Abstract
Experimental treatment strategies using human umbilical cord blood mononuclear cells (hUCB MNCs) represent a promising option for alternative stroke therapies. An important point for clinical translation of such treatment approaches is knowledge on the therapeutic time window. Although expected to be wider than for thrombolysis, the exact time window for hUCB MNC therapy is not known. Our study aimed to determine the time window of intravenous hUCB MNC administration after middle cerebral artery occlusion (MCAO). Male spontaneously hypertensive rats underwent MCAO and were randomly assigned to hUCB MNC administration at 4, 24, 72, and 120 or 14 days. Influence of cell treatment was observed by magnetic resonance imaging on days 1, 8, and 29 following MCAO and by assessment of functional neurological recovery. On day 30, brains were screened for glial scar development and presence of hUCB MNCs. Further, influence of hUCB MNCs on necrosis and apoptosis in postischemic neural tissue was investigated in hippocampal slices cultures. Transplantation within a 72-h time window resulted in an early improvement of functional recovery, paralleled by a reduction of brain atrophy and diminished glial scarring. Cell transplantation 120 h post-MCAO only induced minor functional recovery without changes in the brain atrophy rate and glial reactivity. Later transplantation (14 days) did not show any benefit. No evidence for intracerebrally localized hUCB MNCs was found in any treatment group. In vitro hUCB MNCs were able to significantly reduce postischemic neural necrosis and apoptosis. Our results for the first time indicate a time window of therapeutic hUCB MNC application of at least 72 h. The time window is limited, but wider than compared to conventional pharmacological approaches. The data furthermore confirms that differentiation and integration of administered cells is not a prerequisite for poststroke functional improvement and lesion size reduction.
Keywords
Introduction
Ischemic stroke is one of the most frequent causes of death and the most common reason for permanent disabilities in adults throughout industrialized nations (16). Despite vigorous research during the last decades, treatment opportunities are limited. The only approved therapy to date, vessel recanalization by recombinant tissue plasminogen activator, is limited by a narrow time window of only 4.5 h (19). Within this time window therapeutic efficacy is reduced continuously, accompanied by an increasing probability of serious complications such as hemorrhages (20). Consequently, there is a strong demand for alternative strategies, in particularly for approaches expanding the time for intervention. Among these, transplantation of neonatal or adult stem cell-containing populations seems promising (28,41). Therapeutic effects of intravenous administration of human umbilical cord blood mononuclear cells (hUCB MNCs), the most important source of neonatal stem cells, have already been demonstrated in rodents (7). Later studies confirmed beneficial impact of hUCB MNCs when transplanted between 8 and 48 h upon stroke onset, as reviewed by Yu and colleagues (42). However, it is still unclear whether or not there is a time window for an effective hUCB MNC transplantation. This hypothetical time window is of particular interest for clinical translation. It may be assumed that the therapeutic benefit of hUCB MNC administration following stroke is in fact transient, as integration and neuronal differentiation of grafted cord blood cells in the recipient central nervous system (CNS) seems to be a very rare event (7). Moreover, these processes are possibly not even related to functional improvement and reduction of ischemic lesion size (6). Therapeutic effects of transplanted hUCB MNCs are more likely mediated by the production of molecular mediators in a neuroprotective, antiapoptotic, or immunomodulatory manner (14). These mechanisms in turn are bound to specific temporal and spatial kinetics of pathophysiological processes following stroke (10). This emphasizes the importance of the appropriate transplantation time point. The current study investigates the time-dependent efficacy of a hUCB MNC treatment following experimental brain ischemia.
Materials and Methods
Experimental Subjects
All experiments were performed in accordance with the European Communities Council Directive (86/609/EEC) and with local guidelines for care and use of experimental animals. Animals were housed under constant conditions of temperature (22°C), humidity (80%), and light (12/12-h light/dark cycle). A total of 51 male spontaneously hypertensive rats (SHR, Charles River, Germany), weighting 200–250 g were used in this study.
Cell Sources and Preparation
Cord blood samples from healthy full term neonates were donated after normal pregnancy and delivery for scientific purposes. Donation was approved by the ethics committee of the University of Leipzig. hUCB MNCs were isolated from cord blood samples by density gradient centrifugation (in vivo study), or cryopreserved, stored, and thawed (in vitro study) as described elsewhere (32). Prior to experiments, cells where prelabeled using the fluorescence dye PKH26 (Sigma, USA) according to the manufacturer's instructions, centrifuged at 1250 rpm for 5 min at 4°C, and washed three times with phosphate-buffered saline (PBS). After the final wash, cell viability was assessed using 0.4% trypan blue in a Neubauer counting chamber to exclude dead cells and to ensure an adequate number of vital cells for transplantation or cocultivation. For in vivo transplantation, volume was adjusted for a total amount of 1 × 106 hUCB MNCs per 1 ml and injected immediately. For direct cell application on postischemic hippocampal slices, 2.5 × 104 hUCB MNCs were suspended in 1 μl Neurobasal-A medium and applied on slices.
