Abstract
The low survival rate of graft stem cells after transplantation into recipient tissue is a major obstacle for successful stem cell therapy. After transplantation into the site of spinal cord injury, the stem cells face not only hypoxia due to low oxygen conditions, but also a lack of nutrients caused by damaged tissues and poor vascular supply. To improve the survival of therapeutic stem cells after grafting into the injured spinal cord, we examined the effects of cotransplanting mouse neural stem cells (mNSCs) and adipose tissue-derived mesenchymal stem cells (AT-MSCs) on mNSC viability. The viability of mNSCs in coculture with AT-MSCs was significantly increased compared to mNSCs alone in an in vitro injury model using serum deprivation (SD), hydrogen peroxide (H2O2), and combined (SD + H2O2) injury mimicking the ischemic environment of the injured spinal cord. We demonstrated that AT-MSCs inhibited the apoptosis of mNSCs in SD, H2O2, and combined injury models. Consistent with these in vitro results, mNSCs transplanted into rat spinal cords with AT-MSCs showed better survival rates than mNSCs transplanted alone. These findings suggest that cotransplantation of mNSCs with AT-MSCs may be a more effective transplantation protocol to improve the survival of cells transplanted into the injured spinal cord.
Keywords
Introduction
The low survival rate of graft stem cells after transplantation into recipient tissue is a major obstacle for successful stem cell therapy. Several strategies to improve the survival of transplanted cells have been proposed, such as the induction of neurotropic factors and antiapoptotic genes (8,15,28,30). Moreover, it has been reported that transplantation of the bone marrow mesenchymal stem cells (BMSCs) reduces neuronal apoptosis under ischemic conditions (16) and that human adipose tissue-derived mesenchymal stem cells (AT-MSCs) improves the differentiation potential and cell migration of mouse neural stem cells (mNSCs) in vitro (10). These findings suggest that the growth factors secreted by MSCs play a role in neural stem cell differentiation and protection (5,29).
AT-MSCs are a population of multipotent stem cells (1,3,7,25). The genetic profiles of AT-MSCs and BMSCs are similar (14). Moreover, the phenotype expressed is similar to that of BMSCs, as both cells highly express mesenchymal stem cell markers CD105, CD90, and CD29, but are negative for CD34, CD44, and CD45 (14,20). AT-MSCs, like BMSCs, can differentiate into other cell lineages, such as bone and cartilage (12,13). Additionally, AT-MSCs are able to secrete higher quantities of bioactive factors such as IL-1Rα, IL-6, IL-8, G-CSF, GM-CSF, MCP-1, NGF, and HGF than BMSCs (2).
Transplantation of neural stem cells into the injured spinal cord promotes locomotor recovery (6). Cotransplantation of NSCs with primary fibroblasts into injured spinal cords facilitates cellular guidance and enhances the outgrowth of corticospinal axons at the lesion site (21). Although stem cell transplantation can help the host tissue recover, most of the transplanted cells are dead within a few weeks after being transplanted into the injured region.
In the present study, we hypothesized that coculture of AT-MSCs with mNSCs would enhance the viability of mNSCs under ischemic conditions (serum deprivation, oxidative stress, and a combination of these factors). We also investigated how AT-MSCs enhance the survival of mNSCs in a spinal cord injury cotransplantation model.
Materials and Methods
Isolation and Differentiation of AT-MSCs
This experiment was approved by the Research Ethics Committee at the Severance Hospital in Yonsei University, Seoul, Korea. AT-MSCs were isolated from human fat tissue that was kindly donated by Prof. Dae Hyun Lew (Plastic Surgery Laboratory, Yonsei University, Seoul, Korea). Isolation of AT-MSCs was performed per established protocol (32). Briefly, the obtained samples were digested with 0.1% collagenase (Wako, Osaka, Japan) under gentle agitation for 30 min at 37°C and then centrifuged at 1300 rpm for 10 min to obtain the stromal vascular fraction. The pellet was resuspended in phosphate-buffered saline (PBS) and filtered with 70-μm pore cell strainer (BD Falcon, Bedford, MA). The cell suspension was centrifuged at 1300 rpm for 3 min. The supernatant was discarded, and the pellet was suspended in DMEM/F12 (Invitrogen, Carlsbad, CA) supplemented with 10% fetal bovine serum (FBS, Hyclone, Logan, UT) and 1% penicillin/streptomycin (100 Usol;ml, Invitrogen), and plated in flasks. The medium was replaced every 3 days, and the nonadherent cells were discarded. Cultures were maintained at 37°C in a humidified atmosphere containing 5% CO2. AT-MSC identification was confirmed by flow cytometry analysis. In brief, AT-MSCs were harvested using trypsin/EDTA and then washed with PBS. Cells were then treated with CD90 (Immunotech, Marseille, France), CD105 (Beckman Coulter, Fullerton, CA), CD31 (Immunotech), CD34 (Immunotech), and CD45 (Immunotech) antibodies, and then incubated on ice for 30 min. After a final PBS wash, flow cytometry analysis was performed using the FACS Calibur machine (Beckton Dickson, Mountain View, CA). To confirm whether these cells could differentiate into adipogenic, chondrogenic, and osteogenic lineages, cells were cultured with induction medium then analyzed as described previously (11).
