Abstract
Intraductal administration of a c-Jun NH2-terminal kinase (JNK) inhibitor enhances islet viability. However, its role in reducing the inflammatory response in islets is unknown. It is also unknown whether a JNK inhibitor could act in synergy with statins. We examined if the sequential combination of a JNK inhibitor and simvastatin would reduce islet inflammation and improve islet viability. We performed porcine islet isolation with or without intraductal administration of SP600125, a JNK inhibitor. This was followed by culture medium supplementation with either nicotinamide alone or nicotinamide plus simvastatin. We assessed the viability of islets by flow cytometry, islet loss during overnight culture, graft function in NOD/SCID mice, and expression of inflammation-related genes in islets. The sequential combination of a JNK inhibitor and simvastatin increased the β-cell viability index of porcine islets cultured overnight (p = 0.015) as well as islet viability as assessed by a DNA binding dye staining (p = 0.011). The combination of a JNK inhibitor and simvastatin significantly increased the islet survival rate (p = 0.027) when the histomorphometry of donor pancreas indicated a large islet proportion of greater than 50.55%. When we transplanted the same islet mass per recipient for each group, there was no difference in overall islet graft function. Intraductal administration of JNK inhibitor significantly suppressed mRNA expression levels of interleukin-1β (IL-1β), interferon-γ, tumor necrosis factor-α, IL-6, IL-8, and macrophage chemoattractant protein-1. It also decreased the concentration of IL-1β (p = 0.040) and IL-8 (p = 0.023) in the culture supernatant. In conclusion, the sequential combination of a JNK inhibitor and simvastatin protected porcine islets from peritransplant apoptosis. Inhibition of JNK reduced the inflammatory response and could be considered an alternative target for suppression of porcine islet inflammation.
Introduction
The fragility of adult porcine islets during islet isolation and subsequent short-term in vitro culture remains an important hurdle in the process of adult porcine islet xenotransplantation (27). Moreover, a rapid loss of islet grafts upon transplantation is often observed in adult pig-to-nonhuman primate islet xenotransplantation settings (31). Given that nonspecific inflammation plays a major role in islet graft loss in the early period of transplantation (2,3,10,23), islet cytoprotection carries additional importance for the adult porcine islet xenotransplantation field.
During pancreas preservation and islet isolation, islets are exposed to various stressful insults, including hypoxia, reactive oxygen species, shearing stress, and proinflammatory cytokines. These insults activate the p38 kinases and the c-Jun NH2-terminal kinase (JNK) in islets (1). The activation of these stress-activated protein kinases (4) mainly occurs during the islet preservation, digestion, washing, and purification processes (1,24). Once isolated, the activity of stress-activated protein kinases significantly decreases during culture (1,24). During short-term culture, however, the nuclear factor (NF)-KB pathway is activated and mRNA expression of inducible nitric oxide synthase increases (1). Subsequently, mRNA expression of cyclooxygenase-2, interleukin-6 (IL-6), and macrophage chemoattractant protein-1 (MCP-1) significantly increases during short-term culture despite the inactivation of stress-activated protein kinases during this period (13). The sequential activation of stress-activated protein kinases during the islet isolation process followed by expression of inflammation-related genes in culture may cause the early graft loss mediated by proinflammatory cytokines, such as IL-1β and tumor necrosis factor-α (TNF-α) (3,10). However, it is unknown whether this sequential activation is only a timed occurrence of two independent events or a cause-and-effect relationship.
Recently, intraductal administration of a JNK inhibitor before pancreas preservation was shown to reduce the activation of JNK during islet isolation. This enhanced the in vitro viability of islets, the rate of islet survival during culture, and graft function in immune-deficient mice (24). However, it is unknown if JNK inhibition during the pancreas preservation and islet isolation processes also suppresses the sequential activation of proinflammatory cytokines. Furthermore, it has not been determined if the intraductal administration of a JNK inhibitor could act in synergy with statins (2,5,29) to protect porcine islets from apoptosis. Statins exert antiapoptotic effects on islets via the activation of the phosphoinositide 3-kinase (PI3-K)/Akt pathway (5). Inhibition of JNK is also known to induce Akt phosphorylation in human islets (7). Therefore, supplementation with simvastatin in the in vitro culture stage could extend the antiapoptotic effect of the preceding JNK inhibition.
