Abstract
Because human lungs are unlikely to repair or regenerate beyond the cellular level, cell therapy has not previously been considered for chronic irreversible obstructive lung diseases. To explore whether cell therapy can restore lung function, we administered allogenic intratracheal mesenchymal stem cells (MSCs) in the trachea of rats with chronic thromboembolic pulmonary hypertension (CTEPH), a disease characterized by single or recurrent pulmonary thromboembolic obliteration and progressive pulmonary vascular remodeling. MSCs were retrieved only in high pressure-exposed lungs recruited via a homing stromal derived factor-1α/ CXCR4 pathway. After MSC administration, a marked and long-lasting improvement of all clinical parameters and a significant change of the proteome level were detected. Beside a variation of liver proteome, such as caspase-3, NF-κB, collagen1A1, and α-SMA, we also identified more than 300 resident and nonresident lung proteins [e.g., myosin light chain 3 (P16409) or mitochondrial ATP synthase subunit alpha (P15999)]. These results suggest that cell therapy restores lung function and the therapeutic effects of MSCs may be related to protein-based tissue reconstituting effects.
Keywords
Introduction
Chronic thromboembolic pulmonary hypertension (CTEPH) has emerged as one of the leading causes of severe pulmonary hypertension (PH). It is characterized by single or recurrent unresolved pulmonary thromboemboli obstructing and/or obliterating the pulmonary vascular bed. Pulmonary embolism is, however, thought to be the initiating event followed by progressive pulmonary vascular remodeling. This leads to elevation of pulmonary vascular resistance, progressive pulmonary hypertension, and right ventricular failure and death. Although it has been investigated for years, the exact mechanisms influencing its etiopathology and outcome are still poorly understood (24,30,36,40). Vascular disobliteration by pulmonary endarterectomy (PEA) is the preferred treatment but not all patients are eligible for surgery (11–13,15,21). Other therapeutic alternatives include lung transplantation and balloon pulmonary angioplasty (7,16). Clinical evidence regarding pharmacologic strategy is currently restricted to small, uncontrolled trials with, at most, a moderate outcome (12). The tissue engineering, which is currently applied in several studies, might be the wrong method to design such a highly complex and multifunctional organ like the lung when targeting the clinical application.
Cell-based therapy, latterly used in experimental studies (38,45), might be a more promising therapeutic option for lung diseases and the treatment of CTEPH. The potential role of mesenchymal stem cells (MSCs) as a treatment for CTEPH has not been previously studied. Bone marrow-derived MSCs are so-called multipotent adult stem cells. They have the ability to differentiate into various cell types (5,32,34). When engrafting into the lung they can differentiate into type I and II epithelial cells and fibroblasts. Other lung injury models demonstrated an improvement after MSC administration (27,35) and this was explained not only by their tissue incorporation but also their immunomodulatory capability (1) and paracrine signaling.
We now demonstrate that intratracheal MSC administration in our previously established experimental animal model of CTPEH (14) results in a restoration of both lung and liver tissue and a significant improvement of all clinical parameters. These remarkable changes might be explained via the altered protein expression level.
Materials and Methods
Animals
Male Lewis rats (n = 40) (Charles River GmbH, Sulzfeld, Germany) weighing 200–250 g were used for this experimental study: n = 10 animals as cell donors and n = 30 for three experimental groups. Anesthesia and ventilation of the animals were performed according to our previously described protocol (14). CTEPH was induced by the ligation of the left pulmonary artery (PA) (14). After the surgical intervention a 1-ml syringe was attached to the oropharyngeal tube and a total of 6 × 106 carboxy-fluorescein succinimidyl ester (CFSE; Invitrogen, Cell Trace, Paisley, UK) and bromodeoxyuridine (BrdU, Millipore, Billerica, MA) labeled allogenic MSCs were immediately administered into the trachea. The ventilation of the animals was continued after cell administration for an additional 5 min to provide cell distribution into the distal bronchi. All animal care and experimental procedures were performed under approval from the Animal Care and Use Committee and the Bioethic Committee of the University of Barcelona. Animals were sacrificed 12 weeks after induction of CTEPH and administration of MSCs, and organs were harvested for the investigations.
