Abstract
Combination therapy with a dipeptidyl peptidase-4 inhibitor (DPP-4i) and a proton pump inhibitor (PPI) raises endogenous levels of GLP-1 and gastrin, respectively, and restores pancreatic β-cell mass and normoglycemia in nonobese diabetic (NOD) mice with autoimmune diabetes. The aim of this study was to determine whether a DPP-4i and PPI combination could increase β-cell mass in the adult human pancreas. Pancreatic cells from adult human pancreas donors were implanted in NOD-severe combined immunodeficient (NOD-scid) mice and the mice were treated with a DPP-4i and a PPI for 16 weeks. Human grafts were examined for insulin content and insulin-stained cells. Graft β-cell function was assessed by intravenous glucose tolerance tests (IVGTT) and by glucose control in human cell-engrafted mice treated with streptozotocin (STZ) to delete mouse pancreatic β-cells. Plasma GLP-1 and gastrin levels were raised to two- to threefold in DPP-4i- and PPI-treated mice. Insulin content and insulin-stained cells in human pancreatic cell grafts were increased 9- to 13-fold in DPP-4i and PPI-treated mice and insulin-stained cells were colocalized with pancreatic exocrine duct cells. Plasma human C-peptide responses to IVGTT were significantly higher and STZ-induced hyperglycemia was more completely prevented in DPP-4i- and PPI-treated mice with grafts than in vehicle-treated mice with grafts. In conclusion, DPP-4i and PPI combination therapy raises endogenous levels of GLP-1 and gastrin and greatly expands the functional β-cell mass in adult human pancreatic cells implanted in immunodeficient mice, largely from pancreatic duct cells. This suggests that a DPP-4i and PPI combination treatment may provide a pharmacologic therapy to correct the β-cell deficit in type 1 diabetes.
Keywords
Introduction
Many pancreatic β-cell growth factors have been identified, one of the most promising being glucagon-like peptide-1 (GLP-1), a peptide secreted from intestinal endocrine L-cells in response to nutrient ingestion. GLP-1 is rapidly cleaved and inactivated in vivo by dipeptidyl peptidase-4 (DPP-4), a ubiquitous serine protease. DPP-4 inhibitors (DPP-4i) raise circulating levels of active GLP-1 and thus increase and maintain effective concentrations of this peptide reaching target tissues (4). Studies in humans with type 2 diabetes have shown that DPP-4i therapy improves glucose tolerance (1). In addition, DPP-4i treatments can preserve pancreatic β-cell mass in animal models of type 2 diabetes (9) and stimulate β-cell regeneration in streptozotion (STZ)-induced diabetic rats (11).
Gastrin is another gastrointestinal peptide reported to induce β-cell neogenesis from pancreatic exocrine duct cells in rodents (12,18). The combination of GLP-1 and gastrin was found to increase the β-cell mass in adult human pancreatic cells (14) and restore normoglycemia in nonobese diabetic (NOD) mice (15). In addition, combination therapy with a DPP-4i and a proton pump inhibitor (PPI) raised circulating levels of GLP-1 and gastrin respectively and reversed diabetes in NOD mice (13). Here, we report that combination therapy with a DPP-4i and a PPI raised endogenous levels of GLP-1 and gastrin in immunodeficient mice implanted with adult human pancreatic cells, and this combination therapy significantly expanded the functional β-cell mass in the human pancreatic cell grafts.
Materials and Methods
Human Pancreatic Cells
Human pancreases were obtained, with informed consent of relatives, from brain-dead adult organ donors. Tissue procurement and experimental protocols were approved by the human ethics committee of the University of Alberta Hospitals. Pancreatic islets were isolated as previously described (6) and islet preparations with low endocrine cell purity (<20% dithizone-stained cells) were used in this study. Islets were dissociated into single cells and small clumps (≤5 cells) as previously described (7). Cells from the nine donor pancreases used in this study contained 16 ± 2% β-cells, 6+1% α-cells, 20 ± 3% exocrine duct cells, and 31 ± 4% acinar cells (see Immunohistochemical Studies).