In the present study, we examined whether intraductal administration of a JNK inhibitor before pancreas preservation (24) can suppress the sequential activation of proinflammatory cytokines and act synergistically with subsequent simvastatin supplementation (2,5,29) to protect porcine islets from peritransplant apoptosis. We assessed viability in vitro, islet loss during overnight culture, graft function in NOD/SCID mice, mRNA expression levels of inflammation-related genes in islets, and the concentration of proinflammatory cytokines in the culture supernatant.
Materials and Methods
Porcine Pancreas Procurement and Islet Isolation
After the pancreas was harvested at the local slaughterhouse, cold Seoul National University (SNU) solution (21) with or without 10 μM of the JNK inhibitor SP600125 (BIOMOL Research Laboratories Inc., Plymouth Meeting, PA, USA) was injected intraductally (24). SNU solution was a preservation solution based on University of Wisconsin (UW) solution. To prepare SNU solution in house, hydroxyethyl starch (50 g; Fresenius Kabi AG, Bad Homburg v.d.H., Germany) was dissolved in 500 ml of distilled water and mixed with 100 ml of water containing 35.83 g of lactobionic acid (Sigma-Aldrich, St. Louis, MO, USA), in which pH was adjusted to pH 7.4 with KOH. The mixed solution of hydroxyethyl starch solution and lactobionic acid was then mixed with a solution containing raffinose (30 mM, Sigma-Aldrich), K2HPO4 (25 mM, Sigma-Aldrich), MgSO4 (5 mM, Sigma-Aldrich), glutathione (3 mM, Sigma-Aldrich), adenosine (5 mM, Sigma-Aldrich), and allopurinol (1 mM, Sigma-Aldrich). The pH of the final solution was adjusted to pH 7.4 with NaOH.
Because the intraductal injection of JNK inhibitor into the whole pancreas technically made it impossible to compare the effects of the presence and absence of JNK inhibitor using the same pancreas, we obtained a small piece of donor pancreas tissue, as previously described (15), to characterize the quantitative histomorphometrical parameters of each pancreas. The bias that may occur due to the differences in the donor pancreas was normalized by the histomorphometrical parameters. The definitions of each histomorphometrical parameter were previously defined (15). After transportation, islet isolation was performed using the modified Ricordi method (15). After digestion with Liberase PI (Roche Biochemicals, Basel, Switzerland), the digested tissue was maintained in buffers containing 10 mM nicotinamide (Sigma-Aldrich). Islets were purified with a continuous OptiPrep density gradient (Axis-Shield, Oslo, Norway) and a Cobe 2991 cell separator (Gambro BCT Inc., Lakewood, CO, USA). All animal procedures in this study were approved by the Institutional Animal Care and Use Committee of the Seoul National University Hospital.
Porcine Islet Culture
Purified islets were cultured overnight in Medium 199 (Gibco, BRL, Life Technologies Ltd, Paisley, Scotland, UK) supplemented with 10% porcine serum at 37°C. In all experiments, the medium contained 10 mM nicotinamide (Sigma-Aldrich) (9). The JNK inhibitor was administered before pancreas preservation, and the culture medium for the isolated islets also contained 1 μM of alkaline hydrolysis-activated (5) simvastatin (kindly donated by Pacific Pharma, Seoul, Korea). When the JNK inhibitor was not administered before pancreas preservation, the culture medium for the isolated islets either did or did not contain 1 μM of alkaline hydrolysis-activated (5) simvastatin.
