Abstract
Depletion of pancreatic β-cells results in insulin insufficiency and diabetes mellitus (DM). Single transplantation of mesenchymal stem cells exhibits short-term effects in some preclinical studies. Here, we further investigated the long-term therapeutic effects of multiple intravenous MSC transplantations. In this study, multiple human MSC transplantations (4.2 × 107 cells/kg each time) were performed intravenously at 2-week intervals into streptozocin (STZ)-induced diabetic mice for 6 months. Blood sugar, insulin, renal function, cholesterol, and triglyceride levels were monitored. We demonstrated that compared to single intravenous transplantation, which only transiently decreased hyperglycemia, multiple MSC transplantations effectively restored blood glucose homeostasis. Systemic oxidative stress levels were reduced from the seventh week of treatment. From the 11th week, production of human insulin was markedly increased. When MSC transplantation was skipped after blood sugar level returned to normal at the end of 15th week, a sharp rebound of blood sugar occurred, and was then controlled by subsequent transplantations. At the end of 6 months, histopathology examination revealed MSCs specifically engrafted into liver tissues of the recipients. Fifty-one percent of human cells in the recipient liver coexpressed human insulin, especially those surrounding the central veins. Taken together, intravenous MSC delivery was safe and effective for blood glucose stabilization in this preclinical DM model. Multiple transplantations were essential to restore and maintain glucose homeostasis through decreasing systemic oxidative stress in the early stage and insulin production in the late stage. Liver engraftment and differentiation into insulin-producing cells account for the long-term therapeutic effects of MSCs.
Keywords
Introduction
Intravenous mesenchymal stem cell (MSC) transplantation has been demonstrated to be a useful therapeutic strategy for functional recovery in a variety of diseases such as therapy-resistant graft-versus-host disease (28,42), acute myocardiac infarction (20), stroke (3), and multiple systemic atrophy (33) by various preclinical and clinical trials. In our previously reported preclinical study, intravenous transplantation of MSCs was effective in rescuing lethal fulminant hepatic failure (27). We found that single dose of intravenous MSC transplantation reduced oxidative stress, promoted repopulation of host hepatocytes, and replenished host liver cells by differentiation of transplanted MSCs into functional hepatocytes, suggesting that both reactive oxygen species (ROS)-scavenging and paracrine effects of MSCs contributed to the survival of mice with lethal hepatic failure besides direct hepatocyte differentiation (27).
Depletion of β-cells in the pancreatic islets results in type 1 diabetes mellitus (T1DM) due to insufficient insulin production. Insulin replenishment is essential to treat insulin-dependent diabetes (11,30,38). Exogenous insulin supply fails to respond to plasma glucose level and may result in asymptomatic hypoglycemia, which pushes the therapeutic strategy towards cell transplantation (16,30). Islet cell transplantation has gained some clinical success (5,36,37,44); however, limited donor source is the major hurdle for extensive clinical application. Stem cells, which possess the potentials of self-renewal and multilineage differentiation, may be an ideal source of β-cells (2,39,40). MSCs, which can be isolated from many tissues and can be extensively culture expanded (6,23,50,53), possess the multipotency in vivo (9,39), and immunomodulation abilities because of their low immunogenicity as well as the ability to inhibit T-cell proliferation and to prevent cytotoxic T-cell development (1,4,26,29), indicating the possibility of allogenic MSC transplantation.
It has been reported that in streptozocin (STZ)-induced diabetic animal models, a single dose of intravenous MSC injection diminished hyperglycemia and attenuated diabetic complications in some short-term animal studies (14,45,52). It has been described that single dose of human multipotent marrow stromal cell injection preserved mouse islet cells in nonobese diabetic/severe combined immunodeficient (NOD/SCID) mice (34). In experimental autoimmune DM model, allogeneic MSCs resulted in temporary reversal of hyperglycemia through immunomodulation (17). Besides, the immunosuppressive ability of MSCs not only prevented the rejection of allogeneic islet grafts (12) but also prolonged the survival of semiallogeneic heart (7).
