T-cell lineage that secretes the cytokines IL-17A, IL-17F, IL-21, and IL-22. Its role in acute GvHD is still controversial (19,47). It is reported that ICOS was critical for the differentiation and expansion of human Th17 cells (15,32). Rafei et al. showed that MSCs could ameliorate experimental autoimmune encephalomyelitis by inhibiting Th17 T cells with reduced plasma levels of IL-17 (34). However, Guo et al. found that fetal BM-derived MSCs promoted the expansion of human Th17 cells in vitro (17). It is possible that the interactions of MSCs and Th17 cells are different in vivo and ex vivo, or the inconclusive results were due to different models utilized. In our study, we found a significant decrease in serum IL-17A in MSCICOS-EGFP recipients. We believe that IL-17A might aggravate acute GvHD in this model. Therefore, decreased Th17 polarization may contribute to GvHD alleviation in our study.
In summary, our study demonstrated for the first time that MSCsICOS-EGFP showed protective effects on acute GvHD. Administration of MSCsICOS-EGFP could attenuate GvHD severity and prolong long-term survival. The treatment effects were superior to MSCs or ICOS-Ig fusion protein administration. We believe this protective effect was realized due to MSCICOS-EGFP-mediated ICOS-B7h pathway blockage. Administration of MSCsICOS-EGFP could induce CD4+ T-cell apoptosis and inhibit CD4+ T-cell proliferation at as early as day 2, and the effects could be maintained until day 14 posttransplantation. Importantly, MSCICOS-EGFP-mediated blockage of ICOS/B7h interactions could induce Th2 polarization and suppresses Th1/ Th17 differentiation (Fig. 6).

Roles of MSCsICOS-EGFP on GvHD induced by CD4+ T cells. (a) CD28 and ICOS signaling positively regulates T-cell responses to alloantigens and supports GvHD development. (b) MSCs are able to modulate the immune system and suppress the activation, proliferation, maturation, and effector functions of CD4+ T cells by suppressing Th1 and Th17 polarization and promoting Th2 polarization. (c) ICOS-IgG competitively binds to B7h molecules on mDCs and induces the suppression of CD4+ T cells by suppressing Th1 and Th17 polarization and promoting Th2 polarization. (d) MSCsICOS-EGFP are able to provide the synergistic benefits of MSCs and ICOS-IgG. They competitively bind to B7h molecules on mDCs and induce the suppression of CD4+ T cells by suppressing Th1 and Th17 polarization and promoting Th2 polarization. APC, antigen-presenting cell; TCR, T-cell receptor.
Up to now, adenovirus vectors have been widely used for gene transfer, both in experimental research and in clinical trials (20,41), with the characteristics of highly efficient delivering of the target gene without potential insertion gene mutation. Here we used a chimeric adenovirus Ad5/F11 that can effectively transfer the ICOS gene, mostly to provide an efficient and safe approach for further clinical application. The higher transfection efficiency and no insertion mutation characteristics of this recombinant Ad make the carrier MSCs highly express the target gene ICOS in a certain time after in vivo administration, especially within the first 14 days. Administration of Ad5/F11-transfected MSCICOS-EGFP block the initiation and development of aGvHD at an early stage without a long-term security concern. This unique feature would perfectly meet our aims for the prevention and treatment of aGvHD.
In addition to its effects on aGvHD, interestingly, it was shown that ICOS ligand blockage may enhance cytotoxicity of T cells on hematologic neoplasm cells, thus may significantly enhance graft-versus-leukemia (GvL) effects and therefore prolong survival of leukemia mice after allogeneic BMT (43,44). Therefore, we hope administration of MSCsICOS-EGFP may prevent acute GvHD without compromising GVL effects. More studies are warranted before initiation of MSCICOS-EGFP clinical trials.
Footnotes
Acknowledgments
The authors thank Dr. Min Yu for her valuable comments and assistance in editing and proofreading this article and Dr. Qijun Qian at the Institute of Oriental Hepatobiliary Surgery Hospital, SMMU for assistance in making adenovirus vectors. This work was supported by National Natural Science Foundation of China (NSFC; 30871100, 81090413, 81270638, 81000229), D.Y. was sponsored by Stem Cells and Medical Research Innovation Programs Foundation