Cell Implants in Mice
NOD-severe combined immunodeficient (NOD-scid) female mice, age 6–8 weeks, purchased from Jackson Laboratory (Bar Harbor, ME), were used as recipients of the human pancreatic cell implants. The mice were housed and fed under specific pathogen-free conditions in a biocontainment hood and were cared for according to the guidelines of the Canadian Council on Animal Care. Pancreatic cell implants were placed under the left renal capsule of the mice according to a previously described procedure (17).
Experimental Studies
In the first study, NOD-scid mice were divided into four groups. One group of mice (n = 6) was sham implanted. The other three groups of mice were implanted with human pancreatic cells (2 × 106) obtained from three different human organ donors, and cells from each pancreas were distributed to four mice in each of the three groups (n = 12 mice/group). The human pancreatic cell grafts with a portion of underlying kidney were removed 1 day after implantation in the first group of mice, and the mice were killed. The other two groups of mice with human pancreatic cell grafts were treated for 16 weeks with either i) a DPP-4i, sitagliptin (Merck Frosst, Canada) 100 mg/kg given by oral gavage once daily and a PPI, pantoprazole (Nycomed, Oakville, Canada) 40 mg/kg given by SC injection twice daily or ii) the DPP-4i vehicle, methylcellulose in H2O and the PPI vehicle, PBS. After 16 weeks, blood was collected 8–10 h after treatments for assays of GLP-1 and gastrin. Grafts with a portion of underlying kidney were removed from six mice of each group for immunohistochemical and immunofluorescence studies and from six mice of each group for assay of insulin content.
In the second study, NOD-scid mice were divided into three groups of 6 mice/group. The first group was sham implanted. The other two groups were implanted with human pancreatic cells (2 × 106) obtained from two different human organ donors, and cells from each pancreas were distributed to three mice in each of the two groups (n = 6 mice/group). One group was treated with DPP-4i and PPI and the other with vehicles for 16 weeks as in the first study. All mice were then fasted and treatments stopped for 24–28 h before intravenous glucose tolerance tests (IVGTT) were carried out by IV injection of 1.5 g/kg glucose. Blood was collected for glucose and human C-peptide levels before and 10, 30, and 120 min after glucose injection.
In the third study, NOD-scid mice were divided into three groups. The first group of mice (n = 10) was sham implanted. The other two groups were implanted with human pancreatic cells (2 × 106) obtained from four different human organ donors, and cells from each pancreas were distributed to three mice in each of the two groups (n = 12 mice/group). One group was treated with DPP-4i and PPI and the other with vehicles for 16 weeks as in the first study. Treatments were stopped and all mice were treated with streptozotocin (STZ, Sigma-Aldrich, St. Louis, Mo) 70 mg/kg given by IP injection once daily for 6 days to destroy β-cells in mouse pancreas without damaging β-cells in the human pancreatic cell grafts (2). Blood glucose concentrations were measured every 3–5 days for 34 days after STZ administration, and then the left kidneys with the human pancreatic cell grafts were removed from the anesthetized mice. The mice recovered and blood glucose concentrations were measured daily until they exceeded 25 mmol/L; then the mice were killed.
Blood Glucose
Blood glucose concentrations were measured using a blood glucose meter and test strips (Ascencia Elite, Bayer, Etobicoke, ON, Canada).
Peptide Hormone Assays
Active GLP-1(7–36) amide in mouse plasma was measured using an enzyme linked immunosorbent assay (ELISA) electrochemiluminescence assay (Meso Scale Discovery, Gaithersburg, MD). Serum gastrin was measured using a radioimmunoassay (RIA) for the major circulating forms of mouse gastrin (MP Biomedicals, Orangeburg, NY). Plasma human C-peptide was measured using a RIA with no cross-reactivity to mouse C-peptide (Linco Research, St. Charles, MO). Pancreatic insulin content was measured in ethanolic extracts of the human pancreatic cell grafts using an electrochemiluminescence immunoassay and analyzer (Roche, Mannheim, Germany).