However, DM is a chronic, progressive disease, and long-term follow-up is indispensable to evaluate the therapeutic effects of MSC transplantation. Besides, multiple MSC transplantations may be necessary. However, the preclinical studies of multiple MSC transplantations on DM are lacking. The aim of this study is to investigate the effects and the mechanisms of systemic MSC transplantation via intravenous injection on blood glucose regulation on a long-term basis. In this study, serial intravenous MSC transplantations were carried out in STZ-induced diabetic mice for 6 months at the interval of 2 weeks. Blood sugar, insulin, renal function, cholesterol, and triglyceride (TG) levels were continuously monitored and histopathology studies were performed at the end of the study.
Materials and Methods
Animals
Eight-week-old BALB/c male mice were purchased from the National Laboratory Animal Center (NLAC, Taipei, Taiwan). All the animals were housed following the animal care guidelines of NLAC, and the experimental protocols had been reviewed and approved by the Institutional Animal Care and Use Committee (IACUC) at the Taipei Veteran General Hospital. Body weight of the experimental animals was recorded weekly.
Isolation and Expansion of MSCs
MSCs were isolated from human bone marrow following the previously reported protocol (31) that had been approved by the Institutional Review Board at the Taipei Veteran General Hospital. Informed consent had been obtained prior to the aspiration of bone marrow from patients. Briefly, after negative immune selection and limiting dilution from the mononuclear fraction of the bone marrow aspirates, colonies obtained from single cells were culture expanded in T75 flasks with MesenPro medium (Invitrogen, Carlsbad, CA, USA). When the cell density reached 60—70% of confluence, they were detached from one flask and reseeded to three new flasks after two gentle phosphate-buffered saline (PBS, Gibco BRL, Grand Island, NY, USA) washes. The surface immune phenotype and multipotency of MSCs were confirmed as previously described (22,31,32). An approval from the Institutional Review Board had been obtained prior to the commencement of this study.
Induction of Diabetes and MSC Transplantation
T1DM was induced by intraperitoneal injection of STZ (Sigma-Aldrich, St. Louis, MO, USA) in 25 mM sodium citrate (pH 4.5), with the dosage of 200 mg/kg body weight, into 10-week-old BALB/c mice with the body weight of 23—25 g. Onset of DM was defined as elevated blood sugar values of higher than 200 mg/dl found at two consecutive tests at 3-day intervals. Same volume of phosphate-buffered saline (PBS) was injected into BALB/c mice intraperitoneally as controls.
Diabetic mice were further divided into two groups, STZ group and STZ with MSC transplantation group. In the MSC transplantation group, we performed single and multiple MSC transplantations. For single MSC transplantation, 4.2 × 107 cells/kg body weight in 0.2 ml PBS were injected into diabetic mice via tail vein 7 weeks after STZ injection. For multiple MSC transplantation, the same dosage of cells (4.2 × 107 MSCs/kg body weight in 0.2 ml PBS) were injected into diabetic mice via the tail vein from 1 week after the onset of diabetes, and a series of MSC transplantations were performed with the interval of 2 weeks. In STZ group, 0.2 ml of PBS was injected into diabetic mice via tail vein at the same time points of MSC transplantation. In this study, insulin was not given to any experimental animals.
Measurement of Blood Sugar, Insulin, Cytokines, Growth Factors, and Systemic Oxidative Stress Level
Mice were under starvation for 6 h before the measurement of fasting blood sugar level. Tail capillary blood sugar levels were measured twice a week using One Touch Ultra (LifeScan Inc., Milpitas, CA, USA). Whole blood was collected from facial vein weekly, and the plasma was collected after centrifugation at 7000 rpm for 15 min. Serum interferon-γ (INF-γ) and tissue necrosis factor-α (TNF-α) was detected by ELISA kits (eBioscience, Inc., San Diego, CA, USA). Serum level of mouse insulin was measured by Mercodia Mouse Insulin ELISA Enzyme immunoassay (Mercodia AB, Uppsala, Sweden) and human insulin was detected by Mercodia Ultrasensitive Insulin ELISA (Mercodia AB) as per the manufacturer's instructions. All the ELISA data were analyzed by an ELISA reader (Spectra MAX 250, Spectra Devices, Sunnyvale, CA, USA).