Assessment of Islet Viability In Vitro
The β-cell viability index of islets cultured overnight was determined using flow cytometry. In our previous study, we showed that this method for determining the β-cell viability index predicted posttransplant outcome (15). Briefly, 1,000 islet equivalents (IEQ) of porcine islets were dispersed into single-cell suspensions in 1 ml accutase solution (Innovative Cell Technologies, Inc., San Diego, CA, USA) for each stain. The debris was gated out according to the forward- versus side-scatter light properties, and intact cells were analyzed for emission of green fluorescence (14). The proportion of zinc-binding β-cells among islet cells was assessed by incubating cells with 500 nM FluoZin-3 (Invitrogen, Carlsbad, CA, USA) for 30 min at 37°C (14). The cut-off value for positive staining with FluoZin-3 was determined at a fluorescence intensity corresponding to the 99.5 percentile of unstained samples (15). Mitochondrial membrane potential was assessed by incubating cells with 50 nM tetramethylrhodamine ethyl ester (TMRE; Invitrogen) for 30 min at 37°C (11). We gated FluoZin-3-positive cells and then determined the proportion of TMRE-positive cells to obtain the β-cell fractional viability. The β-cell fractional viability was defined as the proportion of TMRE-positive cells among the FluoZin-3-positive cells. The β-cell viability index was defined using the following formula: (proportion of FluoZin-3-positive cells) × (β-cell fractional viability). In parallel, islet viability was assessed by incubating cells with 7-aminoactinomycin D (7-AAD; Invitrogen), which binds to DNA when cell membrane permeability is altered after cell death. Another aliquot of the same cells used in the β-cell viability index experiment was incubated with 8 μM 7-AAD for 10 min. Islet viability was defined as the percentage of cells not stained with 7-AAD (% 7-AAD-).
Glucose-Stimulated Fold Increment in Oxygen Consumption Rate (OCR)
We obtained the glucose-stimulated fold increment in OCR using the BD oxygen biosensor system (BD Biosciences, San Jose, CA, USA), as described previously (8). The BD oxygen biosensor system was a standard round-bottom 96-well microplate coated with a suspension of silicone rubber containing a ruthenium-based oxygen-sensitive fluorophore, the intensity of which varies inversely with the oxygen concentration at the well bottom. Using a temperature-controlled fluorometric plate reader, the OCR estimate of 500 IEQ of islet per well in Krebs buffer containing either low (2.2 mM) or high (22.2 mM) glucose was determined. The OCR estimate was derived from the fluorescence values using the previously described equations (8). In parallel, we performed DNA quantification, using Pico Green dsDNA probe reagent (Molecular Probes Eugene, OR, USA), of the same islet used for the estimation of OCR. The rdO2 index was defined as the DNA content-normalized OCR estimate of the islet in the high-glucose Krebs buffer divided by the DNA content-normalized OCR estimate of the islet in the low-glucose Krebs buffer (8).
Assessment of Islet Function In Vivo
After overnight culture, 2,500 IEQ of isolated islets per recipient were transplanted under the renal capsule of two to four streptozotocin-induced diabetic NOD/SCID mice. The dose of streptozotocin was 100 mg/kg and injected via intraperitoneal route. Mice were regarded as diabetic when two consecutive blood glucose level readings were higher than 300 mg/dl. The pretransplant cut-off for diabetes was set as over 300 mg/dl of nonfasting blood glucose level.
After islet transplantation, random blood glucose levels were measured twice a week. The cut-off for normoglycemia was 200 mg/dl. Diabetes reversal was defined when two consecutive nonfasting blood glucose levels were below 200 mg/dl and became above 300 mg/dl after removal of graft-bearing kidney. To evaluate early islet graft function, the mean random blood glucose level (mg/dl) obtained between days 1 and 10 after transplantation was also recorded.