Hemodynamic and Blood Gases
Hemodynamic parameters and blood gases were measured at baseline and 12 weeks after surgical intervention. Anesthesia was induced as described and hemodynamic measurement started after the animals had stabilized. The animals were placed in the supine position and an arterial pressure line (Physiological Pressure Transducer, MLT1199; ADInstruments, Oxfordshire, UK) inserted into the left carotid artery. An ultra-miniature microtip catheter (SPR-1000; Millar Instruments, Inc., Houston, TX) was inserted in the left internal jugular vein. Both were connected to a PowerLab 4/30 (ML866/P; ADInstruments) and a Cardiac Output Pod (ML313B; ADInstruments). The following hemodynamic variables were recorded for 3 h: heart rate (HR, bpm), cardiac output (CO, ml/ min), mean pulmonary arterial pressure (mPAP, mmHg), and pulmonary vascular resistance (PVR, mmHg ml−1 min−1). Blood samples were taken to measure the partial arterial pressure of carbon dioxide (PaCO2) and partial arterial pressure of oxygen (PaO2).
Plethysmography
Twelve weeks after PA ligation, the animals were checked via plethysmography for respiratory functions using a plethysmograph animal chamber developed by Buxco (Buxco Electronics, Troy, NY). The chamber was an unrestrained whole body chamber that allows for long-duration, long-term, and anesthesia-free investigations. The animals were placed into the chamber and the respiratory rate and enhanced pause (Penh) were measured for 1 h. Penh was calculated using the formula: Penh = ((Te - Tr)/(Tr x PEF/PIF)), where Penh = enhanced pause (dimensionless), Te = expiratory time (s), Tr = relaxation time (s), PEF = peak expiratory flow (ml/s), and PIF = peak inspiratory flow (ml/s). Penh was measured and averaged approximately every 5 s, and the cumulative values were averaged as the Penh value for each time point.
Forced Swim Test
We used a swim test (“forced swim test”) 12 weeks after ligation to compare the agility between animals of different groups, according to our previously described method (14). Animals were carried over into a container filled with water and left inside for 10 min, to allow for adaption to the water. Then we measured the time of ‘swimming” (for 7 min), which was defined as moving all four extremities under water. This measured time was thereafter subtracted from the total time (=420 s) to obtain immobility. High values were correlated with limited physical strain. To prevent research outcomes from being biased, two independent observers were responsible for the measurement.
MSC Isolation and Culture
We removed the thigh bone from donor animals (n = 10) and placed it into culture medium [l-DMEM (Gibco, Prat de Llobregat, Spain) supplemented with 10% fetal bovine serum (FBS; Biological Industries, Beit Haemek, Israel), 1% antibiotics (Sigma Chemical Company, Barcelona, Spain), and amphotericin B (Sigma)]. Thereafter we gently flushed out the bone marrow and purified the cells by density centrifuge (1500 rpm for 15 min) using Percoll plus (GE Healthcare, Uppsala, Sweden). Cells (1 × 105/ml) were cultivated in l-DMEM supplemented with 10% FBS (Biological Industries). After 24 h of culture (5% CO2, 37°C), nonadherent cells were removed and the medium was changed to MesenPro RS (Gibco) supplemented with 2% FBS, 2 mM l-glutamine, and growth supplement (Gibco). The adherent MSCs were grown to 90% confluency (~7 days, defined as MSCs at passage 0). A fluorescence-activated cell sorter (FACS) was used to analyze and verify the cells as MSCs. Cells were incubated for 10 min at room temperature with 10 μl of mouse serum (BD Biosciences Pharmingen, San Jose, CA), followed by an incubation with 1.6 μl phycoerythrin (PE)-Cy5 anti-rat CD 45 and 1.6 μl fluorescein isothiocyanate (FITC)-labeled anti-rat CD90 (both BD Biosciences). At the studies' end-point, the organs were harvested and the administrated cells were detected via immunochemistry following the manufacturer's instruction.
Labeling and Administration of MSCs
To detect MSCs after intratracheal injection, 2 × 106 cells were labeled using a CFSE cell Tracer kit at a concentration of 500 μg of CFSE and an incubation time of 15 min. MSCs (4 × 106) were also incubated with a final concentration of 10 μM of BrdU at 37°C for 2 h. Cell viability was determined using the trypan blue exclusion method. The cell suspension was decanted into microtubes and stocked on ice until the ligation of the PA was performed. After MSC administration, the animals were reattached to the ventilator for 5 min using a ventilation pressure of 12 cm H2O to aid in the distribution of MSCs to distal bronchi and airspaces. The animals were disconnected from the ventilator and extubated once they started to respire adequately. Labeled cells were then detected after organ harvesting via fluorescence or confocal microscopy.