Tissue growth factors (TGF-α, TGF-β1, TGF-β2, and TGF-β3) and hepatocyte growth factor (HGF) produced by MSCs were measured by RayBio human growth factor antibody array (RayBiotech, Inc., Norcross, GA, USA). MSCs from three independent donors were maintained in MesenPro medium (Invitrogen) for 2 days, and then the conditioned medium was collected for growth factor array analysis according to the manufacturer's instructions. MesenPro medium served as the control.
Systemic oxidative stress was determined by the ratio of glutathione versus oxidative glutathione (GSH/ GSSG) in whole blood using a commercially available kit (Bioxytech GSH/GSSG-412TM kit, OxisResearch, Portland, OR, USA). Higher GSH/GSSG ratios represented lower systemic reactive oxidative stress levels in the circulation.
Histopathology and Immunohistochemical Staining
Tissues/organs were harvested, fixed in formalin, and prepared in paraffin-embedded blocks for sectioning at the thickness of 3—4 μm. Tissue sections were stained with hematoxylin and eosin (H&E) (Sigma-Aldrich). Besides, tissue sections were incubated with rabbit antibodies against human IgG (1:400, Abcam Inc., Cambridge, MA, USA), rabbit antibodies against mouse insulin (1:400, Cell Signaling, Danvers, MA, USA), mouse antibodies against human insulin (1:100, Abcam), or rabbit antibodies against human c-peptide (1:2000, Abcam) at the room temperature for 1 h, followed by adding goat antibodies against rabbit IgG (Dako Cytomation, Glostrup, Denmark), or goat anti-mouse IgG (Dako Cytomation), for another 40—60 min. Tissue sections were assessed by fluorescent microscopy (Leitz, Germany). Image acquisition was performed with SPOT RT Imaging system (Diagnostic Instruments, Sterling Heights, MI, USA).
Western Blot Analysis
MSCs were cultured in MesenPro medium for 2 days. After removal of the medium, MSCs were washed by PBS twice, and cell lysates were prepared. Three different dosages of protein extracted from MSCs lysate (10, 20, and 30 μg) as well as 20 μg of human insulin (Actrapid HM, Novo Nordisk Inc., Princeton, NJ, USA) were separated on 12% sodium dodecyl sulfate-polyacrylamide gel electrophoresis (SDS-PAGE) and blotted onto polyvinylidene fluoride (PVDF) membrane (Amersham Biosciences, Uppsala, Sweden). After blocking, the membrane was then blotted with indicated primary antibodies: rabbit against human insulin (1:200, Cell Signaling) or mouse against human β-actin (1:10000, Sigma-Aldrich). After washes, the membrane was incubated with the horse radish peroxidase (HRP)-conjugated secondary antibody: goat against rabbit IgG (1: 8000, Epitomics Inc., Burlingame, CA, USA) or rabbit against mouse IgG (1:10000, Sigma-Aldrich) at room temperature for 1 h and the protein intensity was determined by ECL chemiluminescence reagent (PerkinElmer Life Sciences, Inc. Waltham, MA, USA), and their intensities were quantitatively measured by a densitometry (LabWorks, UVP Inc., Upland, CA, USA).
Statistical Analysis
Statistical analyses were performed using the Statistical Package for Social Science-12 software (SPSS Inc., Chicago, IL, USA) or GraphPad Prism 5 (GraphPad Software, Inc., La Jolla, CA, USA). Data of INF-γ and TNF-α were analyzed by ANOVA tests with Tukey's post hoc tests at 95% confidence intervals. Data of blood sugar was analyzed at the same time points by ANOVA tests with Bonferroni's post hoc tests at 95% confidence intervals. Different characters represented different levels of significance. Results of secreted growth factors was analyzed by two-tail, nonpaired t-tests, and values of p < 0.05 were considered statistically significant.
