Abstract
Oxygen is a vital nutrient for growth and maturation of in vitro cells (e.g., adult hepatocytes). We previously demonstrated that direct oxygenation through a polydimethylsiloxane (PDMS) membrane increases the oxygen supply to cell cultures and improves hepatocyte functions. In this study, we removed limits on oxygen supply to fetal rat liver cells through the use of direct oxygenation through a PDMS membrane to investigate in vitro growth and maturation. We chose fetal liver cells because they are considered a feasible source of liver progenitor cells for regenerative medicine therapy due to their highly efficient maturation and proliferation. Cells from 17-day-old pregnant rats were cultured under 5% and 21% oxygen atmospheres. Some cells were first cultured under 5% oxygen, and then switched to a 21% oxygen atmosphere. When oxygen supply was enhanced by a PDMS membrane, the rat fetal liver cells organized into a complex tissue composed of an epithelium of hepatocytes above a mesenchyme-like tissue. The thickness of this supportive tissue was directly correlated to oxygen concentration and was thicker under 5% oxygen. When cultures were switched from 5% to 21% oxygen, lumen-containing structures were formed in the thick mesenchymal-like tissue and the albumin secretion rate increased. In addition, cells adapted their glycolytic activity to the oxygen concentrations. This system promoted the formation of a functional and organized thick tissue suitable for use in regenerative medicine.
Keywords
Introduction
The liver is the largest organ in mammals, and is involved in many functions, from detoxification to metabolism and storage. Because it is central to blood detoxification, it is of prime interest for regenerative medicine (32,38). Given the finite donor organs, cell transplantation offers a promising alternative (31). However, it is limited by the small number of available hepatocytes (12) and their low in vitro growth and function (7,14). All liver functions have yet to be replicated at desired levels in vitro. This has prompted ongoing development of new approaches that physiologically mimic liver functions.
Current knowledge indicates that several key factors maintain hepatocyte growth and functions in vitro: cells with high differentiation potential, the culture environment (such as culture medium formulation, spatial organization, interactions between parenchymal and nonparenchymal cells), and the amount of oxygen supplied. Fetal liver cells have a highly efficient maturation capacity and are regarded as a feasible source of liver progenitor cells that will become hepatocytes and bile duct epithelial cells for cell therapy (36). Their greater genetic normality and proliferative activity in vitro (9,10) compared with adults hepatocytes and their capacity to differentiate into various cell types make them very attractive for cell-based alternative treatment approaches for liver disease (12). The growth and differentiation of these cells in vitro are strongly related to the presence of growth factors in the cell culture medium [e.g., hepatocyte growth factor (HGF), fibroblast growth factor-1 (FGF-1), and fibroblast growth factor-4 (FGF-4)] (34). Previously, we combined this cell culture medium and other factors to increase liver-specific functions of fetal liver cells (15).
Oxygen supply is a vital nutrient for in vitro fetal cell development (27). However, oxygen releases reactive oxygen species (ROS) into the cells (13). Under physiological conditions, ROS are detoxified by various antioxidant enzyme (AOE) defense systems (21). Insufficient response from AOE systems of immature cells, such as fetal cells (21), causes oxidative stress that may result in cellular or tissue damage (35). It is therefore necessary to develop several physical and chemical techniques to increase controlled oxygen supply to the cells. In this aim, some gas-permeable materials, such as polydimethylsiloxane (PDMS), can be used as cell culture scaffolds (17). This direct oxygenation methodology is known to significantly improve cell culture and cell function of hepatocellular carcinoma cell line Hep G2 cells (11) and primary hepatocytes (24). However, it has not yet been applied to normal hepatocyte progenitors that are expected to be used in future liver tissue engineering.
We studied the construction of liver tissue from fetal liver cells in vitro when oxygen limitation was completely removed by the use of direct oxygenation through PDMS membranes. We hypothesized that proliferative cells would spontaneously grow to several layers as previously described (11). Because we used full fetal liver, which contains the different types of progenitors cells (parenchymal and mesenchymal) (25), we hypothesized that the cells would organize into a complex liver tissue similar to that seen in vivo. We also supplied a low oxygen concentration to study the effect of limited oxidative stress on fetal liver cell behavior in vitro.
