Abstract
The objective of vascular tissue engineering is to develop tissue-engineered, biocompatible, small-diameter vessels suitable to withstand in vivo systolic pressures as well as be immunologically compatible with the patient, in order to minimize graft rejection. In this study, we present and compare two models of biocompatible, tissue-engineered vascular grafts (TEVG), using chitosan and acellularized rat aortas as options for scaffolds. Human aortic adventitial smooth muscle cells and fibroblasts were seeded onto a fibrin gel and subsequently wrapped around either of the two scaffolds. After several weeks of maturation in standard culturing conditions, the graft models were analyzed and compared by mechanical testing, cell viability, and histology. Histological and viability data showed that both models were viable and developed similarly, with a scaffold surrounded by two layers of cells, the fibroblasts lying on top of the smooth muscle cells. Both models responded to 200 mM potassium chloride (KCl) (tensions of 38 ± 3, 78 ± 13, and 52 ± 7 μN) and 25 mM 8-bromo-cyclic AMP (tensions of −23 ± 4, −39 ± 10, and −31 ± 12 μN) stimulation by vasoconstriction and vasorelaxation (n = 3), respectively; however, the chitosan model was unable to maintain the contracted and relaxed tension. Because the acellularized aorta TEVGs were able to maintain stimulated tension better than chitosan TEVGs, we concluded that the acellularized aorta model was better suited for further development.
Keywords
Introduction
In the year 2004, the American Heart Association reported that close to 430,000 cardiac revascularization surgeries (coronary artery bypass) were performed on nearly 250,000 patients (31). The main motive of these cardiac revascularizations is the development of atherosclerosis, which, if left untreated, leads to myocardial infarction, stroke, and ischemic gangrene (16,35). One of the principal risk factors of atherosclerosis is a high circulating plasma concentration of cholesterol, or low-density lipoproteins (LDLs), which adhere to arterial walls, and along with other secreted inflammatory factors, accumulate within the wall as a lesion, occluding blood flow (33).
Current therapy for congestive heart failure (CHF), a result of ischemic heart disease (IHD) stemming from coronary artery disease (CAD), involves preventing and/or delaying the progression of cardiac dysfunction (2). Such surgical treatments most commonly include coronary artery bypass grafting (CABG), a procedure in which an artery or vein is taken from elsewhere in a patient's body and grafted onto the coronary artery in order to bypass the atherosclerotic narrowing, improving blood supply to the myocardium.
Before discussing the current advances and progress of vascular tissue engineering, it is important to understand the anatomy and physiology of an artery. All arteries stemming from the heart observe the same anatomy, consisting of three layers: the tunica adventitia (outermost layer), the tunica media (middle layer), and the tunica intima (innermost layer). Three main types of cells are involved in the architecture of an artery: adventitial fibroblasts making up the tunica adventitia, smooth muscle cells (SMC) making up the tunica media, and endothelial cells (EC) residing in the tunica intima. SMCs are responsible for the contractile properties of an artery, while the EC layer acts a selectively permeable barrier. The endothelial cells also play an important role in vascular tone, platelet activation, leukocyte adhesion and activation, along with SMC migration and proliferation (23). The fibroblasts occupy the layer of cells that lies above the SMC layer. The mail role of fibroblasts is the production of the extracellular matrix, the supporting scaffold for all cells.
Autologous veins or arteries are sometimes not an option for a patient undergoing a CABG procedure due to complications such as artery disease, trauma, and/or previous surgery (28). In order to avoid these circumstances, previous attempts at engineering blood vessels using synthetic material, such as Dacron and expanded polytetrafluoroethylene (ePTFE), has proven to be useful, yet this option comes with its own set of impediments. For example, using completely synthetic materials may lead to failure of the graft, a result of thrombosis formation, especially in vessels, such as coronary, of 6 mm or less in diameter. Other complications include infection, nondynamic interface in children, and stenosis (11,12,15,39). The field of cardiovascular tissue engineering aims to avoid the problems mentioned above by developing blood vessels using biocompatible materials. The general principle involves seeding cells onto a biocompatible scaffold, culturing the graft in vitro, and then using it to bypass an atherosclerotic occlusion. Within the body, the biocompatible material will eventually degrade, and the patient will be left with cells that have remodeled to form a vessel. To date, many tissue-engineered blood vessels (TEBV) have been used in animal models; however, few have reached the clinical phase.
Several biomaterials have been evaluated for use in vascular tissue engineering for their support of culture of the three cell layers as well as the mechanical properties and the ability to withstand arterial pressure in excess of 2000 mmHg. The biomaterials utilized thus far to engineer vascular grafts have included decelluarized conduits (4), biodegradable hydrogels like collagen (38) and fibrin (13,14), and polymeric scaffolds like poly-glycolic acid (30) and chitosan (27).
