Abstract
Near-infrared (NIR) fluorescent agents hold great promise for noninvasive in vivo imaging. We have recently reported that a NIR fluorescent heptamethine dye, IR-780 iodide, exhibits unique optical properties for biomedical imaging. On the basis of this foregoing work, we further describe here the potential application of IR-780 iodide as a novel NIR agent for stem cell labeling and tracking. The labeling efficiency, subcellular localization, and the effects on cell viability and differentiation of IR-780 iodide were investigated. The in vivo distribution of stem cells after intravenous transplantation was traced by whole-body animal NIR imaging. Our results showed that IR-780 iodide exhibited superior labeling efficiency and biocompatibility with unique optical properties. Following whole-body NIR imaging, the pulmonary passage of stem cells was noninvasively visualized in rats after systemic transplantation of IR-780 iodide-labeled stem cells through intravenous delivery. With this NIR imaging method, we further confirmed that pretreatment with sodium nitroprusside (SNP), a vasodilator agent, significantly reduced the cell trapping in the lung and increased the cell passage through the lung capillaries. Our study suggests that IR-780 iodide may represent an effective NIR fluorophore for stem cell labeling and tracking.
Introduction
Stem cell transplantation has become a promising strategy in encouraging tissue regeneration, repair, and replacement from injuries (22,25,27). Cell imaging and tracking are crucial for understanding the fate and the mechanisms of stem cell-based therapies. Optical imaging is an easy and real-time tool with high sensitivity and nonirradiation exposure for in vivo stem cell tracking (7,26). Recently, the application of near-infrared (NIR) imaging holds great promise in biomedical imaging due to low tissue autofluorescence and high tissue penetration depth in NIR wavelength (26). Organic dyes and inorganic quantum dots (QDs) are two representative fluorescent contrast agents that are currently available. Inorganic QDs have unique optical properties in stem cells labeling (29), but their safety is still a serious concern because there are toxic elements in their cores (23). Polymethine cyanine dyes are most common NIR fluorophores, and indocyanine green (ICG), a FDA-approved contrast agent for clinical application in angiographies and lymph node mapping, has already been used in stem cell labeling and tracking (1).
By summarizing current available cell labeling agents, there is an urgent need to develop new NIR agent for future fluorescent optical imaging technologies. In our previous study, we have identified a NIR fluorescent heptamethine cyanine dye, IR-780 iodide, as a novel NIR agent with unique optical properties in biomedical imaging (30). IR-780 iodide is a lipophilic cation with peak absorption and fluorescence maxima in NIR wavelength, which can be detected conveniently by a NIR fluorescent detection system. On the basis of our foregoing work, we describe here the application of IR-780 iodide as a NIR tracer for noninvasive in vivo imaging and tracking of stem cells.
Materials and Methods
Animals
Sprague-Dawley (SD) rats and green fluorescent protein (GFP) transgenic mice were purchased from Laboratory Animal Center of the Third Military Medical University. All animal studies were performed in accordance with the Animal Care and Use Committee Guidelines of the University for the care and use of animals.
NIR Agents
The heptamethine cyanine dye IR-780 iodide [2-(2-(2-chloro-3-((1,3-dihydro-3,3-dimethyl-1-propyl-2H-indol-2-ylidene)ethylidene)-1-cyclohexen-1-yl)ethenyl)-3,3-dimethyl-1-propylindolium iodide] and IR-786 iodide [2-(2-(2-chloro-3-((1,3-dihydro-1,3,3-trimethyl-2H-indol-2-ylidene)ethylidene)-1-cyclohexen-1-yl)ethenyl)-1,3,3-trimethylindolium iodide] were purchased from Sigma-Aldrich (St. Louis, MO, USA). ICG was purchased from JiShi Company (Shenyang, Liaoning, China). All the dyes were dissolved in DMSO at 10 mM and stored at −20°C before use.
Cells and Cell Culture
Cells used in this study included multipotent stem cells derived from rat skin dermis (termed rDMCs), human bone marrow stromal cells (hBMSCs), rat bone marrow stromal cells (rBMSCs), and GFP transgenic mouse-derived bone marrow stromal cells (mBMSCs). All these cells were established following published protocols (5,13,16,21). These cells were all cultured in ATCC-recommended media, with 10% fetal bovine serum (FBS) at 37°C with 5% CO2.