Induction of Stroke and Cell Administration
Forty-eight animals were subjected to permanent middle cerebral artery occlusion (MCAO) and randomly assigned to six experimental groups (n = 8 each): intravenous transplantation of 1 × 106 hUCB MNCs at 4, 24, 72, and 120 h, or 14 days following stroke onset. Controls received 1 ml PBS after 24 h. Animals were anesthetized with ketamine hydrochloride (100 mg/kg, Merial, Germany), xylacine (10 mg/kg, Bayer, Germany), and atropine (0.1 mg/kg, Ratiopharm, Germany) given as an intraperitoneal injection. Experimental stroke was induced by permanent distal (cortical) MCAO, as described previously (17). During surgery, core body temperature was continuously monitored and maintained using a feedback controlled heating pad connected to a rectal probe (Fine Science Tools, Germany). Heads of the animals were fixed in a flexible face mask over which O2-enriched air was applied. For analgesia postsurgery rats received 25 mg Metamizole-Na (Ratiopharm) in 40 ml of 5% glucose drinking solution for at least 5 days following MCAO. One subject from the 72-h group died during the surgical procedure. Cell suspension or vehicle solution was injected via intravenous catheter into a lateral tail vein within one minute. All experimental procedures were performed by investigators blinded to group allocation.
For histological positive control of intracerebrally located PKH26-labeled cells, three additional rats were used. These animals received intracerebral transplantation of PKH26-labeled hUCB MNCs. Two depots containing 5 μl of cell suspension (with 1.5 × 105 hUCB MNCs in each depot) were transplanted using a stereotaxic frame (TSE Systems, Germany) to the following coordinates: (i) bregma cranial-caudal: −0.2 mm; right temporal: 3.3 mm; depth: 4.0 mm; and (ii) bregma cranial-caudal: −0.2 mm; right temporal: 3.3 mm; depth: 4.4 mm. Control animals were not assessed by behavioral phenotyping or magnetic resonance imaging (MRI).
Behavioral Phenotyping
Sensorimotor functions were regularly assessed using the BeamWalk and modified Neurological Severity Score (mNSS) tests following MCAO. In the BeamWalk test rats had to cross a narrow beam (14 mm diameter, 1 m length) at the end of which the animal's home cage was placed (18). A score of 20 s was recorded if the animals hugged the beam, and 30 s were recorded when they fell off the beam. After a training phase of 7 days prior to surgery, baseline values were recorded as the mean of five trials. The mNSS was performed according to Chen and colleagues (7). Animals were investigated using both tests before experimental stroke for presurgical deficits and after MCAO on days 1, 4, 7, 11, 15, 19, 23, and 27.
To reveal possible differences between groups treated at 4, 24, and 72 h and controls, subjects were further investigated using the RotaRod (TSE Systems, Germany) on days 1, 2, 4, 7, 11, 17, 23, and 29 (7) following a 4-day training phase and baseline assessment. The rod was continuously accelerated from 5 rpm and final rotation speed (40 rpm) was reached after 180 s. A trial ended when the animals fell off the rung or after remaining on the rod for more than 300 s. Mean was calculated from five individual trials per day. Animals were allowed to recover in their home cages for at least 3 min before a new trial was started. Variables measured were quoted as percentage of baseline values.
Magnetic Resonance Imaging (MRI) and Data Analysis
Five animals of each group treated at 120 h or less and controls were randomly selected to undergo MRI investigations at 23 h and on days 8 and 29 following MCAO. Animals were anesthetized intraperitoneally as described above and placed in a 1.5 T MRI scanner (Gyroscan Intera human whole body spectrometer, Philips, The Netherlands) equipped with a small loop radiofrequency (RF) coil (47 mm; Microscopy Coil, Philips). T2-weighted and T2*-weighted [T2*-fast field echo (FFE), to exclude intracerebral bleedings] sequences were performed at each MRI session, consisting of 20 transverse slices (matrix: 224 × 224; field of view: 50 mm; slice thickness: 1 mm). All sequences were measured as turbo-spin echo sequence. Data set evaluation was performed on coronal T2 weighted images using ImageJ (1). Volumes of the lesion, of the ipsilateral and the contralateral side ventricles, and of both hemispheres were assessed. Ischemic tissue loss was indicated by brain atrophy being calculated as follows: the quotient between the size of the ipsilateral hemisphere minus the infarct and the associated ventricle and the contralateral hemisphere subtracted by the contralateral ventricle was determined. For MRI lesion volumetry, a group comparison approach for day 8 and 29 was performed.