Materials and Methods
PDMS Plate Fabrication
We used PDMS plates with a culture surface of covalently bound collagen molecules and collagen-coated tissue culture polystyrene plates (TCPS) to culture cells. The PDMS plates were prepared as previously described (23,24). A PDMS membrane (thickness: 1.5 mm) was attached and clamped between a 24-well polycarbonate frame and a stainless steel board with 24 holes to avoid inhibition of oxygen permeation through the PDMS membrane. Subsequently, PDMS plates were treated by oxygen plasma and coupled with aminosilane (KBE-903; Shinetsu Silicone, Japan). The introduced amino groups were reacted with a photoreactive cross-linker, sulfosuccinimidyl 2-(m-azido-o-nitrobenzamido)-ethyl-1,3′-dithiopropionate (SAND) (PC21549; Pierce, IL, USA), by exposing them to UV light (37). We used this cross-linker to covalently immobilize collagen Type 1-P (Nitta Gelatin, Japan) on the PDMS surface.
Cell Inoculation
Rat fetal liver cells were isolated by the collagenase digestion method, as previously described (15). We removed rat fetuses from pregnant Wistar rats on day 17 of gestation (Sankyo Labo Service, Tokyo, Japan). They were then killed by ether anesthesia and placed on a dish filled with phosphate-buffered saline (PBS). We obtained approximately 4 to 12 fetuses from each pregnant rat. The livers were removed from the rat fetuses, minced, and washed with liver perfusion medium (Invitrogen, CA, USA) in a water bath kept at 38°C for 10 min. After centrifuging them once at 60 × g for 3 min, the minced liver tissues were treated with a liver digest medium (Invitrogen) containing collagenase and dispase for 20 min in a water bath at 38°C. Liver cells were dispersed by gentle pipetting and undigested tissues were removed by filtration on a 70-mm Falcon cell strainer (BD Biosciences; CA, USA). Red blood cells were disrupted with a hemolysis buffer. After filtration by the 70-mm Falcon cell strainer (BD Biosciences, CA, USA) and two centrifugations at 60 × g for 1 min, cells were counted and cellular viability for experiments (over 80%) was measured by trypan blue exclusion. The use of pregnant rats for this study was approved by the Animal Experimentation Committee at the Institute of Industrial Science, University of Tokyo, and the experiments were conducted according to the University of Tokyo guidelines for animal experimentation.
Cell Culture
The fetal liver cells were inoculated in collagen type I-coated tissue culture polystyrene (TCPS) 24-well plates (Asahi Techno Glass; Chiba, Japan) and in PDMS 24-well plates at 1.6 × 105 cells/well. The basal culture medium was Williams' medium E (Sigma-Aldrich, MO, USA) supplemented with 2 mM l-glutamine (Wako, VA, USA), 10−6 M hydrocortisone (Sigma-Aldrich), 10 ng/ml mouse epidermal growth factor (EGF; Peprotech, NJ, USA), 10−7 M insulin (Takara, Shiga, Japan), 10−8 M glucagon (Sigma-Aldrich), 0.5 mM ascorbic acid phosphate (Wako), antibiotic/antimycotic solution (Invitrogen), 1% MEM nonessential amino acid solution (Invitrogen), and 10% fetal bovine serum (FBS; Gemini Bio-Product, CA, USA). After 6 h, we replaced the spent culture medium with fresh medium with 10 mM nicotinamide (Wako) and various soluble factors. These included 10 ng/ml oncostatin M (OSM; R&D Systems, MN, USA), 20 ng/ml FGF-1 (Peprotech), 20 ng/ml FGF-4 (Peprotech), and 20 ng/ml HGF (Peprotech). After 2 days of being cultured, we added 1 mM sodium butyrate (Wako). We changed the culture medium every 2 days, and then sampled the medium from each culture for functional measurements. The media were stored at −20°C for further experimentation.
Cultures on conventional TCPS plates (T) and PDMS plates (P) were incubated under an air atmosphere incubator (21% oxygen—T21 or P21) or a physiological oxygen environment (5% oxygen—T5 or P5). Some cultures were first cultured in a 5% oxygen atmosphere for 1 week, and then transferred to a 21% oxygen atmosphere incubator (5% then 21% oxygen—T5/21 or P5/21).
