Abstract
One of the primary obstacles in the restoration or repair of damaged tissues is the temporospatial orchestration of biological and physiological events. Cellular transplantation is an important component of tissue repair as grafted cells can serve as replacement cells or as a source of secreted factors. But few, if any, primary cells can perform more than a single tissue repair function. Epithelial cells, derived from the choroid plexus (CP), are an exception to this rule, as transplanted CP is protective and regenerative in animal models as diverse as CNS degeneration and dermal wound repair. They secrete a myriad of proteins with therapeutic potential as well as matrix and adhesion factors, and contain responsive cytoskeletal components potentially capable of precise manipulation of cellular and extracellular niches. Here we isolated CP from neonatal porcine lateral ventricles and cultured the cells under a variety of conditions to specifically modulate tissue morphology (2D vs. 3D) and protein expression. Using qRT-PCR analysis, transmission electron microscopy, and gene microarray studies we demonstrate a fine level of control over CP epithelial cell clusters opening further opportunities for exploration of the therapeutic potential of this unique tissue source.
Keywords
Introduction
The choroid plexus (CP) is made up of ciliated columnar epithelial cells that secrete a large, but relatively uncharacterized, number of bioactive factors apically into the cerebrospinal fluid and basally into the circulation (19). While the primary function of the CP is to produce and enrich cerebrospinal fluid in the adult (5), it also plays an instructive role during embryologic development (12,13,17). Because of the wide range of activities encompassed within the CP's complex secretome, cultured CP epithelial clusters are being explored as a therapeutic source of cell-based regenerative molecules. An abundance of regenerative factors secreted from CP have been identified in vitro, including vascular endothelial growth factor (VEGF), connective tissue growth factor (CTGF), fibroblast growth factor 2 (FGF2), transthyretin, insulin-like growth factor II, netrin 1, pentraxin 3, pigment epithelium-derived factor (PEDF), cardiotrophin-1, Wnt-7B, Wnt-10A, nerve growth factor (NGF), neurotrophin-4 (NT-4), transforming growth factor-β (TGF-β), growth hormone (GH), a variety of inflammatory factors, and others (5,8,10,11,16,20,22–24).
Encapsulated within alginate-polyornithine-alginate (APA) microcapsules, transplanted CP clusters are neuroprotective in animal models of Huntington's disease (4,7) and stroke (1,2), and can markedly accelerate the rate and quality of healing of full thickness dermal wounds in rats (22). While these benefits could conceivably be related to secretion of individual factors from CP, it is more logical that such a broad range of therapeutic effects can, in part, be explained by factor synergy within the secreted cocktail. Consistent with this notion is the observation that while delivery of VEGF from hydrogels is potently neuroprotective in a rat model of Huntington's disease (6), comparable benefits have been achieved (3) with CP transplants delivering several log-fold reductions in VEGF together with a cocktail of other CP-secreted neuroprotective factors (23). Together, these data support the notion that CP may exert its therapeutic effects through the secretion of multiple synergistic factors, capable of achieving equivalent potency of monofactor therapy at very low doses.
The therapeutic potential of CP, whether in encapsulated form or as a purified mixture of proteins derived from supernatant, would be significantly enhanced by gaining a greater understanding of the mosaicism of the secreted factors as well as being able to specifically modulate the dose of identified molecules delivered from CP. Indeed, previous studies have shown that regenerative factor candidates in the transcriptome of porcine CP are exceptionally stable over time and that they can be modulated in tissue culture (22). Here we extend these data by examining the effects of media type, extracellular matrix, and CP cluster morphology (three-dimensional cultures). Using VEGF as a candidate factor combined with qRT-PCR analysis, transmission electron microscopy, and gene microarray studies we demonstrate a fine level of control over CP epithelial cell clusters opening further opportunities for exploration of the therapeutic potential of this unique tissue source.
