Abstract
Although human islet transplantation has been established as valid and safe treatment for patients with type 1 diabetes, the utilization rates of human pancreases for clinical islet transplantation are still limited and substantially determined by the quality and composition of collagenase blends. While function and integrity of collagenase has been extensively investigated, information is still lacking about the most suitable supplementary neutral proteases. The present study compared islet isolation outcome after pancreas digestion by means of collagenase used alone or supplemented with either neutral protease (NP), clostripain (CP), or both proteases. Decent amounts of islet equivalents (IEQ) were isolated using collagenase alone (3090 ± 550 IEQ/g), or in combination with NP (2340 ± 450 IEQ/g) or CP (2740 ± 280 IEQ/g). Nevertheless, the proportion of undigested tissue was higher after using collagenase alone (21.1 ± 1.1%, P < 0.05) compared with addition of NP (13.3 ± 2.2%) or CP plus NP (13.7 ± 2.6%). Likewise, the percentage of embedded islets was highest using collagenase only (13 ± 2%) and lowest adding NP plus CP (4 ± 1%, P < 0.01). The latter combination resulted in lowest post-culture overall survival (42.7 ± 3.9%), while highest survival was observed after supplementation with CP (74.5 ± 4.8%, P < 0.01). An insulin response toward glucose challenge was present in all experimental groups, but the stimulation index was significantly decreased using collagenase plus NP (2.0 ± 0.12) compared with supplementation with CP (3.16 ± 0.4, P < 0.001). This study demonstrates for the first time that it is possible to isolate significant numbers of human islets combining collagenase only with CP. The supplementation with CP is an effective means to substantially reduce NP activity, which significantly decreases survival and viability after culture. This will facilitate the manufacturing of enzyme blends with less harmful characteristics.
Introduction
Over the last 15 years, transplantation of isolated human islets has been developed to a valid and safe treatment to restore euglycemia and to cure unawareness of hypoglycemia in prone patients with type 1 diabetes 1 –3 . The outcome of islet transplantation alone for nonuremic patients has now reached outcome levels that are similar to pancreas transplantation alone 2,4 .
Despite these achievements, the utilization rates of human pancreases for clinical islet transplantation are still variable and limited. The relevance of different donor factors for success or failure of human islet isolation have been extensively described 5 –8 . The importance of isolating high yields of islets has recently been underlined by a comprehensive multivariate analysis of recipient, donor and islet variables showing that the number of islets infused into the patient is the only variable that correlates significantly with islet graft function in transplanted patients with type 1 diabetes 9 . The inconsistency in isolation outcome reduces the limited donor pool of human pancreases and increases the costs for this procedure through failed isolations by approximately 50% or more 10 . One of the most important determinants for efficient release of islets from within the acinar tissue is the quality and composition of collagenase blends 11 –14 .
While integrity, function, and effective blending of collagenase class I and class II has been clarified to a significant extent 15 –19 , studies performed to identify the most suitable neutral protease are still ongoing. Experiments in rat and human pancreases clearly suggested that neutral protease from Clostridium histolyticum (NP) has to be accurately measured out to avoid damage of islets during pancreas digestion 20 –22 . In several products for human islet isolation, thermolysin from Bacillus thermoproteolyticus rokko has been used as alternative to NP 23,24 . Since the specific activity of thermolysin is multi-fold higher compared with NP 25 , it is associated with extensive islet fragmentation and significant loss of islets during culture if not carefully adjusted 26 . In a human pancreas split lobe model, the advantage of NP over thermolysin was demonstrated by consistently higher yields of islets fulfilling the release criteria for clinical islet transplantation 27 .
Clostripain (CP) is another protease that may be used as supplementary enzyme for collagenase. CP is responsible for the tryptic-like activity (TLA) in collagenase blends 25 . Only very few studies about the effect of CP on human islet isolation outcome are currently available. Studies in rat and human pancreases demonstrated improved islet isolation outcome when CP/TLA was present in collagenase blends 28 –30 . A most recent study demonstrated a significantly lower harmful effect of CP on the integrity of already isolated and pre-cultured human islets when compared with NP and thermolysin 31 .
