Abstract
Fibroblasts isolated from skin and from anterior cruciate ligament (ACL) secrete type I and type III collagens in vivo and in vitro. However, it is much easier and practical to obtain a small skin biopsy than an ACL sample to isolate fibroblasts for tissue engineering applications. Various tissue engineering strategies have been proposed for torn ACL replacement. We report here the results of the implantation of bioengineered ACLs (bACLs), reconstructed in vitro using a type I collagen scaffold, anchored with two porous bone plugs to allow bone–ligament–bone surgical engraftment. The bACLs were seeded with autologous living dermal fibroblasts, and grafted for 6 months in goat knee joints. Histological and ultrastructural observations ex vivo demonstrated a highly organized ligamentous structure, rich in type I collagen fibers and cells. Grafts' vascularization and innervation were observed in all bACLs that were entirely reconstructed in vitro. Organized Sharpey's fibers and fibrocartilage, including chondrocytes, were present at the osseous insertion sites of the grafts. They showed remodeling and matrix synthesis postimplantation. Our tissue engineering approach may eventually provide a new solution to replace torn ACL in humans.
Introduction
Several types of bioengineered substitutes are under development to provide new alternatives for the replacement of torn anterior cruciate ligament (ACL) (2,4,7,8). The replacement of damaged or torn ACL can be achieved by the transplantation of 1) a portion of a healthy tendon of the host (2), 2) an acellular scaffold (including or not synthetic materials), which will eventually be colonized by the host's cells in vivo (3,4), 3) a scaffold seeded with cells (preferably but not necessarily autologous cells) (14), or an allograft (22). We have developed a bioengineered ACL (bACL) using a collagen scaffold anchored with two bone plugs and seeded with autologous ACL fibroblasts (10–15). When this type of bACL is grafted in a goat knee joint, its strength reaches an average of 23% (±5%) after 11 months and 36% (±5%) after 13 months, compared to the average ultimate strengths of native goat ACL corresponding to 100%. These data demonstrate that implanted bACLs gained considerable mechanical strength postimplantation (14).
Under ideal conditions, a ligament or other tissue substitutes would preferentially be composed of autologous components to avoid any risk of immune reactions, rejections, or transmission of infectious diseases from the donor to the host. If the use of allogeneic grafts is questionable, the possibility of using heterologous stem and adult cells to produce bioengineered substitutes is widely studied (15). In tissue engineering, some choices can be made according to the features and functions of the tissue to be replaced. A reconstructed tissue needs to be integrated in the host postgrafting in order to be regenerated in situ thereafter. Thus, its matrix or scaffold must be strong enough to withstand physiological stresses early postgrafting, but must also be biodegradable to be efficiently remodeled and regenerated in situ to become functional permanently. Interestingly, lyophilized collagen seems to be a good matrix to meet these criteria in vitro and in vivo (14). However, can autologous skin fibroblasts be used, instead of autologous ACL cells, to produce a graftable bACL? Skin fibroblasts secrete type I and III collagens, like ACL fibroblasts (1). Technically, it would be less invasive to collect a microbiopsy from the skin than from the torn ACL of a patient, to isolate autologous live fibroblasts and use them to seed a bACL. We report here the results of experiments that were designed on goats to investigate this key question.
Materials and Methods
Isolation of Autologous Goat ACL and Skin Fibroblasts
To isolate autologous goat ligament fibroblasts (LFs), female adult goat knee ACLs were totally resected from their osseous insertion sites. The two insertion sites on the femur and tibia were carefully scraped down to the mineralized tissue with a scalpel to ensure that no residual pieces of ACL remained in situ. These sites were also marked for the future insertion of an autologous bACL for ACL replacement. The ACL was temporarily maintained at 4°C in serum-free Dulbecco's modified Eagle's medium (DMEM, Gibco BRL, Life Technologies, Grand Island, NY, USA), and LFs were isolated and cultured as previously described (12–14). Briefly, goat ACL biopsies were weighed and cut into small pieces after removal of the periligamentous tissue. The fragments were digested with 0.125% collagenase, containing 2 mM CaCl2 (1 ml of enzymatic solution/mg of tissue) for 20 h, under gentle agitation, at 37°C. A 0.1% trypsin solution (1 ml/mg of hydrated tissue) was then added to the cellular suspension for 1 h. The enzymes were dissolved in DMEM, pH 7.4, containing antibiotics.