Relocalization of Transplanted hUCB MNCs and Appraisal of Glial Reactivity
On day 30, animals were deeply anesthetized and transcardially perfused with 200 ml PBS for 10 min, and thereafter with 300 ml 4% paraformaldehyde (PFA) for 15 min. Brains were carefully removed and fixed in 4% PFA followed by storage in ascending concentrations of sucrose (10%, 20%, 30%) for 24 h each, and finally stored at −80°C. The cryopreserved brains were cut in 30-μm-thick slices and mounted on Superfrost® slides (Resolab, Germany). Immunohistochemistry was performed for astrocytes, using mouse anti-glial fibrillary acidic protein (GFAP, 1:100; Sigma) and for human cells using mouse anti-human nuclei (HuNu (MAB1281), 1:50; Millipore, Germany) as primary antibodies according to manufacturer instructions. The primary antibody was incubated with the slices in 0.9% PBS-Tween and 10% fetal cow serum (FCS) for 24 h at 4°C. Thereafter, slices were washed and incubated with a secondary antibody conjugated with fluorescence isothiocyanate-conjugated anti-mouse (FITC, 1:200; Sigma-Aldrich, for GFAP) or cyanine 3 (Cy3, 1:200, Sigma-Aldrich, for human nuclei), 0.9% PBS-Tween, and 10% FCS at room temperature for 1 h. After the final wash, the slides were air-dried and cover-slipped. Nuclei were counterstained with DAPI (Sigma-Aldrich) for colocalization of PKH26 signals with cells in the host brain. Slices were screened using an Axioplan fluorescence microscope (Carl Zeiss, Germany) and, for detection of intracerebrally located cells, using a Zeiss LSM710 confocal laser scanning microscope (Carl Zeiss). Spleen slices from animals that did not receive hUCB MNCs served as negative controls.
For appraisal of glial reactivity, the lesioned hemisphere was photographed up to 750 μm from the edge of the infarct border using Axioplan fluorescence microscope (Carl Zeiss). GFAP-positive cells were quantified in this area using an automated counting software (Axiovision, Carl Zeiss), and quoted in to the exact size of the investigated area (events/mm2).
hUCB MNC Cocultivation with Oxygen/ Glucose-Deprivated Hippocampal Slice Cultures
Organotypic hippocampal slice cultures were prepared from postnatal Wistar rats (P7-P9; Harlan Winkelmann, Germany) as described previously (31). Briefly, hippocampi were dissected from brains and transversely cut into 350 μm thick sections. Slices were maintained in 1 ml serum-based medium (50% MEM-Hanks, 25% HBSS, 17 mM HEPES, 5 mM glucose, 1 mM l-glutamine, 25% horse serum, and 0.5% gentamycin) at 37°C for 3 days and subsequently transferred to serum-free medium (50% MEM-Hanks, 25% HBSS, 17 mM HEPES, 5 mM glucose, 1 mM l-glutamine, 25% Neurobasal-A, 0.5% B27, and 0.5% gentamycin) and kept at 37°C for further 7 days. Oxygen glucose deprivation (OGD) was started by transferring the slices into 1 ml glucose-free Ringer solution containing 10 mM mannitol. Oxygen deprivation was induced by incubating the slices under 95% N2/5% CO2 for 50 min at 37°C in a gas-tight chamber. Normoxic control cultures were maintained for the same time in glucose-containing Ringer solution. Following OGD, slices were transferred to normoxic culturing conditions and cocultured with 2.5 × 104 hUCB MNCs in 1 μl Neurobasal-A medium, given directly onto the slices.
Analysis of PI Incorporation and Caspase-3 Expression
Propidium iodide (PI, 2 μg/ml; Sigma) was added to the culture medium 24 h prior to the experimental use. Only PI-negative slices were selected for OGD. In the experimental course, PI was added to the culture medium at 22, 46, or 70 h after OGD and slices were densitometrically analyzed 2 h after PI was added. The evaluation of caspase-3 expression was performed in formalin-fixed und cryosectioned slices (4% formalin for 45 min, 20 μm). Briefly, slices were incubated with rabbit anti-active caspase-3 antibody (1:5000; PBS/10% FCS, R&D Systems, Germany) for 48 h at 4°C. As secondary antibody biotinylated goat-anti-rabbit (1:500; PBS/10% FCS Dianova, Germany) was added for 1 h. Subsequent to the reaction with alkaline phosphataseavidin-biotin complex (1:400; Vectastain Elite ABC-kit, Vector Laboratories, USA), the complex was visualized by 3′,3-diaminobenzidine tetrahydrochloride (DAB, Dako, Germany). Slices were air dried and counterstained with standard Nissl method. Quantification of caspase-3-positive cells was performed in photographes of the dentate gyrus (DG) using Zeiss Axiovision microscopy image analyzing software (Carl Zeiss).