However, it is difficult to draw a final conclusion from this low number of studies available as CP was always used in combination with NP or thermolysin, but never applied as sole supplementary protease for collagenase. The aim of this prospective approach was to dissect the effects of collagenase, NP, and CP on the liberation, morphological integrity, and in vitro function of islets isolated from human donor pancreases.
Materials and Methods
Organ Procurement
Research grade pancreata were retrieved from 30 human multiorgan donors with ethical approval and consent for research. Antihypotensive donor treatment with dopamine, adrenaline, noradrenaline, and norepinephrine is termed as vasopressor administration. Once legal consent had been given, pancreata were procured from multiorgan donors with brain death utilizing cold perfusion with University of Wisconsin solution (UW, ViaSpan, DuPont Pharmaceuticals, Herts, UK) or with histidine-tryptophan-ketoglutarate (Custodiol, Köhler Chemie, Alsbach, Germany). Explanted pancreata were shipped to the central isolation facility (Uppsala, Sweden) in 400 mL of cold UW or Custodiol.
Enzyme Blending and Administration
Human islets were isolated for research purposes as previously described
32
. The isolation procedure was always performed by the same isolation team. Briefly, after removal of the duodenum, the main pancreatic duct was cannulated at the distal part of the pancreatic head. Prior to manual distension with cold (8°C) Hank´s balanced salt solution (HBSS, Gibco-Invitrogen, Stockholm, Sweden) using a ratio of 1.2 mL/g trimmed pancreas weight, the distension solution was mixed with collagenase and additionally supplemented with either NP, CP, a combination of both proteases, or without proteases (w/o). The different enzyme blends are listed in Table 1. The enzyme mixtures were defined by the presence of 22 phenylazobenzyloxycarbonyl-
Enzyme Blending.

w/o: without protease supplementation; NP: neutral protease activity (DMC-U, dimethyl-casein-units); CP: clostripain activity (BAEE-U, benzoyl-
Donor Characteristics.

w/o: without protease supplementation; NP: neutral protease; CP: clostripain; BMI: body mass index; CIT: cold ischemia time.
Islet Isolation
Pancreas digestion was performed in a 350 mL digestion-filtration device filled with HBSS continuously utilizing a digestion temperature of 37°C
34
. During recirculation, samples were frequently assessed for amount, cleavage, and integrity of released islets. Digested tissue was collected in 250 mL-centrifuge tubes prefilled with 25 mL of cold (4°C) newborn calf serum, and centrifuged twice for 1 min at 150 x g. After 60 min of storage in 250 mL of 1.2-fold-concentrated UW (Apoteket, Stockholm, Sweden
35
) the digested tissue was centrifuged for 5 min at 2400 rpm in a Cobe 2991 cell processor (Gambro, Lakewood, CO, USA) using a continuous hyperosmolaric Ficoll gradient for separation of islets from exocrine tissue
32
. Purified islet fractions were washed twice in HBSS supplemented with 10% newborn calf serum, and collected finally in 100 mL of Connaught Medical Research Laboratories (CMRL) 1066 medium supplemented with 25 mM 2-[4-(2-hydroxyethyl)piperazin-1-yl]ethanesulfonic acid (HEPES), 1 mM pyruvate, 10 mM nicotinamide (all culture media supplements supplied by PAA, Pasching, Austria), 10% fetal calf serum, 2.5 mM
Islet Characterization
Subsequent to purification and after culture, islet equivalent (IEQ) yield was quantified as previously described in detail 37,38 . Islet morphological integrity was categorized using a fragmentation score from 0 (no fragmentation) to 3 (extensive fragmentation). Isolated islets were distinguished from exocrine tissue using insulin-specific dithizone staining (Sigma-Aldrich, Stockholm, Sweden) 38 .