Materials and Methods
Choroid Plexus Isolation
All procedures adhered to NIH and Society for Neuroscience guidelines for the use of research animals. Surgical procedures were conducted under aseptic conditions and all efforts were made to minimize animal suffering and to reduce the number of animals used. Neonatal Yorkshire pigs aged 7–10 days, anesthetized initially with ketamine (500 mg/kg) and xylazine (0.15 mg/kg), were maintained on 3% isoflurane during exsanguination. Following a midline incision, the overlying skull was removed with rongeurs, and the brain was hemisected to expose the CP in the lateral ventricles. The CP were acquired and placed in Hanks balanced salt solution (HBSS, 4°C) supplemented with ciproflaxocin (Sigma, USA, 3 μg/ml), 2% human serum albumin and digested as previously described (3,7). After mechanically disrupting the tissue with scissors, CP epithelial clusters were generated with light enzymatic digestion over 20 min (Liberase HI, Roche, NJ). Clusters of CP epithelial cells were washed and maintained in RPMI-1640 (Sigma, St. Louis, MO) with 2% neonatal porcine serum and 3 μg/ml ciproflaxocin (RPMI-CPN) and placed in nonadherent flasks at 37°C with 5% CO2. Half of the media was removed and replaced with fresh media after 24 h, 48 h, and every other day thereafter. After 0, 2, and 8 days in RPMI, clusters were counted and separated into individual flasks. After 14 days, the CP clusters were divided into groups incubated in RPMI-CPN as a suspension, immobilized in Matrigel-HC (BD Biosciences), or suspended in serum-free endothelial cell culture medium (ENDO-SFM, Gibco, USA).
Tissue Culture Media Screen and Formation of 3D Hollow Shells
After 2 weeks in RPMI-CPN, cultures were divided into groups in RPMI-CPN, ENDO-SFM, Matrigel-HC in RPMI-CPN, or Matrigel-HC in ENDO-SFM. Briefly, clusters were sedimented, counted, and placed into groups of 65,000 clusters each into four nonadherent plates in either ENDO-SFM or RPMI-CPN. 3D culture groups were created in 24-well plates loaded with 3,000 clusters per well suspended in 50 μl Matrigel-HC diluted with 50 μl RPMI-CPN or ENDO-SFM at 4°C. Matrigel was allowed to gel at 37°C for 15 min, and then 1 ml medium was added to each well. All groups were maintained in an incubator at 37°C with 5% CO2 for either 7 or 14 days with at least three media changes per week, and then samples were assessed for VEGF secretion by incubation in 1 ml fresh medium for 24 h, followed by extraction of RNA for PCR and microarray. Samples cultured in Matrigel were photographed and the diameter of developing epithelial shells was measured using ImageJ image analysis software. At each time point, at least 100 cell clusters were analyzed. In each culture group, a sample cohort was processed for transmission electron microscopy, wherein epithelial shells (capsuloids) or epithelial clusters were immersion fixed in Karnovsky's fixative, embedded in Spurr epoxy, and thin sectioned for viewing at 80 kV on a Hitachi microscope.
Detection of VEGF
VEGF was measured as a marker of potency as previously described (23). Samples containing 3,000 clusters were incubated with 1 ml fresh medium (37°C, 5% CO2), and the supernatant was removed and frozen for subsequent analysis. Thawed samples were analyzed in triplicate using a human VEGF ELISA (Quantikine®, R&D Systems, USA). Sequence homology between porcine and human VEGF is greater than 90% (18). The value obtained, expressed in pg/ml, was compared to a standard curve of recombinant human VEGF. Background samples including culture media and Matrigel-HC showed negligible signal and were subtracted to obtain final VEGF values. Groups were statistically compared in a one-way ANOVA and post hoc Dunnett's test against the group cultured in RPMI-CPN using Prism software (GraphPad, USA).
Isolation of RNA, Quantitative Real-Time PCR (QRT-PCR), and Microarray Analysis
QRT-PCR was used to confirm the expression of VEGF and transthyretin. RNA was extracted using RNAeasy (Qiagen, USA) per manufacturer's protocol and optimized for real-time PCR. Total RNA (5 μg) was treated with DNase (Invitrogen, Carlsbad, CA) before reverse transcription with iScript Reverse Transcriptase (Bio-Rad, Hercules, CA). A method was optimized for real-time PCR wherein samples were run on Bio-Rad's iCycler iQ thermocycler at 95°C for 4.5 min, then cycled 40 times at 95°C for 10 s and 59.5°C for 45 s, with a final hold at 95°C for 1 min. Samples were run in triplicate in 25-μl reactions with 25 ng/well of sample (based on total RNA) using iQ SYBR green supermix (Bio-Rad). The primer sequences were: VEGF, 5′-gca gaaggagaccagaaacc-3′ and 5′-actcgatctcatcggggtact-3′; transthyretin, 5′-aggagggagctctttgaagg-3′ and 5′-ttgtctct gcccgagtttct-3′; and β-actin (housekeeping gene), 5′-gg gcagtagcatcgctttag-3′ and 5′-ggctgcccactcaaaataaa-3′. Relative expression of VEGF was calculated using the mathematic model previously described (23). The primer efficiency was calculated by the BioRad iCycler program and is based on the slope of Ct values obtained from standard curves of control tissue samples. Primer efficiencies for the genes of interest (VEGF and transthyretin) and the housekeeping gene (β-actin) were used to determine the relative expression of VEGF between samples from Ct values using the following equation: [2.068^(Ct FACTOR control – Ct FACTOR samples)]/ [2.087^(Ct β-actin control – Ct β-actin samples)],where Ct FACTOR (or Ct β-actin) is the cycle number in which the fluorescent signal from VEGF or transthyretin primers (or β-actin) crosses the noise threshold in the “control” tissue (or unknown “samples”). Statistical analysis was performed between groups using a one-way ANOVA and post hoc Dunnett's test against the group cultured in RPMI-CPN with Prism software (GraphPad, USA).