Results
Streptozocin (STZ) Induced Diabetes with Disruption of Islet Structure and Human MSCs Did Not Trigger Immune Reactions in Recipient BALB/c Mice
Intraperitoneal injection of STZ rapidly disrupted the balance of sugar homeostasis in BALB/c mice. It was found that the onset of DM (Fig. 1A) and weight loss (Fig. 1B) occurred within 1 week after STZ treatment. Before STZ injection, insulin producing cells were abundant in the islets (Fig. 1C, D). Two weeks after STZ injection, decreased cell numbers in the islets (Fig. 1E) and the dramatic loss of insulin-producing cells were found (Fig. 1F). Subsequently, β-cell depletion was shown 7 weeks after STZ injection (Fig. 1G, H).

Streptozocin (STZ)-induced diabetes and weight loss by disruption of islet structure and β-cells. (A) Within 1 week of STZ administration, blood sugar was increased by intraperitoneal (IP) injection of STZ (200 mg/kg). (B) Body weight was dramatically decreased after IP injection of STZ. (C) The anatomic structure of islets was normal without STZ injury and (D) stained strongly positive by antibody against insulin. (E) Two weeks after STZ injection, cell numbers in islets were slightly decreased. (F) Moreover, the numbers of insulin-producing cells were markedly decreased. Both the islet structure (G) and β-cells (H) had been markedly destroyed 7 weeks after STZ injection (n = 3).
Human bone marrow-derived MSCs were transplanted via intravenous injection into control and STZ-induced diabetic mice to investigate whether they exerted immune reactions in experimental animals. We monitored the changes of serum INF-γ and TNF-α, two crucial cytokines for innate immune response in acute rejection (15,35) in this xenotransplantation model. It was shown that in the first 3 days after MSC injection, STZ was not immunogenic as INF-γ was only transiently increased in the first 12 h in control mice (Fig. 2A), and TNF-α levels were not altered (Fig. 2B). Around 6 months later before sacrifice, serum level of INF-γ (Fig. 2C) and TNF-α (Fig. 2D) did not show significant difference between diabetic mice and diabetic mice with multiple MSC transplantations. In addition, it is well known that MSCs possess the ability of immunomodulation and may be well tolerated in allogenic transplantation (1,4,7,12,26). Surface antigens of MSCs were analyzed using cell-based array (BD lyoplate); the MSCs we transplanted lacked major histocompatibility complex (MHC) class II (antigen-presenting cell; APC), CD40, CD80, and CD86 expression (the costimulatory factors) and minimal expression of MHC class I (data not shown). Moreover, compared to MesenPro medium, MSCs secreted TGF-α, TGF-β1, TGF-β2, TGF-β3, and HGF (Fig. 2E), the critical factor for inhibition of T cell proliferation (13).

Tolerance of human mesenchymal stem cells (MSCs) by BALB/c mice. Human MSCs (4.2 × 107/kg) were injected via tail vein (IV) into BALB/c mice and mice with previous streptozocin (STZ) injection. (A) MSCs transiently increased the serum level of interferon-γ (INF-γ) in BALB/c mice without STZ injection, and rapidly returned to normal 24 h after injection. (B) MSC transplantation did not increase tissue necrosis factor-α (TNF-α) up to 72 h after injection. Six months after transplantation, multiple MSC transplantation neither altered serum levels of INF-γ (C) nor TNF-α (D). (ANOVA, Tukey's post-hoc tests at 95% confidence intervals, different characters represented different levels of significance, n = 3.) (E) MSCs secreted transforming growth factors (TGFs) and hepatocyte growth factor (HGF). (t-test, *p < 0.05, n = 3.)
Multiple Intravenous MSC Transplantation Gradually Stabilized Blood Glucose But Single Transplantation Only Exerted Transient Effects
To test the effect of MSCs on β-cell-depleted diabetic mice, a single dose of MSCs was injected via tail vein 7 weeks after STZ injection when the vast majority of β-cells and the structure of islets have been destroyed (Fig. 1G, H). According to our previous experimental study, 1.4 × 106 to 4.2 × 107 MSCs/kg body weight could effectively rescue acute lethal fulminant hepatitis (27). In this study, various dosages from 1 × 107 to 7 × 107 MSCs/kg body weight were injected into STZ-induced diabetic mice, and dosages of 3 × 107 MSCs/kg body weight and below failed to reduce plasma glucose (data not shown). Therefore, the dosage of 4.2 × 107 MSCs/kg body weight per injection was chosen.