For the vertical cross-section, we cultivated some cells on a PDMS surface in TCPS 24-well plates. Changes in oxygen concentration produced no significant differences in the morphology, function, or metabolism of TCPS and PDMS-TCPS plate cultures (data not shown).
DNA Measurement
The cellular DNA amounts in each culture well after 2 weeks (day 14) were measured by 4′6-diamidino-2-phenylindole (DAPI—Dojindo, Kumamoto, Japan) with fluorescence at an excitation wavelength of 350 nm and an emission wavelength of 450 nm.
Glucose Consumption and Lactate Production Measurement
Every 2 days, we measured glucose concentration on the culture medium with an automated glucose analyzer (A&T, GA05; Japan). Glucose consumption was calculated by subtracting the measured glucose from the total amount of glucose. The lactate produced by the cells was measured at the same time by an enzymatic automated analyzer (YSI-7100MBS; YSI Life Sciences, OH, USA).
Albumin Measurement
Secreted albumin in the culture medium was measured by the sandwich-type enzyme-linked immunosorbent assay (ELISA). Anti-rat albumin goat IgG (MP Biomedicals-Cappel Products, CA, USA) was used as the primary antibody, and horseradish peroxidase-conjugated anti-rat albumin sheep antibody (MP Biomedicals-Cappel Products) was used as the secondary antibody.
Cross Section, H&E Staining, AZAN Staining, GST-pi, CK18, and CK19 Immunostaining
The cells were fixed overnight on day 14 with 4% paraformaldehyde (Sigma-Aldrich) in PBS, and embedded in paraffin. Vertical sections (7 μm thick) of the cell layers were obtained from the center region of each specimen, and stained with hematoxylin and eosin (H&E) or Azan (Muto, Tokyo, Japan).
Some paraffin-embedded vertical cross sections were used for immunostaining. In these cases, paraffin was removed by successive washing with xylene, ethanol (100%, 95%, 80%, and 70%), and PBS. We retrieved glutathione S-transferase-pi (GST-pi), cytokeratin-18 (CK18), and cytokeratin-19 (CK19) antigens by performing trypsin digestion (15 min at +37°C). Slides were then treated with immunostaining kits [anti-rabbit horseradish peroxidase (HRP)-3-amino-9-ethyl-carbazole (AEC) system (R&D Systems; MN, USA); anti-goat HRP-3, 3-diaminobenzidine (DAB) system (R&D Systems); and anti-mouse HRP-DAB system (R&D Systems)]. Staining agents included peroxidase, serum, avidin, and biotin blocking; first antibody incubation (anti-GSTpi (MBL, Japan) dilution = 1:1000; anti-CK18 (Sigma-Aldrich) dilution = 1:50; anti-albumin (MP Biomedicals-Cappel Products, CA, USA) dilution = 1:100; anti-CK19 (Novocastra, Wetzlar, Germany) dilution = 1:10 overnight at +4°C; biotinylated second antibody incubation; high sensitivity streptavidin conjugated to HRP incubation; and AEC or DAB revelation. Positive cells were stained either red (AEC) or brown (DAB). Counterstaining was performed using a conventional hematoxylin counterstaining technique that stains the nucleus in blue. We performed a negative control with an isotype antibody and PBS instead of a first antibody.
Statistical Analysis
Data were expressed as mean ± SD. Statistical comparisons were done using one-way ANOVA followed by the Student-Newman-Keuls test. Differences of p < 0.05 were considered to be statistically significant. All errors bars were presented as SDs.
Results
Morphologies of Fetal Liver Cells in the Different Oxygen Conditions
Cultures on TCPS plates under 5% oxygen (T5) conditions, including those switched to 21% (T5/21) on day 8, showed a lack of differentiation in hepatocytes and formation of a fibrous tissue on day 14 (Fig. 1A, C), whereas the 21% oxygen (T21) condition partly supported emergence of hepatocyte-like epithelial cells (Fig. 1B, arrows). Under direct oxygenation using PDMS plates, we observed an apparent increase in the hepatocyte-like epithelial cell population regardless of the oxygen concentration (P21, P5, and P5/21) (Fig. 1D–F). From days 10 to 12, the cell layers on P21 sometimes detached from the surfaces and became wound together to form a huge floating aggregate containing all the detached cells of the relevant well. P5 and P5/21 cultures remained attached to the PDMS membrane.