Optimization of Stem Cell Labeling
The optimal labeling condition was determined using the rDMC cell model. When cells reached 70–80% confluency, they were harvested and washed three times with phosphate-buffered saline (PBS). We first evaluated the effect of various labeling time on cells. In brief, cells were detached using trypsin and resuspended at a concentration of 5 × 106 cells/ml in serum-free medium and then labeled with IR-780 iodide at 20 μM for 1, 5, 10, 15, and 20 min at 37°C before washing in PBS for monitoring of fluorescent signal. Imaging of IR-780 iodide-labeled cells was performed using Kodak In-Vivo FX Professional Imaging System (New Haven, CT, USA) equipped with fluorescent filter sets (excitation/emission, 770/830 nm). The field of view (FOV) was 150 mm in diameter. The frequency rate for NIR excitation light was 2 μW/cm2. The camera settings included maximum gain, 2×2 binning, 1024 × 1024 pixel resolution, and an exposure time of 20 s. We then chose the optimized labeling time at 15 min, and cells at 5 × 106/ml in serum-free medium were further incubated with IR-780 iodide at the final concentrations of 0, 5, 10, 20, 40, 80, and 160 μM, respectively. After incubation, cells were washed three times with PBS to remove free dyes. The cell viability was measured by cell counting kit (CCK-8) assay (Dojindo, Kummoto, Japan) (10). In brief, labeled cells were seeded at a density of 4 × 103 cells/well in 96-well plates and cultured in 100 μl culture medium for 48 h at 37°C with 5% CO2. Then, 10 μl of the CCK-8 solution was added to each well of the plate, incubated for 2 h, and measured at a wavelength of 450 nm by a Microplate Spectrophotometer (Beckman, CA, USA).
Flow Cytometric Analysis
For detecting the labeling efficiency, IR-780 iodide-labeled cells were washed three times with PBS and fixed using 4% paraformaldehyde. After sufficient washing, cells were measured by using a FACScalibur (Becton Dickinson, CA, USA). Finally, data were analyzed using CELLQuest software (Becton Dickinson).
Optical Imaging of IR-780 Iodide-Labeled Stem Cells
The rDMC cells were labeled with 20 μM IR-780 iodide for 15 min as described above. After sufficient washing, a total of 2 × 105 labeled and unlabeled cells in 100 μl PBS in a 96-well plate were imaged. In addition, two other reported cell labeling agents, ICG and IR-786 iodide, were also applied for cell labeling and imaging with the same labeling condition (20 μM, 37°C with 5% CO2, 15 min).
Then, IR-780 iodide-, ICG-, and IR-786 iodide-labeled cells were cultured at a density of 2 × 104 cells/well in a 35-mm petri dish at 37°C with 5% CO2 for 6 h. The nucleus was stained by Hoechst (Sigma, St. Louis, MO, USA) at the concentration of 5 μg/ml for 2 min. Fluorescence microscopy was performed on a laser scanning confocal microscope (Leica, Wetzlar, Germany) using 633 nm (excitation) and 700–800 nm (emission) filter set to collect NIR fluorescent signal.
To compare the labeling capability of IR-780 iodide with different cell types, we further labeled hBMSCs, rBMSCs, and GFP-positive mBMSCs with IR-780 iodide, respectively. In addition, to investigate whether the fluorescence of IR-780 iodide altered after fixation and immunofluorescence staining, we also fixed IR-780 iodide-labeled GFP-positive mBMSCs and stained with GFP antibody and second antibody following published protocols (16). After staining, cells were imaged by a laser scanning confocal microscope as described above.
Subcellular Localization of IR-780 Iodide
To detect intracellular localization of IR-780 iodide, labeled cells were stained with Mito-Tracker Green (Molecular Probes, OR, USA) at a concentration of 200 nM for 20 min at 37°C with 5% CO2, washed three times with PBS, and imaged by a laser scanning confocal microscope.
As the sufficient labeling of stem cells can be achieved by simple incubation with IR-780 iodide, we further studied the possible mechanisms for IR-780 iodide labeling. Before labeling, cells were pretreated with sulfobromophthalein (BSP), a competitive inhibitor of Na+-independent organic-anion-transporting polypeptide (OATP), at a concentration of 250 μM for 5 min (2,6,18). In addition, to determine whether the uptake of IR-780 iodide was energy dependent, we also incubated cells with IR-780 iodide at 0°C. The uptake of IR-780 iodide was determined by a laser scanning confocal microscope.