Cultivation of NSCs and Transfection
Stable cell lines expressing the DsRed protein were established. The mNSC line used in this study was purchased from the American Type Culture Collection (ATCC, Catalog #CRL-2925). Approximately 2 × 105 mNSCs were suspended in DMEM/F12 supplemented with 10% fetal bovine serum (FBS, Hyclone), 1% penicillin and streptomycin (100 U/ml, Invitrogen), and plated in cell culture dishes. The cultures were maintained at 37°C in a humidified atmosphere containing 5% CO2.
The pBudCE4.1-DsRed plasmid was constructed by inserting the DsRed fragment (BamHI/HpaI) from a pDsRed-monomer-golgi Vector (Clontech, Palo Alto, CA) into the compatible sites (BglII/PmeI) in pBudCE4.1 (Invitrogen). The detailed experimental protocol used was as follows. Step 1: plasmid DNA (pBudCE4.1-DsRed, 1 μg/μl) was mixed with 100 μl opti-MEM (Invitrogen). Step 2:2.5 μl Lipofectamine™ 2000 (Invitrogen) was mixed with 100 μl opti-MEM and incubated for 5 min at room temperature. Step 3: The solutions from step 1 and step 2 were combined and incubated for 20 min at room temperature. Step 4: The mixed solution of step 3 was added to the cultured cells, and cells were then maintained at 37°C in a humidified atmosphere containing 5% CO2 for 4 h. After 4-h incubation, medium was changed to the growth culture medium. After 24-h incubation, Zeocin (200 μg/ml, Invitrogen) was added to the growth culture medium. Single Zeocin-resistant cells were isolated by cloning rings and expanded. Cell populations with intense DsRed expression were used for experiments.
Growth Rate Measurement
The cellular proliferation of normal mNSCs and DsRed expressing mouse neural stem cells (mNSC-DsRed) was measured to determine whether normal mNSCs are altered by transfection with DsRed. A population of 1.5 × 106 cells from both cell types was seeded into culture dishes and maintained at 37°C in a humidified atmosphere containing 5% CO2 for different time points (6, 9, 12, 18, and 24 h). At each time point, cells were washed with PBS and then treated with 0.25% trypsin/EDTA. DMEM/F12 containing 10% FBS was added to inhibit trypsinization, and the cell suspensions were collected in 15-ml tubes. After centrifugation at 1500 rpm for 5 min, the supernatant was discarded followed by re-suspension of the pellet in media. Equal volumes of cell suspension and trypan blue (Sigma, St. Louis, MO) were mixed, and 10 μl of the mixture was transferred to a hemocytometer. The hemocytometer was placed under a phase-contrast microscope (IX71, Olympus, Japan) to count cells.
Establishment of an In Vitro Injury Model
To test whether the AT-MSCs coculture system can improve the viability of mNSC-DsRed under serum deprivation (SD), hydrogen peroxide (H2O2), and combined injury (SD + H2O2) for 24 h under normoxia (21% O2) and hypoxia (1% O2), an in vitro injury model was established. Two groups of cells, comprising mNSCs alone and mNSCs cocultured with AT-MSCs, were subjected to the following injuries; Injury 1: SD under normoxia; injury 2: H2O2 (500 μM) under normoxia; injury 3: combined injury (SD + H2O2) under normoxia; injury 4: SD under hypoxia; injury 5: H2O2 (500 μM) under hypoxia; injury 6: combined injury (SD + H2O2) under hypoxia. AT-MSCs and mNSC-DsRed were seeded in six-well plates (Nunc, Roskilde, Denmark) and maintained at 37°C in a humidified atmosphere containing 5% CO2 for 24 h. Cells were then washed with PBS and the media was changed to media containing H2O2 (500 μM), serum-free media, or serum-free media plus H2O2 (500 μM). Cells were cultured as described above. Photographs of each plate were taken at five random areas at a wavelength of 575 nm using a fluorescence microscope, and the optical density of each sample of mNSC-DsRed was measured using MetaMorph software (Universal Imaging, West Chester, PA). The in vitro and in vivo experimental models are illustrated and summarized in Figure 1.

Schematic representation of in vitro and in vivo experiments. (A) The in vitro injury experiment was performed using SD, H2O2 (500 μM), and combined (SD + H2O2) under normoxia (21% O2) and hypoxia (1% O2). (B) The in vivo injury experiment was performed using mNSCs transplanted with/without AT-MSCs in a rat spinal cord injury model.