Morphologies of the rat fetal liver cells on day 14 when cultured under T5 (A), T21 (B), T5/21 (C), P5 (D), P21 (E), or P5/21 conditions (F). The arrows indicate immature hepatocyte-like epithelial cells. Scale bar: 50 μm. T5 = physiological oxygen environment (5% O2) on tissue culture polystyrene (TCPS) plates; P21 = air atmosphere incubator (21% O2) polydimethylsiloxane (PDMS) plates.
Multilayered Organization Observed by Vertical Cross Section of the Cell Layers
Histological Staining
From the H&E staining of the thin sections vertical to the culture surfaces, we observed that cells on TCPS plates grew as a spread monolayer regardless of the concentration of atmospheric oxygen (Fig. 2A–C). In contrast, when cultured on our PDMS plates, all cultures grew to thick multilayered tissue (Fig. 2D–F). Respective thicker tissue specimens measuring from 40 to 75 μm and from 75 to 130 μm were observed in the cultures consistently exposed to 5% oxygen (Fig. 2D) or those initially exposed to 5% oxygen and later to 21 % oxygen (Fig. 2F). A thinner tissue specimen (from 15 to 50 μm) was seen in the culture consistently exposed to 21% oxygen (Fig. 2E). In all the PDMS plate cultures, inoculated cell populations organized as an epithelium-like monolayer above a thick multilayered tissue composed of collagen (blue color) and cells (red color) in the Azan stainings (Fig. 2G-I). This outcome indicates that those P5 and P21 cells organized as mesenchymal tissue, and not as epithelium (Fig 2G, H). On P5/21 cultures, lumen-containing structures developed in the thick mesenchymal-like tissue (Fig. 2F, I).

Hematoxylin and eosin staining (A–F) and Azab staining (G–I) of vertical cross sections of the rat fetal liver cell cultures, on day 14 when cultured under T5 (A), T21 (B), T5/21 (C), P5 (D, G), P21 (E, H), or P5/21 conditions (F, I). The cell layers fronted to culture medium above and to TCPS (A–C) or PDMS (D–I) below. Scale bar: 50 μm.
Immunohistochemical Staining
Upper cell monolayers facing the culture medium were positively stained (Fig. 3) by anti-CK18 and anti-albumin antibodies (in brown), two hepatocyte markers, and by anti-GSTπ antibody (in red), a specific protein of the bile canaliculi area formed by the tight junction of two hepatocytes (20) in the three cultures [P5 (Fig. 3A, E, J); P21 (Fig. 3B, F, K); P5/21 (Fig. 3C, G, L)]. This suggests formation of an epithelium of hepatocytes. An immunohistochemistry analysis of lower layer tissues was unable to precisely identify the cellular populations. Nevertheless, the spread shape of the cells indicates the presence of mesenchymal cells even in the lowest layer facing the PDMS surface. These cells show the same staining characteristic as the upper layer. Inside the thick mesenchymal layer, CK18 and albumin were observed as cellular components (arrowhead in Fig. 3), or deposited into the extracellular matrix (ECM, asterisk in Fig. 3). The lumen structure observed in the P5/21 condition was negative for CK19, which is a marker for bile epithelial cells (Fig. 3I) and albumin (Fig. 3H). It was positive with CK-18 (Fig. 3M), thus suggesting that cells organizing these lumen structures are immature hepatocytes.