Statistical Analysis
Statistical analyses were performed by an independent biostatistician using the SPSS statistical software (Version 11.5.1). Data were tested for Gaussian distribution. Normally distributed data of two independent variables were analyzed using Student's t-test (MRI data sets, experiments with organotypic slices). Ill behaved data (all other data sets) were subjected to the Mann-Whitney test. ANOVA or ANOVA on ranks were used in case of three or more variables. Post hoc analysis was performed using Tukey's test. Statistical significance was considered for values of p < 0.05 according to standard conventions and indicated by single symbols (*, #). For values of p < 0.01 double symbols (**, ##) are shown. All data are presented as mean ± SD.
Results
Transplantation of hUCB MNCs Within 72 h Induces Functional Recovery
Statistical analysis of functional deficits before and 1 day after stroke onset displayed uniform experimental groups. All groups showed a considerable reduction of sensorimotor abilities compared to baseline in all behavioral tests applied (p < 0.01), but time course and amount of regression were different between the groups. Defect amelioration after MCAO appeared homogenously within experimental groups treated after 4 (Fig. 1A, B), 24 h (Fig. 1C, D), and 72 h (Fig. 1E, F). Those subjects showed an early onset of sensorimotor function amelioration within 4–7 days after hUCB MNC administration, leading to sustained recovery in all tests (p < 0.05) (Fig. 1A–F). Although showing lower mean values compared to controls, cell administration at 120 h (Fig. 1G, H) and 14 days (Fig. 1I, J) after MCAO did not induce statistically significant functional benefits in the BeamWalk. In the mNSS test, a slight recovery was detected at days 23 and 27 (p < 0.05) (Fig. 1H). However, both groups already differed significantly at day 1 (p = 0.04) and 4 (p = 0.01) post-MCAO, but not thereafter until day 23, with the control group showing worse outcome. Thus, the particular group results have to be interpreted very cautiously.

Effect of human umbilical cord blood mononuclear cell (hUCB MNC) administration in the BeamWalk and modified Neurological Severity Score (mNSS) tests. Baseline values and postoperative data at day 1 did not show statistically significant differences between the groups. Cell transplantation within 72 h (A, B: 4 h; C, D: 24 h; E, F: 72 h) after middle cerebral artery occlusion (MCAO) improved functional recovery (p < 0.05) in both tests. In the BeamWalk test, animals reached baseline values by the end of the experimental period. Delayed cell transplantations at 120 h (G, H) and day 14 (I, J), despite minor improvements in the mNSS test in the late testing stage after hUCB MNC application at 120 h, failed to show efficacy. In this regard, please note significant differences in the mNSS 120 h control group on days 1 and 4 (H). *p < 0.05 versus controls.
The RotaRod test was performed to assess possible slight differences in recovery after cell administration within 72 h (Fig. 2A–C) in comparison to control treatment and late cell administration (day 14) (Fig. 2D). In accordance to BeamWalk and mNSS testing, cell administration was effective within 72 h (p < 0.05), but not thereafter. Animals that received hUCB MNCs 24 h after MCAO tended to stay on the RotaRod compared to animals that received cells at all other injection time points (Fig. 2B). However, this difference was not statistically significant.
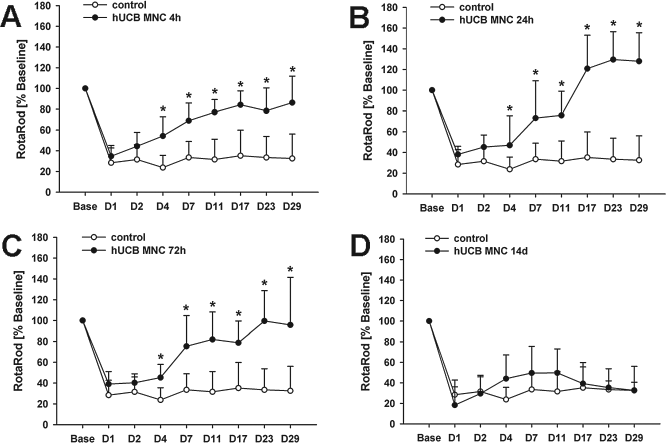
Effect of hUCB MNC administration in the RotaRod test. The RotaRod test was used to detect differences between groups treated successfully by hUCB MNCs within 72 h (A: 4 h; B: 24 h; C: 72 h) in comparison to control treatment and late cell application (D: 14 days). Similar to the BeamWalk results, transplantation of hUCB MNCs at 4, 24, or 72 h effectuated a significant improvement of functional defects (p < 0.05). This was observed within 3 days (p < 0.05) upon cell administration. Later cell therapy (day 14) did not show statistically significant differences compared to the control group. Despite best mean performance of animals treated after 24 h from day 17 on, no statistical difference was evident to hUCB MNC injection at 4 and 72 h. *p < 0.05 versus controls.