Separately, RNA was isolated from three of the four groups (RPMI-CPN, ENDO-SFM, Matrigel in RPMI-CPN) followed by Affymetrix Microarray Analysis. RNA sample integrity was confirmed on electropherograms acquired from a Bioanalyzer 2100 (Agilent Technologies, USA). Once samples were confirmed to be suitable for array analysis, an Affymetrix Porcine Microarray was used. Sample preparation and hybridization protocols were used as supplied and recommended by Affymetrix. Samples were run in duplicate and compared in Microsoft Excel against RPMI-CPN controls. Data presented meets the criteria of having fold-changes less than or greater than 2, with p < 0.05.
Results
Morphology
Epithelial clusters ranging between 50 and 200 μm were obtained from the collagenase digestion of fresh CP, with a yield of approximately 100,000 clusters per CP after 8 days in RPMI-CPN. Following a transition to Matrigel-HC, epithelial shells formed that increased in size throughout the course of the experiment. As shown in Figure 1, CP clusters deposited in Matrigel-HC in RPMI-CPN remained as solid masses for only 1 day, as the outer layer of epithelium formed projections toward the basement membrane-rich substrate. After 2 or 3 days, a fluid-filled space was formed in the interior of the cluster that increased in size over the next several days. By 5 days, most of the remaining epithelium that spanned the interior migrated toward the outer shell, causing further overall expansion. Eventually, epithelial shells in close proximity joined to create very large cell assemblies or complex tubular networks, as shown on day 14 in Figure 1. This can also be seen in Figure 2, which shows a grouping of capsuloids at low magnification over the course of 2 weeks. Table 1 shows a morphometric comparison of capsuloids formed in Matrigel-HC cultured in RPMI-CPN or ENDO-SFM. Capsuloids increased in diameter by almost twofold in RPMI-CPN and 1.7x in ENDO-SFM, with relatively consistent overall growth kinetics between groups.
Capsuloid Diameter (μm) as Measured by Image Analysis From a Sample Set of 100 Capsuloids


Light micrographs of choroid plexus (CP) capsuloids formed in Matrigel-HC. Scale bars: 50 μm (top row; day 1–5) and 100 μm (bottom row, day 7–14).

Light micrographs of CP capsuloids in Matrigel-HC after 4, 7, 11, or 14 days. Scale bars: 100 μm.
At the ultrastructural level, the reorientation of the CP epithelium was evident through the inverted polarity of cilia and microvilli. In Figure 3A, these structures are seen on the outside of the cluster, which in this illustrated case is 4–5 cells deep and bound by tight junctions (Fig. 3B). In contrast (Fig. 3C), epithelial shells formed in RPMI-CPN contain a fluid-filled interior with motile cilia and microvilli, also with apparent tight junctions within intercellular borders (Fig. 3D). Epithelial shells formed in ENDO-SFM also displayed similar morphology, but with a marked increase in the density of cytoplasmic vesicles and secretory granules (Figs. 3E, F).

Transmission electron micrographs of CP epithelial clusters maintained in RPMI-CPN (RPMI + ciproflaxocin; A, B), CP epithelial capsuloids maintained in RPMI-CPN (C, D), and CP epithelial capsuloids maintained in ENDO-SFM (serum-free endothelial cell culture media; E, F). Scale bars: 100 μm (A, C, D, E), 1 μm (B), 5 μm (F).