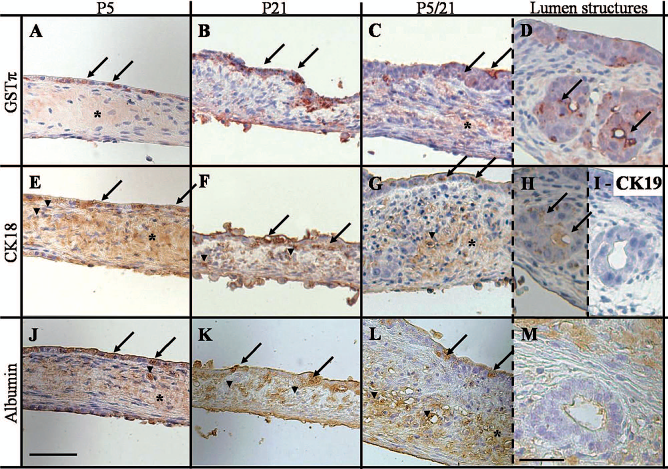
Immunohistochemistry staining of vertical cross sections of the rat fetal liver cell cultures on day 14, under P5 conditions (A, E, J), P21 conditions (B, F, K), or P5/21 conditions (C, D, G, H, L, M). The cell layers fronted to culture medium above and to PDMS below. Lumen structures observed in P5/21 cultures were also stained (D, H, I, M). Cross sections were stained with antibodies directed against glutathione S-tranferase-pi (GSTπ; A–D), cytokeratin [CK18 (E–H), CK19 (I)], and albumin (J–M). Positive epithelial cell (arrow), positive mesenchymal cell (arrow head), and positive extracellular matrices (asterisk) are stained in red (A–D) or brown (E–M). Scale: 50 μm except for lumen structures (D, H, I, M). Scale bar: 25 μm.
Growth, Functional Maturation, and Metabolism of Fetal Liver Cells; Differences Between PDMS and TCPS Plates
DNA Measurement
To measure the growth of the culture, we measured the total DNA content in each condition on day 14 (Fig. 4). First we observed that cultures on conventional 24-well plates did not show any significant differences regardless of the oxygen concentration. In contrast, direct oxygenation produced a significant increase in cell numbers (up to six times more for the P5 culture; p < 0.001). In addition, when cultured in PDMS, a lower oxygen concentration (5%) induced a significantly higher cell proliferation compared with a high oxygen (21%) concentration (p < 0.01). Changes in oxygen concentration on the P5/21 culture did not induce significant differences in tissue growth compared with tissues consistently cultured in a 5% oxygen atmosphere.

Cellular DNA contents of the rat fetal liver cell cultures on day 14 when cultured under T5, T21, T5/21, P5, P21, or P5/21 conditions. Inoculated DNA amount per well is represented by the dot line. The columns and the error bars represent the mean ± SD of six wells from two independent experiments. *p < 0.001, §p < 0.005, ‡p < 0.05, †p < 0.01.
Albumin Secretion
We measured albumin secretion of the culture medium as an index for functional maturation (Fig. 5). In conventional cultures (T21), the albumin secretion rate increased to a maximum on day 6, and then decreased until the end of the culture. When exposed to 5% oxygen, even partially, TCPS plate tissues secreted a very low amount of albumin with no significant variation. When cultured on PDMS plates, albumin secretion increased for the first 6 days then reached a stationary phase for 2 days regardless of oxygen concentration. The albumin rate was constant in 5% oxygen cultures, but it increased in the cells with 21% oxygen (P21 and P5/21). On day 10, albumin secretion of P21 cultures strongly decreased, while the rate of P5/21 cultures continued to increase.

Albumin secretions of the rat fetal liver cell cultures under T5 (solid line/square), T21 (solid line/circle), T5/21 (solid line/triangle), P5 (dashed line/square), P21 (dashed line/circle), or P5/21 conditions (dashed line/triangle). Data are expressed as the mean values ± SD of six wells from two independent experiments. Vertical dashed line indicates the time point where oxygen concentration was changed from 5% oxygen to 21% oxygen.
Glucose Consumption and Lactate Production
Glucose consumption and lactate production were measured to analyze the basic metabolism for energy production of the cells. Because of the significant growth differences observed between TCPS and PDMS plates; both were normalized by the amount of DNA on day 14. Glucose consumption and lactate production by cells were higher when cultured in TCPS plates than in PDMS plates (up to 8 times more for P5/21; Fig. 6A). When focusing on the secreted lactate per consumed glucose ratio (Fig. 6B) under 5% oxygen atmosphere in TCPS or PDMS plates, the molar ratio of produced lactate over consumed glucose increased in time to reach 2 mol of lactate produced for 1 mol of consumed glucose (which is the anaerobic glycolytic ratio) (2). The ratio in the T21 condition increased strongly for the first 4 days, but then slowed to 1.5. Under the same oxygen condition, P21 cultures showed the same increase, but after day 4, the ratio decreased below 1. Kinetics of the ratios in T5/21 and P5/21 cultures followed those in the 5% oxygen condition when cultured under 5% oxygen. On day 8, they followed those in the 21% oxygen condition immediately after the change in oxygen concentration.