The 3-(4,5-Dimethylthiazol-2-yl)-2,5-Diphenyltetrazolium Bromide (MTT) Assay
The cell viability of mNSC-DsRed after SD, H2O2, and combined injury under normoxia (21% O2) and hypoxia (1% O2) was investigated using the MTT assay (Sigma). mNSC-DsRed cells (3 × 105 cells) were seeded in six-well plates and cultured for 24 h. After injury for 24 h under normoxia or hypoxia, 5 mg/ml MTT solution was added to the cells in wells and they were incubated for an additional 5 h. The reaction was stopped by removing the MTT solution, and formazan crystals were dissolved by adding dimethyl sulfoxide (DMSO, Sigma) to each well. After transferring the dissolved products to 96-well plates (Nunc, Roskilde, Denmark), the absorbance was measured at 550 nm.
Flow Cytometry
To confirm whether the AT-MSCs coculture system can inhibit the early apoptosis of mNSCs under different injury models under normoxia (21% O2) and hypoxia (1% O2), flow cytometry analysis was performed using the ApoScan™ Annexin V FITC apoptosis detection Kit (BioBud, Seoul, Korea). In brief, cells were harvested using trypsin/EDTA after injury. Cell pellets were washed with ice-cold PBS and then 500 μl 1× binding buffer was added. Annexin-V solution (250 ng/μl) was added to the 1× binding buffer and then the mixture was incubated for 15 min in the dark at room temperature. Cells were then analyzed using a FACS Caliber (Becton Dickson) and Cell Quest software (Becton-Dickson). Approximately 1 × 104 cells were analyzed per sample.
Immunofluorescence Staining
Bax is a well-known proapoptotic member of the Bcl-2 family, while Bcl-2 has been widely shown to have antiapoptotic effects. To confirm the expression of Bax (proapoptotic factor) and Bcl-2 (prosurvival factor) in neural stems cells after injury, cells were fixed in methanol for 10 min at −20°C. After washing with ice-cold PBS, cells were incubated with 0.5% Triton X-100 (Invitrogen) in PBS for 15 min at room temperature. Samples were blocked with 3% bovine serum albumin (BSA, Sigma) in PBS for 1 h at room temperature. Chambered slides containing the cells were incubated with rabbit anti-Bax antibody (1:2000, Millipore, Milford, MA) and rabbit anti-Bcl-2 antibody (1:2000, Millipore) for 1 h. After incubation, cells were washed with PBS and incubated with FITC-conjugated anti-rabbit immunoglobulin G (1:250, Jackson Immuno Research, West Grove, PA) for 1 h at room temperature. The cells were then washed three times with ice-cold PBS and then covered with a cover slip after drops of vectashield mounting medium with 4′,6′-diamino-2-phenylindole (DAPI, Vector, Burlingame, CA) were added. Samples were analyzed using a BX51 Olympus fluorescence microscope (Olympus, Japan). After pictures of each group were taken, the expression of Bax and Bcl-2 in mNSC-DsRed was measured by quantifying the optical density of Bax- and Bcl-2-positive cells. To confirm the location of cotransplanted AT-MSCs in the injured spinal cord, immunohistochemistry was performed, as outlined above, using mouse anti-human mitochondria (1:500, Millipore) and FITC-conjugated anti-mouse immunoglobulin G (1:250, Jackson Immuno Research).
Reverse Transcription Polymerase Chain Reaction (RT-PCR)
To confirm whether coculture with AT-MSCs inhibits apoptosis of mNSCs, RT-PCR was performed using mouse-specific primers of Bax and glyceraldehyde-3-phosphate dehydrogenase (GAPDH). After injury, total RNA was isolated from mNSCs and mNSCs cultured with AT-MSCs using RNasy kit (Qiagen, Hilden, Germany) and cDNA was synthesized using Accupower RT/PreMix (Bioneer, Seoul Korea). DNA was amplified with mouse-specific primers (Bax forward: CTGCA GAGGATGATTGCTGA, Bax reverse: GATCAGCTC GGGCACTTTA, GAPDH forward: AGACTGTGGAT GGCCCCTCT, GAPDH reverse: TCAGATCCACGA CGGACACA) following these steps: 94°C for 5 min, 30 cycles of 94°C for 30 s, 51°C for 30 s, 72°C for 30 s, and 72°C for 10 min.
Treatment with Neutralizing Anti-VEGF
After coculture with AT-MSCs, cells were incubated under SD conditions for 24 h. At this time, the AT-MSC coculture group was treated with neutralizing anti-VEGF, yielding the following four groups: Group 1: Control (mNSC-DsRed); group 2: AT-MSCs cocultured with mNCSs; croup 3: AT-MSCs cocultured with mNCSs treated with neutralizing anti-VEGF (500 ng/ml, Millipore); and group 4: AT-MSCs cocultured with mNCSs treated with neutralizing anti-VEGF (5 μg/ml). After 24 h, cells were stained using the ApoScan™ Annexin V FITC apoptosis detection Kit (BioBud) followed by flow cytometry analysis.
