Abstract
Obtaining viable islets is a crucial step for successful islet transplantation. Adenosine triphosphate (ATP) is a marker of cell viability. However, little is known about any changes in the energy status of the tissues that are being digested during the digestion phase. We herein examined whether the ATP content in serially digested pancreatic tissue samples could be specific objective parameters that signal the optimal point to stop the digestion process. We obtained partial pancreata (body to tail) from 4- to 5-year-old pigs from a slaughterhouse. The tissue samples were preserved in M-Kyoto solution for less than 3 h. They were digested using an automated enzymatic and mechanical dissociation system at 37°C for 90 min following intraductal injection of Liberase HI. Samples were collected from the digestive circuit every 5 or 10 min to determine the ATP level, total adenine nucleotide (TAN) level, islet count (count/g), and yield of islet equivalent (IEQ) in the serial digestive fluids. The ATP and TAN levels, IEQ and islet count were increased and then decreased during digestion process. The profile of these parameters differed from case to case. However, when ATP changing ratio (respective value/precedent value) was compared with IEQ changing ratio, a greater than threefold increase in the ATP changing ratio followed by an increase in the islet count changing ratio within 5 min was consistently observed, indicating the optimal time to stop the digestion. The ATP levels of the handpicked islets in the digested samples were lower in the overdigested phase in comparison to those in the earlier digested phase. These results indicate that the ATP level in digested fluid could be an effective indicator to estimate the viability of cells as well as determine the optimal time to terminate the digestion process in order to obtain viable islets.
Introduction
During the past few decades, pancreatic islet transplantation has become a treatment option for patients with type 1 diabetes mellitus (7,25). However, it is still difficult to recover the entire quantity of islets contained in a pancreas (16). Given that islet isolation is crucial for the success of this transplantation procedure, many attempts to obtain more viable islets have been reported in the literature (1,12,18). In rodent models, collagenase digestion can be performed for a specific period of time to obtain a peak islet yield; however, in humans, the timing of dilution is empirically determined by human expertise and experience (1,16–18). If the timing is delayed, then poor results are inevitable. Various factors can affect the quantity and quality of islets during the digestion phase (17).
The adenosine content is known to reflect viability and is considered useful for outcome prediction in islet transplantation (5,6). We previously reported a new method for the determination of adenosine levels for isolated islets (8,9). This methodology, using the bioluminescent enzymatic cycling assay, produces rapid measurements with only a small sample volume (8,9), thus enabling us to determine the adenosine levels from a small volume of samples collected during the collagenase digestion phase of islet isolation.
The collagenase digestion phase results in the release of endogenous pancreatic enzymes from the exocrine tissues (3,15,20–23). A strong increase in such enzyme activity involved in the digestion process has been shown to be detrimental to the islet isolation process (3,15,20–23). However, little is known about any changes in the energy status of the tissues that are being digested during the digestion phase.
The goal of this study was to examine the serial changes in the energy status of digesting fluid containing exocrine and endocrine cells of the pancreas in relation to the islet count and yield. To address this issue, pancreatic tissue samples from retired breeder pigs were subjected to continuous mechanical and enzymatic digestion using a Ricordi chamber with Liberase-HI until all pancreatic tissue disappeared (this process is termed the “overdigestion model”). Using this model, we then tried to determine the specific objective parameters that signal the optimal point to stop the digestion process in relation to the adenosine triphosphate (ATP) content of the digested fluid, islet count, and islet yield.