Immunohistochemical Studies
Composition of the human pancreatic cells before implantation in mice was determined according to a previously described method (14). Briefly, β-cells were identified by staining with a guinea pig anti-insulin antibody, α-cells by staining with a rabbit anti-human glucagon antibody, exocrine duct cells by staining with a mouse anti-human CK19 antibody, and acinar cells by staining with a rabbit anti-α-amylase antibody. Primary antibodies were followed by secondary biotinylated antibodies and streptavidin enzyme conjugates and substrate kits. The β-cell mass in human pancreatic cell grafts removed from the mice was determined by point-counting morphometry on insulin-immunostained sections of the grafts according to a previously described method (15). Briefly, five sections from each graft were scanned using a Scanscope Imagescope system (Aperio Technologies, Vista, CA) at a magnification 40×. The digital images were analyzed with Scanscope software and the number of positive signals (insulin-stained cells) as a percent of the total (positive plus negative) signals representing the total graft tissue area scanned was determined by Positive Pixel Count Algorithm (Aperio). The β-cell area per graft was calculated by multiplying the percent of insulin-positive signals by the total area of the graft tissue scanned.
Immunofluorescence Studies
Tissue samples were fixed overnight in Zinc Formal-Fixx (Fisher Scientific, Ottawa, Canada) and then processed and embedded in paraffin, after which 3-μm sections were cut and placed on histobond slides (Fisher Scientific). The sections were rehydrated to water, followed by the quenching of endogenous peroxide by a solution of 20% hydrogen peroxide in methanol for 6 min. Blocking was performed with 20% normal goat serum (Jackson ImmunoResearch Laboratories, West Grove, PA) for 15 min at room temperature. Primary antibody concentrations were as follows: 1:1000 guinea pig anti-insulin (Dako Cytomation, Missisauga, Canada), 1:50 mouse anti-CA19–9 (AbD Serotec Raleigh, NC). The primary antibody incubation was 1 h at room temperature, followed by three washes in PBS. This was followed by secondary antibody staining for 30 min at room temperature using goat anti-guinea pig AlexaFluor 488 and goat anti-mouse AlexaFluor 594 (Invitrogen, Burlington, Canada). Slides were then cover slipped using Prolong Gold antifade plus DAPI (to stain nuclei), and visualized on an Axioplan 2 fluorescent microscope using Axioplan 4.1 multidimensional acquisition software (Carl Zeiss Canada, Toronto, Canada).
Statistical Analyses
Data are expressed as means ± SE. The Mann-Whitney rank sum test was used to compare two groups, and a one-way ANOVA followed by Dunnett's multiple comparison tests was used when more than two groups were compared. A value of p < 0.05 was considered significant. Analyses were done using InStat 3 Windows (Graph Pad, Software, San Diego, CA).
Results
Plasma GLP-1 and serum gastrin levels were elevated two- to threefold in NOD-scid mice implanted with human pancreatic cells and treated with DPP-4i and PPI compared with levels in mice implanted with pancreatic cells and treated with vehicles, or mice not implanted or treated (Fig. 1a, b). Immunohistochemical examination of the human pancreatic cell grafts revealed a striking expansion in the size of the graft in mice treated for 16 weeks with DPP-4i and PPI (Fig. 1e) compared with grafts initially implanted (Fig. 1c) and grafts in mice treated for 16 weeks with vehicles (Fig. 1d). Importantly, the area of insulin-stained cells (mm2 × 10−3) was significantly increased in human pancreatic cell grafts in mice treated with DPP-4i and PPI (60.0 ± 6.3) compared with grafts initially implanted (4.6 ± 0.4) and grafts in mice treated with vehicles (11.3 ± 1.7) (Fig. 1f). Also, insulin content (μg/graft) was significantly increased in human pancreatic cell grafts in DPP-4i- and PPI-treated mice (15.2+1.8) compared with grafts initially implanted (1.6 ± 0.2) and grafts in mice treated with vehicles (2.5 ± 0.2) (Fig. 1g). The area of the human pancreatic cell grafts occupied by cells stained for the CK-19 duct cell marker (mm2 × 10−3) was also greatly increased in the human pancreatic cell grafts in DPP-4i-and PPI-treated mice (57.2 ± 10.0) compared with grafts initially implanted (5.4 ± 1.1) and grafts in mice treated with vehicles (2.8 ± 0.5). Immunofluorescence study of the human pancreatic cell grafts in DPP-4i-and PPI-treated mice revealed that essentially all insulin-stained cells were colocalized to cells stained for the pancreatic exocrine duct cell marker CA19–9 (Fig. 2). Plasma human C-peptide basal levels and responses to IVGTT were significantly higher in DPP-4i- and PPI-treated mice with human pancreatic cell grafts compared with vehicle-treated mice with grafts (Fig. 3b) and blood glucose clearance was more rapid (Fig. 3a).