Several models of vascular grafts has been reported in the literature (6,7,13,14,19–21,25,26,29,30,32,34,36,38), with an overall objective to fabricate vascular conduit utilizing a suitable biomaterial followed by cellularization with smooth muscle cells followed by the luminal attachment of endothelial cells. Different cellularization approaches have been utilized: direct layering of cell sheets around the peripheral surface or embedding the cells at the time of graft formation.
One of the first models of vascular grafts was reported by Bell and colleagues (38). Primary cells were isolated from a bovine source and mixed with collagen. A three-layer structure was developed to resemble normal vessel anatomy consisting of a medial layer of smooth muscle cells, an adventitia layer of fibroblasts, and with a lining of endothelial cells on the luminal surface of the vascular graft. Later, Nerem and colleagues demonstrated the feasibility of using hydrogels for support of vascular grafts, utilizing collagen, fibrin, and a mixture of the two gels in conjunction with primary rat aortic SMCs to generate small-diameter vascular grafts (7). Niklason and colleagues showed the effects of pulsatile flow from custom designed bioreactors on formation and maturation of vascular grafts (30). Also, a burst pressure of over 2000 mmHg was reported.
L'Heureux and colleagues developed a multiple layered model of small-diameter vascular grafts (26). Human umbilical vein endothelial cells and vascular smooth muscle (VSM) cells were obtained from healthy newborns donors and fibroblasts were obtained from skin specimens from normal adult during breast surgery. SMCs and fibroblasts were cultured independently for 30 days under conditions to promote the formation of extracellular matrix (ECM) and subsequently a cohesive cell monolayer. A sequential approach was utilized for the formation of TEBVs, which consisted of physically wrapping the SMC sheet around a tubular support structure. This was followed by physical wrapping of the fibroblast cell monolayer. ECs were injected in the luminal surface of the vascular graft. The entire process for the formation of TEBV required a time period of 3 months with 3 weeks for the formation of SMC sheets, 1 week for media maturation, 7 weeks for adventitial maturation, and 1 week for endothelization.
Experiments such as the ones listed above provide invaluable insight into the demands of development and maturation of functional vascular grafts to be used in patients.
The current study aims to describe two developing TEBV models in terms of viability and contractile performance. In the first model, we used chitosan, a biocompatible derivative of chitin that is found in the exoskeletons of arthropods, as a supporting scaffold. This material has been previously described as a good scaffold due to its biocompatibility and degradation kinetics (3). In the second model, an acellularized adult rat aorta was used as a support for cells. In both models, a layer of human SMCs embedded within a fibrin gel was wrapped around and cultured for several weeks. Following this incubation, a subsequent layer of human aortic adventitial fibroblasts was wrapped around the SMC layer. Following a 3-week incubation, the vascular grafts were evaluated for viability and contractile performance, using potassium chloride (KCl) as a vasoconstrictor and 8-bromo-cyclic AMP (8-bromo-cAMP) as a vasodilator.
Materials and Methods
NIH guidelines for the care and use of laboratory animals have been observed. All materials were purchased from Sigma (St. Louis, MO) unless otherwise specified.
Plate Preparation
The detailed method for preparing the culture surface has been described in detail (9,10,17,24). Briefly, 35-mm culture plates were coated with 1.5 ml of a SYLGARD polydimethylsiloxane (PDMS) elastomer (Dow Chemical Corporation, Midland, MI). After 2 weeks of curing, the plates provided a layer in which anchor material was able to be pinned. Anchor points, 6-mm-long segments of size 0 braided silk sutures (Ethicon, Cornelia, GA), were pinned 12 mm apart in the center of the culture surface. The SYLGARD coating does not promote cell adhesion, and thus a layer of fibrin gel was added to the surface. Briefly, 500 μl of 10 U/ml thrombin was added to each plate surface and agitated until the entire surface was coated. Next, 200 μl of 20 mg/ml fibrinogen was added and gently swirled. The plates were placed in a 37°C incubator, supplemented with 95% humidified air/5% CO2, for 15 min to allow complete fibrin polymerization and gel formation (Fig. 1).

Vascular graft protocol. The figure outlines the basic protocol for tissue-engineered vascular grafts (TEVGs). First, the scaffolding material is made; chitosan is dissolved in acetic acid, frozen, and lyophylized within a tubular mold, an aorta is excised from an adult rat and subjected to various solutions within a 2-week period in order to remove all cells. Next, a fibrin gel is made from thrombin and fibrinogen in a 35-mm petri dish and cell layer #1 (HuSMC) is added to the gel. After a day, the HuSMC/fibrin gel layer begins to delaminate towards the middle, at which point a tubular scaffold (either acellularized aorta or chitosan tubule) is placed parallel to the two sutures and the layer of cells is wrapped around the scaffold. The same procedure is repeated 2 weeks later with the aortic fibroblasts (HAoAFB), except that the graft already containing a layer of SMCs is used as the supporting scaffold. After 2 weeks in culture, a ring segment is cut out of the graft and analyzed.