Differentiation of IR-780 Iodide-Labeled Stem Cells
The effect of IR-780 iodide on the multilineage differentiation capacity of rDMCs was also assessed. After labeling by IR-780 iodide at the concentration of 20 μM for 15 min, cells were seeded at a density of 3 × 104 cells per well in six-well plates and cultured in either expansion medium or one of three differentiation medium to induce adipogenic, osteogenic, or myofibrogenic differentiation. For adipogenic differentiation, cells were cultured in adipogenic induction medium containing IMEM with 10% FBS, supplemented with dexamethasone 1 μM, indomethacin 0.1 mM, insulin 10 μg/ml, and 3-isobutyl-l-methyl-xanthine 0.5 mM (all from Sigma). For osteogenic differentiation, cells were cultured in the osteogenic induction mediums containing IMEM plus 10% FBS and freshly added β-glycerophosphate 10 mM, dexamethasone 100 nM, and ascorbate 2 μM (all from Sigma). For myofibrogenic differentiation, cells were cultured in an myofibrogenic induction medium containing IMEM plus 10% horse serum and freshly added 5-Aza (Sigma) at 3 μM for 24 h and then changed to the expansion medium without 5-Aza. All media contained 50 μg/ml gentamycin, 100 U/ml penicillin, and 0.1 mg/ml streptomycin. Cells were cultured in differentiation medium for 3 weeks during which period the medium were refreshed twice a week. Adipogenesis of cells were stained with oil red O and counterstained with hematoxylin. The mineralized nodule formation of osteogenic differentiation was identified by alizarin red staining. Myofibrogenic differentiation was identified by morphological identification and immunofluorescence staining of rat-myosin heavy chain (MHC) (Santa Cruz, CA, USA).
Whole-Body Animal NIR Fluorescence Imaging
NIR imaging of small animals was performed using Kodak In-Vivo FX Professional Imaging System. Rats were anesthetized with pentobarbital and maintained in an anesthetized state during imaging procedure. Briefly, rats were shaved and a total of 2 × 106 labeled cells in 500 μl PBS were injected intravenously. Then, rats were imaged at time points of 5 min, 1 h, 24 h, 48 h, and 7 days postinjection, respectively. After imaging, rats were sacrificed and dissected organs were measured ex vivo to verify the in vivo imaging results. In order to confirm the fluorescent signal detected was derived from intravenously injected IR-780 iodide-labeled cells, we also used IR-780 iodide-labeled GFP-positive mBMSCs as a positive control for histological analysis.
Considering the low survival rates of implanted cells in vivo (9,28), we also investigated the distribution of IR-780 iodide-labeled cell lysate. A total of 2 × 106 labeled rDMCs were disrupted by an ultrasonic cell disintegrator, Uibra cell (Sonic material, CT, USA) and then implanted in rats by intravenous injection. The NIR imaging was achieved at 5 min and 3 h after administration.
To further investigate the role of lung as a major obstacle for intravenous stem cells delivery, we also treated rats with (n = 3) or without (n = 3) sodium nitroprusside (SNP), a vasodilator agent, at the dose of 1 mg/kg, 10 min before cell administration and imaged 5 min after cell administration.
Histological Analysis
After imaging, dissected organs were harvested from rats and fixed by 4% paraformaldehyde for 24 h. Then, organs were embedded in optimal cutting temperature compound (OCT) (Zsbio, Beijing, China), snap-frozen, and cryosectioned at 8 μm. Sections were stained with Hoechst and then were examined by laser scanning confocal microscope and light microscope.
Statistical Analysis
Means and SDs were used to describe the variation in fluorescent signal and cell viability for the following groups: 1) viability of rDMCs compared to nonlabeled controls; 2) fluorescent signal for IR-780 iodide-labeled rDMCs compared to nonlabeled controls, ICG-labeled and IR-786 iodide-labeled rDMCs; 3) fluorescent signal for resected lungs of SNP-treated rats compared to lungs of rats without SNP treatment. One-way ANOVA was used to describe variation in viability for rDMCs compared to nonlabeled controls and fluorescent signal for the various labeling protocols. Multiple comparison p-values were determined by the LSD test. Comparison of fluorescent signal for resected lungs of SNP-treated rats and lungs of rats without SNP treatment were analyzed using Student' t-test. A value of p < 0.05 was considered statistically significant. All analyses were formed by SPSS 10.0 software.