Harvesting and Analysis of Organs
Animals were sacrificed 12 weeks after surgical procedure with a bolus IV injection of pentobarbital (200 mg/kg). Lung, heart, and liver tissue samples were taken from each animal group and placed in formalin for 18 h and then in 70% methanol for 24 h or snap-frozen. Paraffin-embedded tissue sections measuring 5 μm were mounted on slides and stained with hematoxylin/eosin (Merck, Darmstadt, Germany) and examined by a histologist blinded to the experimental protocol. Lung injury was scored according to alveolar congestion, hemorrhage, infiltration, or aggregation of neutrophils in airspace or vessel wall, and thickness of alveolar wall/ hyaline membrane formation. Items were graded with 5-point scales: 0 = minimal (little) damage, 1 = mild damage, 2 = moderate damage, 3 = severe damage, and 4 = maximal damage. Lung injury score was calculated as the sum of the items and thus, maximum and minimum possible scores were 0 and 16, respectively.
2D-PAGE and Gel Imaging
Two-dimentional electrophoresis (2DE) was performed as described by Klose and Kobalz (17) combining nonequilibrium pH gradient electrophoresis (NEPHGE) and sodium dodecyl sulfate polyacrylamide gel electrophoresis (SDS-PAGE) from WITA GmbH (Teltow, Germany). The first-dimension was performed by loading 120 μl of the sample on the anodic side of a poured 20 × 0.15-cm rod gel and focusing in a vertical isoelectric focusing (IEF) chamber. After first dimensional IEF, the gels were equilibrated for 10 min with 1 M Tris, 75 mM dithiothreitol (DTT), 40% glycerol, and 3% SDS. The second-dimensional separation was done by a vertical 15% 30 × 23-cm SDS-PAGE gel according to Laemmli (18). After 2DE, all gels were silver stained according to the procedure of Blum et al. (3). Spot detection, quantification, and analysis were performed using Melanie 7.0 (Swiss Institute of Bioinformatics). Afterwards, selected spots were excised and proteins were digested with trypsin for further identification.
Mass Spectrometry
Matrix-assisted laser desorption/ionization (MALDI) mass spectrometry (MS) was performed on a Bruker Reflex III MALDI mass spectrometer. The range of measurement was set at 0–4 kDa. Main MALDI measurement parameters were: reflector positive ion mode, 25 kV and matrix deflection 0–600 Da. Analysis and postprocessing of spectra were performed employing XMASS/NT 5.1.5 and Flex Analysis 2.0 software (Bruker Daltonics) (29). For nano-liquid chromatography electrospray ionization tandem mass spectrometry (LC-ESI-MS/MS) the total volume of each of the samples was injected into a Bruker esquire HCT mass spectrometer. Data collection was performed using the HyStar 2.3 and esquireControl 5.1 software. Analysis and postprocessing of spectra were performed employing HyStar PP 2.3 and DataAnalysis 5.4 (Bruker Daltonics) (26). The identification of proteins was realized by Bio-Tools 2.2 (Bruker Daltonics) and Mascot Server 2.0 in the peptide mass fingerprint or in the MS/MS ion search mode (Matrix Science) using the NCBInr database.
Quantitative Real-Time PCR
Total RNA was purified from liver tissue using Trizol reagent (Invitrogen, Karlsruhe, Germany). Total RNA (1 μg) was used to synthesize cDNA using Super-Script First-Strand Synthesis System (Invitrogen) and was resuspended in 100 μl of H2O. cDNA samples (5 μl) were used for real-time PCR, in a total volume of 20 μl using SYBR Green Reagent (Finzyme, Espoo, Finland) and specific primers on a qPCR machine (Opticon 2, MJ Research Inc., San Francisco, CA). Realtime PCR reactions were performed in triplicate. Primer sequences are available upon request. All values were normalized to the level of ubiquitin mRNA.
Apoptotic Cell Detection (via TUNEL)
Snap-frozen tissue sections (4–6 μm) were fixed in 10% buffered formalin for 10 min. The TUNEL (terminal deoxynucleotidyl transferase-mediated dUTP nick end labeling) was performed to detect apoptotic cells, using the in situ cell death detection Kit POD (Roche Diagnostics Corporation, IN) according to the manufacturer's instructions. Analysis involved confocal microscopy (Zeiss, Germany) to identify fluorescently labeled cells that were considered positive for DNA fragmentation. Values represent percentages from at least 1,000 counted apoptotic and nonapoptotic cells.
Stromal Cell-Derived Factor-1 (SDF1)
For immunohistological detection of stromal cell-derived factor 1 (SDF-1), we employed monoclonal mouse anti-SDF-1 (clone 79018, R&D Systems, Minneapolis, MNA) and Alexa-Fluor 488 conjugated goat anti-mouse IgG (Invitrogen) as primary and secondary antibody, respectively. Nuclei were counterstained with TOPRO3 (Invitrogen). Labeled sections were observed using a Leica DM 2500 Confocal Microscope (Leica, Hamburg, Germany).