Spinal Cord Injury and Cotransplantation
The spinal cord injury (SCI) animal model involved adult male Sprague-Dawley rats (250–300 g; OrientBio Kyungki-do, Korea). All protocols were approved by the Animal Care and Use Committee of the Medical Research Institute of Yonsei University College of Medicine. All experiments were performed according to international guidelines on the ethical use of animals, and the number of animals used was minimized. Animals were anesthetized by sodium penthobarbital (20 mg/kg; Choongwae Pharma, Seoul, Korea). After anesthesia, laminectomy was performed at the T9 level. The spinal cord was injured by clip compression at the T9 level for 10 min.
At day 7 after surgery, mNSC-DsRed alone or mNSC-DsRed plus AT-MSCs were transplanted into the injured spinal cord to yield two groups. Group 1 rats only had mNSC-DsRed (3 × 105 cells) transplanted, while group 2 rats had AT-MSCs (3 × 105 cells) and mNSC-DsRed (3 × 105 cells) transplanted. Cells were injected into the epicenter of the injured spinal cord using an electrode microneedle. All animals received cyclosporine (10 mg/kg, Chong Kun Dang Pharm, Seoul, Korea) every day after transplantation.
Measurement of the Survival of Transplanted mNSC-DsRed
To investigate whether cotransplantation with AT-MSCs improved the survival of mNSC-DsRed transplanted into injured spinal cords, viable mNSCs were measured. After 2 and 4 weeks of transplantation, rats were sacrificed and then perfused with saline and 4% paraformaldehyde (PFA, Merck, Germany). Spinal cord tissue including injury epicenter and transplantation site was removed. A total of 100 longitudinal sections with a thickness of 10 μm were made from all spinal cord tissues. Because mNSC-DsRed fluoresce red, we did not use immunohistochemistry to identify mNSCs. To distinguish between the injected mNSC-DsRed and host cells, DAPI staining (Vector Laboratories, Inc.) was performed as counterstaining and stained cells were observed at a wavelength of 575 nm under a fluorescence microscope. The mNSC-DsReds were counted from 20 serial sections slide out of a total of 100 sections.
Statistical Analysis
Student's t-test was used to compare data between two experimental groups. One-way ANOVA was used to compare three experimental groups with a Student-Newman-Keuls test for pairwise comparisons of the subgroups. Data were expressed as mean ± SD. A value of p < 0.05 was considered statistically significant.
Results
Characterization and Differentiation of AT-MSCs
Before using AT-MSCs, flow cytometry analysis was performed to confirm the identity of the primary cell culture using CD90 and CD105 antibodies. The AT-MSC preparations showed high numbers of mesenchymal stem cell markers CD90-positive (>97.90%) and CD105-positive (>98.26%) cells and did not show endothelial and hematopoietic cell markers such as CD31, CD34, and CD45 (Fig. 2A). Moreover, these cells exhibited the potential to differentiate into adipogenic, chondrogenic, and osteogenic lineages (Fig. 2B–D).

Characterization and differentiation potential of AT-MSCs. (A) Phase contrast micrograph displaying the typical fibroblast morphology of plated AT-MSCs. Flow cytometry analysis demonstrated that AT-MSCs express the MSC markers CD90 and CD105 on the cell surface, but not endothelial and hematopoietic cell markers CD31, CD34, and CD45. Images showing that AT-MSCs differentiated into adipogenic (B), chondrogenic (C), and osteogenic (D) lineages. Cells were photographed by phase contrast microscopy at 100× magnification. Scale bars: 100 μm.
Characterization of mNSCs and Coculture with AT-MSCs
To determine whether mNSC-DsRed still had the same phenotype as normal neural stem cells, their growth rates and morphology were compared. mNSCs expressed DsRed continuously, and there was no difference in morphology between mNSCs containing plasmid (Fig. 3B) and those without plasmid (Fig. 3A). The growth rate of DsRed-expressing mNSCs was not significantly slower than that of normal mNSCs (Fig. 3C). The culture conditions of mNSCs alone and with AT-MSCs are shown in Figure 3D–F and G–I, respectively. Neither cell contamination nor rejection was observed between mNSC-DsRed and AT-MSCs. When grown alone, mNSC-DsRed cells attached to the bottom of the culture dish. In contrast, when cultured with AT-MSCs, mNSC-DsRed cells were attached to both AT-MSCs and the culture dish.