Islet viability was assessed immediately after isolation and after 3–4 days of culture at 37°C utilising 25 µmol/L of Syto-13 (Invitrogen, Carlsbad, CA, USA) and 50 µmol/L of ethidium bromide (Sigma-Aldrich, St. Louis, MO, USA) for staining of viable and dead cells, respectively 39 . The fluorescence of Syto-13 and ethidium bromide was quantified at 545 nm and 490 nm, respectively. Recovery of viable cells only was calculated as overall survival.
Mitochondrial functional activity was evaluated in duplicate samples measuring the conversion of the tetrazolium compound 3-(4,5-dimethylthiazol-2-yl)-5-(3-carboxymethoxyphenyl)-2-(4-sulfophenyl)-2H-tetrazolium (MTS, Promega, Mannheim, Germany) into formazan as previously described 40 . Formazan formation was measured at 490 nm and expressed as optical density (OD) per 100 IEQ.
Islet in vitro function was assessed during static glucose incubation of islets precultured for 3–4 days at 37°C. Twenty hand-selected islets with an average diameter of 150–200 µm were sequentially incubated in duplicate, first for 45 min in bicarbonate-free CMRL 1066 (Applichem, Darmstadt, Germany) supplemented with 2 mmol/L glucose, followed by 45 min of incubation in 20 mmol/L glucose, finally followed by a second 45-min period in 2 mmol/L glucose. Basal and stimulated insulin secretion was normalized to the intracellular insulin content measured as described below. The glucose stimulation index was calculated by dividing the insulin release at 20 mmol/L glucose by the average of the two basal periods. After incubation, islets were recovered and sonified prior to overnight incubation in acid ethanol for subsequent extraction and determination of intracellular insulin 41 .
Statistical Analysis
All statistical analyses were performed utilizing Prism 7.0d for MacIntosh (GraphPad, La Jolla, CA, USA). Analysis of data was carried out by the nonparametric Kruskal Wallis test followed by Dunn’s test for multiple comparisons. The data for glucose-stimulated insulin release at low and high glucose concentrations were compared using the Friedman test followed by Dunn’s test. Islet viability measured post-isolation and post-culture was compared using the Wilcoxon test. Categorized variables such as gender and vasopressor administration were analyzed by the Chi-square test. Differences were considered significant at P < 0.05. P-values >0.05 are termed nonsignificant (NS). Results are expressed as mean ± standard error of the mean (SEM).
Results
The donor characteristics are presented in Table 2. No significant differences were found between experimental groups regarding gender, age, BMI, cold ischemia time (CIT), serum amylase, administration of vasopressors and trimmed pancreas weight.
Islet Isolation Outcome
As demonstrated in Table 3, recirculation time was decreased from 26 ± 2 min to 21 ± 1 min when collagenase was supplemented with NP. This reduction became significant when NP was compared with CP (25 ± 1 min, P < 0.05). A minor variability (NS) was observed with respect to packed tissue volume that was lowest when pancreases were digested with collagenase without additionally supplementing proteases (Table 3). In turn, the proportion of undigested tissue remaining in the chamber after pancreas digestion was significantly higher (21.1 ± 1.1%, P < 0.05) when compared with adding NP only (13.3 ± 2.2%) or with the combination of NP plus CP (13.7 ± 2.6%) (Table 3).
Digestion Variables.