Timely Administration of hUCB MNCs Reduced Brain Atrophy Rate After Experimental Stroke
Morphologically, all groups showed a homogenous diffusion disturbance, indicating the lesion at 23 h following MCAO in MRI (Fig. 3A). At day 29, control animals and subjects that received hUCB MNCs at 120 h and later following MCAO showed a more pronounced enlargement of side ventricles (Fig. 3B, white arrows), indicating more extensive brain atrophy.
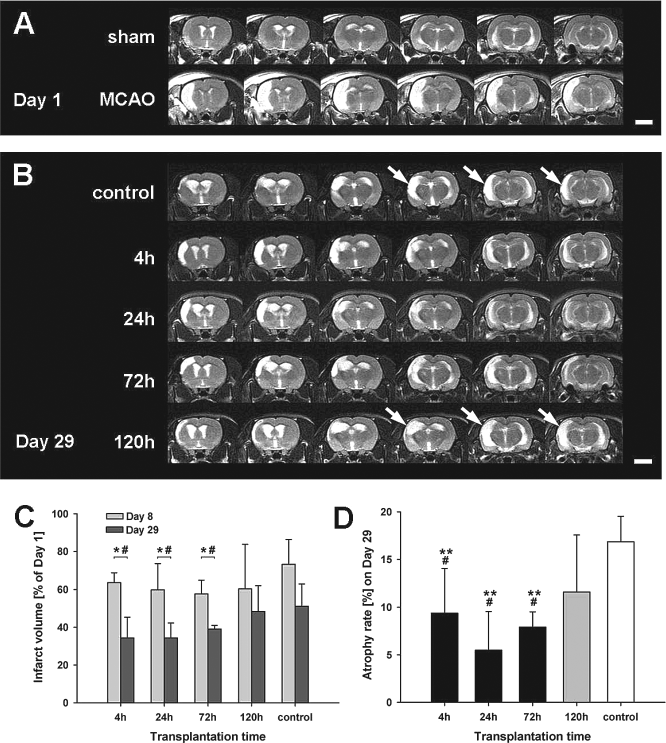
MRI assessment of lesion development. Exemplary T2-weighted sequences of untreated MCAO animals taken at day 1 after MCAO showed distinct T2 hyperintense regions within the right cortex. Those regions were not detectable in sham-operated subjects (A). Morphological MRI data analysis revealed a more pronounced enlargement of lateral ventricles in control animals and those subjects treated 120 h after MCAO on day 29 (B, white arrows). There was no difference in lesion size reduction between day 1 and day 8 in any group (C). Animals treated with hUCB MNCs showed a statistically significant reduction of lesion size between day 8 and day 29 (C). Significant decrease of brain atrophy was detected in groups transplanted 4, 24, or 72 h following MCAO compared to controls (p < 0.01) and the transplantation at 120 h (p < 0.05). No difference in brain atrophy was observed between groups treated at 4, 24, or 72 h (p > 0.05) at day 29 (D). Scale bars: 5 mm. *p < 0.05 versus control; **p < 0.01 versus control; #p < 0.05 versus 120 h.
Lesion volumetry revealed no differences in lesion size between the groups at day 1 (data not shown). Group comparison of lesion volumetry on later days showed a pronounced reduction of lesion sizes in groups that received hUCB MNCs within 72 h after stroke (p < 0.05) (Fig. 3C), but not thereafter. Further, analysis of MR data sets revealed a significantly smaller brain atrophy on day 29 within the experimental groups 4 h (9.4 ± 4.7%), 24 h (5.4 ± 4.1%), and 72 h (8.4 ± 3.0%) compared to the control group (16.9 ± 2.7%, p < 0.01) and subjects treated at 120 h after stroke onset (11.6 ± 6.0%, p < 0.05) (all Fig. 3D). Although a clear mean difference between the group receiving hUCB MNCs at 120 h after stroke onset and controls, a statistically significant difference does not exist between these groups (p > 0.05). We did neither observe any signs of tumor formation nor secondary bleedings at any day in any group (data not shown).