Goat dermal fibroblasts (DFs) were isolated from a small skin biopsy (1 mm2) removed under local anesthesia from the back of the neck of each goat, after shaving the skin at the site of sampling. The skin biopsies were processed according to the method established in our laboratory using thermolysin prior to trypsin digestion of the epidermis (9), which allows complete separation of the epidermis from the dermal components. The epidermis was discarded and DFs were isolated from dermis using 0.2 IU/ml collagenase H (Boehringer Mannheim, W. Germany) overnight at 4°C (1,9). All procedures were approved by the local Ethics Committee.
Culture of Autologous Goat Ligament and Skin Fibroblasts
The LFs and the DFs were cultured in DMEM supplemented with 10% fetal calf serum (FCS) and antibiotics. When primary cultures reached 85% confluence, the cells were detached from their culture flasks using 0.05% trypsin-0.01% EDTA solution (pH 7.8), for about 10 min at 37°C. Cell suspensions were centrifuged twice at 200 × g for 10 min. The cell pellets were resuspended in culture medium and the cells were counted with a Coulter counter and Multisizer analyzer. The cellular viability was determined using the trypan blue exclusion method (always >90% viable cells) (9). The culture medium was changed three times a week. Both LFs and DFs maintained their morphology for at least 7 passages in culture and secreted types I and III collagens and glycosaminoglycans in monolayers (data not shown). Cells from passages 3 to 5 were used in the experiments.
Preparation of Graftable bACLs
The methods that were developed to produce and graft bACLs in goat knee joints were reported previously (14). However, all previous animal experimentation involved the grafting of bACLs seeded with autologous LFs.
To achieve the permanent fixation of the ACL to the bones (14,21), cylindrically shaped porcine bone plugs were prepared (1 cm diam. by 2 cm long) and pierced with a transverse hole (3.2 mm diam.). They were rinsed and stored in 100% ethanol for 2–3 days to eliminate traces of blood and to achieve sterilization. A polyglyconate MAXON surgical thread (size 3-0; Sherwood-Davis & Geck, St-Louis, MO), resorbable within 4–6 weeks postsurgery, was passed through the holes in the two bone plugs and tied. The bones and thread were counter-rotated to provide a single, twisted-thread link between the plugs. This bone/thread scaffolding was transferred to a sterile plastic tube and kept extended in a central, suspended position by passing two metal pins across the tube and through the transverse holes in the bone plugs.
For casting the bACLs, DMEM containing FCS and 1.0 mg/ml of bovine type I collagen (isolated in our laboratory from healthy Canadian beef skin, tested for its purity by electrophoresis, and solubilized in acetic acid diluted 1,000 times with sterile water) was quickly mixed with a suspension of autologous goat LFs or DFs (2.5 × 105 cells/ml). The mixture (total of 10 ml) was poured into 12-ml sterile plastic tubes containing the bone plugs linked by the surgical thread. The collagen polymerized in the mixture within 20 min at room temperature, under a sterile culture flow hood, while maintained without any agitation. These bACLs were cultured in DMEM supplemented with 10% FCS, 50 μg/ml ascorbic acid, and antibiotics.
After 24 h, the bACLs were frozen in sterile petri dishes overnight at −70°C and subsequently lyophilized. They were transferred back into new sterile plastic tubes and fixed again with pins as previously described. Following rehydration in fresh DMEM to produce a semi-rigid central core, a second coating of collagen seeded with LFs or SFs was applied as described above. A bilayered bACL was obtained with a lyophilized core and an outer layer. The bACLs were viable prior to implantation, because the cells progressively contracted the outer collagen layer in vitro over 24 h and thereafter. All bACLs were kept in culture for 6–8 days until grafted into their respective hosts (14).