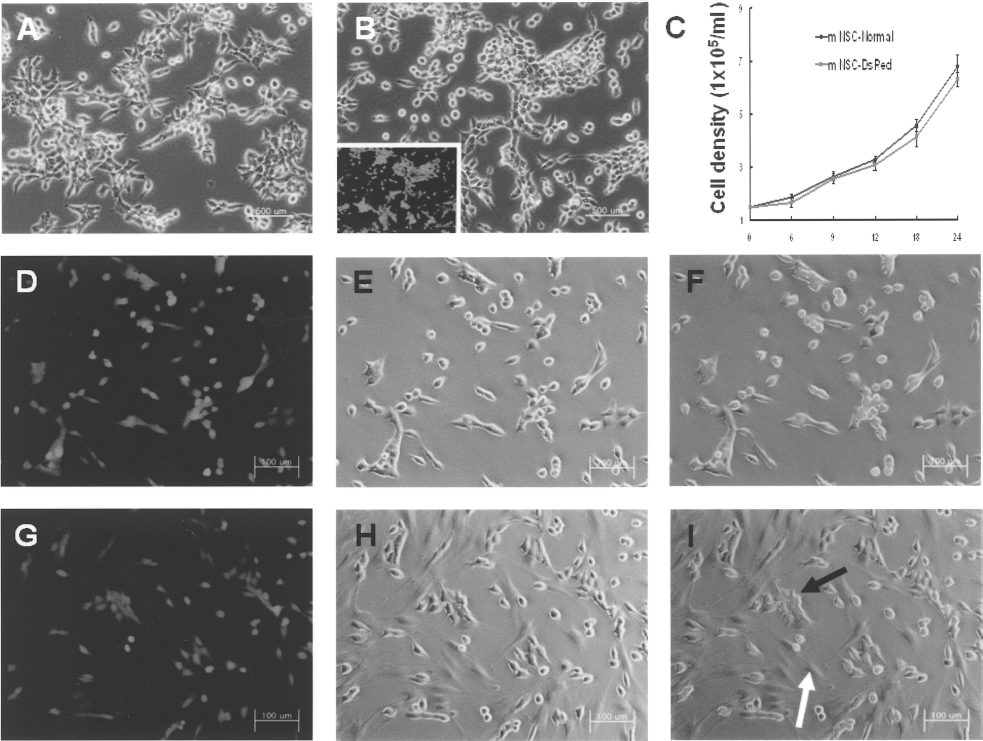
Characterization of mNSC-DsRed and Co-culture with AT-MSCs. Nontransfected control mNSCs (A) and mNSC-DsRed (B) have similar morphologies. mNSC-DsRed express red fluorescence under a fluorescence microscope (the box in B). (C) mNSC-DsRed cells have similar growth rates compared to normal mNSCs. The doubling time of both mNSC-DsRed and normal NSCs is around 12 h. Images show the condition of mNSC-DsRed when cultured alone (D–F) or with AT-MSCs (G–I). White arrows indicate AT-MSCs and black arrows point to mNSC-DsRed cells. Cells were photographed using phase contrast and fluorescence microscopy at 100× magnification. Scale bars: 100 μm.
Viability of mNSC-DsRed Under Serum Deprivation, Oxidative Stress, and Combined Injury
The viability of mNSC-DsRed in each of the injury models decreased according to injury intensity (Fig. 4A). Furthermore, the viability of mNSC-DsRed during the three different injuries under hypoxia also decreased.

Viability of mNSC-DsRed in the in vitro injury model. (A) To evaluate the in vitro injury model, mNSC-DsRed were treated with SD, H2O2, or combined (SD + H2O2) injury under normoxia (21% O2) or hypoxia (1% O2). The viability of mNSC-DsRed was investigated using the MTT assay. The viability of mNSC-DsRed decreased significantly under SD, H2O2, and combined (SD + H2O2) injury conditions. The combined injury group had lower viability than the single injury (SD or H2O2) groups. Similar patterns of decreased mNSC-DsRed viability were noted in a hypoxic environment. (B, C) Coculture of mNSC-DsRed and AT-MSCs significantly improved the viability of mNSC-DsRed under SD, H2O2, or combined SD plus H2O2 injury in normoxia (B) or hypoxia (C). Data are presented as mean ± SD. **p < 0.01 compared to the control group.
Twenty-four hours after injury, the number of mNSC-DsRed cocultured with AT-MSCs was significantly greater than that in the control group under normoxia (Fig. 4B) and hypoxia (Fig. 4C). This result indicates that coculture with AT-MSCs can improve the survival of mNSCs in SD, H2O2, and combined injury under normoxia and hypoxia.