Intravenously Injected hUCB MNCs Were Not Detected in Host Brains After 30 Days
Intracerebrally located PKH26-positive cells were observed 30 days after local cell administration in the positive-control group (Fig. 4A). In contrast, PKH26-positive signals were not observed in brain specimens of animals from all experimental groups that received intravenous hUCB MNC transplantation (Fig. 4B). Although very rare, red fluorescence signals were observed in single slices, no PKH26-like fluorescence signal was found in close spatial relation to DAPI-stained nuclei. These signals were therefore considered as scattered autofluorescence. PKH26-positive signals after local hUCB MNC injection were not located within astrocytes (Fig. 4C). We alternatively used an antibody against human nuclei to locate hUCB MNCs in the brain. However, this was not successful as unspecific staining was observed in negative controls (spleen slices of rats that did not receive human cells) (Fig. 4D, E), prohibiting a reliable detection of intracerebrally located hUCB MNCs. Summarizing, there was no valid evidence for graft-derived resident cells in host brains after intravenous application.
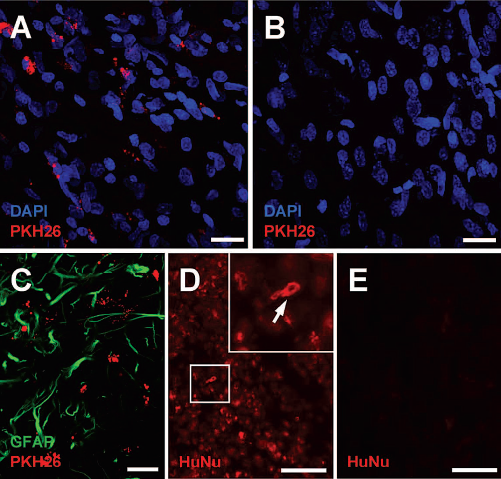
Failure of detection of intracerebrally located hUCB MNCs after intravenous administration. The intracerebral PKH26-signal was identified in close relation to DAPI-stained nuclei (A) 30 days after local stereotaxic administration. However, PKH26-positive cells could not be observed 30 days after intravenous application of hUCB MNCs (B). Intracerebral PKH26-signals were not located in astrocytes (C). Further attempts to identify the hUCB MNCs using antibodies directed against human nuclei (HuNu) were not successful as control staining reproducibly led to false-positive results. In particular, after applying an antibody against human nuclei (MAB1281) in spleens of animals that did not receive human cells, unspecific signals were even observed in cell somata (D, white arrow in inset), thus questioning the specificity of the particular antibody batch used. Omission of primary antibodies did not lead to any staining (E) in those slices. (A), (B), and (C) represent superpositions of 20 μm confocal z-stacks. Scale bars: 20 μm (A–C) and 50 μm (D, E).
Decreased Glial Reactivity After hUCB MNC Administration Within 72 h
The investigation of glial reactivity 30 days upon MCAO displayed statistically significant differences in the amount of GFAP-positive cells within a 750-μm broad area adjacent to the infarct (p < 0.01) (Fig. 5A). The density of GFAP-positive cells was significantly reduced in subjects treated 4, 24, and 72 h following MCAO compared to the 120 h (p < 0.01) (Fig. 5B) and control groups (p < 0.01) (Fig. 5B). The number of GFAP-positive cells was not significantly altered in animals treated with hUCB MNCs at 120 h compared to the control group (Fig. 5A). Interestingly, diminished astroglial reactivity was not observed in a small strip directly bordering the central necrosis (Fig. 5C–G).

Effect of hUCB MNC transplantation on gliosis. Glial scar analysis was performed by quantitative evaluation of glial fibrillary acidic protein (GFAP)-positive events within a 750-μm broad area close to the infarct border (A). The density of GFAP-positive cells is given in (B). Frame from (A) is given with higher magnification in (C) to (G). Reactive gliosis was significantly higher in subjects treated with hUCB MNCs at 120 h (C) and controls (D) compared to those treated at 4 (E), 24 (F), or 72 h (G). **p < 0.01 versus controls; ##p < 0.01 versus 120 h. Scale bars: 100 μm (A, C–G).
Cocultivation with hUCB MNCs Reduced Neural Necrosis and Apoptosis in Hippocampal Slices
On each day of investigation OGD injured organotypic hippocampal slice cultures showed significantly increased leveles of PI incorporation, indicating necrosis compared to the normoxic control slices (Fig. 6A). However, cocultivation with hUCB MNCs caused a significant reduction of PI incorporation at days 1, 2, and 3 (p < 0.05) (Fig. 6A) with a level of necrosis being comparable to normoxic controls.