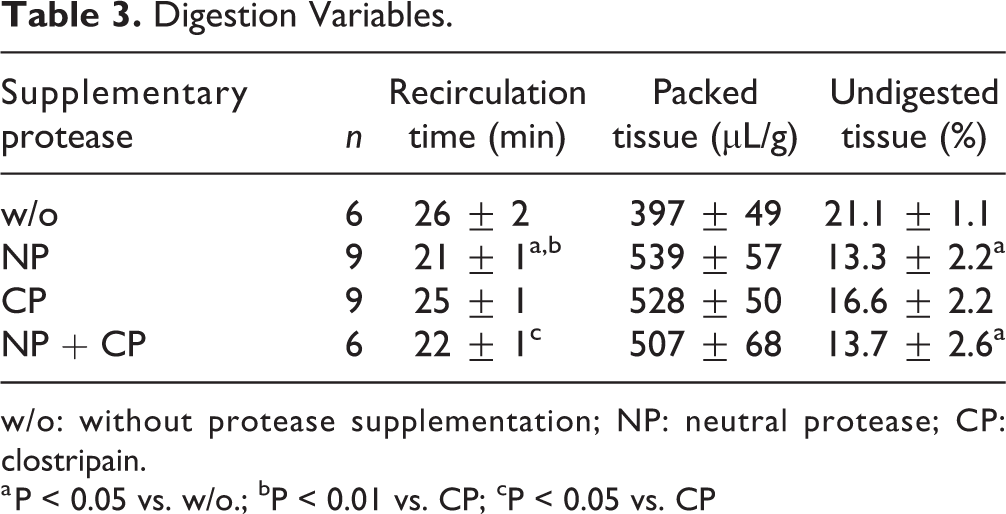
w/o: without protease supplementation; NP: neutral protease; CP: clostripain.
a P < 0.05 vs. w/o.; bP < 0.01 vs. CP; cP < 0.05 vs. CP
As shown in Table 4, nearly identical islet yields were isolated per organ when pancreases were digested without adding significant amounts of proteases (222250 ± 67510 IEQ) or when adding NP (211790 ± 58750 IEQ) or CP (209430 ± 29030 IEQ). In contrast, a slightly higher islet yield was obtained when combining NP and CP (283230 ± 56660 IEQ, NS). This trend towards higher yields by using a combination of NP plus CP was also observed when islet yield was calculated per gram trimmed pancreas weight (3430 ± 630 IEQ, NS vs. 3090 ± 550, 2340 ± 450 and 2740 ± 280 IEQ/g). The supplementation of collagenase with NP only (1.2 ± 0.3, NS) or with NP plus CP (1.3 ± 0.4, NS) was associated with slightly higher islet fragmentation when opposed to no supplementation (0.8 ± 0.3) or to CP only (0.8 ± 0.2) (Table 4).
Islet Isolation Outcome.

w/o: without protease supplementation; NP: neutral protease; CP: clostripain; IEQ: islet equivalent.
a P < 0.01 vs. w/o; bP < 0.05 vs. CP.
The proportion of islets embedded in exocrine tissue was highest in pancreases digested by collagenase without additional protease supplementation (13 ± 2%) and lowest using a combination of NP and CP (4 ± 1%, P < 0.01) (Table 4). No significant difference was found comparing NP (9 ± 2%) with CP (11 ± 4%, NS). These data did not correlate with islet purity which was lowest in islets isolated by collagenase supplemented with NP (49 ± 6%), and highest adding CP (64 ± 6%, P < 0.05 vs. NP) or CP plus NP (63 ±10%, NS) (Table 4).
The viability of freshly isolated islets varied marginally between islets isolated without additional protease supplementation (78.9 ± 1.7%) and when supplementing collagenase with NP (75.7 ± 2.5%) or CP (81.4 ± 2.3%). A slight decline of viability was measured in islets when CP was combined with NP (73.1 ± 4.3%, NS vs. CP).
Islet Culture Outcome
After 3–4 days of culture at 37°C, islets were harvested and assessed. The outcome of this assessment is shown in Table 5. When collagenase was supplemented with NP added either alone (67.6 ± 6.1%) or in combination with CP (66.5 ± 4.6%), the post-culture recovery was significantly lower in comparison with CP only (94.4 ± 5.2%, P < 0.01). Nevertheless, performing enzymatic pancreas digestion without additionally supplementing proteases, post-culture islet recovery was still lower (70.2 ± 6.1%, P < 0.05) compared with supplementing CP. After culture, a decrease of islet purity was noted in all experimental groups. However, this reduction reached statistical significance only in islets isolated without additionally adding NP or CP (59 ± 5% vs. 52 ± 5%, P < 0.05).