Quantitative Real-Time Polymerase Chain Reaction (qRT-PCR)
After overnight culture, islets were washed twice with phosphate-buffered saline (PBS) and stored in RNAlater solution (Ambion, Austin, TX, USA) at −80°C. Total RNA was isolated from these islets using an RNeasy Mini Kit (Qiagen, Valencia, CA, USA). Using the ABI Prism 7700 Sequence Detection System (Applied Biosystems, Foster City, CA, USA) in combination with SYBR Green Master Mix protocol (Applied Biosystems), qRT-PCR analysis of RNA was performed according to the manufacturer's recommendations. Primer sequences for porcine genes were as follows: IL-1β forward, 5′-AAGCATCCAGCTGCAAATCT-3′ and reverse, 5′-ACGATGGGCTCTTCTTCAAA-3′; TNF-α forward, 5′- TCCTCACTCACACCATCAGC-3′ and reverse, 5′-CCCAGGTAGATGGGTTCGTA-3′; interferon-γ (IFN-γ) forward, 5′-CCATTCAAAGGAGCATGGAT-3′ and reverse, 5′-ACGATGGGCTCTTCTTCAAA-3′; IL-6 forward, 5′-AGCAAGGAGGTACTGGCAGA-3′ and reverse, 5′-CAGCCTCGACATTTCCCTTA-3′; IL-8 forward, 5′-AAAGTGGGTGCAGAAGGTTG-3′ and reverse, 5′-CAGCACAGGAATGAGGCATA-3′; tissue factor (TF) forward, 5′-CCATCCCGAAGAGTTAACCA-3′ and reverse, 5′-GGACACAGCTTTCTCTTGGC-3′; caspase-1 forward, 5′-GAAG-GACAAACCCAAGGTGA-3′ and reverse, 5′-TGGGCTTTCTTAATGGCATC-3′; β-actin forward, 5′-TG GACATCAGGAAGGACCTC-3′ and reverse, 5′-ACAT CTGCTGGAAGGTGGAC-3′. After normalization with porcine β-actin, a comparative threshold cycle (CT) method was used for relative quantification of mRNA expression. Data are shown as fold changes in the gene expression of the islets relative to that of an arbitrary control chosen within the JNK inhibitor-treated group.
Enzyme-Linked Immunosorbent Assay (ELISA)
After overnight culture of 30,000 IEQ in 30 ml of medium, 1 ml of supernatant from the culture was obtained. After centrifugation, aliquots were frozen at −80°C. After thawing, the concentrations of IL-1β, IL-6, IL-8, TNF-α, and IFN-γ were determined using the Quantikine porcine IL-1β/IL-1F2 kit, the Quantikine porcine IL-6 immunoassay kit, the porcine CXCL8/IL-8 kit, the porcine TNF-α/TNFSF1A kit, and the Quantikine porcine IFN-γ immunoassay kit (all from R&D Systems, Minneapolis, MN, USA) according to the manufacturer's recommendations.
Statistical Analysis
Values are expressed as the mean ± SD or median plus quartiles as appropriate. Means of more than three groups were compared using one-way analysis of variance (ANOVA) using Bonferroni's post hoc test. Continuous variables of two groups were compared using Mann-Whitney U-test. We performed Kaplan-Meier survival analysis to compare the diabetes reversal rate of each group, and the difference between groups was determined by log rank test. To compare the rates of diabetes reversal among the groups as a categorical variable, we performed linear-by-linear association test to obtain a p-value for trend. A value of p < 0.05 was considered statistically significant. All analyses were performed using SPSS version 12.0K (SPSS Inc., Chicago, IL, USA).
Results
Baseline Donor Histomorphometrical Characteristics and Isolation Parameters
The inflation of the whole pancreas by intraductal administration of a preservation solution during pancreas procurement made it impossible to examine the effect of the JNK inhibitor using the same pancreas. Therefore, the bias possibly caused by the donor factor and islet isolation parameter needed to be normalized. For this reason, we compared the quantitative histomorphometrical parameters obtained with the frozen section of the donor pancreas. These parameters are predictive of islet isolation outcome and islet function in vitro and in vivo, as evaluated in our recent study (15). In terms of the histomorphometrical parameters, there were no differences in the donor characteristics (Table 1). The isolation parameters did not significantly differ between groups (Table 1). Although the digestion time of the simvastatin (+) JNK inhibitor (–) group trended to be shorter than that of the simvastatin (–) JNK inhibitor (–) group, the difference was not significant (p = 0.052).
Baseline Donor Histomorphometrical Characteristics and Islet Isolation Parameters

IEQ, islet equivalent; N.S., not significant (p > 0.05).
One-way analysis of variance with post hoc test using Bonferroni's method.
IEQ divided by actual total number of islets obtained from frozen sections of biopsied donor pancreas tissue.
Proportion of islets larger than 100 μm in diameter among all islets larger than 50 μm in diameter appearing in the frozen sections of biopsied donor pancreas tissue.
Simvastatin (–) JNKI (–) versus simvastatin (+) JNKI (–), p = 0.052.