Cell Culture
Primary human aortic vascular smooth muscle cells (HuSMCs) were purchased from Cambrex Inc (Cat.#CC-2571, Lot# $F1292; Walkersville, MD), isolated from a 57-year-old Caucasian male donor obtained under informed consent. Primary human aortic adventitial fibroblasts (HAoAFb) were purchased from PromoCell (Cat.# C-12380, Lot#6090101.22; Heidelberg, Germany), isolated from a 47-year-old Caucasian male donor under informed consent. Both cell types were cultured in DMEM (high glucose) medium (Invitrogen, Carlsbad, CA) supplemented with 1% antibiotic-antimycotic (AA) (Invitrogen) and 17% fetal bovine serum (FBS) (Invitrogen) in 100 times; 20-mm culture plates. For the vascular graft, we used HuSMCs at passage 11 (we did not notice a decrease in proliferative potential between passage 11 and lower passages, and decided that these cells would be sufficient for the current, preliminary study) and HAoAFb at passage 5.
Support Scaffold Preparation
Acellularization of Adult Rat Aorta
The protocol for vascular acellularization has been previously described (5). Briefly, five solutions were made with the following compositions: 1) 80% glycerol, 0.9% NaCl, 0.05% NaN3, 25 mM EDTA; 2) 4.2% sodium deoxycholate, 0.05% NaN3; 3) 1% SDS, 0.05% NaN3; 4) 0.05% NaN3; and 5) 3% Triton X-100, 0.05% NaN3. The aorta was excised from adult Sprague-Dawley rats, and washed with DPBS (Invitrogen) to get rid of blood. Next, over a 16-day period, the aorta was exposed to the solutions in the following order: days 1–3 in solution 1; days 3–6 in solution 2; days 6–8 in solution 1; days 8–10 in solution 3; days 10–12 in solution 5; days 12–14 in solution 3; and days 14–16 and indefinitely in solution 4. Before use, the aortas were incubated in 70% ethanol and washed three times in sterile DPBS for 1 h.
Tubular Chitosan Scaffold Preparation
The protocol for chitosan scaffold fabrication has been previously described (3,26). Briefly, chitosan (Cat.#4-00559, Carbomer, San Diego, CA) was solubilized in 1% glacial acetic acid at a concentration of 2% with continuous mixing at room temperature. The mixed chitosan was then poured into a manifold, in between two plastic tubes. This manifold was frozen at −80°C for 2 h and then lyophilized overnight. The dried chitosan scaffold was carefully removed and rehydrated using 0.1 M NaOH for 30 min. The samples were then washed in PBS three times, sterilized in 70% ethanol for 1 h, and stored in PBS until use. The tubular grafts were cut to approximately 15 mm in length.
The tubular samples were cut along the longitudinal axis, placed on a metal stub, and coated with graphite at the contacting ends. The samples were dried in a vacuum dessicator for 6 h and then sputter coated with gold using a Polaron E5100 Sputter Coater. Scanning electron microscopy (SEM) was performed using an AMRAY 1000-B scanning electron microscope using an accelerating voltage of 3 kV.
Fabrication of Vascular Graft
Plates were prepared as described above. Primary HuSMC cells, after being expanded separately in 100 times; 20-mm culture plates, were used at passage 11. Passage 10 HuSMC cells were detached from the culture plates using 3 ml 0.25% trypsin-EDTA (Invitrogen), resuspended in HuSMC/HAoAFb growth media, and counted using a hemocytometer. Each 35-mm culture dish containing polymerized fibrin gel was plated with 500,000 P11 HuSMC cells.
Two days after seeding, the media was removed, and either a 1-cm-long chitosan tube or a 1-cm-long acellularized aorta was placed in between the two sutures, parallel to the delaminating HuSMC cell layer. The two sides of the delaminating layer were taken and physically wrapped around either the chitosan tube or accelularized aorta. These plates were then cultured for 3 weeks in an incubator (37°C, 95% humidified air, 5% CO2). The growth media was modified to include ascorbic acid (50 μg/ml), insulin (2 μg/ml), and transforming growth factor-β (TGF-β) (2.5 ng/ml).
While the first-stage grafts described above were in culture, the same seeding procedure was used to engineer a HAoAFb layer. Briefly, 650,000 HAoAFb (passage 5) were seeded on a fibrin gel-coated 35-mm culture plate. Two days postseeding, the grafts containing the first HuSMC layer were cut at the ends of the inserted support matrix (chitosan tube or accellularized aorta). This was then placed in the same way between the two sutures within the HAoAFb layer. This second layer was subsequently wrapped around the graft and allowed to culture for 3 weeks in an incubator, using growth media described previously, but without any factors added to it.