Results
In Vitro Labeling and Imaging of Stem Cells by IR-780 Iodide
IR-780 iodide-labeled cells were observed by Kodak In-Vivo FX Professional Imaging System to determine the optimal conditions for optical imaging. As shown in Figure 1A, the uptake and retention of IR-780 iodide was time dependent and most of the dye was taken up within the initial 10 min after incubation and approached a plateau after 15 min. So, in this study, we used the optimized incubation time of IR-780 iodide at 15 min and examined the effects of various dye concentrations on cell viability. By incubating at different concentrations ranging from 5 to 160 μM, we analyzed the cytotoxicity of IR-780 iodide by CCK-8 assay. There was essentially no significant loss of cell viability when incubated with IR-780 iodide at concentrations up to 20 μM (Fig. 1B). However, higher concentrations of IR-780 iodide showed a significant decrease of cell viability. Therefore, these results demonstrated that an incubation time of 15 min with a concentration of 20 μM of IR-780 iodide was appropriate for stem cell labeling. The labeling efficiency of IR-780 iodide was more than 99% at this labeling condition (Fig. 1c) and this condition was applied for further in vitro and in vivo testing. In addition, the fluorescence intensity of IR-780 iodide could be greatly increased after cell labeling due to the binding with proteins as a result of the rigidization of these fluorophores. A similar effect of IR-780 iodide upon binding with serum proteins has also been reported in our previous study (30).
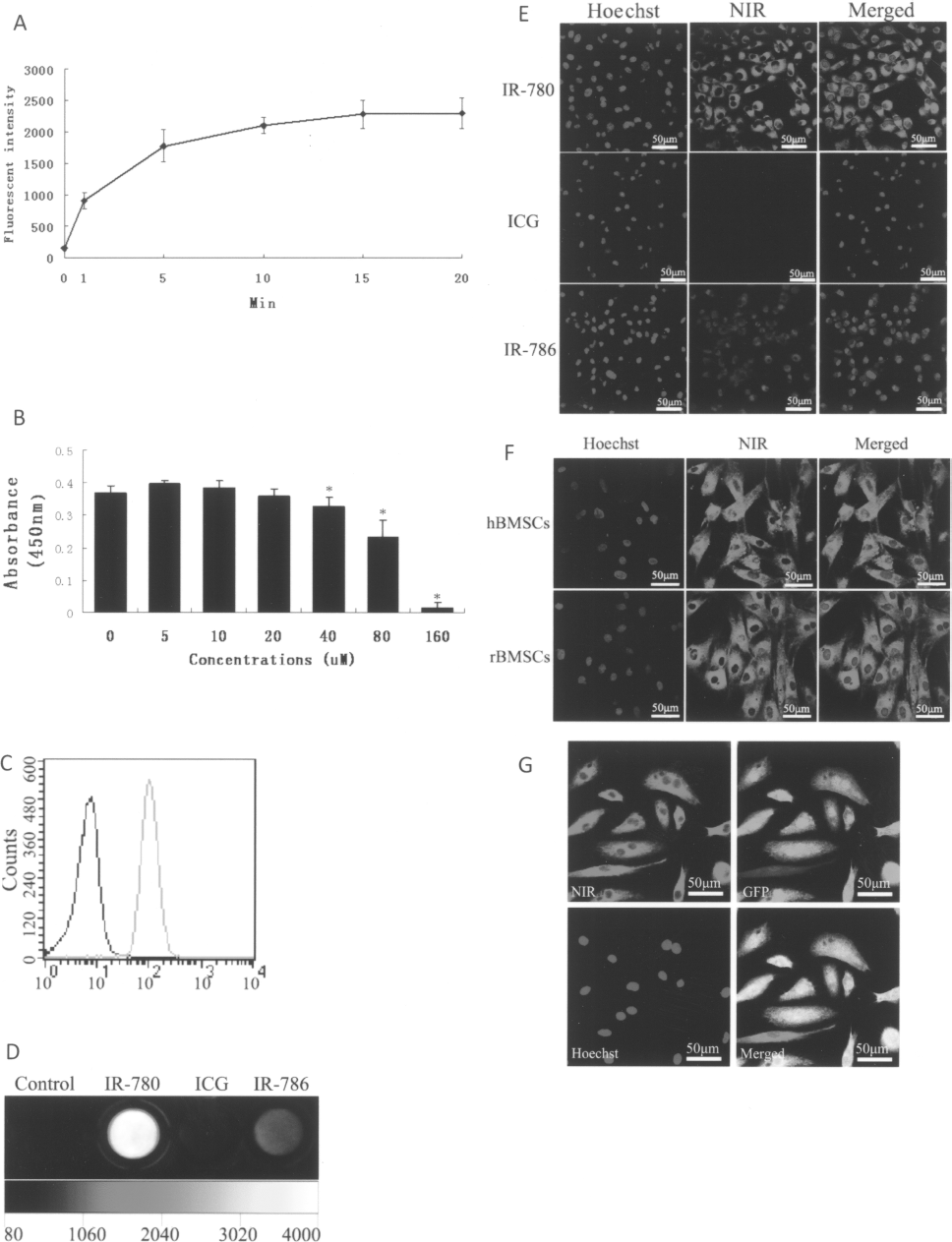
In vitro stem cell labeling and imaging with IR-780 iodide. (A) Kinetic uptake of IR-780 iodide by rDMCs exposed to 20 μM of IR-780 iodide at 37°C. (B) The viability of rDMCs labeled by different concentrations of IR-780 iodide (compare to control, ∗p < 0.05). (C) Fluorescent histograms of the unlabeled rDMCs (black) and IR-780 iodide labeled rDMCs (green) by flow cytometry. (D) NIR fluorescence images of rDMCs labeled by IR-780 iodide, ICG, and IR-786 iodide, respectively. (E) Fluorescence microscopy of rDMCs labeled by IR-780 iodide, ICG, and IR-786 iodide, respectively. The NIR fluorescence image was obtained by using a laser confocal microscope. (F) Fluorescence microscopy of IR-780 iodide-labeled hBMSCs and rBMSCs. (G) Immunofluorescent staining of GFP antibody on IR-780 iodide-labeled GFP-positive mBMSCs. The cell nucleus was displayed blue by Hoechst.