Combination of Intraductal Administration of JNK Inhibitor and Culture Medium Supplementation with Simvastatin Increases the β-Cell Viability Index and Islet Viability of Porcine Islets Cultured Overnight
The simvastatin supplement alone (p = 0.011) or in combination with JNK inhibitor (p = 0.009) significantly increased the proportion of FluoZin-3-positive cells among all the islet cells (Fig. 1A). The proportion of TMRE-positive cells among all the islet cells did not differ between groups (Fig. 1B). β-Cell fractional viability, defined as the proportion of TMRE-positive cells among FluoZin-3-positive cells, did not differ between groups (Fig. 1C). The β-cell viability index, defined as the product of the proportion of FluoZin-3-positive cells and the β-cell fractional viability, was not increased by either simvastatin alone or JNK inhibitor alone. However, the combination of intraductal administration of JNK inhibitor and culture medium supplementation with simvastatin significantly increased the β-cell viability index of porcine islets cultured overnight (Fig. 1D, p = 0.015). We also compared the viability of islet cells using a DNA binding dye because TMRE staining only provides information on early apoptosis (Fig. 1E). The combination of intraductal administration of JNK inhibitor and culture medium supplementation with simvastatin increased the islet viability when compared to the simvastatin (–) JNK inhibitor (–) group (p = 0.011) and simvastatin (+) JNK inhibitor (–) group (p = 0.003). The rdO2 index, which represents the glucose-stimulated fold increment in OCR normalized to DNA content, did not significantly differ between groups (p = 0.351) (Fig. 1F).

(A) Proportion of cells stained with the zinc-binding dye FluoZin-3. (B) Proportion of cells stained with the active mitochondrial membrane potential indicator, TMRE, among the cells positive for FluoZin-3. (C) β-Cell fractional viability, defined as the proportion of cells stained with TMRE among all the islet cells regardless of FluoZin-3 staining. (D) β-cell viability index, defined using the formula: (proportion of FluoZin-3-positive cells) × (β-cell fractional viability). (E) Islet viability, defined as the percentage of islet cells not stained with 7-aminoactinomycin D (% 7-AAD–). (F) The rdO2 index, which represents the glucose-stimulated fold increment in oxygen consumption rate normalized to DNA content.
Combination of Intraductal Administration of JNK Inhibitor and Culture Medium Supplementation with Simvastatin Increases Islet Survival Rate
We defined the islet survival rate as the IEQ after overnight culture divided by the IEQ right after islet purification. Overall, intraductal administration of the JNK inhibitor did not significantly increase the islet survival rate, although the post hoc comparison between the simvastatin (+) JNK inhibitor (–) group and the simvastatin (+) JNK inhibitor (+) group showed a borderline significant difference in islet survival (p = 0.050) (Fig. 2A). To exclude the influence of the donor factor, we defined the donor subgroup according to the histomorphometrical parameters of the donor pancreas. Because islet potency is optimal when the frozen section of the donor pancreas has a large islet proportion greater than 50.55% (15), we analyzed the selected cases in which the donor pancreas histomorphometry had a large islet proportion of more than 50.55% (Fig. 2B). This analysis indicated a clear difference was achieved by the combination of intraductal administration of JNK inhibitor and culture medium supplementation with simvastatin (–) JNK inhibitor (–) group versus simvastatin (+) JNK inhibitor (+) group (p = 0.018); simvastatin (+) JNK inhibitor (–) group versus simvastatin (+) JNK inhibitor 0.027) (Fig. 2B).

(A) Islet survival rate of all the cases. The islet survival rate was defined as islet mass in islet equivalents after overnight culture divided by islet mass in islet equivalents right after purification of islets. (B) Islet survival rate in the selected cases in which the donor pancreas histomorphometry had a large islet proportion greater than 50.55%.