VEGF Expression
Secretion of VEGF from cultured epithelial clusters ranged significantly over the 2-week period. Wells containing 3,000 CP clusters were analyzed in each group, followed by RNA extraction for PCR or microarray. Successful RNA extraction prohibited the destructive measurement of DNA for normalization by cell number, and thus the results are reported as gross secretion per 3,000 cell clusters. Overall, as shown in Figure 4, secreted VEGF was increased in response to capsuloid formation in Matrigel, and was further enhanced with the addition of ENDO-SFM, as previously reported (23). In suspension culture, ENDO-SFM was responsible for an increase in VEGF detected in supernatant by 194% after 7 days and 38% after 14 days, both in comparison to RPMI-CPN. In contrast, Matrigel in RPMI-CPN was associated with a 337% increase in VEGF production after 7 days and 240% after 14 days. The addition of ENDO-SFM to the Matrigel system resulted in a further improvement in VEGF secretion, by 721% after 7 days and 405% after 14 days.

Vascular endothelial growth factor (VEGF) secretion as determined by hVEGF ELISA based on incubation of 3,000 CP clusters for 24 h in fresh supernatant. Values represent average ± SE. Filled bars: 7 days, open bars: 14 days. ∗p < 0.05.
The relative abundance of transthyretin and VEGF were calculated in relation to β-actin expression using real-time PCR. As shown in Figure 5, values were relatively consistent between suspension culture groups, with ENDO-SFM producing a 110% increase in transthyretin expression and 275% increase in VEGF expression compared to the RPMI-CPN group after 2 weeks in culture. This finding is consistent with the increase in secreted VEGF measured after 2 weeks (240%). However, groups cultured as epithelial shells in Matrigel showed dramatic increases relative to suspension culture groups, on the order of 1000% after 2 weeks.

Relative expression of transthyretin and VEGF in suspension or 3D culture. Black bars: suspension culture (RPMI-CPN); open bars: suspension culture (ENDO-SFM); light gray bars: 3D culture (Matrigel/RPMI-CPN); dark gray bars: 3D Culture (ENDO-SFM). ∗∗p < 0.01.
Gene Microarray
Porcine whole genome microarray was used to characterize changes to individual genes following exposure to the culture conditions under study. Tables 2 and 3 summarize the genes with statistically significant changes (>2 or <2, p < 0.05) in expression levels between CP clusters cultured in RPMI-CPN and ENDO-SFM (Table 2) or Matrigel-HC in RPMI-CPN (Table 3). In a comparison against CP clusters cultured in RPMI-CPN, the CP cluster transcriptome in ENDO-SFM exhibited 3,093 statistically significant changes in gene expression after 7 days and 4,691 after 14 days (p < 0.05). Similarly, in comparison to RPMI-CPN suspension cultures, CP capsuloids formed in Matrigel-HC in RPMI-CPN showed 4,890 changes in expression after 7 days and 1,828 after 14 days (p < 0.05).