Materials and Methods
Animals and Isolation Methods
Partial pancreata (body to tail) from retired breeder pigs were obtained from a local slaughterhouse and preserved using cold M-Kyoto solution. M-Kyoto solution is an extracellular-type organ preservation solution developed at Kyoto University (19). It contains the cytoprotectives disaccharide, trehalose, and ulinastatin (Mochida Pharmaceutical. Co. Ltd., Tokyo, Japan). Trehalose has a cytoprotective effect against stress, and ulinastatin inhibits trypsin. All the pancreata were procured within 17 min of warm ischemic time (15.4 ± 1.1 min). The partial pancreata were carefully handled and a cannula was inserted into the main pancreatic duct and fixed with a purse suture. Thereafter, 0.5 ml/g of cold M-Kyoto solution was carefully infused into the pancreatic duct (21). The pancreas was preserved in cold M-Kyoto solution and transported to our laboratory within 3 h. In the laboratory, the connective tissue surrounding the pancreas was removed and the pancreas was distended for 15 min with cold Hank's balanced salt solution (HBSS) (Nissui Pharmaceutical Co. Ltd., Tokyo, Japan) containing 0.5 mg/ml of collagenase (Liberase HI; Roche Diagnostics Corporation, Indianapolis, IN, USA). After distention, the pancreas was cut into seven pieces and then they were placed in a Ricordi chamber. The pieces were subsequently digested using an enzymatic and mechanical dissociation system at 37°C for 60–90 min until all pancreatic tissue disappeared. The mechanical dissociation procedures were performed by the same one person in cases 1 to 4, and case 5 was performed by another.
We took samples from the digestive circuit every 5 or 10 min. We analyzed the samples to determine the values of ATP, total adenine nucleotides (TAN), islet equivalent (IEQ), an index based on the size of islets (1 IEQ equals an islet of 150 μm diameter), and islet yield in serial digestive fluids. For ATP and TAN measurements, we took 1 ml of digestive fluid from the digestive circuit and immediately treated it with 100 μl of 5 N perchloric acid (HClO4) to inactivate various enzymes (including ATPase) before measurement. The fluid was frozen at −80°C until it was assayed.
Measurement of Adenosine Nucleotides
We performed a bioluminescent enzymatic cycling assay using synthetic firefly luciferase, pyruvate kinase (PK) and pyruvate orthophosphate dikinase (PPDK) to measure the levels of ATP and TAN in the samples (25). This method makes it possible to obtain results quickly and it also has a high sensitivity for detecting the ATP content in even one islet cell (8,9). We mixed 20 μl of a 5 N HClO4-treated sample with 80 μl of Tris-HCl buffer (pH 8.4) and 100 μl of reagent containing firefly luciferase (Kikkoman Corp., Chiba, Japan). The reagent reacted with ATP or TAN and emitted light in proportion to the quantities of ATP or TAN in the samples. The time required to complete the reaction between ATP or TAN and luciferase was 90 s. The light generated from the samples was measured with a Mithras LB 940 luminometer (Berthold Technologies KmBH & Co. KG, Germany).
To determine the appropriate condition to assay the adenine nucleotide levels, we measured dilution rates of samples at various concentrations of HClO4. Finally, we adopted the concentration of HClO4 at 5 N and a dilution rate at 100 times for the measurement of adenosines.
Measurement of Islet Equivalent and Yield
To measure the IEQ and the islet yield, we took 200-μl samples from the circuit with a syringe. These samples were placed in a dish filled with 1 ml of HBSS, and 200 μl of dithizone was added to stain any islets present in the sample. In all five exams, one experienced person analyzed the islet yield, IEQ, purity, and morphology. The values of the islet yield and IEQ/g were calculated based on the values of the islet yield or IEQ of a 200-μl sample and the dilution factor (3,250×).
Measurement of ATP Content in Islets
We also measured the ATP content of the islets picked up from the digestive fluid. Zinquin (10,26), a specific probe for zinc, enabled us to identify the islets from the digestive fluid. A 1-ml sample of digestive fluid was taken from the digestive circuit and then it was washed two times in HBSS. We subsequently added 20 μl of zinquin to the fluid. We next picked up 10 islets, 100–150 μm in diameter, from the digestive fluid at various points. They were placed in 900 μl of HBSS with 100 μl of 5 N HClO4 and frozen until assayed.