Cell Viability
Cell viability of TEVGs was assessed using the 4-[3-(4-iodophenyl)-2-(4-nitrophenyl)-2H-5-tetrazolio]-1,3-benzene disulfonate (WST-1) Reagent assay (Roche Applied Science, Indianapolis, IN). A 4-mm ring section was taken from three TEVGs engineered with acellularized aortas and placed into 3 ml of culture media in a 35-mm culture dish. Next, 100 μl of the WST-1 reagent was added to each culture dish containing the TEVG and two blank controls (3 ml of culture media). After an overnight incubation, the absorbencies were read at 485 nm on a spectrophotometer.
Contractile and Relaxation Function
The method for evaluating the contractile and relaxation properties of the vascular grafts was adapted from previous work on engineered cardiac muscle strips and vascular smooth muscle strips (9,10,18). All vascular grafts were tested in a modified Krebs-Hensleit Buffer (KHB) consisting of 114 mM NaCl, 5 mM KCl, 24 mM NaHCO3, 1 mM MgCl2*6H2O, 2.2 mM CaCl2*2H2O, 10 mM HEPES, 0.25% bovine serum albumin (BSA), and 7.8 mM glucose. The buffer was adjusted to pH 7.35 and temperature was adjusted to 37°C before each use.
To test the functionality of the vascular grafts, approximately a 4-mm-thick ring segment was taken from the middle of a vascular graft, testing three different grafts (N = 3), for both the acellularized aorta grafts and chitosan grafts. The ring segment was placed in 3 ml of KHB and fixed at one end with a pin. The other end was also attached to a pin (suspended in the KHB), which was affixed at the top to a custom-built optical force transducer (resolution of 1.4 μN and range of 2 mN) with canning wax (Fig. 2).

Force test set-up. After a ring section is cut out from the center of a tissue engineered vascular graft (TEVG), it is placed in modified Krebs-Hensleit Buffer (KHB) within a 35-mm plate coated with SYLGARD polydimethylsiloxane (PDMS) elastomer. Two pins were inserted within the lumen of the vascular ring, one pinned to the elastomer and the other attached to a force transducer with canning wax. The contraction/relaxation of the ring moves the pin attached to the force transducer needle. The force tension is converted and recorded as a μN force on the computer.
For the mechanical contractility measurements of the TEVGs, the culture dish containing 3 ml of KHB was placed on a heated (37°C) aluminum platform. A standard protocol was followed for measuring spontaneous basal tone of the TEVGs: stretching of the vascular ring, followed by an equilibrium period, approximately 15 min, to allow the tissue to stabilize. This resulted in a new stable baseline of tension.
Spontaneous function was determined after each ring segment stabilized in KHB buffer for about 15 min. The force transducer was turned on to record spontaneous activity for 60 s without any external stimulation.
Next, the contractile and relaxation properties of TEVGs were tested using chemical and mechanical stimulation. The active force was measured using the optical force transducer and force tracings were digitally recorded using Lab-VIEW data acquisition software (National Instruments Corporation, Austin, TX). Force measurements were collected at 100 samples/s for 60 s. The cut out TEVG ring was placed in 2.4 ml of KHB, attached to the force transducer, as described above. Ring tension stabilized over a period of roughly 6 min and reached a stable baseline. To optimize the concentration of KCl to be used, we started with a concentration of 66 mM, as discussed by Hecker et al. (18). We sequentially added 200 μl of 1 M KCl to the TEVG until we were able to measure a contraction. At this point, we stopped addition of KCl and determined that a minimal final concentration of 200 mM (three sequential additions of 1 M KCl) were enough to record a contraction of the TEVG. After an optimal concentration was determined, the contraction of each TEVG was recorded three times with the addition of 600 μl of 1 M KCl (200 mM). Between each contraction, the TEVG ring was washed with KHB three times and allowed to stabilize back to baseline, which required approximately 20 min. Ring segments were used from three different TEVG constructs in order to test contractility.
Following contraction evaluation, we stimulated the TEVG rings with 8-bromo-cAMP to induce vasorelaxation. The rings were washed with KHB three times and allowed to stabilize to baseline before 8-bromo-cAMP was added. The protocol for optimizing the concentration of 8-bromo-cAMP followed the same protocol as for the KCl concentration optimization. Briefly, we started with the addition of 12.5 mM 8-bromo-cAMP (addition of 150 μl of 0.25 M stock to a final volume of 3 ml). We then added another 150 μl of 8-bromo-cAMP for a final concentration of 25 mM. At this concentration, we noted a force decrease (vasodilation) and therefore used 25 mM as the final concentration to induce relaxation of the TEVGs. As with contractile force testing, the TEVG ring segments were allowed to stabilize at baseline tension in KHB, approximately 10 min. Next, 300 μl of 0.25 M stock 8-bromo-cAMP was added (final concentration of 25 mM). A measurable force decrease was recorded and the ring segments were washed three times with KHB and allowed to stabilize back to baseline tension. Addition of 8-bromo-cAMP was repeated two more times. Ring segments were used from three different TEVG constructs.