Statistical Analysis
All values were analyzed and tested with a commercial statistical package (SPSS 14.0, Inc, Chicago, IL). Continuous variables were compared with independent-samples t-test and sample correlation coefficient. Results are presented as percentages or means ± SD. Differences were considered significant at the 95% level [p < 0.05 (or p > 0.995 using the protein quantification parameter in Melanie)]. To account for the multiple testing in the same sample we adjusted the alpha = 0.05 with the simple Bonferroni approach.
Results
Animal Model and MSC Isolation
To investigate whether intratracheal (IT) MSC administration might improve the outcome of CTEPH, we induced CTEPH according to our previously described protocol in a rat model via ligation of the left pulmonary artery (14). Animals (n = 30) were randomly divided into three groups of n = 10 animals each. Group I: left thoracotomy only (control); group II: ligation of the left pulmonary artery (PA) and IT saline administration (sham); group III: left thoracotomy with ligation of the left PA and an IT administration of MSCs. Allogenic MSCs were isolated from thigh bones according to standard protocol. Mechanical disruption easily dissociated both the stromal and the hematopoietic cells into the same cell suspension. Due to the plastic adherence, MSCs were readily separated from the hematopoietic stem cells (HSCs) by multiple washing. Figure 1A shows the spindled, fibroblast appearance morphology of MSCs in MesenPro RS (Gibco, Prat de Llobregat, Spain) culture. Based on our previous findings in a cardiac infarction animal model, only normal shaped MSCs that stained positively for CD90 (but negatively for CD45) were used for the present study (9). To detect MSCs later in harvested organs, we labeled cells with CFSE at a concentration of 100 μM and BrdU at a concentration of 50 μM.

Histological evidence of the effect of MSCs. (A) Bone marrow-derived mesenchymal stem cells (MSCs) with spindled, fibroblast appearance in MesenPro culture. Scale bar: 200 μm. (B, C) Group I. H&E staining of healthy lung parenchyma. Scale bar: 200 μm (B) and 100 μm (C). (D, E) Group II showed CTEPH pulmonary vasculopathic abnormalities and bronchopulmonary artery hypertrophy (1) and inflammatory signs such as hemorrhage, edema, and granulocytes migration (2). Scale bar: 200 μm (D) and 100 μm (E). (F, G) H&E staining of lung sections demonstrated improved lung injury in animals given MSCs; reduction in the degree of hemorrhage, edema, granulocytes migration, and vascular remodeling. Scale bar: 200 μm (F) and 100 μm (G).
MSC Administration Results in CTEPH Amelioration and Clinical Improvement
After the animals' chest was closed, we disconnected them from the mechanical ventilator for 20 s. Thereafter we attached a 1-ml syringe directly to the endotracheal tube and injected 300 μl of the MSC suspension (containing 6 × 106 cells) rapidly but gently into the trachea to enhance the engraftment of the cells. We recorded the following hemodynamic variables to demonstrate the impact of MSCs on induced CTEPH: heart rate (HR, bpm), cardiac output (CO, ml/min), mean pulmonary arterial pressure (mPAP, mmHg), and pulmonary vascular resistance (PVR, mmHg ml−1 min−1). Blood samples were taken to measure partial arterial pressure of carbon dioxide (PaCO2) and partial arterial pressure of oxygen (PaO2). Baseline parameters of the three experimental groups were equal and group I values remained constant during the study (data not shown).
Table 1 displays the impact of inducing CTEPH in rats and its attenuation when MSCs were applied. MSC administration resulted in a significant improvement (p < 0.025) of the pulmonary hemodynamic (mPAP: 25 ± 3 vs. 30 ± 4 mmHg; PVR: 35 ± 3 vs. 45 ± 4 mmHg/ml/min; and CO: 84 ± 4 vs. 74 ± 5 ml/min) and blood gases (PaO2: 155 ± 21 vs. 92 ± 14 mmHg; and PaCO2: 45 ± 4 vs. 38 ± 4 mmHg), although their values were still significantly altered compared to control animals. The respiratory function of the animals was measured in a Buxco unrestrained whole body chamber, allowing for long-duration, long-term, and anesthesia-free investigations. The animals were placed into the chamber and respiratory rate and their enhanced pause (Penh) of the animals was measured for 1 h. Both respiratory rate (125 ± 12 vs. 168 ± 17 breaths/minute) and Penh (0.35 ± 0.04 vs. 0.92 ± 0.04) improved significantly in animals treated with MSCs.