SDS-PAGE
Sodium dodecyl sulfate polyacrylamide gel electrophoresis (SDS-PAGE) was carried out with 5–20% acrylamide gels (SuperSep; Wako Pure Chemical Industries, Ltd., Osaka, Japan) as described by Laemmli (14). Protease inhibitor cocktail (Complete; Roche Diagnostics GmbH, Roche Applied Science, Mannheim, Germany) was added immediately when samples were taken from digestive circuit to prevent proteolysis. Samples were divided into supernatants and pellets by centrifugation and then supernatants were analyzed. Supernatants were mixed with SDS buffer, containing of 0.35 mM Tris-HCl, pH 6.8, 10% SDS, 9.3% dithiothreitol (DTT), and 30% glycerol, and heated at 95°C for 5 min. Then the gels were run at 300 V until the prestained molecular marker running through the gel. Samples were made visible with silver staining reagent (2D silver stain II; Daiichi Pure Chemicals Co., Ltd., Tokyo, Japan).
Statistical Analysis
Data are expressed as the mean ± SD. The ATP and TAN contents are expressed as the amount of luminescence. The ATP increment of the sample was determined by comparing the values between the serial samples. The islet increment (count and IEQ) was also determined in this manner. The “ATP changing ratio” and the “islet count changing ratio” were calculated to determine the changing ratios of the ATP and the islet count by the following equations: ATP changing ratio = ATP content in 1 ml sample/precedent ATP content in 1-ml sample; islet count changing ratio = islet count in 200-μl sample/precedent islet count in 200-μl sample. The ATP contents and ATP/TAN ratio were compared using the unpaired Student's t-test with Bonferroni correction. Values of p < 0.05 were considered to be statistically significant. The relationship between the TAN level and IEQ was compared using Pearson's correlation coefficient. All statistical analyses were performed using the Statview J 4.58 for Windows software program (Abacus Concepts Inc., Berkeley, CA, USA).
Results
Changes in the ATP Level, TAN Level, Islet Count, and IEQ During the Digestion of Pancreatic Tissue
Five separate experiments were performed, with the ATP and TAN contents, islet count, and IEQ analyzed in the digestive fluid. The digestion time was 90 min in three experiments, 70 min in one experiment, and 60 min in another. Table 1 shows the basic information of the digested pancreata. The average weight of the digested pancreata was 42.8 ± 10.8 g. The average warm ischemic time was 15.4 ± 1.1 min, and the average cold ischemic time was 167.4 ± 24.0 min.
Basic Information of the Pancreas Grafts and Outcome of Isolation in Five Cases

ATP, adenosine triphosphate; TAN, total adenine nucleotide; IEQ, islet equivalent.
Table 1 also shows the four parameters measured in this study. The time required to reach the peak ATP value ranged from 20 to 50 min, with an average time of 33.0 ± 12.0 min. The required time to reach the peak TAN value ranged from 20 to 70 min, with an average time of 45.0 ± 20.6 min. The islet yield peaked at 20–80 min (44.0 ± 22.7 min), and the IEQ/g peaked at 20–70 min (43.0 ± 18.6 min) from the beginning of digestion.
The peak values are shown in Table 1 as well. The peak values of the amount of luminescence of ATP ranged from 38 to 873 nmol/μl, with an average of 293.5 ± 347.1 nmol/μl. The peak values of the TAN luminescence ranged from 203 to 5,354 nmol/μl, with an average value of 1615.2 ± 2128.3 nmol/μl. The maximum values of the islet yield ranged from 6,920 to 83,942/g of pancreas digested, with an average value of 28,496 ± 31,388/g. The maximum values of the islet yield ranged from 2,846 to 26,984 IE/g, with an average value of 12,684 ± 9,992 IE/g.
The chronological course of the ATP level, TAN level, islet count, and IEQ during pancreatic digestion in five cases are shown in Figure 1. The ATP levels, TAN levels, islet counts, and IEQ differed from case to case. The time required to reach the peaks of these parameters also differed from case to case.