Islet Culture Outcome.
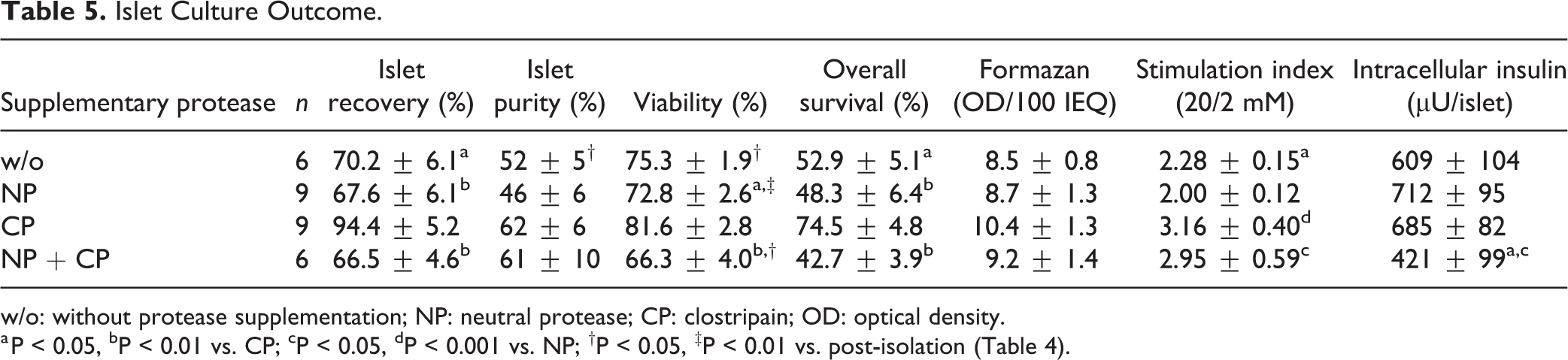
w/o: without protease supplementation; NP: neutral protease; CP: clostripain; OD: optical density.
a P < 0.05, bP < 0.01 vs. CP; cP < 0.05, dP < 0.001 vs. NP; †P < 0.05, ‡P < 0.01 vs. post-isolation (Table 4).
After culture, also a change of islet viability was observed. Except islets isolated by means of CP (81.6 ± 2.8%), viability decreased significantly during culture in all experimental groups (Tables 4 and 5). When comparing viability post-culture it became obvious that the presence of NP during pancreas digestion added either alone (72.8 ± 2.6%, P < 0.05 vs. CP) or in combination with CP (66.3 ± 4.0%, P < 0.01 vs. CP) reduced viability when compared with CP only. No significant difference was found opposing CP to no protease supplementation (75.3 ± 1.9%, NS). Calculating the post-culture recovery of viable cells only revealed highest overall survival in islets isolated with CP-supplemented collagenase (74.5 ± 4.8%), and lowest overall survival after adding a combination of NP plus CP (42.7 ± 3.9%, P < 0.01 vs. CP) (Table 5). Collagenase used without additionally supplementing proteases released islets characterized by an overall survival (52.9 ± 5.1%, P < 0.05) which was still significantly lower compared with CP-supplemented collagenase.