For the functional testing of this experiment, we used three different TEVGs from both scaffolding conditions (acellularized aorta and chitosan), with ring segments taken from the middle of each graft and tested separately.
Histology
After the TEVGs were sectioned into several rings, one of the rings was used for histological evaluation. Histological evaluation was performed at two time points: postculturing with just HuSMC layer and postculturing with HuSMC and HAoFb layers. Briefly, the TEVG rings were fixed in 10% formalin solution for 24 h. Following fixation, the rings were washed in 1× DPBS three times, for 30 min each. The rings were then stored in 70% ethanol until paraffin embedding. The TEVGs were sectioned into 4-μm sections and then evaluated using a standard hematoxylin and eosin staining protocol.
Burst Pressure
We assembled a pressure monitoring system to measure the small pressure changes within the lumen of the TEVGs. The system consists of a pressure transducer (MLT0670 Disposable BD Transducer, PowerLab, ADInstruments Inc., Colorado Springs, CO), signal conditioning amplifier and data acquisition input/output (I/O) board (PCI-6040E, National Instruments, Austin, TX). The pressure transducer has a working range of −50 to 300 mmHg. The response of the pressure transducer is 5 μV/mmHg per volt of excitation. The data I/O board was controlled by a custom made LabView (National Instruments, Austin, TX) software program running on a personal computer. In order to measure burst pressure, the pressure transducer was secured in the lumen and one of the TEVG secured. A syringe was inserted in the open end of the TEVG and secured using sutures. The luminal pressure was gradually increased using the syringe and changes in pressure were recorded until graft failure. A total of six TEVG were tested.
Statistics
We used the two-sample t-test with Bonferroni correction and Minitab V13.31 (State College, PA) was used for statistical analysis.
Results
TEVG Formation, Cell Viability, and Histology
TEVGs were made using a layering approach, with sequential addition of cell layers onto a biocompatible scaffold, either an acellularized aorta or chitosan tubule (Fig. 3). For the acellularized aortas, histological evidence showed complete removal of cells and an intact extracellular matrix after exposure to the acellularization solutions (Fig. 4). An SEM was taken of the chitosan tubule to show its porous structure (Fig. 4). Within a course of 2 months, approximately 20 TEVGs were constructed, half using aorta as scaffold and the other half using chitosan as scaffold, with a success rate of 100%, with success being defined as complete formation of a bilayer (smooth muscle and FB) vascular graft, evaluated via macroscopic gel compaction.
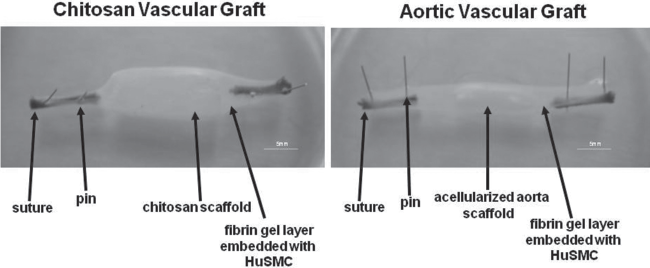
Vascular grafts. Representation of the final vascular graft products in culture, one with chitosan as a supporting scaffold (right) and the other with an acellularized aorta as a supporting scaffold (left).

Material characterization. The scaffolding material for the vascular graft support matrix is shown here. A native aorta taken from an adult Sprague-Dawley rat is contrasted from an acellularized version. An H&E stain of the native vessel shows extracellular matrix (ECM) and cells embedded within the matrix (purple). When acellularized, only the extracellular matrix can be seen in an H&E stain (pink). Lower panel shows a scanning electron microscopy (SEM) image of a chitosan tubule after lyophilization.
In order to determine whether the layered cells had survived the handling and culturing conditions, we performed a standard WST-1 assay. A previous experiment showed that an overnight incubation with the WST-1 reagent provided sufficient time for color development (data not shown). In this study, our main objective was to determine that there are viable cells within the TEVG, compared to blank media. Our results showed a significant difference in absorbance values at 485 nm between blank media (0.15 ± 0.004) and TEVG with aorta (0.27 ± 0.045) and chitosan (0.25 ± 0.017), p = 0.034 and p = 0.008, respectively (N = 3) (Fig. 5). Also, we did not see a significant difference in viability absorbance between aorta scaffolds and chitosan scaffolds.
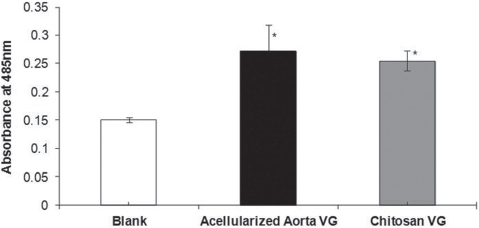
WST cell viability assay. To measure cell viability, we used a standard WST-1 Assay Reagent, measuring mitochondrial activity within metabolically active cells. This figure shows a significant difference in absorbance values at 485 nm between blank media (0.15 ± 0.004) and tissue-engineered vascular grafts (TEVG) with aorta (0.27 ± 0.045) and chitosan (0.25 ± 0.017), p = 0.034 and p = 0.008, respectively (N = 3). Also, we did not see a significant difference in viability absorbance between aorta scaffolds and chitosan scaffolds.