Hemodynamic and Clinical Findings in the Study Groups

mPAP, mean pulmonary arterial pressure (mmHg); CO, cardiac output (ml/min); PVR, pulmonary vascular resistance (mmHg ml−1 min−1); RR, respiratory rate (breaths/ min); VT, tidal volume (ml); Penh, enhanced pause (dimensionless)—measured for 1 h, calculated using the formula: Penh = [(Te - Tr)/(Tr x PEF/PIF)], where Te = expiratory time (s), Tr = relaxation time (s), PEF = peak expiratory flow (ml/s), and PIF = peak inspiratory flow (ml/s), Penh was measured and averaged approximately every 5 s; PaO2, partial arterial pressure of oxygen (mmHg); PaCO2, partial arterial pressure of carbon dioxide (mmHg); FiO2, fraction of inspired oxygen; I.T., immobility time (s)—the time of “swimming” during total time (t.t. 7 min; 420 s) was defined as moving all four extremities under water and subtracted from the t.t. to obtain immobility time; RV to LV/septum, expressed as the ratio of the right ventricle over the free LV weight and ventricular septum weight (LV + S).
p < 0.025 versus control (group I).
p < 0.025 versus MSCs injection (group III).
To evaluate the animals' physical strength we used a special forced swimming test before sacrificing animals at the 12th week (19). The animals treated with MSCs showed lower values of immobility time (76 ± 9 vs. 123 ± 11 s) during the swimming stress test compared to sham-treated animals (group II). Although the described changes were statistically significant (p < 0.025) compared to sham-treated animals, parameters such as Penh, PaO2, and immobility time still differed from control group. Two animals (belonging to group II) died before studies end point (72 ± 3 days) due to cardiac arrest.
MSC Administration Led to Local and Systemic Histological Changes
After evaluating the clinical outcome of the animals, their organs were harvested to investigate for the morphological correlate. Figure 1B and C shows healthy lung parenchyma. Typical postobstructive pulmonary vasculopathic abnormalities and bronchopulmonary artery hypertrophy and initial signs of small vessel arteriopathy and capillaritis were histologically detected in group II (Fig. 1D, E) but were clearly less in animals given MSCs (group III; Fig. 1F, G). Lung tissue injury and its improvement were scored according to alveolar congestion, hemorrhage, infiltration, or aggregation of neutrophils in airspace or vessel wall, and thickness of alveolar wall/hyaline membrane formation. We graded items with 5-point scales: 0 = minimal (little) damage, 1 = mild damage, 2 = moderate damage, 3 = severe damage, and 4 = maximal damage. Lung injury score was calculated as the sum of the items and thus, maximum and minimum possible score was 0 and 16, respectively.
The described lung injury model revealed significant differences (p < 0.025) between the groups: group I (2 ± 1) versus II (12 ± 2) versus III (7 ± 2) (Fig. 2). CFSE-labeled (Fig. 3A) and BrdU-labeled (Fig. 3B, C) MSCs were detected only in the nonligated right lung, primarily at the lung parenchyma, which was most likely caused by the SDF-1α/CXCR4 pathway. In effect, the significant high expression of anti-SDF-1α (Fig. 3D, E) is an indicator for inflammatory processes and represents a strong homing factor for MSCs and other cell types (46). Aside from local effects, MSC administration led to notable systemic changes. Persistent CTEPH is linked with right ventricle (RV) hypertrophy resulting, sooner or later, in right heart dysfunction and finally in cardiac arrest. An indication for impaired right heart function is the hepatojugular reflux, which leads to hepatic blood congestion, chronic hepatocyte damage, and finally cardiac cirrhosis. To demonstrate the systemic impact of MSCs, the animal's heart and liver were harvested for analysis. Concerning the heart, we separated RV from the left ventricle (LV) wall and determined wet weight of the RV, free LV wall, and ventricular septum. RV hypertrophy developed in CTEPH-induced animals but was significantly less when MSCs were applied (0.54 ± 0.05 vs. 0.63 ± 0.06, p < 0.025). Regarding the liver we stained tissue slides with H&E to evaluate structural changes.
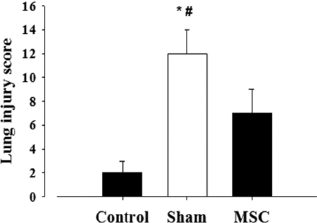
Lung injury score. Lung injury score according to alveolar congestion, hemorrhage, infiltration, or aggregation of neutrophils in airspace or vessel wall, and thickness of alveolar wall/hyaline membrane formation. Score ranges from a maximum of 16 (maximal damage) to a minimum of 0 (minimal damage). ∗p < 0.01 versus control; #p < 0.025 versus MSC.