Log2 Gene Expression Values of CP Clusters in Suspension Culture Obtained From Affymetrix Porcine Microarray
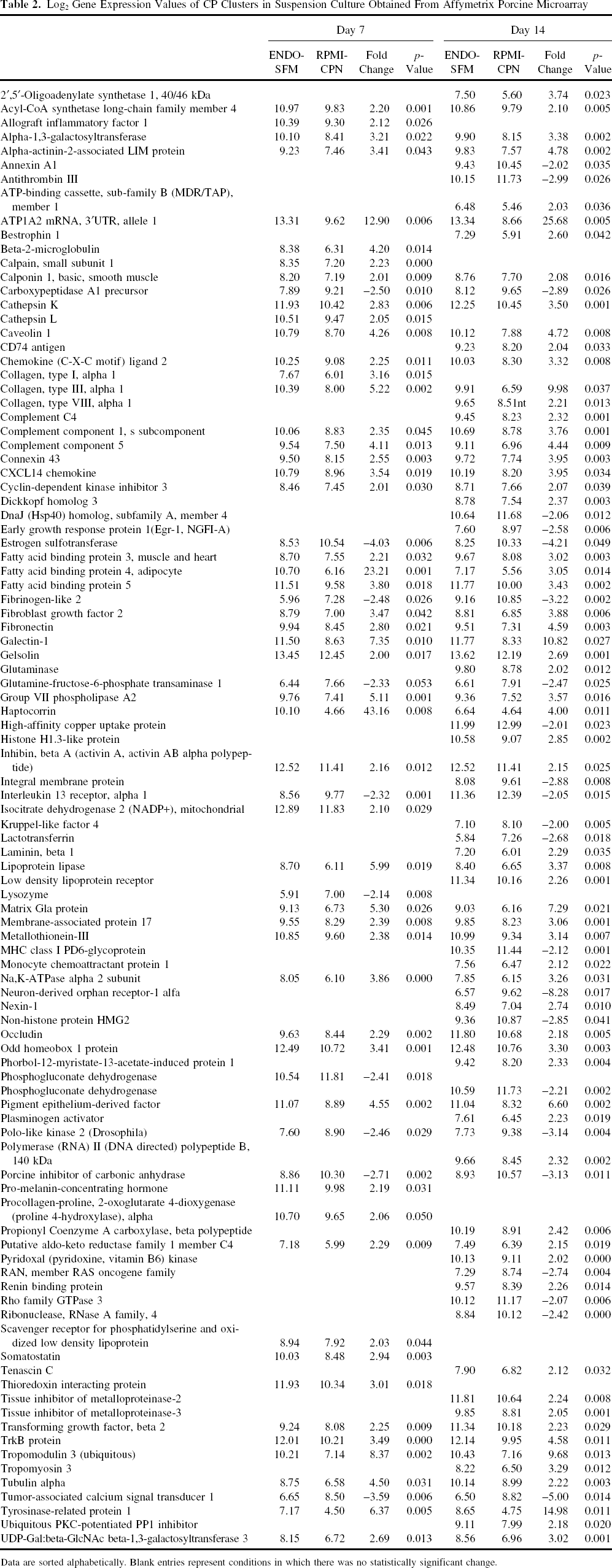
Data are sorted alphabetically. Blank entries represent conditions in which there was no statistically significant change.
Log2 Gene Expression Values of CP Clusters Cultured in 3D Within a Matrigel-HC Matrix, Obtained From Affymetrix Porcine Microarray

Data are sorted alphabetically. Blank entries represent conditions in which there was no statistically significant change.
Table 2 shows selected gene changes in suspension culture (RPMI-CPN vs. ENDO-SFM) with magnitude greater than 2. The largest change after 7 days was in the gene for the cobalamin-binding protein haptocorrin, a vitamin B12 carrier (15). Factors with significant changes having substantial therapeutic potential include TGF-β2, tissue inhibitor of metalloproteinases 2 (TIMP-2), PEDF, FGF-2, and galectin-1, among others. After 7 days in culture, these factors show variable expression, but by 14 days, they were all upregulated by greater than twofold. A number of extracellular matrix constituents also showed increased expression in ENDO-SFM, including nearly a 10-fold increase in the expression of collagen type III after 14 days and a 4.6-fold increase in fibronectin. Other notable changes after 2 weeks included a 15-fold increase in the expression of tyrosinase-related protein 1, a protein localized in the epidermis associated with melanocytes (25), a 9.7-fold increase in tropomodulin 3, a regulator of cell migration (9), and a 25.7-fold increase in ATP1A2, an Na+/K+ transporter (14).
Epithelial cells formed into capsuloids exhibited a differential expression profile that showed many significant changes after 7 days, but very few after 2 weeks. In this comparison, both culture systems included equivalent biomass in RPMI-CPN medium, with Matrigel-HC added to form fluid-filled shells. As shown in Table 3, the most abundant changes occurred after 7 days in culture, as many genes were downregulated by up to nearly 14-fold (amylase). Other genes of note with decreased expression (less than -2-fold, p < 0.05) included various components of the complement system (-7.2 to −10.2-fold), aquaporin 1 (2.7-fold), optineurin (6-fold), solute carriers (2.5 to 8.8-fold), tropomodulin 3 (9.9-fold), and others. Genes upregulated after 7 days (more than 2-fold, p < 0.05) included adrenomedullin (2.4), cytochrome p450 1A1 (3.0), fatty acid binding protein 5 (2.1), and several others. However, after 14 days, the system appeared to reach equilibrium, with only 21 statistically significant changes greater than 2-fold or less than -2-fold. These changes included upregulation of several cytoskeletal elements and extracellular matrix proteins including vascular smooth muscle α-actin (18.3), matrix GLA protein (3.8), and collagen type III (10.5), and decreased expression of fatty acid synthase (-2.1-fold) and low-density lipoprotein (LDL) receptor (-2.1-fold).