We found that the concentration of blood sugar only transiently returned to normal levels 2—3 weeks after MSCs injection (Fig. 3A). On the other hand, upon the performance of serial MSC transplantation via tail vein every 2 weeks on STZ-induced diabetic mice, blood sugar gradually decreased after three doses of MSC injection. After seven doses of MSC transplantation, fasting blood sugar level was the same as the control group. Fasting blood sugar in STZ-induced diabetic mice remained high (Fig. 3B).

Gradual stabilization of blood glucose homeostasis and decrease in oxidative stress by multiple MSC transplantations. (A) Single injection of MSCs transiently lowered the blood sugar level in STZ-induced diabetic mice (n = 4). (B) Multiple MSC transplantation at the interval of 2 weeks resulted in the gradual decrease of blood sugar after three doses of MSC injections. (C) STZ not only damaged the islet cells and induced DM, but also increased the systemic oxidative stress. During the first 15 weeks, the gradual reduction of blood sugar by multiple IV MSC transplantation was accompanied by the gradual decrease in systemic oxidative stress. (ANOVA, Bonferroni's post-hoc tests at 95% confidence intervals, different characters represented different levels of significance among the Control, STZ, and STZ+MSCs groups at the same time points, n = 3.)
MSCs Restored Blood Glucose Homeostasis Through Inhibition of Systemic Oxidative Stress at the Early Stage
In our previous study, tail vein injection of human MSCs reduced the systemic oxidative stress elevated by CCl4-induced acute liver injury (27). Based on the previous findings, we first measured the in vivo GSH/GSSG ratio, which inversely represented systemic ROS levels. Similar to what we found with the blood sugar level, systemic oxidative stress in STZ-induced diabetic mice was gradually increased, while systemic oxidative stress decreased after three doses of MSC injection (Fig. 3C).
MSCs Maintained Blood Glucose Homeostasis by Insulin Production at Later Stage
Next, we measured the serum levels of mouse and human insulin. In the control mice, the serum mouse insulin levels were at 10—15 mU/L (data not shown); however, serum mouse insulin was gradually decreased and became undetectable 13 weeks after STZ injection (Fig. 4A). Although human insulin levels were low in the first 10 weeks after MSCs transplantation in diabetic mice, the levels markedly increased from the 11th week upon multiple MSC transplantations (Fig. 4B). At the same time, the blood sugar began to return to the normal range (Fig. 4C).
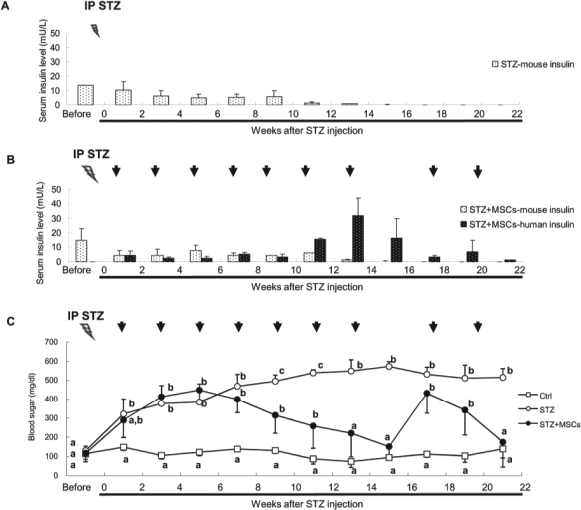
Persistent maintenance of blood glucose homeostasis by insulin production of transplanted MSCs. (A) Mouse insulin was gradually undetectable after STZ injection. (B) Mouse insulin gradually decreased and was no more detectable 13 weeks after STZ injection. However, human insulin became detectable but at very low level in the first 10 weeks, and the concentration of human insulin was dramatically increased from the 11th weeks after multiple rounds of IV MSC transplantation. When MSC transplantation was discontinued, human insulin level was decreased. (C) IV MSC transplantation was discontinued after blood sugar returned to normal level, and rapid rebound of diabetes was noted, which was then controlled by subsequent MSC injection. (ANOVA, Bonferroni's post-hoc tests at 95% confidence intervals, different characters represented different levels of significance among the Control, STZ, and STZ+MSCs groups at the same time points, n = 3.)