Chronological course of the ATP/TAN level, islet count, and IEQ during pancreas digestion in five cases. The solid line, bold dashed line, thin dashed line, and double line indicate the adenosine triphosphate (ATP) content, total adenine nucleotide (TAN) content, islet equivalent (IEQ)/g pancreas, and islet count/g pancreas, respectively. The correspondence table is in the bottom-right corner. These parameters differed from case to case, and the duration to reach the peaks of these parameters was also different.
Appropriate Timing to Stop the Digestion
One experienced person performed serial observations of dithizone-stained samples under a microscope. We determined the point in time at which the digestion process should be stopped according to conventional criteria, including the presence of a number of individual acinar cells and the emergence of at least 10 islets free from the surrounding exocrine tissue within one visual field (40x). The stop points of five separate digestions were matched with the peaks of the islet count changing ratios, as shown in Figure 2. These peaks were preceded by the peak of the ATP level. Therefore, a greater than threefold increase in the ATP changing ratio followed by an increase in the islet count changing ratio within 5 min indicated the optimal time to stop the digestion.

Chronological changes in the islet count changing ratio and the ATP changing ratio. The solid line with open circles indicates the ATP changing ratios. The dashed line with open triangles indicates the islet count changing ratio. The stop points of five separate digestions were matched with the peaks of the islet count changing ratios, and these peaks were preceded by the peak of the ATP level.
The relationship between TAN (nmol/μl) and IEQ (×102/ml) in five digestions is shown in Figure 3. It indicates a significant correlation between these two parameters (Pearson's correlation coefficient, r2 = 0.71, p < 0.01). The ATP/TAN ratio of the digestion fluid increased rapidly as the temperature of the circuit rose. This increase lasted for 15 min until reaching a plateau. The levels declined significantly 40 min after the start of digestion (p < 0.05).

The relationship between the TAN level and IEQ in different five digestions. A significant correlation was observed between these two parameters (Pearson's correlation coefficient, r2 = 0.71, p < 0.01).
The Energy Levels of the Digesting Fluid and Islets During Appropriate Digestion and Overdigestion
To determine the energy levels of digesting fluid and islets during appropriate digestion and overdigestion periods, we compared the ATP/TAN ratio and the ATP content of 10 islets at 25 and 50 min after the start of digestion (Fig. 4). The ATP/TAN ratio, which indicates cell viability, decreased significantly at 50 min compared to the ratio at 25 min (p < 0.05) (Fig. 5A). The ATP content of 10 islets at 50 min, expressed as a percentage of 25 min, was significantly less than that at 25 min after the beginning of digestion (p < 0.01) (Fig. 5B).
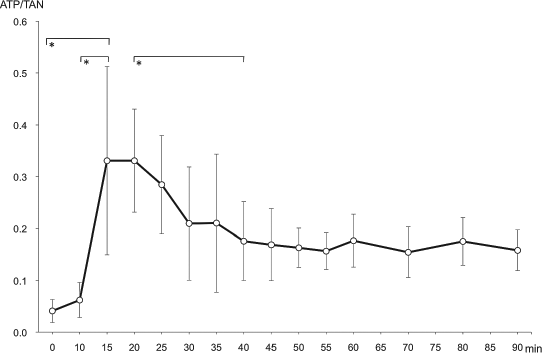
Chronological changes of ATP/TAN ratio. The ATP/TAN ratio indicates cell viability, and had increased significantly at 15 min in comparison to the beginning of digestion and also at 10 min (p < 0.05). Thereafter, it was observed to have significant decrease at 40 min.*Significant difference between the digestion periods, p < 0.05.

ATP/TAN ratio (A) and ATP content of 10 islets (B) at 25 and 50 min after the beginning of digestion. The ATP/TAN ratio had decreased significantly at 50 min (p < 0.05). ATP content of 10 islets at 50 min, which is expressed as a percentage to 25 min, was also significantly lower (p < 0.01). *Significant difference between the digestion periods, p < 0.05, **p < 0.01.