hUCB MNCs attenuate necrosis and apoptosis after oxygen glucose deprivation (OGD) in hippocampal slices. Following OGD, increased levels of necrosis (A) and apoptosis (B) were observed in hippocampal slices. Application of 2.5 × 104 hUCB MNCs immediately after OGD decreased amount of necrotic cells over the entire observation period (A, p < 0.05). Cell application transiently decreased caspase-3 expression as a marker for apoptosis on days 1 and 2 (B, p < 0.05). Representative micrographs of propidium iodide (PI) uptake (C to C″) and caspase-3 staining (D to D″) are given for day 2. Indicated by the fluorescence signal, the overall amount of necrotic cells was reduced in hippocampal slices (C) and only few caspase-3-positive cells were observed in the dentate gyrus after hUCB MNC application (D″, white arrows). *p < 0.05 versus OGD. Scale bar: 500 μm (C″) and 100μ (D″).
The investigation of caspase-3 activity showed a distinct increase of caspase-3-positive apoptotic cells within the DG following OGD at days 1, 2, and 3 (Fig. 6B). Number of caspase-3-postive apoptotic cells was significantly smaller in normoxic control slices during the entire observation period and in hUCB MNC-treated hippocampal slices at days 1 and 2, but not at day 3 (Fig. 6B). Representative micrographs of PI incorporation (Fig. 6C–C″), caspase-3 staining (Fig. 6D–D″) after OGD, normoxic cultivation, and OGD slices in coculture with hUCB MNCs are given for day 2.
Discussion
Time Dependency of Functional Recovery After hUCB MNC Application
Sensorimotor assessment using BeamWalk and mNSS showed a significant functional improvement when hUCB MNCs were administered within 72 h after MCAO. In animals treated after 120 h upon MCAO onset, delayed functional improvement was detected in the mNSS. Later administration completely failed to induce recovery. Previous studies gave clear indications for a wider time window of hUCB MNC application compared to thrombolysis (7), ranging up to 48 h (24). However, even longer time windows exist for effective transplantation of other adult cell populations after experimental stroke. For bone marrow stromal cells, also being a promising option for stroke therapy (33), a therapeutic time window of even 1 month was reported (29), raising the question whether there is a comparable window for hUCB MNC treatment. The behavioral data obtained in this study for the first time clearly indicate a time frame of at least 72 h for beneficial hUCB MNC administration after experimental stroke. In addition, comparison of RotaRod results of groups that received treatment within 72 h after MCAO did not reveal any superiority of a certain transplantation time point. However, transplantation after 24 h resulted in best mean performance in the RotaRod from day 17 and in the mNSS from day 7 onwards. Although not being statistically significant, this tendency may indicate 24 h as the optimal time point for transplantation. The number of subjects per group might be too small to reveal small effect differences at a statistically significant level. Nevertheless, the observed time window is considerably wider than the time frame of conventional pharmacological approaches, particularly thrombolysis.
Sustained Functional Recovery Is Related to Lesion Size Reduction But Not to Cell Engraftment
In accordance with results of neurofunctional tests, there was a clear decrease of brain atrophy measured by MRI at day 29 in all groups that received hUCB MNC treatment within 72 h after MCAO. Brain atrophy reduction was not observed after later cell administration and sustained behavioral improvement was absent in these groups. Although neural differentiation of hUCB cells was previously shown (43), graft-derived cells were not reliably detected in brain specimens in this study. Several other groups also reported beneficial effects of hUCB MNC administration at singular time points after stroke without cerebral homing of grafted cells (6,38). Some authors even presume that central nervous system entry of administered cells is not needed to induce beneficial effects (6). Thus, neurofunctional improvements could also be explained by absence of administered cells at later stages and may be related to processes outside the brain (38). It might be assumed that reduction of lesion size in relation to late transplantation and controls, obviously not based on differentiation and integration of grafted cells in a larger scale, could be due to prevention of acute or delayed cell death in the recipient brain.