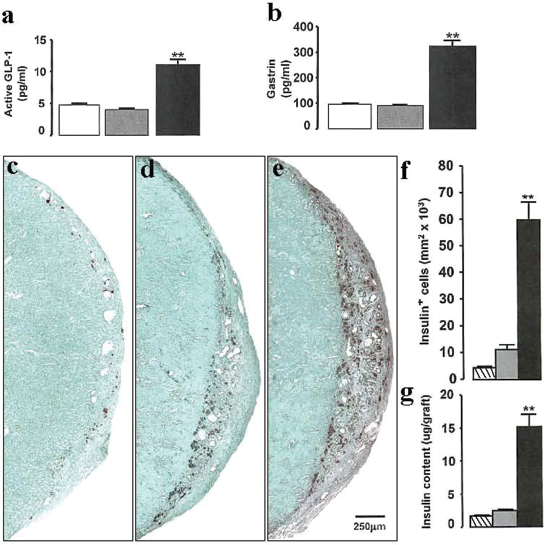
Effects of dipeptidyl peptidase-4 inhibitor (DPP-4i) and proton pump inhibitor (PPI) in nonobese diabetic severe combined immunodeficient (NOD-scid) mice implanted with human pancreatic cell grafts. Plasma active glucagon-like peptide-1 (GLP-1) (a) and serum gastrin (b) levels in mice that received grafts and were treated for 16 weeks with DPP-4i and PPI (black bars) or with vehicles (shaded bars) and in mice sham-implanted and not treated (white bars). Photomicrographs of histological sections of human pancreatic cells implanted under the renal capsule of NOD-scid mice show few insulin-containing cells (stained dark brown) in grafts at day 1 (c), more insulin-containing cells in grafts of vehicle-treated mice at 16 weeks (d), and many more insulin-containing cells in grafts of DPP-4i- and PPI-treated mice at 16 weeks (e). Insulin-stained cells (f) and insulin contents (g) in the grafts at day 1 (striped bars), and at 16 weeks after treatment with vehicles (shaded bars), or DPP-4i and PPI (black bars). Mean ± SE values are shown for six mice in each group. **p < 0.01 for DPP-4i and PPI versus vehicle-treated mice.

Photomicrographs of histological sections of human pancreatic cell grafts under the renal capsule of NOD-scid mice treated with DPP-4i and PPI for 16 weeks. Immunofluorescence double staining for insulin-positive cells in green (A) and cells expressing a pancreatic exocrine duct cell marker, CA19–9 positive in red (B) reveals that essentially all insulin-positive cells were colocalized with CA19–9-positive duct cells, with the double positive cells appearing yellow in the merged photomicrograph (C). Insets delineate corresponding areas in each panel.

NOD-scid mice were implanted with human pancreatic cell grafts and treated with DPP-4i and PPI (triangles) or vehicles (solid circles) for 16 weeks and other NOD-scid mice were sham implanted and not treated (open circles). Mean ± SE blood glucose (a) and plasma human C-peptide (b) concentrations are shown before (0 min) and after (10, 30, and 120 min) IV injection of 1.5 g/kg glucose for six mice in each group. *p < 0.05, **p < 0.01 for DPP-4i and PPI versus vehicle-treated mice.
To determine the impact of insulin secretion from the human pancreatic cell grafts on blood glucose levels independent of insulin secretion from host mouse pancreatic β-cells, we treated the mice with STZ to destroy mouse pancreatic β-cells without affecting human β-cells in the grafts (2). STZ treatment rapidly induced severe hyperglycemia (>25 mmol/L) and death in mice that had not been implanted with human pancreatic cell grafts, whereas mice that had received grafts were relatively protected from diabetes development (Fig. 4). Blood glucose levels rose to 10–15 mmol/L after STZ treatment in mice with human pancreatic cell grafts and treated with vehicles, but to only 7–8 mmol/L after STZ treatment in mice with human pancreatic cell grafts and treated with DPP-4i and PPI. Severe hyperglycemia rapidly developed when the human pancreatic cell grafts were removed from mice that had received either vehicle or DPP-4i and PPI treatments, indicating that blood glucose control in the STZ-treated mice depended on functional β-cells in the human pancreatic cell grafts, and that this was significantly better in mice that had been treated with DPP-4i and PPI than with vehicles (Fig. 4).