Histological evaluation was performed at two time points, 3 weeks after the HuSMC layer was added and 3 weeks after HAoFb cells were added (6 weeks from initial time point). H&E sections from time point 1 (3 weeks) showed an intact layer of either the acellularized aorta or chitosan surrounded by a layer of purple-stained cells (HuSMC) within a pink-stained fibrin gel. The second time point H&E stain showed a similar pattern, only with an additional layer of cells (FBs) within a fibrin gel matrix surrounding the HuSMC layer (Fig. 6).

Histology: hematoxylin and eosin stain. After 2 weeks in culture, postaddition of just the HuSMC cell layer or addition of both the human-derived smooth muscle cells (HuSMC) and human-derived, aortic, adventitial fibroblasts (HAoAFB) layers, the tissue-engineered vascular grafts (TEVBs) were fixed in 10% formalin solution overnight, embedded within paraffin, and sectioned into 4-μm sections. The top two figures show an H&E stain following 2 weeks in culture with the scaffold (acellularized aorta, left, and chitosan tubule, right) and the first cell layer, HuSMC. The bottom two figures also show an H&E stain, 2 weeks after the addition of the second layer of cells, HAoAFB to the circumference of HuSMC layer.
Functional Testing
Spontaneous Activity
Spontaneous activity was determined by attaching a ring segment of the vascular graft fabricated from a chitosan scaffold or acellularized aorta to the force transducer. Figure 7 shows that TEVGs made with chitosan scaffold supports as well as the TEVGs made with acellularized aortas displayed spontaneous contractile activity.
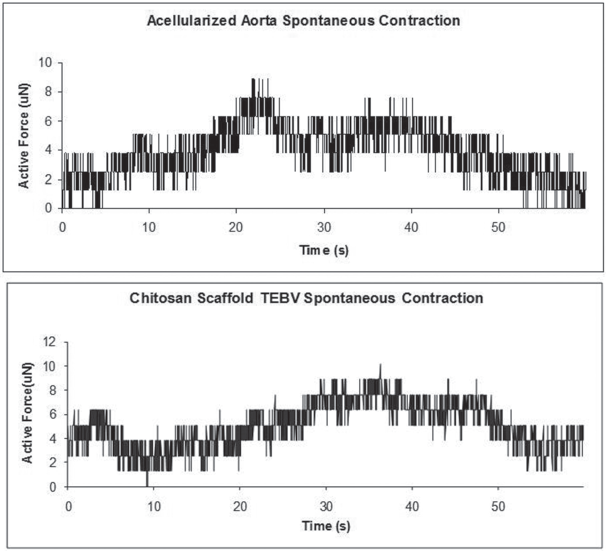
Spontaneous contractions. Representative images of spontaneous contractions exhibited by the two models we evaluated of tissue-engineered vascular grafts (TEVGs).
Contraction with KCl
Upon addition of KCl to the chitosan TEVGs, we saw an increase in force (contraction); however, the graft was unable to maintain the tension. After the initial increase in tension, the force began to decline and continued to decline for several minutes thereafter. Successive additions of KCl and testing of several other rings showed the same pattern and, because of this, we were unable to obtain accurate contractile force readings for the vascular grafts fabricated with a chitosan scaffold.
The vascular grafts made from acellularized aortas were able to maintain tension after stimulation with KCl. The following contractile force was obtained for ring segments 1, 2, and 3, respectively: 38 ± 3, 78 ± 13, and 52 ± 7 μN (N = 3) (Fig. 8). Figure 8 also shows a representative tracing of a typical contraction that we were able to obtain after KCl stimulation.

KCl-stimulated contraction. Ring segments from three vascular grafts made from acellularized aortas were evaluated in terms of their response to the addition of 200 mM KCl. The top panel represents the maximal contractile tension achieved from ring segments, taken from three tissue-engineered vascular grafts: 38 ± 3, 78 ± 13, and 52 ± 7 μN. The bottom panel shows a representative tracing of a typical contractile response we obtained.
Relaxation with cAMP
The TEVG ring segments were washed with fresh KHB, allowed to stabilize to baseline tension, and stimulated to relax with 25 mM 8-bromo-cAMP. For the vascular grafts fabricated with chitosan, the rings were again unable to maintain a tension and thus accurate relaxation readings were difficult to record. Again, ring segments from several different grafts were tested, showing a similar pattern.