To ask whether multiple transplantations are essential, we discontinued the MSC transplantation when blood sugar level returned to normal levels in the 15th week. Without further MSC transplantation, blood sugar levels sharply increased and human insulin decreased. Upon the continuation of MSC transplantations, blood sugar reduced and human insulin production increased (Fig. 4B, C). In this study, serum total cholesterol and creatinine were not affected by STZ and MSC transplantation. However, STZ increased blood urea nitrogen (BUN) and serum TG. Although the elevated BUN could not be reversed by MSC transplantation, serum TG level was decreased and stabilized after three doses of MSC transplantation (data not shown).
Engraftment of MSCs and Differentiation Into Insulin-Producing Cells in Mouse Liver
Six months after multiple MSC transplantations, the mice were sacrificed. Histopathology showed that compared to control mice, pancreatic tissues were damaged by STZ (Fig. 5A, B) and MSC transplantation did not rescue the damaged pancreas tissues (Fig. 5C). Besides, insulin-producing cells could no longer be found in pancreas of diabetic mice (Fig. 5B, C). However, insulin-producing cells could be found in the liver of the diabetic mice after multiple MSC transplantations (Fig. 5D—F) but not in other organs and tissues such as adipose tissues, skeletal muscles, heart, and kidney (Table 1). To distinguish the origin of insulin-producing cells, immunostaining for human IgG and human insulin were performed. As shown in Fig. 6, engrafted human cells as well as human insulin-producing cells could be found in the liver parenchyma, especially near the central vein areas (Fig. 6A, B). Some foci of human cell fragments were noted in the portal vein (Fig. 6A), which also expressed human insulin (Fig. 6B). Moreover, we found that most of the engrafted human MSCs located near the central vein differentiated into insulin-producing cells (Fig. 6C, D); while engrafted cells near the portal vein (Fig. 6E) rarely differentiated into insulin-producing cells (Fig. 6F). We further counted the number of human cells in 12 different high-power fields (HPF, 400x). It was found that the mean number of engrafted human MSCs was 23.6 ± 14.96 cells/HPF, and the mean number of human insulin-producing cells was 12.1 ± 13.40 cells/HPF (Fig. 6G). Most engrafted human cells were mononuclear cells, and only 6% of those cells were double nuclear cells (Fig. 6H). The frequency of double nuclear cells in human insulin-producing cells was similar to that of human IgG expressing cells (Fig. 6I).

Insulin-producing cells found in liver rather than in islets after MSC transplantation. (A—C) Compared to the normal insulin-producing beta islet cells (A), STZ injection resulted in disruption of islets 6 months after STZ injection (B), and could not be reversed by multiple IV MSC transplantations (C). (D—F) In STZ-induced diabetic mice, multiple foci of insulin-producing cells were found in liver after multiple IV MSC transplantations (F), which were not found in liver of control mice (D) or STZ-induced diabetic mice (E).

Human MSCs engraftment into liver and differentiation into insulin-producing cells. (A, B) Human MSCs were engrafted in liver parenchyma and some cell fragments aggregated in the portal vein (PV), evidenced by expression of human IgG (A, white arrow) and human insulin (B, white arrow). (C, D) Most of the human cells were located near the central vein (CV) (C), which expressed human insulin (D). (E, F) However, most of the human cells near the PV (E) did not express human insulin (F). (G) Fifty-one percent of human MSC-derived cells in the recipient liver coexpressed human insulin. (H) Six percent of human IgG-expressing cells were double-nuclear cells. (I) Six percent of human insulin-producing cells were double-nuclear cells.