Protein Expression
SDS-PAGE revealed that a marked change was thus apparently observed in the digestive fluid from 10 to 20 min, and degradations of large protein were observed as time passed (Fig. 6).

Sodium dodecyl sulfate polyacrylamide gel electrophoresis (SDS-PAGE) of the digestive fluids of the porcine pancreas. The numbers below the columns indicate the times from the beginning of digestion. From 10 to 20 min a marked change was thus apparently observed in the digestive fluid.
Discussion
Obtaining a higher mean islet mass from a pancreatic graft is crucial for a successful outcome in the islet isolation procedure (7,25). Although several methods for obtaining a higher islet mass have been reported (12), the optimal timing at which to terminate the collagenase digestion and start dilution usually depends on the appearance and number of islets in the serially digested samples (4,13,25). Further objective parameters are therefore needed to determine the digestion termination point and promote successful outcomes (4). In this study, we measured the adenosine contents in relation to the islet count and islet yield using an overdigestion model. We observed the conventional termination of digestion to be consistent with the peak of the islet count changing ratio, which was preceded by the peak of the ATP changing ratio. The ATP changing ratio is also a useful objective indicator for terminating the digestion phase; within 5 min, a greater than threefold peak is always followed by an increase in the islet count changing ratio.
We previously reported the utility of the ATP bioluminescence assay for the evaluation of isolated islets. It enables us to measure the ATP content even in a small sample volume within several minutes (8,24). The ATP content has been identified as a marker for the viability of a pancreatic graft and isolated islets (2,5,8,9,11). We observed a rapid increase in the ATP level after 15–20 min of digestion, thus indicating the presence of a number of individual acinar cells and free islets with good viability. Furthermore, measuring the ATP level is more sensitive than conventional cell counting.
The TAN content in the digested fluids significantly correlated with the IEQ and it also displayed a peak value after 20 min of digestion. Meanwhile, the ratio of ATP/TAN gradually decreased with the progression of digestion, and then it significantly decreased after 40 min (in comparison to the ratio at 20 min of digestion). This outcome indicates the toxic effect of the digestion process on exocrine and endocrine cells in the digested fluid. Many factors, including the activation of endogenous serine proteases, have recently been reported to influence the outcome of islet isolation (3,13,15,20,22,23). In fact, we observed a greater number and variety of proteins at 20 min or later in the digestion process than at 0–20 min (Fig. 6).
We also examined the toxicity of the environment against islets in the digestion circuit during the digestion phase. We selected 40 islets from the digested fluid at 25 and 50 min and compared the ATP content from each time period. The ATP content of the islets selected at 50 min was significantly less than that obtained at the 25-min point. Therefore, prolonged digestion damages endocrine as well as exocrine cells in the digestion circuit. These results support the idea that collagenase digestion should be stopped earlier to obtain viable islets.
We measured adenosine contents in relation to islet count and yield using an overdigestion model of the porcine pancreas. Our findings clarified the chronological changes of these parameters and demonstrated that the conventional time for the termination of digestion correlated with the peak of the islet count changing ratio. Furthermore, the conventional time for the termination of digestion was preceded by a greater than threefold increase in the ATP changing ratio, which is therefore considered to be a useful objective indicator for the termination of the digestion phase. Further study is needed to compare the yield of islets and the in vivo and in vitro function of islets between the findings obtained with the conventional isolation method and islet isolation using the ATP measurements.
Footnotes
Acknowledgments
This work was supported in part by grants from the Japanese Ministry of Education, Culture, Sports, Science and Technology and in part by a Grant-in-Aid for Research on Human Genome, Tissue Engineering Food Biotechnology, Health Sciences Research Grants, Ministry of Health, Labor and Welfare of Japan. The reagents of adenosine nucleotides and luciferase were provided by Kikkoman Corp. The authors declare no conflict of interest.