Combination of Intraductal Administration of JNK Inhibitor and Culture Medium Supplementation with Simvastatin Has Only Subtle Effects on Islet Graft Function
We compared the diabetes reversal rate of each group using Kaplan-Meier survival analysis. There was no difference in the diabetes reversal rate between groups (log rank test, p = 0.730) (Fig. 3A). In addition, we analyzed the posttransplant blood glucose levels of the subgroup that received islets isolated from pancreases with large islet contents less than 50.55%. In this subgroup analysis, the proportion of mice that achieved normoglycemia during the follow-up period of 56 days was 3/10 (30%) (Fig. 3B), 5/10 (50%) (Fig. 3C), and 9/13 (69%) (Fig. 3D) for the simvastatin (–) JNK inhibitor (–) group, simvastatin (+) JNK inhibitor (–) group, and simvastatin (–) JNK inhibitor (+) group, respectively (p for trend of 0.066 by linear-by-linear association test). The simvastatin (–) JNK inhibitor (+) group was not included in this analysis because all eight mice of this group received islets isolated from the pancreas subgroup with large islet contents greater than 50.55%. Because we did not administer JNK inhibitor or simvastatin to the recipients after islet transplantation, we also compared the mean posttransplant blood glucose levels obtained in the initial 10 days after transplantation of each group. This was significantly lower in the JNK inhibitor (+) simvastatin (+) group (200 ± 84 mg/dl) than in the JNK inhibitor (–) simvastatin (–) group (347 ± 109 mg/dl, p = 0.005) and in the JNK inhibitor (–) simvastatin (+) group (313 ± 115 mg/dl, p = 0.039).

(A) Kaplan-Meier survival curve showing the proportion of diabetic mice over time. The p-value was obtained with the log rank test. S, simvastatin; J, JNK inhibitor. (B, C, D) Posttransplant blood glucose level of the subgroup that received islets isolated from the pancreases with large islet contents less than 50.55%. (B) Simvastatin (–) JNK inhibitor (–) group. (C) Simvastatin (+) JNK inhibitor (–) group. (D) Simvastatin (+) JNK inhibitor (+) group. *The mean blood glucose level of each group obtained in the initial 10 days; p < 0.05 by one-way analysis of variance with a post hoc test using Bonferroni's method.
JNK Inhibition During Islet Isolation Subsequently Suppresses the Expression of Inflammation-Related Genes in Islets After Overnight Culture
To investigate whether JNK inhibition during pancreas preservation and islet isolation also suppresses the sequential activation of inflammation-related genes, we compared the mRNA expression levels of the following genes after the islets were cultured overnight: IL-1β, caspase-1, IFN-γ, TNF-α, IL-6, IL-8, MCP-1, and TF. The qRT-PCR results (Fig. 4) demonstrated significantly decreased mRNA expression levels of IL-1β (p < 0.001), IFN-γ (p = 0.004), TNF-α (p = 0.004), IL-6 (p < 0.001), IL-8 (p = 0.006), and MCP-1 (p < 0.001). The concentrations of IL-1β and IL-8 in the culture supernatant after overnight culture were significantly lower in the JNK inhibitor-treated group when compared to the control group (p = 0.040 and p = 0.023, respectively) (Fig. 5). The culture supernatant concentrations of IFN-γ, TNF-α, and IL-6 did not differ significantly. For both groups with and without the JNK inhibitor treatment, all the mRNA expression levels of these genes and culture supernatant concentrations of cytokines were similar between the cases with and without simvastatin treatment (data not shown).

Relative mRNA expression level of each inflammation related gene as measured by qRT-PCR. Data are shown as fold changes in gene expression in islets relative to that of an arbitrary control chosen within the JNK inhibitor-treated group (*p < 0.05, **p < 0.01, ***p < 0.001, Mann-Whitney U-test). JNKI, JNK inhibitor.
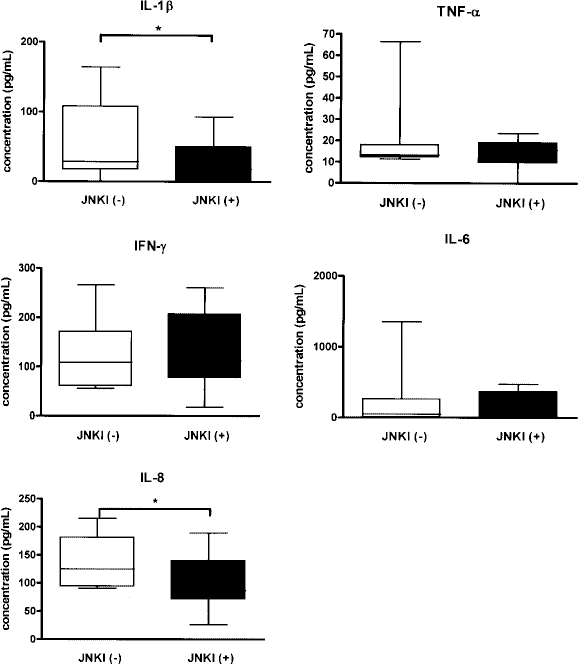
Concentrations of cytokines as measured by ELISA using the culture supernatant after overnight culture (*p < 0.05, Mann-Whitney U-test). JNKI, JNKI inhibitor.