Potential Role of Antiapoptotic Processes for Lesion Size Reduction
Necrotic cell death following stroke is an immediate event taking place in the ischemic core within the first 4–12 hours (4,12), leaving only limited time for successful intervention. Cell death at later time points is mostly determined by antiapoptotic mechanisms, particularly in the ischemic penumbra (5). The in vitro investigation in this study showed that hUCB MNCs can strongly diminish apoptosis and necrosis in postischemic hippocampal slices. Similar effects of hUCB MNCs had been described in other model systems (13,14) and the antiapoptotic effect itself is likely mediated by the release of neurotrophic factors and recovery enhancing effects from the grafted cells (11). Apoptotic processes following focal cerebral ischemia in rats occur within 72 h after vessel occlusion (35,39). Thus, prevention of delayed cell death may result in reduced lesion volumes and brain atrophy at later time. However, MRI-based lesion size comparison did not detect any differences in lesion size between the groups at day 8. Albeit relevant antiapoptotic capabilities of hUCB MNCs in comparison to hUCB stem cell fractions were found in vitro (14,25), assessment of impact of hUCB MNC administration 24 h after MCAO on lesion size and caspase-3-dependent apoptosis during the first 96 h following MCAO did not reveal a statistically significant effect (26). Thus, antiapoptosis in penumbral tissue may contribute to lesion size reduction to a minor extent but is unlikely the sole reason for decreased brain atrophy at day 29, which may also be attributed to other processes diminishing cerebral tissue loss after stroke at later stages. It may further be concluded that novel in vitro systems providing a higher predictive power for the in vivo situation are demanded.
Further Beneficial Effects of hUCB MNC Application After Stroke
In fact, a clear regenerative potential beyond preventing necrosis in acute and after ischemic conditions was reported for hUCB MNCs. Cells were able to increase oligodendrocyte survival (27), diminish peripheral inflammatory (21), or microglial (15) response and stimulated neurogenesis (3). Moreover, hUCB and in particular MNC subfractions promote angiogenesis (37) and may prevent oxidative damage (2). These processes can lead to an overall reduction of lesion size and behavioral improvements also at alter stages of cerebral ischemia, but might need relatively early induction following stroke.
Possible Explanation of the Time Window by Diminishing Gliosis
Transplantation of hUCB MNCs influenced the formation of reactive gliosis. Quantification of GFAP-positive reactive astrocytes in the present study showed a limitation of the glial scar extent when hUCB MNCs were administered within 72 h. Interestingly, astrocytic reactivity is known to develop within a time span of up to 5 days while the time course of glial response was found peaking 72 h after stroke onset (8,36). It is well known that glial cells respond to ischemic damage and form the glial scar, but it is still uncertain whether this response promotes or limits endogenous repair processes in the central nervous system. The glial scar constitutes a chemical and physical barrier against sprouting axons after injury (9). Inhibitory elements within the glial scar as chrondrotoin sulphate proteoglycans are upregulated in reactive astrocytes and are presumed to constrict neural outgrowth, a key mechanism of functional regeneration, both in vitro and in vivo (23,30). A limitation of astrocytic reactivity therefore might ameliorate the endogenous regeneration potentials, thus leading to beneficial effects of hUCB MNCs as observed in this study. Assuming that the vast majority of glial scar tissue was formed in the host brain 120 h upon MCAO would explain the minimal cell effect on functional outcome and absent impact on lesion size and astroglial reactivity at this time.
Current Controversies About the Beneficial Effect of hUCB MNCs After Experimental Stroke
Some well designed studies were not able to reproduce the beneficial effects of hUCB MNCs, especially when using more demanding behavioral tests (22). However, there are possibly relevant differences to our study. First is the use of comorbid animals in our trial, which, even after cortical MCAO, show stronger defects and larger infarct volumes compared to control strains (34). This possibly allows better detection of a therapeutic impact. Second, fresh cells were administered in our in vivo study while the use of cells being stored for 1 year or longer was reported by Mäkinen and colleagues (22). Although hematopoietic potential and cell recovery rate of hUCB cells are not affected even after 10 years of cryopreservation (40), equivalent information regarding the neuroprotective or regenerative capacity following stroke is missing so far. Strain differences, freezing time, and so far unconsidered differences in the freezing/thawing protocols potentially may have contributed to differing results. This clearly emphasizes the importance to thoroughly address even “minor” or “technical” points during a later translational process into the clinic.
Optimal Time Window for hUCB MNC Transplantation Following Stroke
Summarizing, beneficial effects of hUCB administered within 72 h after MCAO were clearly shown by three independent methods. The failure to induce sustained functional recovery, lesion size reduction, and limitation of glial scaring in animals treated 120 h following MCAO and thereafter indicates a time window of at least 72 h for efficient cell application. Any therapeutic impact between these time points cannot be excluded, but could be considered minor and/or declining based on the data obtained in our experiments. Although limited, the time span for hUCB MNC administration after experimental stroke is far beyond that of thrombolysis. Thus, hUCB MNCs are expected to be a promising tool for acute stroke treatment, which, however, might require comparatively early intervention in the subacute phase of stroke.
Footnotes
Acknowledgments
The authors want to thank Dr. Holger Kirsten for professional raw data review and assistance in statistical analyses and interpretation. This work was supported by structural founds of the European Union, granted by the Development Bank of Saxony, Dresden, Germany. The authors declare no conflict of interest.