We further compared the labeling efficiency of IR-780 iodide with IR-786 iodide, an analog of IR-780 iodide that has been applied in other studies (11,14), and a FDA-approved fluorescent dye, ICG. The results showed that the fluorescence emission of IR-780 iodide-labeled rDMCs was significantly higher than the control cells and cells labeled with either ICG or IR-786 iodide (Fig. 1D). More intense fluorescence emission from IR-780 iodide-labeled than IR-786 iodide- or ICG-labeled cells was further confirmed by fluorescence microscopy (Fig. 1E).
Under the same culture and labeling conditions of rDMCs, effective labeling with IR-780 iodide was also observed in many other stem cells such as hBMCs, rBMCs, and mBMCs just by simple incubation without additional need of transfection agents. The uptake of IR-780 iodide dye by different cells is shown in Figure 1F and these results showed no obvious differences in the intensity and specificity with IR-780 iodide between these cells. Furthermore, the fluorescence of IR-780 iodide after cell labeling persisted after fixation and permeabilization of immunofluorescence staining, allowing further histological and immunofluorescence evaluation of the labeled cells (Fig. 1G).
Subcellular Localization and Effects on Stem Cell Differentiation of IR-780 Iodide
Unlike other lipophilic dyes such as DIO, DII, and DIR (3,17,19), IR-780 iodide is not a membrane stain and did not diffuse laterally to stain the entire cell. It showed a predominant distribution in the cytosol, with a specific localization in the mitochondria. By a laser scanning confocal microscope, the intracellular accumulation of IR-780 iodide in the subcellular compartments was visualized. We found that most IR-780 iodide localized in the mitochondria as confirmed by the colocalization with the mitochondria-specific fluorophore, Mito-Tracker green (Fig. 2A). We further observed that IR-780 iodide was transported into cells through an active and energy-dependent pathway, because the uptake and accumulation of the dye was significantly inhibited in cells at 0°C or by treatment of BSP, a competitive inhibitor of Na+-independent OATP (Fig. 2B).

Subcellular localization of IR-780 iodide and cell differentiation. (A) Localization of IR-780 iodide within mitochondria in rDMCs. Cells were costained with IR-780 iodide and mitochondrial tracker, then imaged under confocal microscope. (B) Energy-dependent labeling of IR-780 iodide. The cellular uptake of IR-780 iodide was significantly inhibited at 0°C or by treatment with sulfobromophthalein (BSP), a competitive inhibitor of Na+-independent organic-anion-transporting polypeptide (OATP). (C) Multilineage differentiation of IR-780 iodide-labeled rDMCs. Oil red O staining for adipogenic differentiation (left panels), alizarin red staining for osteogenic differentiation (middle panels), and immunostaining of anti-MHC for myofibrogenic differentiation (right panels) was confirmed in labeled cells.