Discussion
We report here that sequential combination of a JNK inhibitor and simvastatin during the pancreas preservation and islet isolation/culture process improved islet viability, and the intraductally administered JNK inhibitor suppressed the subsequent activation of inflammation-related genes in islets.
To the best of our knowledge, this is the first report that JNK inhibitor and simvastatin can act synergistically to improve islet viability. The β-cell viability index was improved by the combination of JNK inhibitor and simvastatin, but not by JNK inhibitor alone. In the subgroup in which the donor histomorphometry showed a large islet content greater than 50.55%, only the combination group showed significant improvement in islet survival during overnight culture. Statins have been reported to have an antiapoptotic effect on human islets (5) and anti-inflammatory effects in the canine islet autotransplantation model (2) and rodent streptozotocin-induced type 1 diabetes model (29). The activation of the PI3-K/Akt pathway is involved in the antiapoptotic effect of simvastatin on β-cells (5). We speculate that activation of the PI3-K/Akt pathway by inhibition of JNK during islet isolation (7) is further extended by simvastatin supplementation during the in vitro culture period. In addition, simvastatin increased the proportion of cells positively stained with the zinc-binding dye FluoZin-3. This was observed irrespective of the presence of JNK inhibitor. Given that the proportion of zinc-containing cells represents the proportion of insulin-containing cells (11) and that this proportion of cells significantly decreases in a short-term in vitro culture period (6), we can infer that simvastatin preserves the cells with insulin-containing capacity.
In this study, intraductal injection of JNK inhibitor during the islet isolation process suppressed the subsequent activation of inflammation-related genes, irrespective of the addition of simvastatin. This result indicates that the inhibition of JNK may be an alternative target for the suppression of porcine islet inflammation, such as proinflammatory cytokine production by resident macrophages (2,3,20). This alternative target is important because the other possible solution of applying immunosuppressants to the islet culture system to prevent nonspecific inflammation (19,21) inevitably deteriorates islet viability (16,26).
The anti-inflammatory effect of JNK inhibitor in this study is consistent with the studies using Jnk1-deficient mice (30,32). In the study by Varona-Santos et al., Jnk1–/– islets produced less MCP-1 than wild-type (WT) islets. Other cytokines, such as IFN-γ, IL-10, TNF-α, IL-6, RANTES, MIP-1α, MIP-1β, and VEGF were either not detected or not different between groups. Varona-Santos et al. also found that Jnk1–/– islets did not retain a survival advantage after transplantation, while Jnk1 deficiency in diabetic recipients of WT islets was beneficial for graft performance (32). They also observed that WT islets cocultured with Jnk–/– macrophages produced less IL-1β, MCP-1, IL-6, and TNF-α than islets cocultured with WT macrophages. Although it was not studied in the setting of islet transplantation, Solinas et al. demonstrated that Jnk1 deficiency in hematopoietic cells decreased obesity-induced inflammation, whereas Jnk1 deficiency in nonhematopoietic cells increased energy expenditure and improved insulin sensitivity (30). Collectively, those findings indicate that the anti-inflammatory effect of Jnk1 deficiency observed in Jnk1-deficient mice was achieved mainly by the Jnk1 deficiency in the recipient's hematopoietic cells, such as macrophages. On the other hand, the present study shows that JNK inhibition in the donor islet using a chemical inhibitor also decreases the expression of inflammation-related genes in the islet. This is important because recipient treatment with JNK inhibitor would not be clinically achievable in the near future, whereas local intraductal injection of some JNK inhibitor peptides are already in clinical trial (24).