Only a marginal variability between experimental groups was observed with respect to mitochondrial activity measured as formazan production after culture (Table 5). In contrast, assessment of islet in vitro function during sequential static incubation at 2, 20, and again 2 mmol/L of glucose, revealed a significant effect of supplementary proteases on the secretory capacity of cultured human islets (Table 5). The glucose stimulation index was significantly reduced when islets were isolated by means of NP (2.0 ± 0.12) and compared with CP (3.16 ± 0.40, P < 0.001) or with CP plus NP (2.95 ± 0.59, P < 0.05). Isolation without adding proteases did not increase the stimulation index (2.28 ± 0.15, P < 0.05 vs. CP) in comparison with NP-supplemented digestion. These observations corresponded with the pattern of insulin release during glucose incubation as shown in Fig 1. While basal insulin release was similar in all experimental groups, a slightly higher proportion of insulin was discharged at stimulatory glucose concentrations when islets had been isolated with CP (NS). The intracellular insulin content varied only marginally between islets isolated without additional protease supplementation (609 ± 104 µU/islet) and isolated with NP (712 ± 95 µU/islet) or CP (685 ± 82 µU/islet). In contrast, when collagenase was supplemented with NP plus CP, a significantly lower intracellular insulin content was measured (421 ± 99 µU/islet, P < 0.05 vs. NP).

Glucose-stimulated insulin secretion of human islets isolated with collagenase used without supplementary proteases (w/o, n = 6) or supplemented with NP (n = 9), CP (n = 9) or a combination of NP plus CP (n = 6). After culture for 3–4 days at 37°C, islets were sequentially incubated for 45 min at 2 (white bars), 20 (grey bars) and again 2 mmol/L of glucose. Insulin release is normalized to intracellular insulin content. Dunn’s test following Friedman test revealed *P < 0.05, **P < 0.01, ***P < 0.001 for insulin secretion at 20 vs. 2 mmol/L glucose. Dunn’s test following Kruskal–Wallis test revealed
Discussion
Previous experiments in rats demonstrated that the fast and effective release of islets from within the acinar tissue essentially requires the presence of collagenase class-I, class-II, and at least one neutral protease 15,16,42 . However, a growing number of studies indicate that the process of islet cleavage is significantly improved when collagenase is additionally supplemented with CP 28 –30 . It has been assumed that the islet-releasing potency of CP is related to its capability to convert pro-elastase into elastase assisting the degradation of elastin as a component of the extracellular matrix 43 . Since significant expression of elastin has been demonstrated only in organs such as lung, heart, and vessels that are continuously exposed to stretching forces, the presence of elastin fibers in the pancreas seems to be unlikely 44 . A possible anatomical location of elastin in the pancreas might be within the walls of the vascular and ductal system. In addition, considering the harmful effects of activated elastase on acinar and islet tissue 45,46 , a negative rather than a positive effect would be expected if CP is administered for islet isolation. Indeed, a negative effect of CP was described by Bertuzzi’s group calculating an inverse correlation between CP and islet yield 47 .
The increased cleavage of islets through CP had also been explained by a synergism when CP is specifically combined with NP as described in rat islet isolation 28,29,48 . The present data confirm these observations with respect to human islet yield and amount of embedded islets, but not when considering recirculation time or proportion of undigested tissue, which were already lowest when only NP was added to collagenase. Moreover, a boosting effect on the islet release from the human pancreas was also noted when CP had been added as third component to a blend composed of collagenase and thermolysin 30 . These findings do not support the existence of a specific synergism between CP and NP as proposed by Dendo et al 29 .
Surprisingly, similar islet yields were isolated when pancreases were processed without adding significant amounts of proteases. As commercial large-scale collagenase production does not have the technical capacity to reduce protease contaminations to virtually zero, it cannot completely be excluded that the collagenase-contaminating traces of NP and CP contributed to pancreas dissociation to a certain extent. The activities of these contaminations are approximately 10- to 20-fold lower when compared with the NP activities used in previous human islet isolation studies 19,27,49 –52 causing a higher percentage of undigested tissue, a lower amount of packed tissue volume, and a higher proportion of embedded islets in comparison with the combined addition of NP plus CP. This may also affect postculture outcome reducing the overall survival of cultured islets isolated without additionally supplementing proteases. Vice versa, when significant amounts of NP were involved in pancreas digestion, a trend towards a higher fragmentation score and lower islet purity was noted. Overall, these findings suggest that it is principally possible to isolate significant amounts of intact human islets with very low activities of NP and CP. The higher disintegration of exocrine tissue and islets isolated by means of NP or NP plus CP did not seem to correlate with viability of freshly isolated islets, which varied in a very narrow range between experimental groups. Although it is speculative so far, it has to be discussed whether the alignment of islet viability between experimental groups was caused by the addition of 10% bovine serum for collection of pancreatic digest, which might have reduced the activity of NP or CP. Conversely, it cannot be excluded that supplementation with human serum albumin alone would reveal larger differences regarding islet viability 53 .