Apoptosis of mNSC-DsRed Under Serum Deprivation, Oxidative Stress, and Combined Injury
Flow cytometry analysis was performed to investigate whether coculture of mNSC-DsRed with AT-MSCs can inhibit the apoptosis of mNSC-DsRed after injury under normoxia and hypoxia. The number of Annexin-V-positive mNSC-DsRed increased according to the injury intensity. In contrast, the number of Annexin-V-positive cells significantly decreased in cocultures of mNSC-DsRed and AT-MSCs. Coculturing of AT-MSCs and mNSCs protected approximately 50% of mNSCs from apoptosis in SD, H2O2, and combined injury under normoxia (Fig. 5A) and hypoxia (Fig. 5B). Additionally, immunofluorescence staining of Bax and Bcl-2 was performed to identify how coculture with AT-MSCs decreased apoptosis in mNSCs (Fig. 6A, B). Bax-positive mNSCs cocultured with AT-MSCs were significantly lower relative to the control group (Fig. 6C), and Bcl-2-positive mNSCs in the coculture group were significantly higher compared to the control group (Fig. 6D). In addition, RT-PCR results show that the expression of Bax was significantly reduced in mNSCs cocultured with AT-MSCs compared to mNSCs cultured alone (Fig. 6E). These results suggest that coculture of mNSCs with AT-MSCs significantly inhibits the apoptosis of mNSCs.

Effect of coculture of AT-MSCs and mNSC-DsReds on the apoptosis of mNSC-DsRed under normoxia (A) and hypoxia (B). (A) Treatment with SD, H2O2, or SD + H2O2 clearly induced apoptosis of mNSC-DsRed as assessed by Annexin V staining (9.57% in SD, 24.43% in H2O2, 29.56% in SD + H2O2). However, coculture of mNSCs with AT-MSCs significantly inhibited apoptosis induced by in vitro injury in mNSCs (5.29% in SD, 12.94% in H2O2, 13.50% in SD+ H2O2). The bar graph summarizes the results from five independent experiments. The dot blots are representative flow cytometry analysis data from a single experiment. (B) Under hypoxic conditions, treatments with SD, H2O2, SD + H2O2 clearly induced apoptosis as assessed by Annexin V staining (14.43% in SD, 24.62% in H2O2, 24.79% in SD + H2O2). However, coculture of mNSCs with AT-MSCs significantly inhibited apoptosis induced by in vitro injury models (4.77% in SD, 9.30% in H2O2, 7.14% in SD + H2O2). The bar graph summarizes the results from five independent experiments. The dot blots are representative flow cytometry analysis data from a single experiment. Data are presented as mean ± SD. *p < 0.05, **p < 0.01 compared to the mNSC-alone group.

Expression of Bax and Bcl-2 in mNSC-DsRed after SD. The expression of Bax and Bcl-2 in mNSC-DsRed after SD injury for 24 h in normoxic and hypoxic conditions was evaluated using immunofluorescence staining. mNSC-DsRed were immunostained with anti-Bax antibody or anti-Bcl-2 antibody, and FITC-conjugated secondary antibody. (A) The expression of Bax in mNSC-DsRed was decreased in the coculture group both under normoxia and hypoxia. (B) In contrast, the expression of Bcl-2 was higher in mNSC-DsRed cocultured with AT-MSCs under normoxia and hypoxia. (C, D) Graph showing the quantification of Bax (C) and Bcl-2 (D) expressed in mNSCs alone and cultured with AT-MSCs. (E) In SD injury under both normoxia and hypoxia, the Bax expression of mNSC-DsRed was significantly reduced in cells cultured with AT-MSCs. The cells were photographed by fluorescence microscopy at 400× magnification. Data are presented as mean ± SD. *p < 0.05 compared to the mNSC-alone group. Scale bars: 25 μm.
Effect of VEGF on the Apoptosis of mNSC-DsRed
To identify the role of AT-MSC-secreted VEGF in mNSC viability, cells were treated with neutralizing anti-VEGF (500 ng/ml or 5 μg/ml). Coculture with AT-MSCs significantly decreased the apoptosis of mNSC-DsRed compared to the control group (Fig. 7). In contrast, treatment with neutralizing anti-VEGF (500 ng/ml or 5 μg/ml) attenuated the antiapoptotic effect of coculture of mNSCs with AT-MSCs in a dose-dependent manner.

Inhibition of the neuroprotective effect of coculture with AT-MSCs using neutralizing antibodies against VEGF. Cells were incubated for 24 h in the SD condition under normoxia and hypoxia. Neutralizing anti-VEGF (500 ng/ml or 5 μg/ml) was administered during SD injury under normoxia and hypoxia. Apoptosis was measured by flow cytometry after Annexin V immunostaining. Treatment with neutralizing anti-VEGF resulted in an increase in the number of apoptotic mNSCs. Data are presented as mean ± SD. *p < 0.05 compared to the coculture group.