The Distribution of Human Cells in STZ-Induced Diabetic Mice

Values are the number of histological sections with positive human IgG staining/the total number of histological sections tested. Cell fragment: no viable cells were found but residual cell fragments expressed human IgG.
To rule out the possibility of MSCs picking up insulin from culture medium and then releasing it in vivo, we stained human c-peptide in STZ-induced diabetic mouse liver after multiple MSC transplantation. In line with what we found in human insulin staining, engrafted c-peptide-expressing cells were found in the liver parenchyma (Fig. 7A). Comparing the areas surrounding portal vein (Fig. 7B) and central vein (Fig. 7C), the number of human c-peptide-positive cells was higher in central vein area than in portal vein area. We further checked whether MSCs expressed or picked up human insulin before transplantation, as shown in Figure 7D, MSCs did not produce or contain insulin before transplantation.

Differentiation of MSCs into insulin-producing cells after transplantation in vivo. (A) Human c-peptide-expressing cells were found in liver parenchyma. Most of the c-peptide-expressing cells were located near the CV (B), rather than the PV (C). (D) Before transplantation, MSCs did not express or contain human insulin in culture.
Discussion
For the first time, we demonstrated the importance of multiple transplantations of MSCs to regulate blood glucose homeostasis. Multiple MSC transplantations via intravenous injection restored glucose homeostasis in STZ-induced insulin-deficient diabetic mice, but a single MSC transplantation only exhibited a transient effect on blood sugar reduction. MSCs gradually decreased hyperglycemia after three doses of transplantation, which was associated with reduced systemic oxidative stress. From the 11th week, multiple MSC transplantations stabilized blood sugar by the engraftment of MSCs and subsequent differentiation into insulin-producing cells in the liver.
STZ-induced murine diabetic model has been extensively used (21,25,43,51). In our study, STZ-induced diabetes was indeed a consequence of progressive insulin exhaustion (Fig. 1). However, we noted that upon STZ administration, the damage of insulin-producing cells was not associated with marked local inflammatory cell infiltration in pancreatic islets (Fig. 1E—H); STZ administration did not result in alteration of systemic inflammatory cytokine levels such as TNF-α and INF-γ either (Fig. 2A—D). These observations suggest that STZ-induced diabetic model is not the equivalent of T1DM. Also, whether MSC transplantation works on autoimmune-induced DM requires further investigation.
Unsurprisingly, human MSCs did not induce an inflammatory response in recipient mice (Fig. 2), indicating that MSCs possessed low immunogenicity and the transplanted MSCs were tolerated in this xenotransplantation model. For the DM patients who are not suitable for harvesting autologous MSCs, allogenic MSCs may therefore be an alternative choice.
In this study, blood glucose homeostasis could not be maintained by a single MSC transplantation and blood sugar level rebounded 1 month after a single MSC transplantation (Fig. 3A). Multiple transplantations gradually stabilized blood glucose homeostasis (Fig. 3B), and the change of sugar level was in line with that of systemic oxidative stress in the first 15 weeks (Fig. 3C). In the first 10 weeks after STZ injection, both mouse and human plasma insulin levels were low (Fig. 4B), but sugar level was gradually reduced (Fig. 4C) after multiple MSC transplantation, suggesting that reduction of systemic oxidative stress may contribute the blood sugar homeostasis in the early stage when the production of insulin of transplanted cells is still insufficient (Fig. 3B, C). Moreover, gradual increase in human insulin levels from the 11th week after a series of human MSC transplantations indicated that insulin production from MSC-differentiated cells restored blood glucose homeostasis in STZ-induced diabetic mice at this later stage (Fig. 4B, C). Although serum levels of TNF-α and INF-γ were not changed by multiple MSC transplantations (Fig. 2C, D), we still could not rule out the possibility of anti-human antibodies produced by BALB/c mice in the absence of TNF-α or INF-γ increase in this xenotransplanted model. Consequently, high dosage of MSCs and multiple transplantations were used in this study. To elucidate the role of multiple transplantations in regulation of blood glucose homeostasis, we discontinued MSC transplantation after the sugar level stabilized. As shown in Figure 3, when MSC transplantation was discontinued, a decrease in human insulin (Fig. 4B) and a sharp rise in blood sugar (Fig. 4C) within 1 month, illustrated that continuous systemic MSC transplantation was indispensible. After further transplantation, blood sugar could be restabilized and human insulin level was detectable and remaining relatively low, suggesting the possibility of increased insulin sensitivity in recipients after a series of MSC transplantations. Since insulin-producing cells differentiated from MSCs play a crucial role for regaining blood glucose homeostasis, the niche that allows long-term engraftment of MSCs is equally important. It is known that stem cells can find their own niches in vivo (46). In many animal models of injury, MSCs can home and engraft into damaged tissues (8,10,19,41). In this study, we have detected the existence of human β2 microglobulin DNA to distinguish human cells from mouse cells by PCR. However, human cells were barely detectable in the pancreas (data not shown), showing that the microenvironment in the pancreas was not suitable for MSCs after STZ treatment. This was further confirmed by histopathology since no insulin-producing cells could be found in the diseased pancreas after STZ treatment (Fig. 5B, C).