In agreement with a previous study in human islets, the detrimental effect of NP on islet morphological and functional integrity, as well as overall survival, became more apparent when assessed after culture 22 . A harmful effect of NP on human islet integrity was also revealed by the Edmonton group. However, Kin et al. used a collagenase batch that was contaminated by a relatively large proportion of NP suggesting that an excess of this protease was present when additionally added as supplement for collagenase 54 .
In contrast, the administration of CP as sole supplementary protease was characterized by a significantly higher post-culture viability, survival, and glucose stimulation index when compared with NP. It is remarkable that the impact of NP on post-culture parameters was not compensated by the simultaneous addition of CP. This phenomenon may be explained by the different affinities of NP and CP for laminin-511. Together with collagen-IV, this extracellular matrix protein is the major component of the islet basement membrane 55,56 , forming a protective barrier to preserve the morphological integrity of underlying islets during enzymatic pancreas digestion 57 . In contrast to NP or thermolysin, inducing a strong degradation of laminin-511, the laminin-511 molecule remained nearly intact when treated with CP alone or collagenase alone 48 . Because the enzymatic degradation of laminin-511 mainly concerns the cleavage of the α5-chain, survival signaling between basement membranes and integrins or other cellular receptors is interrupted after pancreas digestion 55,58 . As a consequence, islets detached from the basement membrane undergo apoptosis and lose functional potency after isolation and during subsequent culture 59 . On the other hand, the presence of perislet basement matrix proteins provide protection of islet integrity and preservation of metabolic capacity 60,61 .
In summary, our prospective study describes the islet cleavage efficiency of NP and CP, used alone or in combination as supplementary proteases for human islet isolation. We could demonstrate that it is possible to isolate significant numbers of human islets combining collagenase only with CP. Although NP appears to be essential for effective pancreas digestion and fast islet release, it significantly reduces human islet survival and viability during culture.
The extent of the present study essentially required large scale collagenase production and purification, which do not have the technical capacity to reduce protease contaminations to virtually zero. We can therefore not completely exclude that marginal amounts of NP and CP might have contributed to pancreas dissociation to a certain extent. Although we cannot quantify the impact of these contaminations in the current setting, the present observations demonstrate that it is principally possible to isolate significant amounts of intact human islets with very low activities of NP and/or CP. However, this does not diminish the finding that supplementation with CP appears to be an effective means to substantially reduce NP activity, thereby increasing the quality of isolated islets. This will facilitate the manufacturing of enzyme blends with less harmful characteristics. Future experimental studies about the effect of supplementary proteases should focus on the use of recombinant collagenase.
Footnotes
Acknowledgments
The authors wish to thank Karin Andersson and Sana Asif for their excellent technical assistance.
Ethical Approval
This study was approved by the Research Ethics Committee of the Uppsala University, Uppsala, Sweden (Ups03-577).
Statement of Human and Animal Rights
All of the experimental procedures of this study were conducted in accordance with the protocols approved by the Research Ethics Committee of the Uppsala University, Uppsala, Sweden (Ups 03-577). Consent for use of islets for research purposes was verbally obtained from legally authorized representatives of the patients.
Statement of Informed Consent
Legal consent for organ donation and subsequent research was verbally obtained from a legally authorized representative of the patient.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research and/or authorship of this article: This study was supported by grants from the Diabetes Wellness Network Sweden, the Swedish Medical Research Council (K2015-54X-12219-19-4) and the Juvenile Diabetes Foundation International.