The TEVG ring segments made from the acellularized aorta, however, were able to maintain baseline tension as well as their new tension post-8-bromo-cAMP stimulation. The same ring segments utilized for contraction were again utilized to determine the relaxation tension decrease. Briefly, ring segments from three different vascular grafts were utilized, with each ring stimulated three times. The following tension decreases were observed for ring segments 1, 2, and 3, respectively: −23 ± 4, −39 ± 10, and −31 ± 12 μN (N = 3) (Fig. 9). Figure 9 also shows representative images of relaxation that were obtained during stimulation with 8-bromo-cAMP.

8-Bromo-cAMP-mediated relaxation. Ring segments from three vascular grafts made from acellularized aortas were evaluated in terms of their response to the addition of 25 mM 8-bromo-cAMP. The top panel represents the tension achieved from ring segments, taken from three tissue-engineered vascular grafts: −23 ± 4, −39 ± 10, and −31 ± 12 μN. The bottom panel shows a representative tracing of a typical relaxation response we obtained.
Myogenic Responses
TEVGs fabricated using acellularized aortas as scaffold matrices were tested for their myogenic response to an increase and decrease in tension. Figure 10 shows that as we increased the tension of the ring by moving the two pins further apart, the ring segment relaxed. Consequently, when we relaxed the tension on the ring by moving the pins closer together, the ring segment contracted.

Myogenic response. Two representative tracings of myogenic responses of acellularized aorta vascular grafts. The arrows indicate the stimuli: an increase in tension (top) and decrease in tension (bottom), resulting in a relaxation and a contraction of the vascular graft, respectively.
Burst Pressure
The burst pressure was evaluated for TEVGs using acellularized aortas and was found to be 137 ± 14 mmHg (N = 6).
Discussion
The main objective of this study was to develop a viable model of a vascular graft, utilizing biocompatible materials that support the culture of two cell layers found within a native vessel, smooth muscle cells, and fibroblasts. All of our cells were human derived, providing clinical applicability for the model. The success of developing a viable vascular model was measured using several key functional metrics such as histology, cell viability, and contraction/relaxation responses to chemical and mechanical stimulation.
For scaffolding material, we chose to use two biocompatible scaffolds, an acellularized adult-derived aorta and chitosan tube. The acellularized aorta represents an anatomically matched scaffold and can be utilized as a positive control for the chitosan model. The acellularized aorta, as the name implies, contains no cells, and theoretically should not elicit an immune response if placed back into an animal of the same species. The extracellular matrix and its composition should also be biologically the same within a species, further providing support for the biocompatibility of our model. Among the aorta being biocompatible, its strong extracellular network is already fully developed and mature; with the cells being the only part missing. Thus, the culturing time to fully construct a functional vascular graft using an acellularized aorta would be relatively short for patients needing emergency bypass; the graft would also be able to withstand systolic blood pressures that autologous vessels encounter.
Chitosan, a linear polysaccharide, is obtained from the exoskeletons of crustaceans. After the powder is dissolved in acetic acid, it is poured into a mold of any shape and lyophilized. The size of the pores can be controlled by the freezing temperature, with a higher freezing temperature leading to a decrease in pore size (27). Thus, pores can be made large enough to ensure fitting of cells within them. A favorable property of chitosan is that it is easily degraded through the addition of lysozyme to the culture media. As the scaffold gets recellularized, the cells will start to make their own extracellular matrix, concomitantly degrading the chitosan scaffold, leaving the cells and its own proteins to support and withstand autologous blood pressures.
The fabrication of the scaffolds proved to be a relatively simple process. For the acellularized aorta, as described previously, the aorta was taken out of an adult rat and exposed to various solutions within a 2-week period. The chitosan scaffold is also easily fabricated, usually taking about a day to complete. Based on all these observations, we chose to proceed with the comparison of these two scaffolding materials, to determine which one had more superior qualities to make a viable and functional bioengineered vascular graft.
To fabricate the vascular grafts, we utilized a layering approach, by first seeding the cells onto a fibrin gel, and then wrapping the gel with the cells around each of the scaffolds. This method of cellularization proved to be simple and involved little handling. We chose fibrin gel as the support matrix for the cells because of our extensive knowledge with the way it interacts with cells (17, 24). Also, it is a biocompatible material that is easily degraded by the cells themselves, as they begin to form their own extracellular matrix. Between the subsequent layering of cells we allowed the constructs to stabilize for a period of 3 weeks. Within that time, we noticed that the fibrin gel began to degrade as the cells laid down their own extracellular matrix, based on the macroscopic observation that the thickness of the graft decreased with culture time.
After the vascular grafts matured in culture for approximately 6 weeks, we wanted to determine if the scaffold supports (both the acellularized aorta and chitosan tubule) were biocompatible enough to support the culture of the cells that we added. To verify cell viability, we used a standard WST-1 assay, which measures the byproduct of mitochondrial metabolism. We found that the absorbance readings for the chitosan vascular graft (0.25 ± 0.017) and the acellularized aorta vascular graft (0.27 ± 0.045) were comparable, with no significant difference. The results from the assay confirmed that the scaffolds were able to support the viability of cells in culture for an extended period of time and with this information, we were able to proceed with histology and functional testing.