In this study, intraductal administration of JNK inhibitor alone was not sufficient to significantly improve islet viability or islet survival during the in vitro culture period. This contrasts with the results of the study performed by Noguchi et al. (24). One of the differences in the experimental setting between this study and the study by Noguchi et al. (24) is that the control groups of this study were already treated with nicotinamide. Nicotinamide has an antiapoptotic and anti-inflammatory effect on human islets (12,22) and has been introduced to the porcine islet isolation and culture process (9,17). Thus, the strength of the benefit derived from JNK inhibition would be more prominent if nicotinamide were not administered to the control, as in the study by Noguchi et al. (24). Moreover, their research design for the evaluation of in vivo porcine islet function was different from ours. They cultured the same number of islets from the JNK inhibitor-treated group and control group and then transplanted the remaining islets of each group after culture. Because more islets remained from the JNK inhibitor-treated group, they could not confirm the superiority of islets from the JNK inhibitor-treated group in terms of in vivo performance of the same number of islets transplanted to each recipient mouse. In addition, they evaluated the in vivo islet function with a limited number (n = 5 for the JNK inhibitor group and n = 13 for the control group) of porcine pancreas (24), which may have caused a biased result, depending on individual pig variation. In our study, the evaluation of relatively large numbers of porcine pancreases and subgroup analysis according to the histomorphometrical characteristics of donor pancreases excluded the bias that can result from allowing the innate characteristics of individual pigs to influence the islet isolation yield and islet viability (15).
Although the expression of inflammation-related genes was suppressed by intraductal administration of JNK inhibitor, the loss of islets during overnight culture was still observed in the JNK inhibitor-treated group, and the in vivo outcome was not significantly improved. This discrepancy may be explained by several factors. First, the proinflammatory cytokines released during the culture period would be important, but they might not be the only noxious stimuli to cultured islets. For example, upon isolation from the pancreas, islets lose vascularization and are exposed to hypoxic conditions in culture because proper oxygenation of the center of large islets is impossible by simple diffusion (18). Therefore, various noxious stimuli other than proinflammatory cytokines may contribute to the loss of islets in the overnight culture period. Second, the benefit on the in vivo outcome from JNK inhibition would be attenuated because the benefit of JNK inhibition and simvastatin was reflected only during the overnight culture period. Because we transplanted 2,500 IEQ that were counted after the overnight culture, it is possible that more potent islets were selected and transplanted in the control group (28). This would be truer in the subgroup that received islets isolated from the pancreases with large islet contents greater than 50.55%, where the benefit in islet survival was significant. In this regard, we additionally analyzed the in vivo islet graft function of the subgroup that received islets isolated from the pancreases with large islet contents less than 50.55%. Third, JNK inhibitor was administered only before islet isolation, according to the purpose of this study. Because we did not inject the JNK inhibitor into the NOD/SCID mouse recipients, we analyzed the early islet graft function in terms of the mean posttransplant blood glucose level obtained in the initial 10 days after islet transplantation. Even in the subgroup that received islets isolated from the pancreases with large islet contents less than 50.55%, the in vivo benefit of the JNK inhibitor and simvastatin was observed only in this peritransplantation period. Because JNK is activated in the early posttransplant period, the benefit would be extended if the JNK inhibitor were added again immediately before transplantation (25). Likewise, treatment of recipients with JNK inhibitor could also have extended the in vivo benefit. In a study using Jnk1-deficient mice, Jnk1 deficiency in recipients of islets, but not Jnk1 deficiency in donor islets, was beneficial for graft performance (32). Fourth, we used the chemical inhibitor of JNK SP600125 instead of ATP-noncompetitive JNK inhibitors. A better demonstration of an in vivo benefit may be achieved by the use of peptide inhibitors that are not competitive inhibitors of ATP (24,25).
In conclusion, the sequential combination of JNK inhibitor and simvastatin protected porcine islets from apoptosis. Intraductal administration of JNK inhibitor reduced islet inflammation. Although the benefit was confined to the peritransplant period, this indicates that the inhibition of stress-activated protein kinases can be an alternative target for the suppression of porcine islet inflammation.
Footnotes
Acknowledgments
This work was supported by a grant from the Korea Health 21 R & D Project, Ministry of Health & Welfare, Republic of Korea (Project A040004).