Recruitment of MSCs via SDF-1. MSCs were incubated with carboxy-fluorescein succinimidyl ester (CFSE) and intratracheally administered. MSCs were only detected by confocal microscopy in nonligated right lung, primarily at the lung parenchyma (A, scale bar: 400 μm), and via fluorescence microscopy bromodeoxyuridine-labeled (B) MSCs (Alexa-488, green) only detected in right lung next to resident cells (red) (C) (scale bars: 100 μm). (D) Right lung stained for anti-SDF-1 (stromal cell derived factor-1; stained red), nuclei (stained blue) were counterstained with TOPRO3, fluorescence microscopy view (scale bar: 100 μm). (E) Confocal microscopy view, nuclei (blue) and anti-SDF-1 (green) (scale bar: 200 μm).
Figure 4a and b-LH (liver histology) shows healthy liver tissue from group I animals. Initial histological signs of blood congestion with broadened and erythrocyte-filled hepatic sinuses could be detected in H&E-stained liver tissue from sham-treated animals (Fig. 4c, d-LH). However, MSC-treated animals showed less hepatojugular reflux linked histological changes (Fig. 4e, f-LH). Finally, we applied the apoptotic cell detection (TUNEL) on healthy liver tissue [Fig. 4a–c-LI (liver immunostaining)] and the other study groups. We observed a high number of apoptotic liver cells in rats with induced CTEPH (15–23%) (Fig. 4d–f-LI) and this was remarkably reduced (5–8%; p < 0.025) upon MSC treatment (Fig. 4g–i-LI).

Liver tissue evaluation LH (liver histology). Liver tissue stained with H&E showing (a, b) normal tissue. (c, d) Initial histological signs of blood congestion with broadened and erythrocyte-filled hepatic sinuses. (e, f) MSC-treated animals showed less hepatojugular reflux linked histological changes. Scale bars: 100 μm (a, c, e) and 200 μm (b, d, f). LI (liver immunostaining). Apoptotic cells (green) in liver tissue were in situ identified by TUNEL method using terminal deoxynucleotidyl transferase (TdT) to transfer biotin-dUTP to strand breaks of cleaved DNA. Nonapoptotic cells were labeled via DAPI (blue). (a–c) Control group I showed low numbers of apoptotic cells; (d–f) sham group II, high degree of apoptotic cells; (g–i) significant decrease of apoptotic cells in animals given MSCs. Scale bars: 200 μm (a–i).
Reduced Inflammation and Fibrosis in Livers of MSC-Treated Animals
We investigated whether MSC treatment might have a secondary impact on liver inflammation and fibrosis. Western blot using a phospho-specific antibody against the cleaved form of caspase-3 (Cell Signaling Technology, Inc., Danvers, MA) revealed a marked reduction of caspase cleavage (Fig. 5A) normalized to glyceraldehyde-3-phosphate dehydrogenase (GAPDH; Santa Cruz Biotechnology, Inc. Heidelberg, Germany) in MSC-treated rats, confirming the previous data showing a reduced number of TUNEL-positive cells in these animals (see Fig. 4a–i-LI). The NF-κB pathway is the main signaling pathway driving inflammation in the liver (19). MSC-treated animals showed a marked reduction in expression of the NF-κB target gene IκBα by qRT-PCR (Fig. 5B), suggesting a reduced inflammatory response in these animals. Finally, qRT-PCR analysis of genes typically expressed during liver fibrosis revealed a significant reduction of collagen1A1 and α-smooth muscle actin (α-SMA) expression in livers from MSC-treated rats (Fig. 5C, D), suggesting that MSC treatment has a beneficial effect on liver inflammation and fibrosis in a setting of liver damage induced by right-heart failure.

Comparison of liver protein expression level. Western blot revealed a marked reduction of caspase cleavage (A) in MSC-treated rats. The NF-κB pathway is a main signaling pathway driving inflammation in the liver. MSC-treated animals showed a significant reduction in expression of the NF-κB target gene IκBα (B), suggesting a reduced inflammatory response. The qRT-PCR analysis revealed a significant reduction of collagen1A1 and α-smooth muscle actin (α-SMA) expression in livers from MSC-treated rats (C, D), genes typically expressed during liver fibrosis. ∗p < 0.025 versus control and MSCs. Data are expressed as mean ± SD.