Histological analysis further supported our findings that both of the scaffolding materials were able to support cellularization. Figure 6 shows two separate time points, one is 3 weeks after cellularization with HuSMC and the second time point is 3 weeks after cellularization with HAoAFbs (6 weeks after start of study). The H&E sections at the first time point showed either a chitosan or acellularized aorta in the middle of the graft, surrounded by a layer of fibrin gel (stained pink) and cells (stained purple). The H&E stains for the second time point showed a similar result, only with the addition of one more layer of fibrin gel with cells. We did not see very much migration of cells from the fibrin gel layer into the scaffold, a possible result of not enough time in culture.
In order to further compare the two models, we proceeded with functional assessment, in response to chemical and mechanical stimulation. First, we evaluated the spontaneous contractions of the grafts after stabilization of baseline tension. Next, we determined whether the grafts will respond to chemical stimulation; more specifically, if a contraction will result after addition of KCl, and if a relaxation will result after addition of 8-bromo-cAMP.
Both of the vascular graft models displayed spontaneous activity after baseline tension stabilization, as can be seen in Figure 7. Upon addition of KCl and 8-bromo-cAMP, both of the vascular grafts displayed contraction and relaxation, respectively. For the TEVG made with acellularized aortas, the average contractile tensions for three constructs were 38 ± 3, 78 ± 13, and 52 ± 7 μN, while the average decrease in tensions in response to 8-bromo-cAMP were −23 ± 4, −39 ± 10, and −31 ± 12 μN. In comparison to a young adult abdominal aorta, Delp et al. recorded a force of roughly 1.4 g/mm2 (~13 mN) when stimulated with KCl (8). Although the forces that we obtained were not in the same magnitude as the forces that are found in literature of native rat aortas, we believe that the presence of small forces is enough evidence to show and improve on the vascular graft model that we propose.
During chemical stimulation, we noticed that the vascular graft made from chitosan was unable to maintain its contracted and relaxed tension as the vascular graft made from acellularized aorta. Primarily, when we added KCl to the buffer, we saw an instant contraction. However, as we allowed the graft to stabilize, the tension did not maintain at the contracted level. After roughly 20 min of stabilization, we were still unable to record a steady level of tension. The same results were obtained when we added 8-bromo-cAMP; the decreased tension would not stay constant, and continued to decrease. Because of this, we were unable to acquire accurate readings and, using this observation, concluded that the more superior vascular graft is one that uses an acellularized aorta as a supporting scaffold. Also, the amount of time needed to stabilize the chitosan vascular graft was much greater than the time needed to stabilize the acellularized aorta vascular graft, and thus this point also supported our decision to proceed with further model development of the acellularized aorta vascular graft. The results attained from the chemical stimulation of the chitosan vascular graft model does not necessarily mean that vascular graft research should be regressed from biomaterials; it simply pushes researchers in a direction to develop more suitable biomaterials, ones that match the properties of native blood vessel scaffolds (37).
This preliminary experiment narrowed down the vascular graft model to one that utilizes acellularized aortas as a support scaffold, a result of cell viability, histology, and functional data. We have compared our chosen vascular graft model with the model described previously (1,26). The acellularized aorta model that we describe here takes less time to mature during the initial steps as the model described by L'Heureux et al. (26). For example, with our vascular model, a cell layer is ready to be wrapped around a scaffold 1 day after seeding the fibrin gel with cells, compared to 30 days of individual cell maturation and laying down of extracellular matrix before being able to wrap the cell layers around a mandrel. With this information at hand, we will proceed to further characterize this model in subsequent experiments. Within these studies, we will add the final endothelial layer to the lumen of the graft and determine the grafts functionality using adrenergic stimulation for vasoconstriction and endothelial function for vasorelaxation. We will also measure burst pressure, as it is crucial for a vascular graft to be able to withstand native systolic pressures. Along with further model characterization, our goal is to develop a novel perfusion/culturing/bioreactor system that will provide adequate pulsatile flow to the graft, in order to further promote cellular organization. The last several factors that we will look into will be longevity studies, in order to determine for how long the vessels will be able to generate contractile force in culture, and, finally, the last step would be to put the vessel back into a rat model, to make sure that there is no immunological response from the animal. After model development, we will switch to the use of mesenchymal stem cells, differentiating them into the three layers present in a native vessel. By using mesenchymal stem cells, the patient can the cell donor, thus eliminating the need for immunosuppressant therapies postimplantation (22,40).
Footnotes
Acknowledgments
This work was supported by funds from the Section of Cardiac Surgery at the University of Michigan. The authors declare no conflicts of interest.