Discussion
Cultured choroid plexus epithelium is a malleable source of therapeutic proteins that has enormous potential in regenerative medicine. Its transcriptome is extremely diverse, encoding for secreted factors that are active across multiple pathways at biologically relevant levels of expression. In the current study, modulation of the cellular output was accomplished through manipulation of the local microenvironment, as CP epithelium formed into fluid-filled capsuloids expressed markedly higher levels of VEGF than CP clusters in suspension. Similarly, exposure to ENDO-SFM tissue culture medium enhanced the potency compared to the RPMI-CPN controls. A comparison of ultrastructure revealed that the change in expression was accompanied by a robust increase in the density of secretory vesicles, while other morphological features of healthy CP such as tight junctions were maintained. In capsuloid form, CP epithelium apparently reversed polarity, with cilia and microvilli oriented inward in a fluid-filled space, evident in real time occasionally entrapping motile CP clusters within. Presumably, this reversal of orientation was induced by an increased affinity of the basal epithelium for the extracellular matrix (ECM) proteins contained in the surrounding Matrigel.
The formation of capsuloids was accompanied early by a number of significant changes in the expression of genes shown in Table 3, which was dramatically reduced after 2 weeks. The ability of the CP epithelium to transport fluid through aquaporin channels (26), in conjunction with its sensitivity to ECM substrates, may combine to serve as the basis for the mechanism of capsuloid formation. As individual cells begin to bind to the 3D matrix, the movement of fluid through the epithelium into the hollow core could create radial forces that drive the growth of the capsuloid until equilibrium is reached, dictated by the number of cells present in the original cluster. The downregulation of aquaporin-1 in capsuloids after 7 days could reveal a shift in the equilibrium of fluid transport established in CP clusters initially cultured in RPMI-CPN.
The natural polarity of CP epithelium at the blood– cerebrospinal fluid interface serves a number of biological purposes, one of which is the regulation of growth factor composition at both sides of the interface. This is evident in the differential composition of factors that are secreted basally into the blood with respect to the composition in the cerebrospinal fluid regulated by apical secretions (19), as well as the localization of Na+,K+-ATPase to the apical membrane and GLUT-1, the facilitative glucose transporter, to the basolateral membrane (27). Certainly the upregulation of the Na+,K+-ATPase ATP1A2 by more than 25-fold after 2 weeks in the capsuloids in conjunction with increased protein secretion is indicative that this in vitro culture system has achieved biological regulation at the level of cellular trafficking.
Aside from the intuitively logical attraction of using CP as a secretory vehicle for multiple proteins effective in a number of human diseases, it is also worth considering CP as a tool for engineering 3D tissue constructs. 3D tissues require an extremely complex and difficult to achieve mixture of cells, vasculature, and ECM components to enable precise integration and proper physiological function. Together with previous studies (21), the data shown here support the CP as an active mediator of tissue assembly, with both autocrine and paracrine activity. The epithelium secretes a myriad of proteins with therapeutic potential as well as matrix and adhesion factors, and contains very responsive cytoskeletal components potentially capable of precise manipulation of cellular and extracellular niches. For instance, cultured CP expresses high levels of intercellular adhesion molecules (ICAMs), collagens, and integrins that could be used to control and modulate the extracellular niche of forming vascular networks, including secondary sources of exogenous cells such as endothelium. Likewise, the high abundance of matrix remodeling factors such as TIMP-2 and connective tissue growth factor could promote multicellular tissue assembly through modulation of ECM composition as well as migration and chemotaxis of fibroblasts and endothelial cells. With the high and controllable levels of VEGF secretion, as well as various other tissue-tropic factors, one could envision using CP as a critical component of 3D tissue construction for in vivo grafting or in vitro diagnostic and testing purposes.
The appeal of using the choroid plexus as a platform for cellular therapy has been extended to its role as a protein factory in vitro, collecting and purifying the supernatant for use as an acellular therapeutic (22). Developing a manufacturing process for such a product depends on a thorough identification and characterization of parameters that contribute to its composition, as well as the health and functionality of the cells themselves. In this manner, longitudinal process control can be achieved, generating acceptable ranges of culture variables that are used to manufacture the product, while also providing a means of manipulating the drug to potentially have crossover efficacy into alternative disease indications. The data presented here demonstrate this potential, as modulation of the transcriptome and secreted factor doses was possible through alteration of media composition as well as immobilized 3D ECM culture. A continued effort into deciphering the processes and factors involved in the CP as well as their mechanism of action may provide critical insight into regenerative mechanisms that drive tissue repair and neuroprotection.
Footnotes
Acknowledgments
C.T., B.B., M.G., K.B., and S.H. all have founder's shares in CytoSolv.