Effects of MSC Administration on Protein Expression Profile in Lung Tissue
Effects of cell therapy on protein expression profiles in lung tissue were tracked by 2D-PAGE and mass spectrometry. An overview in change of protein expression as response to MSC treatment with or without ligation of the left pulmonary artery can be given by comparing the pattern of the 2D gel spots (Fig. 6a and b). Overall, the proteomic analysis revealed a rather consistent number of more than 2,000 spots expressed in lung tissues. In addition, four main variant regions could be found by visual inspection (A–D), comparing the different groups. The ligation of the pulmonary artery without treatment (group II) results in approximately 25 variations of spots and is mainly accumulated in the acidic region A and in the slightly basic region D (p > 0.995). Moreover, around 300 obvious variations in the expression pattern of right lung tissue of animals treated with MSCs were detected compared to all other groups and lung side (p > 0.995). The comparison of the proteomic approaches after MSC treatment allowed the identification of regulated proteins. The identification of singular regulated proteins was carried out by peptide mass fingerprint (PMF). Therefore, the target proteins were digested in-gel and the resulting peptides were analyzed by mass spectrometry. However, only a few of them have been identified so far concerning their name and function. All of them were allocated to the species Rattus norvegicus. Notably, beside from non-tissue-specific proteins such as mitochondrial creatine kinase S-type (P09605) or mitochondrial aconitate hydratase (Q9ER34), proteins have been detected usually expressed in cardiac and liver cells (Table 2). Thus, proteins such as myosin light chain 3 (P16409) or mitochondrial ATP synthase subunit alpha (P15999) gave strong evidence for the multipotency and the in vivo differentiation of the used cells. We also detected an increase in albumin level (albumin isoform CRA_a) in lung tissues after MSC treatment. We were also able to detect an unnamed protein sequence from Rattus norvegicus (UPI000157F7A1). This protein sequence was postulated as a result of combined whole genome shotgun assembly of Rattus norvegicus (8,42).

Proteomic approach. Representative nonequilibrium pH gradient electrophoresis and two-dimensional sodium dodecyl sulphate polyacrylamide gel electrophoresis (NEPHGE 2D-SDS PAGE) of proteins from right lung of CTEPH-induced and MSC-treated rats. Overall approximately 2,000 most abundant proteins were detected after silver staining. Circles represent affected proteins singular detected for group III, identified by MS. Main variant regions in between different groups are assigned as dashed rectangles (comparing wit b). (b) Quantitative analysis comparison of main variant regions (A–D) detected and analyzed in NEPHGE 2D-SDS PAGE using Melanie 7.0. Group I: both lungs (matched protein profile) from thoracotomy-only-received animals. Group II: left and right lungs from left pulmonary artery ligated with sham treatment. Group III: left and right lungs from left pulmonary artery ligated and MSC-treated animals.
Highly Expressed Proteins of Right Lungs From Left Pulmonary Artery Ligated and MSC-Treated Animals (Group III) Compared to Other Groups, Identified in 2D-SDS-PAGE by MS

Discussion
The present study shows the significant impact of administration of intratracheal MSCs on CTEPH. Aside from an improvement of all clinical relevant parameters such as hemodynamics, blood gases, and physical strain abilities, we detected histological changes in lung and liver tissue as well as significant changes in protein expression in animals treated with MSCs. These findings rise hopes for the development of new treatment strategies in this field. Etiopathology of CTEPH is still not completely understood. Abnormalities of coagulation, pulmonary endothelium, or fibrinolytic pathways have not been identified (4) and a satisfactory treatment does not currently exist. Previous studies evaluated the effect of MSC administration in bleomycin- or endotoxin-induced acute lung injury models (27,35). Also, monocrotaline-induced pulmonary hypertension attenuated via MSC administration (2). However, the use of intratracheal administration of MSCs in postobstructive PH has so far not been studied.
In our previously described animal model that already demonstrated its efficiency of mimicking the CTEPH (14), we applied the MSCs via the intrapulmonary route to maximize cell delivery. Although intravenous application showed an effect on lung injury despite the low cell engraftment level (27,35), others used the IT method (2,10). The recently pronounced cell instillation by performing a tracheotomy might be too invasive for human treatment (31). We therefore propose the method described herein of IT administration via an endotracheal tube under general anesthesia to make cell-based therapy clinically practicable and optimize cells engraftment. The labeled MSCs were only detected in the right high-pressure-exposed lungs (group III). This might be explained by the expression of SDF-1/CXCR4-positiv cells in this area.
Aside from the current studies concerning the interrelation of SDF-1α/CXCR4 and the recruitment of stem cells, we already demonstrated that tissue damage and inflammation represent a strong homing factor for MSCs via the mediated SDF-1/CXCR4 pathway (43). Moreover, SDF-1 mediates chemokinesis, chemotaxis, and homing on a plurality of cell types, such as monocytes, macrophages, B- and T-lymphocytes, and platelets, as well as endothelial progenitor cells (EPCs). Detailed knowledge about mechanisms that lead to MSC-related improvement remains speculative. The described effects of MSCs vary from paracrine mechanisms such as neovascularization via releasing vascular endothelial growth factor (VEGF), endothelial function improvement, to cell differentiation and modulation of inflammatory processes (1,2,22,23,27,35). Immunostained MSCs could only marginally be detected in the walls of the pulmonary vessels but primarily in parenchyma surrounding airway.