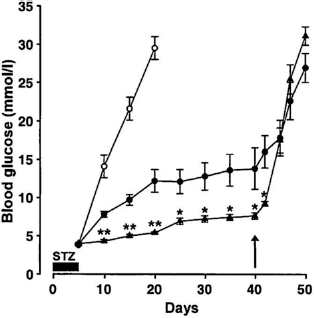
NOD-scid mice were implanted with human pancreatic cell grafts and treated with DPP-4i and PPI (triangles) or vehicles (solid circles) for 16 weeks. Treatments were stopped and these mice as well as sham-implanted and untreated NOD-scid mice (open circles) received streptozotocin (STZ) over 6 days. Blood glucose levels are shown after STZ administration and after removal of the grafts at day 40 (arrow). Mean ± SE values are shown for 10–12 mice in each group. *p < 0.05, **p < 0.01 for DPP-4i and PPI versus vehicle-treated mice.
Discussion
Hyperglycemia in both type 1 and type 2 diabetes results from a relative or absolute deficit in the pancreatic β-cell mass; therefore, β-cell regeneration is an area under active investigation. Clinical interpretation of β-cell regenerative therapies is limited; however, because these studies have been almost exclusively carried out in rodent models of diabetes or under in vitro conditions.
In the present study, we found that the DPP-4i and PPI combination of sitagliptin and pantoprazole raised circulating levels of GLP-1 and gastrin in immunodeficient mice, and insulin-stained cells and insulin content in adult human pancreatic cells implanted in the mice were increased 9- to 13-fold over 16 weeks. The elevations of circulating levels of GLP-1 and gastrin resulting from DPP-4i and PPI treatments were likely responsible for the increased β-cell mass in the human pancreatic cell grafts because we have previously found that combination therapy with GLP-1 and gastrin expanded the β-cell mass in human pancreatic islet grafts in immunodeficient diabetic mice, and this was largely from exocrine duct cells associated with the islets (14). Similarly, in the present study the increased β-cell mass induced by DPP-4i and PPI therapy appeared to be derived from pancreatic exocrine duct cells. This finding is consistent with previous reports that β-cells can be generated by transdifferentiation from pancreatic exocrine duct cells in the adult human pancreas (3,7,10,19). Expression of glucose transporters and other markers of β-cell maturity would be required, however, to establish the phenotypic maturity of the ductal cell-derived β-cells in the present study. Nevertheless, the expanded human β-cell mass in the grafts of DPP-4i- and PPI-treated mice was functional, as shown by human plasma C-peptide responses to IVGTT and by maintenance of normoglycemia in the human pancreatic cell-engrafted mice whose own pancreatic β-cells were destroyed by STZ.
Interestingly, we observed a modest expansion of functional β-cells in human pancreatic cell grafts in immunodeficient mice treated with vehicle only for 16 weeks. This suggests that the environment in vivo contributed in part to the expansion of β-cell mass in the human pancreatic cell grafts in DPP-4i- and PPI-treated mice. Similarly, several studies report that engrafting human embryonic stem cell-derived pancreatic endocrine progenitor cells into immunodeficient mice promotes their maturation in vivo into functional β-cells; however, with a limited capacity to regulate blood glucose levels (8). In this study, we found that elevation of circulating levels of GLP-1 and gastrin in mice with human pancreatic cell grafts expanded the mass of functional β-cells in the human cell grafts to a degree sufficient to regulate blood glucose levels.
The results of this study with adult human pancreatic cells, together with our findings in acutely diabetic NOD mice (13), suggest that a DDP-4i and PPI combination treatment that raises GLP-1 and gastrin levels may provide a pharmacologic therapy to restore pancreatic β-cell mass and function in patients with recent-onset type 1 diabetes. Also, immunosuppressive drugs may not be required because reversal of autoimmune diabetes in NOD mice is accompanied by effects of GLP-1 and gastrin on immunoregulatory cytokines (15) and by effects of DPP-4 inhibitors on T-cell subsets (5,16), in addition to stimulation of β-cell regeneration.
Footnotes
Acknowledgments
This research was supported by a Juvenile Diabetes Research Foundation International grant 17-2007-1043. The authors declare no conflict of interest.