Glucose consumption and lactate production rates normalized to cellular DNA contents on day 14 (A). Changes in the molar ratios of lactate production over glucose consumption of fetal liver cells (B) under T5 (solid line/square), T21 (solid line/circle), T5/21 (solid line/triangle), P5 (dashed line/square), P21 (dashed line/circle), or P5/21 conditions (dashed line/triangle). Vertical dashed line indicates the time point where oxygen concentration was changed from 5% oxygen to 21% oxygen. Horizontal dashed line indicates the stoichiometric ratio of anaerobic glycolysis. Data are expressed as the mean values ± SD of six wells from two independent experiments. *p < 0.0001.
Discussion
In this study, we investigated whether fetal liver cells construct liver tissue in vitro when oxygen limitation is completely removed by the use of direct oxygenation through PDMS membranes. Cells proliferated and spontaneously grew to several layers. The use of full fetal liver was pertinent as the different types of progenitor cells organized themselves into complex in vivo-like liver structures. We also showed that controlling the oxygen concentration supplied to the cells was important to control the functional, structural, and metabolic behavior of the culture.
The improvement in the oxygen supply induced a multilayered growth of the cells. The use of a flat PDMS membrane as a cell support is the simplest system for providing a continuous supply of direct oxygen to the cells. This avoids the quick impoverishment of oxygen in the culture medium (23). The silicone elastomer is a good candidate to cultivate mammalian cells due to its biocompatibility (3,28) as well as its high gas diffusion (4.1 × 10-5 cm2/s) (6) and solubility [10.6 nmol/(ml mmHg)] (19) compared with culture medium [respectively, 4.1 × 10-5 cm2/s and 1.19 nmol/(ml mmHg)] (18,22). The sufficient supply of oxygen to the cells promoted the formation of a thick tissue, while the use of the TCPS plate induced a severe hypoxia responsible for an absence of fetal liver cell development (Fig. 2). This outcome has been observed in human fetal liver cells in vitro (27).
The behavior of rat fetal liver cells was associated with oxygen concentration. When oxygen supply was enhanced, the cells self-organized in an in vivo-like arrangement made up of an epithelial monolayer of hepatocytes above a mesenchymal layer. The oxygen concentration directly influenced the thickness of the latter layer; a low oxygen environment induced mesenchymal cell growth and ECM secretion as observed in vivo (8,33), making the tissue thicker (Fig. 2). However, the exact cell composition of the mesenchymal-like tissue is not yet fully elucidated. A thick mesenchymal-like layer was maintained to be attached to the PDMS membrane, while a thin mesenchymal-like layer, as observed under high oxygen supply, was not stably attached despite the use of covalent-binding PDMS membrane. Furthermore, oxygen conditions in the vicinity of the cells influenced albumin secretion, which increased with the higher oxygen concentration (Fig. 5). In addition, the presence of mesenchymal cells in the PDMS plates allowed interaction between parenchymal and nonparenchymal cells. In vitro data show that enhanced oxygen supply to human hepatoma HepG2 cells (11) and cocultures of adult mouse or rat hepatocytes with nonparenchymal cells (4, 30) both independently improve cell function. This suggests that both parameters have a synergic influence on albumin secretion, as previously observed on HepG2-3T3 fibroblast cocultures on a PDMS membrane (23). In the present study, oxygen had a direct as well as indirect influence on albumin secretion. Enhanced albumin secretion and self-organization of the cells also suggest that the antioxidant process in the fetal liver cells is sufficient. It handled the increase of oxygen species formed after the abrupt oxygen exposure immediately after cell inoculation on PDMS membranes, then culture under high oxygen atmosphere.