To determine the effect of IR-780 iodide on the multipotential differentiation capability of stem cells, the in vitro differentiation capacity was tested in IR-780 iodide-labeled and unlabeled rDMCs by treatment with specific inducing media. Following adipogenic differentiation induction, both labeled and unlabeled rDMC cells with Oil red O positive lipid droplets were presented in clusters (Fig. 2C, left panels). When cultured in osteogenic medium, both labeled and unlabeled cells formed mineralized nodules that were confirmed by alizarin red staining (Fig. 2C, middle panels). For myofibrogenic differentiation, the formation of multinucleated myotubes and positive staining of MHC were observed in both labeled and unlabeled cells (Fig. 2C, right panels). Taken together, these results suggested that there was no significant effect on differentiation capability of rDMCs after IR-780 iodide labeling.
In Vivo Imaging of IR-780 Iodide-Labeled rDMCs
Because IR-780 iodide showed superior properties for stem cells labeling, we next aimed to investigate its feasibility for in vivo cell imaging and tracking. Rats received intravenous injections of 2 × 106 labeled rDMCs demonstrated a detectable fluorescence signal with NIR imaging. Directly after cell injection, fluorescence signals associated with rDMCs were detected primarily in the lungs, apparently as a function of transient pulmonary cell trapping. The dorsal view illustrated the presence of two distinct red spots that corresponded to the location of two lung lobes. The lateral view showed an enhanced fluorescence signal of the lung when compared to the dorsal view, which was attributed to the larger exposure area of the lung and the absence of heart interference (Fig. 3A). After the initial detection of labeled cells in the lung, fluorescence signal enhancement was also visualized in liver at 1 h postinjection. The fluorescence signals remained visualized in lung and liver at 24 and 48 h postinjection (Fig. 3B). The ex vivo assessments of dissected organs confirmed high lung signals in contrast to other organs, which generally corresponded to the result of the whole-body animal imaging studies (Fig. 3C). At day 7, there was no detectable fluorescence signal observed by whole-body imaging (data not shown). Histological analysis of the lung tissue further revealed that many IR-780 iodide-labeled stem cells were still trapped in the lung capillaries 24 h after injection (Fig. 3D, upper and middle panels). Although there was no detectable fluorescence signal by whole-body NIR imaging at day 7, there were also some IR-780 iodide-labeled cells in the lung capillaries (Fig. 3D, bottom panels). We further applied IR-780 iodide-labeled GFP-positive mBMSCs through intravenous injection and confirmed that the detected fluorescent signal from IR-780-labeled cells was derived from GFP-positive cells by colocalization of IR-780 fluorescent signal and GFP staining (Fig. 3E).