Besides the pancreas, we found that the liver has provided a favorable environment for MSCs to engraft (Fig. 6), evidenced by multiple human IgG-positive cells in the murine liver (Fig. 6A, C, E). The finding of human insulin- as well as human c-peptide-positive cells in the recipient liver indicated that the insulin-producing cells were of donor origin (Figs. 6B, D, F, and 7A—C). Moreover, the finding that MSCs did not contain human insulin before transplantation has ruled out the possibility of insulin ingestion in culture followed by subsequent release after transplantation (Fig. 7D). Clinically, liver parenchyma is the niche for pancreatic islets to engraft in islet cell transplantation (44,47,48,49). In our study, we demonstrated that 51% of the transplanted human cells in the recipient liver parenchyma expressed insulin (Fig. 6B, G), illustrating that half of MSCs in diabetic liver could differentiate into insulin-producing cells. These human insulin-producing cells aggregated near the central vein (Figs. 6D and 7B) rather than the portal vein (Figs. 6F and 7C), suggesting that the environmental cues in the central vein areas were crucial for insulin-producing cell differentiation. However, the differentiation of human MSCs into fully functional β-islet cells has yet to be proven.
Fusion of donor and host cells is always an important issue for adult stem cell plasticity (24). It has been reported that hematopoietic stem cells develop into hepatocytes through cell fusion (18). In this study, the frequency of human- and mouse-cell fusion was very low (Fig. 6H, I), showing that cell fusion was not the main mechanism when MSCs engrafted into liver. Besides, the percentage of cell fusion in human IgG-expressing cells (Fig. 6H) and human insulin-producing cells (Fig. 6I) were the same, indicating that the role of cell fusion for MSCs is negligible during differentiation into insulin-producing cells in liver.
In summary, we concluded that multiple transplantations of MSCs were essential to effectively restore and maintain blood glucose homeostasis in the preclinical model of insulin-insufficient diabetes due to β-cell depletion. Liver offered a favorable microenvironment to support long-term MSC engraftment and differentiation into insulin-producing cells. Multiple intravenous MSC transplantations may therefore serve as a new therapeutic strategy for DM patients.
Footnotes
Acknowledgments
The authors acknowledge financial support from the Wan Fang Hospital, Taipei Medical University (100swf03, to J.H.H.), as well as the support of research grants from the Taipei Veterans General Hospital (V99E1-014, V99C1-097, and V99S4-001, to O.K.L.), and the National Science Council (NSC-99-2120-M-010-001, to O.K.L. and J.H.H.; NSC98-2314-B-010-001, NSC98-2627-B-010-004, and NSC98-3111-B-010-003, to O.K.L.; and NSC98-2314-B-038-010, to J.H.H.). The authors also acknowledge the research grant support from Steminent Biotherapeutics Inc. (Grant number: W184 to JHH). This study was under the support of the Ministry of Education, Aim for the Top University Plan. The animal experiments were assisted in part by the Division of Experimental Surgery of the Department of Surgery, Taipei Veterans General Hospital. The authors declare no conflict of interest.