Effect of Cotransplantation of mNSCs and AT-MSCs in a Spinal Cord Injury Model
Rat spinal cord tissue was obtained 2 and 4 weeks after transplantation into the injured spinal cord. Optical density analysis showed a greater number of mNSCs in the spinal tissue of rats that had both AT-MSCs and mNSCs transplanted than the mNSCs-only transplantation group. The higher survival of mNSCs cotransplanted with AT-MSCs was maintained 4 weeks after transplantation (Fig. 8A–C). In the experiment to confirm the location of AT-MSCs transplanted with mNSCs, it was observed that AT-MSCs remained close to mNSC-DsRed cells in the lateral (Fig. 8D) and middle (Fig. 8E) part of the injured spinal cord.
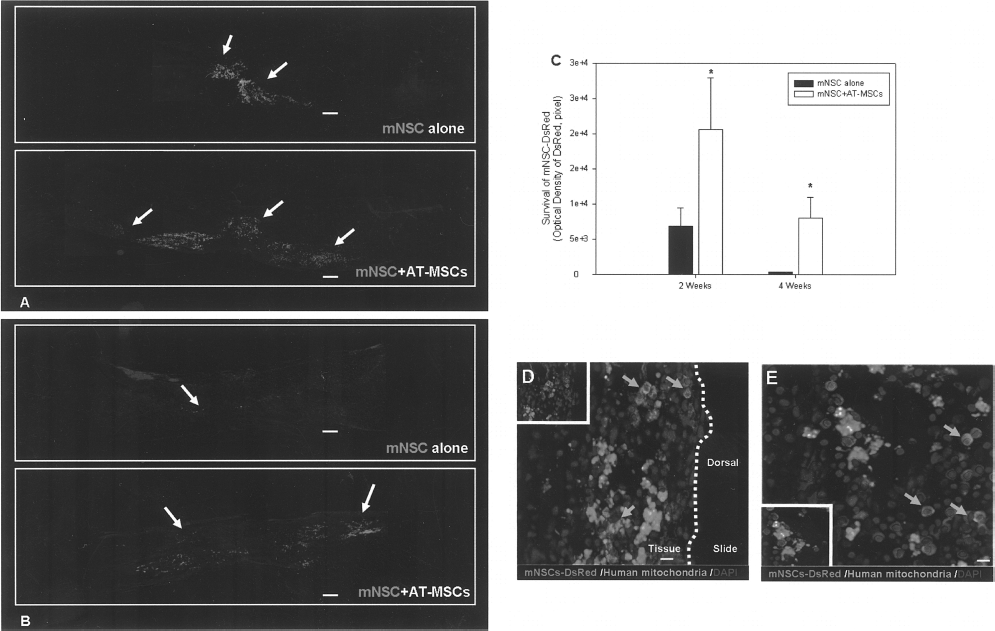
Cell survival of mNSC-DsRed 2 and 4 weeks after transplantation into SCI model. (A) Spinal cord tissue was obtained 2 weeks after transplantation into the injured spinal cord. Optical confocal microscopic findings demonstrate that the number of mNSCs in the AT-MSC cotransplanted group increased compared to the number of mNSCs transplanted alone. Moreover, distance of migration of mNSCs in cotransplantation group was higher than the mNSCs-alone group. Arrows indicate transplanted mNSC-DsRed. Counterstaining was performed by nucleus staining with 4′,6′-diamino-2-phenylindole. (B) Spinal cord tissue was obtained 4 weeks after transplantation into the injured spinal cord. Optical confocal microscopic findings demonstrate that the number and distance of migration of mNSCs in the AT-MSC cotransplanted group increased compared to the number of mNSCs transplanted alone. Arrows indicate transplanted mNSC-DsRed. (C) Optical density analysis showed a greater number of mNSCs in the spinal tissue of rats in both AT-MSCs and mNSCs cotransplanted than the mNSCs-only transplantation group. The higher survival of mNSCs cotransplanted with AT-MSCs was maintained 4 weeks after transplantation. The graph shows the total number of viable cells in 20 slices selected from 100 slices in total. (D, E) AT-MSCs transplanted into the spinal cord with mNSCs remained near mNSCs in the lateral (D) and middle (E) parts of the injured spinal cord. The cells were photographed by fluorescence microscopy at 40× (A, B) and 400× (D) magnification. Data are presented as mean ± SD. *p < 0.05 compared to the mNSC-alone group. Scale bars: 250 μm (A, B), 25 μm (D).
Discussion
In the present study, we established a gene delivery platform comprising neural stem cells transfected with a nonviral vector and the reporter gene DsRed. Maintaining the viability of graft stem cells is a critical factor of success in stem cell therapy. To increase stem cell viability, genes related to the survival and proliferation of stem cells have been overexpressed. In our study, we first confirmed whether mNSCs expressing a reporter gene had similar morphological and growth characteristics to normal mNSCs. DsRed-expressing mNSCs had similar morphological and growth characteristics to normal untransfected mNSCs. Therefore, our gene delivery platform using neural stem cells is suitable for stem cell therapy combined with gene therapy. However, to confirm the safety of using mNSCs for stem cell therapy, the genetic stability of mNSC chromosomes and the risk of malignant transformation still have to be evaluated.