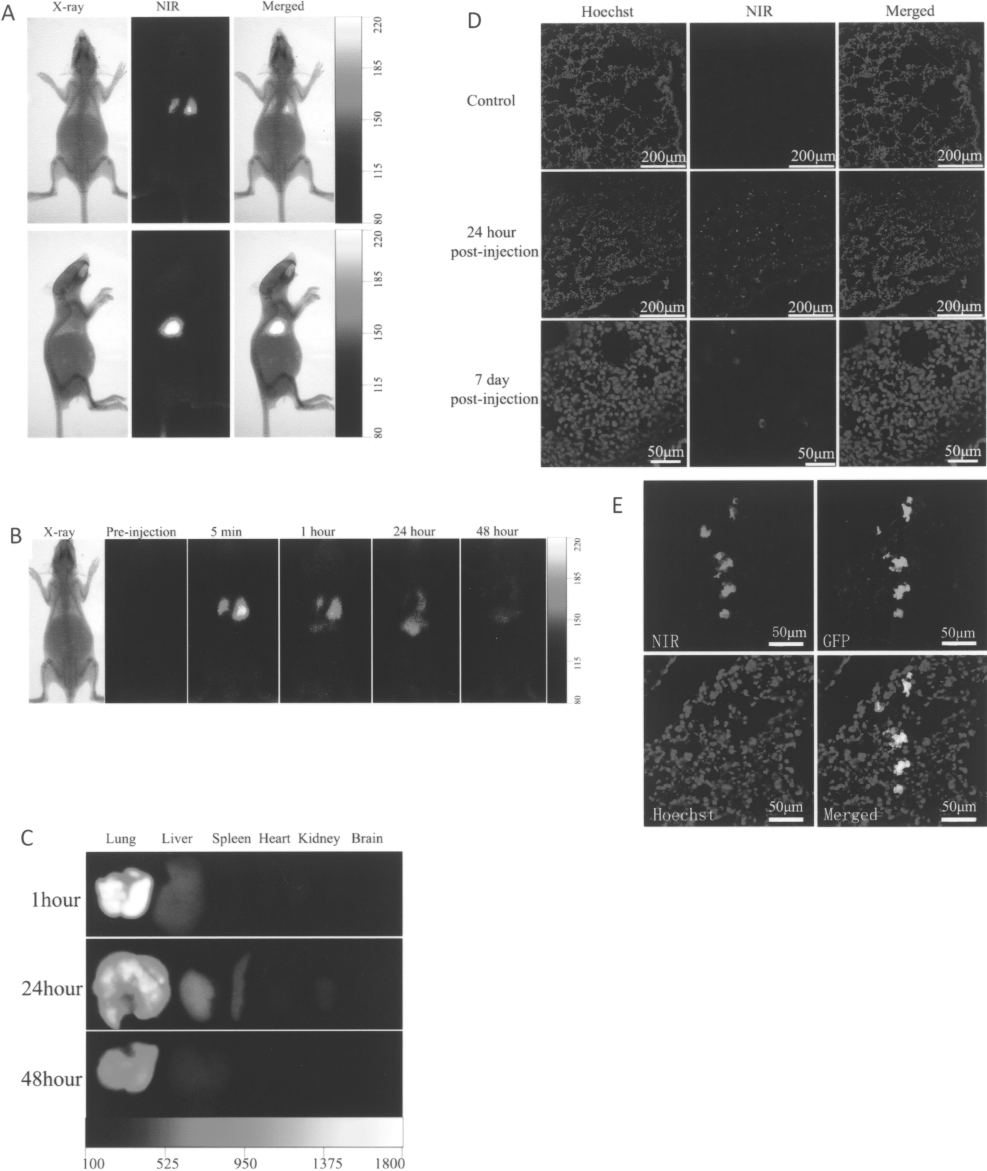
In vivo imaging and tracking of IR-780-labeled rDMCs in rats. (A) The NIR and X-ray images of rats at 5 min after systemic administration of IR-780 iodide-labeled rDMCs through tail vein. The two images were merged for lung localization of implanted cells. (B) NIR images of rats with IR-780 iodide-labeled DMCs before and at 5 min, 1 h, 24 h, and 48 h postinjection. Labeled cells were detected quickly in the lung and eventually localized in the liver. (C) NIR image of organs collected from rats receiving IR-780 iodide-labeled rDMCs 1 h, 24 h, and 48 h postinjection. Fluorescence signals confirmed that the majority of transplanted cells were in the lung regions. (D) Histological investigation indicated that IR-780 iodide-labeled rDMCs in the lung at 24 h and even 7 days after systemic administration using a confocal microscope. (E) Immunofluorescent staining of GFP protein indicated that the fluorescent signal detected in lung was derived from IR-780 iodide-labeled cells.
It has been reported that some engrafted stem cells may not survive after transplantation (17,19). We further tested the metabolism of the potential debris from the dead cells. The disrupted lysate of IR-780 iodide-labeled rDMCs by using an ultrasonic cell disintegrator were injected into rats through tail vein and its distribution was investigated. After injection, whole-body NIR imaging demonstrated an increased fluorescence signal in the liver and consequent in the intestine (Fig. 4A). The ex vivo assessments of organs showed high signals derived from liver and intestine in contrast to other organs, especially to lung that had negligible fluorescence signal (Fig. 4B, C). This result further supports that the signals from the lung were derived from the live cells after transplantation. Interestingly, the bright fluorescence signal from the intestine was able to be eliminated by removal of the fecal matter. Therefore, this high fluorescence signal was possibly attributable to the secretion of bile from the liver into the intestine. This assumption was consistent with the metabolic pathway of most heptamethine cyanine dyes such as ICG, which has FDA approval for assessment of hepatic function (12).

In vivo distribution of the lysate of IR-780 iodide-labeled cells by NIR imaging. (A) Whole body imaging of rats with disrupted IR-780 iodide-labeled rDMCs at 5 min and 3 h postinjection. An initial localization in the liver (arrows) and eventual localization in the intestine (arrowheads) were detected. (B) NIR imaging of dissected organs 3 h after intravenous infusion of the disrupted IR-780 iodide-labeled rDMCs. Most fluorescence signal was detected in the liver. (C) NIR imaging of the intestinal tract 3 h after intravenous infusion of the disrupted IR-780 iodide-labeled rDMCs. The fluorescence signal was significantly eliminated after removal of the fecal matter.