Switching the oxygen concentration from low to high improved the formation of in vivo liver-like structures in the mesenchyme-like tissues (Fig. 3D, H, I, M). The structures were similar to those described in in vitro mouse liver organ culture: bile duct poorly differentiated and formed by hepatocyte aggregates with large lumina, and surrounded by connective tissue (29). To date, such structures have not been observed in individualized cell cultures. Those cells were negatively stained by antibodies to bile duct cytokeratin (Fig. 3I). In the previous study, they were positively stained by antibodies to albumin (29), while our cultures were negative to albumin (Fig. 3M). Further experiments are needed to explain this difference and determine the developmental stages of hepatocytes. Under such conditions, albumin secretion greatly increased in the high oxygen environment (Fig. 5), probably due to a synergistic effect from oxygen and maintenance of the tissue integrity. Together, those results show that switching the oxygen concentration combined the advantages of both cultures (thick and stable functional tissue), and was favorable to, but not sufficient, to form a developmental environment for in vivo liver-like structures.
We focused on lactate production at different concentrations of oxygen supplied to the cells. Lactate is the degradation product of anaerobic respiration of the cells. We demonstrated that the PDMS membrane consistently reduced anaerobic glucose catabolism by the cells. Indeed, in each oxygen environment, the use of the PDMS membrane dramatically decreased the rate of glucose consumption and lactate production (Fig. 6A). Molecular ratios of the lactate production over the glucose consumption were between 1.5 and 2.3. Data suggest the ratio should be 2 during the lactate fermentation that occurs in anaerobic glycolysis (2). The ratio decreased to less than 0.8 when oxygen supply to the PDMS membrane was increased. The lactate/glucose ratio analysis of PDMS cultures strongly suggests that use of the PDMS membrane allowed us to mimic the zonal metabolism observed in vivo in liver acini. Pericentral hepatocytes are supplemented by a low oxygen environment (4%), use anaerobic energy metabolism, and have a low albumin secretion rate (5,16). We observed this in P5 cultures. Perivenous hepatocytes, however, are supplemented by a high oxygen environment (13%), use aerobic energy metabolism, and secrete high levels of albumin (5,16). We observed this in P21 and P5/21 cultures (Figs. 5 and 6). Further experiments are needed to confirm this hypothesis as nonparenchymal cells, which were abundant in our system, secrete more lactate than adult hepatocytes (1). This may influence our results and interpretations. So far, we hypothesize that the use of a highly gas-permeable PDMS membrane allowed us to maintain the actual oxygen concentration in the cellular vicinity at values nearly equal to the atmospheric oxygen concentration described by the Henry's law. In the TCPS plate cultures, the actual oxygen concentration at the cellular vicinity may have been lower than atmospheric oxygen concentration due to the low diffusion and solubility of oxygen. This hypothesis may explain how atmospheric oxygen concentration can dramatically influence cell metabolism on PDMS membranes compared to TCPS plates. We also demonstrated the reversibility of those phenotypes by switching oxygen concentration. This showed the importance of oxygen concentration on the future phenotype of rat fetal liver cells. This has been demonstrated in vitro in adult hepatocytes (26).
In conclusion, we are introducing a new approach for the progenitor cell-based construction of thick liver tissue through the use of a simple flat PDMS membrane and control of oxygen concentration. We demonstrated that in such culture systems, rat fetal liver cells spontaneously grow to several layers (thickness directly depends on the oxygen concentration) and organize themselves into complex in vivo-like liver tissue. We also observed that increasing the oxygen supply raised the cellular albumin secreted, which indicates early hepatocyte maturation. Our simple system may offer a promising approach for studying and improving in vitro organ development from stem or progenitor cell-based liver tissue.
Footnotes
Acknowledgments
This work was performed in the framework of LIMMS (Laboratory for Integrated Micro-Mechatronic Systems), a joint laboratory between the CNRS (Centre National de la Recherche Scientifique) and IIS (Institute of Industrial Science), the University of Tokyo. This work was supported by Grants-in-Aid for Scientific Research from the Ministry of Education, Culture, Sports, Science, and Technology, Japan, by CREST from Japan Science and Technology Agency, Japan, and by the CNRS, France. The authors declare no conflict of interest.