We investigated whether cotransplantation of AT-MSCs with DsRed-expressing mNSCs increased mNSC survival after transplantation into injured spinal cords. Before performing the in vivo experiment, we examined in vitro whether the AT-MSC coculture system could protect mNSCs from SD, H2O2, and combined injury (SD + H2O2), all conditions that mimic the local environment within the spinal cord injury site. We found that coculture of mNSCs with AT-MSCs not only significantly improved the viability of mNSC-DsRed (Fig. 4), but also inhibited early apoptosis of mNSC-DsRed (Fig. 5) under serum deprivation, H2O2, and combined injury models. These findings suggest that biomolecular substances secreted by AT-MSCs improve mNSC survival and inhibit mNSC apoptosis. Kang's group reported about the interaction between AT-MSCs and mNSCs in vitro (10). They reported mNSCs more strongly attached to AT-MSC-coated dishes than laminin-coated dishes and that direct contact of mNSCs with AT-MSCs led to increased neuronal differentiation when compared with mNSCs in AT-MSC condition media and AT-MSCs grown on filter. Coupled with our results, the data suggest that mNSCs physically interacting with AT-MSCs positively impacts both viability and neuronal differentiation.
During hypoxic injury, MSCs secrete higher amounts of neuroprotective cytokines such as VEGF. These results support our hypothesis that neuroprotective factors secreted by AT-MSCs during hypoxia with SD might improve mNSC viability. To verify whether VEGF produced by AT-MSCs is one of the neuroprotective factors that protect mNSCs, we treated cocultures of AT-MSCs and NSCs with a neutralizing anti-VEGF antibody. VEGF is a well-characterized angiogenic factor that is induced by hypoxic stimulation (22). In addition to its classical role in angiogenesis (31), recent studies have revealed that VEGF is also an important neurogenic (9) and neuroprotective agent (17,23,24). Moreover, VEGF released from transplanted BMSCs was demonstrated to improve neurological function in a stroke animal model (4). These findings suggest that VEGF is a major mediator of neuronal protection under hypoxia. In this study, we found that VEGF-neutralizing antibodies inhibited VEGF activity and decreased mNSC viability. Therefore, we assert that VEGF is an important neuroprotective cytokine produced by AT-MSCs that improves the survival of NSCs cocultured with AT-MSCs during SD injury.
Bax and Bcl-2 mediate cell survival and death via the p53 signaling pathway. In a myocardiac infarction model, the control group expressed higher amounts of the proapoptotic Bax than the MSC transplantation group (26). However, the expression of the antiapoptotic Bcl-2 gene was significantly increased in the MSC treatment group (27). In this study, we found that coculture of mNSCs with AT-MSCs inhibited Bax expression and increased Bcl-2 expression in mNSC-DsRed. These findings support the hypothesis that neuroprotective factors secreted from AT-MSCs could induce the antiapoptosis cascade, thereby increasing mNSC viability.
It has been reported that cotransplantation of BMSCs and spinal cord progenitor cells into injured spinal cords increased the viability of the graft spinal cord progenitor cells (19). Moreover, it has been demonstrated that MSCs form tissue bridges at day 7, thereby facilitating axonal sprouting and stem cell attachment (18). In the present study, we transplanted AT-MSCs and mNSCs simultaneously at day 7 after SCI, and demonstrated that the viability of mNSCs increased when they were cotransplanted with AT-MSCs. It indicates that the survival improvement of transplanted NSCs might be improved by molecules secreted from AT-MSCs around NSCs. These findings suggest that a cotransplantation strategy involving AT-MSCs and mNSCs is an attractive therapeutic model for improving stem cell survival. Further studies to investigate the neurologic functional outcomes achieved using mNSCs alone and mNSCs cotransplanted with AT-MSCs are required.
Conclusions
In this study, we demonstrated that coculture of mNSCs with AT-MSCs significantly improved the viability of mNSCs in an in vitro injury model. Cotransplantation of AT-MSCs and mNSCs also enhanced the survival of graft mNSCs in an SCI animal model. These findings imply that cotransplantation of mNSCs with AT-MSCs could improve the outcome of NSC therapy in a neural injury model.
Footnotes
Acknowledgments
This study was supported by a grant from the Stem Cell Research Center of the 21st Century Frontier Research Program funded by the Ministry of Education, Science and Technology, Korea (Grant No. SC-4180) and KRIBB Research Initiative Program (Grant No. KGM2210911). We would like to thank Y. J. Choi at the Cell Therapy Center, College of Medicine, Yonsei University for her guidance in flow cytometry analysis.