As the majority of stem cells are initially trapped inside the lungs after intravenous infusion, the increase of pulmonary passage of transplanted cells will help their engraftment in the target tissues (4,20). We then examined the potential use of IR-780 iodide for monitoring the pulmonary first-pass effect of the transplanted stem cells by the noninvasive whole-body imaging. Rats were pretreated with SNP, a vasodilator agent, and then IR-780-labeled stem cells were injected through the tail vein. Our results showed that pretreatment with SNP significantly reduced the stem cell trapping in lungs and increased the cell passage through the lung capillaries (Fig. 5A). The average fluorescence signal from the resected lungs of SNP-treated rats (673 ± 203.9) was significantly lower than lungs of rats (1186 ± 150.0) without SNP treatment (p < 0.05) (Fig. 5B).

Whole-body imaging of the pulmonary passage of IR-780 iodide-labeled stem cells. (A) NIR imaging of rats receiving the intravenous administration of IR-780-labeled rDMCs with or without SNP treatment. (B) NIR imaging of dissected organs from the rats 5 min after intravenous stem cell infusion with or without SNP treatment. Compared with control, a significant decrease of the fluorescence signal in the lung was detected in rats receiving SNP treatment.
Discussion
Optical imaging provides an effective means of tracking cells noninvasively and in real time. However, the interference from tissue background fluorescence and the limited tissue depth of light penetration are expected to be two major obstacles to optical imaging. Recently, NIR fluorescence imaging has emerged as a promising tool for optical imaging. Wavelengths in the NIR range penetrate relatively deeply into living tissue with much lower background interference and the NIR fluorescent emission can be detected even below up to 5 cm of the lung (8,24).
NIR fluorescent heptamethine dyes have been utilized as fluorescent probes for ex vivo and noninvasive in vivo imaging. In this study we have demonstrated a methodology for the labeling and noninvasive in vivo tracking of stem cells using a novel NIR fluorescent heptamethine dye, IR-780 iodide. As a lipophilic cation possessing a delocalized positive charge, IR-780 iodide is concentrated in the mitochondria in response to negative inside transmembrane potentials. This property is similar with another organic dye, IR-786 iodide, which has been used in cell labeling and tracking (11,14). However, in comparison with IR-786 iodide and ICG, IR-780 iodide has superior biocompatible and optical properties (1,11,14,15). IR-780 iodide has higher fluorescence emission and stability than ICG and IR-786 iodide. Like some of the other organic dyes, the fluorescence emission of IR-780 iodide shows a remarkable increase after cell labeling due to the binding with proteins as a result of the rigidization of these fluorophores. In addition, the fluorescence of IR-780 iodide can persist after fixation and immunofluorescence staining, raising the possibility of developing new and sensitive means of detecting targeting cells in harvested tissues.
By incubation with IR-780 iodide, multiple cell types can be labeled easily and efficiently with no adverse effects on the proliferation and differentiation of cultured cells under the optimal labeling conditions. Although there may be a concern for the transfer of the dye from labeled cells to surrounding unlabeled cells, we have not found any detectable staining of cells by intravenous injection of free IR-780 iodide dye or the lysate of the labeled cells in our in vivo studies, suggesting that the leaked dye from labeled cells can be cleared from the circulation and its effect on unlabeled cells is insignificant.
By monitoring distribution of rDMCs after systemic administration, IR-780 has shown a unique advantage for the imaging of deeper tissues, especially in lung. It has been reported that following systemic administration, the majority of stem cells are initially trapped inside the lungs (7). Therefore, NIR imaging provides a useful tool to imaging the lung trapping of stem cells. The localization of IR-780 iodide-labeled cells is detected easily in lung by NIR imaging. Although there is no obvious fluorescence signal in whole-body NIR imaging at day 7 after injection, cells still can be detected by fluorescence microscopy. As the cell size plays a crucial role in the pulmonary capillary passage (4,20), our results further confirmed that treatment with SNP for vasodilation increased pulmonary passage by whole-body NIR imaging with IR-780 iodide.
Conclusions
The noninvasive NIR fluorescence imaging using IR-780 iodide enables us to monitor the distribution and migration of stem cells in living animal models in real time. To our knowledge, this is the first report of IR-780 iodide as an NIR optical imaging agent to track stem cells in vivo for whole-body animal studies. The superior biocompatibility and unique optical properties of IR-780 iodide may provide a useful noninvasive imaging tool for stem cell-based therapies.
Footnotes
Acknowledgments
This work was supported by Chongqing Municipal Natural Science Foundation Program (CSTC2009BA5043), State Key Basic Research Development Program (2005CB522605), National Natural Science Foundation Program (30870966), and State Education Ministry Program (FANEDD200777). This research is also funded by intramural grants from the Third Military Medical University and State Key Laboratory of Trauma, Burns and Combined Injury.