This low level of cell engraftment into the vessel wall is in line with findings of other studies, and supports the hypothesis of paracrine-induced pathways rather than cell differentiation (2,10). The effect of the MSC application was not only restricted to the lung and its hemodynamics but also systemic impact was detected. However, the improvement of cardiac function and RV hypertrophy might be mostly explained as a consequence of decreased mPAP and PVR by ameliorating lung pathology (41). Our proteomic analysis revealed cardiac cell-specific proteins in right lung (group III), which might influence cardiac remodeling. Liver protein analysis for untreated PH animals showed an increase of caspase 3 (key protein of apoptosis), IκBα (a complex protein responsible for inflammtory and apoptosis processes), α-SMA (involved in processes mediating liver fibrosis), and collagen. All these proteins were significantly lower in animals treated with MSCs after inducing the PH.
Histological and protein expression changes in liver tissue were most likely attributed to the improvement of CTEPH. However, Popp et al. (33) reported about the impact of MSCs on liver regeneration capacity and their immunomodulatory properties. The performed proteomic analysis detected also liver-specific proteins in right lung of MSC-treated animals and this probably affected liver function. The present investigation, which to our knowledge is the first animal model evaluating not only the impact of MSCs on CTEPH but also the protein expression profile, detected more than 2,000 spots expressed in lung tissues. Notably, proteomics revealed around 300 obvious variations in expression of right lung of MSC-treated animals compared to other groups, and this is directly associated to cell administration. In addition, approximately 25 proteins showed significant changes between group I and II, which could be ascribed to dislocation of the artery.
So far, we only evaluated a marginal quantity of the overall protein gel spots concerning their real identity. The curious finding is that the hitherto analyzed proteins are usually associated with liver and cardiac tissue. This might be explained by a differentiation of MSCs into different cell types due to their multipotency. In this context a detected increase of albumin in lung tissue after MSC treatment could be caused by the proliferation of MSCs as well as to their differentiation into liver tissue. Furthermore the detected albumin isoform CRA_a might be due to restoration of endothelial integrity in response to MSC treatment, resulting in limitation of fluid transudation (6). The new found protein sequence (UPI000157F7A1) seems to be a further isoform of myosin, considering its amino acid sequence, molecular weight, and the according gene sequence, respectively (25), which is supposedly induced by systemic circulating mediators such as cytokines released by the CTEPH-related cardiac and liver tissue damage. One has to investigate whether these lung tissue-abnormal proteins have a systemic protective quality and thus improve outcome, or are they merely useless? Although CTEPH and clinical signs improved in animals given MSCs, the parameters were still significantly different to the control group and therefore treatment should be optimized.
The evaluation of the whole protein profile of both CTEPH-induced and MSC-treated animals will potentially provide us with significant findings to elucidate pathways of the disease and the applied cell-based treatment. Probably MSCs need to be transduced with modulatory genes such as prostacyclin synthase (39) to increase their potential. It might also be possible that there is a necessity of adding specific growth factors/cytokines to help MSCs targeting their destined tissue and differentiating into it. Some mechanisms that might influence stem cell differentiation were recently discovered (41). It has been shown that MSC administration can induce and/or promote cancer both positively and negatively (28,37,44). Based on our clinical experience with a fully tissue engineered windpipe seeded with autologous MSCs (20) with a follow-up of more than 2 years, in which we were unable to detect cancer development, we hypothesized that the administration of MSCs in inflammatory or damaged conditions does not lead to an uncontrolled cell proliferation or differentiation. However, at this moment we cannot exclude cancer development and further studies are needed to answer this question.
The findings of the current study demonstrate that the IT application of MSCs in an animal model of CTEPH is highly effective and led to a significant restoration of lung tissue as demonstrated by the amelioration of all relevant parameters. The MSC administration resulted in a significant change of lung tissue protein expression profile, and this might be mediated by both contact-dependent and -independent mechanisms. We showed the multipotency of MSC after intratracheal application by their expressed and/or recruited proteins. The effective meaning of these lung tissue foreign proteins concerning the outcome and the developing of the CTEPH has to be elucidated. There might be a supposable necessity of adding differentiating/growth factors to induce and support cell differentiation into the target tissue. The here described method is easy and safe to apply even in severe health conditions, which makes it highly potential for the clinical use as a treatment in patients suffering from CTEPH.
Footnotes
Acknowledgments
We acknowledge the assistance of Laura Morte for her excellent organization and trouble-shooting. Furthermore, we would like to express our gratitude to Heike Nave and Frank Tacke for their kindly providing laboratory facilities. Finally, we are indebted to Christian Klopsch for his help at the animal laboratories. The authors declare no conflict of interest.