Surgical Procedures for Implantation of Autologous bACLs Into Goats
All surgical implantation procedures were performed under general anesthesia on three groups of three 45-kg goats whose native ACLs were resected at the time of implantation of the bACLs. Autologous LFs (n = 3) and autologous SFs (n = 3) populated -bACLs were grafted in goats' knee joints. The surgical procedure was performed as reported previously (14).
Immunohistological Analysis of bACLs After Implantation
Immunohistological studies were performed on bACLs before implantation and on whole bACLs at 6 months postimplantation. The bACL samples were fixed in an aldehyde-containing solution, embedded in either paraffin or LR White acrylic resin, and sectioned and stained by either Masson's trichrome method (14), hematoxylineosin (9,12), or toluidine blue. Histological sections stained by Masson's trichrome method were examined to assess averaged cell density in the bACLs. Micrographs of the sections, taken under phase contrast microscope, were transferred to a Dell computer. Morphometric analysis of each section was performed using the MetaMorph® imaging system (Universal Imaging Corporation™, Downingtown, PA, USA), by counting the cells on different view fields (n = 4–5), on three different biopsies taken at the midlength of bACLs, grafted for 6 months (14).
A polyclonal antibody was raised in a New Zealand rabbit against bovine type I collagen. A sample of our bovine type I collagen was used to immunize the rabbit using the standard protocol established and approved from the Animal Care Ethics Committee at Laval University. This antibody does not cross-react with human and mouse type I collagens when tested on skin tissue microsections. It does not cross-react with types II, III, IV, and V collagens, when tested on human cartilage, skin, and bronchial tissue sections. For immunohistological labeling experiments, the second antibody was a polyclonal anti-rabbit IgG antibody raised in the swine and florescein conjugated (#CL6200F, Cedarlane Laboratories Ltd., Hornby, Ontario, Canada) (12). A mouse monoclonal antibody directed against neurofilament-M and neurofilament-H phosphorylated forms was purchased from Chemicon Int. (Temecula, CA, USA, #MAB1592). The second antibody was included in a catalyzed signal amplification (CSA) kit (K1500), purchased from Dako Diagnostics Canada Inc (Mississauga, Canada). The positive staining was revealed by a peroxidase reaction, using DAB as substrate.
Statistical Analyses
The statistical comparison of the data on cell density was performed according to the Student t -test, using native ACLs as controls.
Results
Host's Cell Migration Postimplantation in bACLs Seeded with Autologous LFs and DFs
In the bACLs, cell density decreased with matrix remodeling, as was observed previously (14). Similar observations were reported using allografts for ACL replacement (22). An average cell density of 1900 ± 320 cells/mm2 (n = 12) was determined in bACLs seeded with LFs or with SFs before grafting, and it diminished to an average of 860 ± 108 cells/mm2 (p < 0.01 vs. t = 0 with LFs, n = 12) and 945 ± 122 cells/mm2 (p < 0.01 vs. t = 0 with SFs, n = 12), 6 months later, respectively. However, it was not possible to discriminate cells that migrated into the implants in situ postgrafting from the fibroblasts that were already included before implantation. Cell density at the midlength of native ACLs, used as controls for statistical analyses, corresponded to an average of 415 ± 40 cells/mm2 (n = 10). Collectively, these data suggest that the bACLs underwent progressive remodeling and regeneration to become eventually competent ACLs.
Vascularization of the Grafts
The blood supply of the cruciate ligaments arises from the middle geniculate artery. From the synovial sheath, the blood vessels penetrate the ACL in a horizontal plane (20). Within the ligament, most of the blood vessels are oriented in a longitudinal direction (20). Vascularization of the graft, which occurred as early as 1 month postgrafting in bACLs seeded with the host's ACL cells (14), was also observed in several ex vivo sections of the bACLs seeded with DFs that showed blood vessels (Fig. 1a), notably between the fasciculations (Fig. 1b).

Light microscope histology of paraffin-embedded sections, stained with Masson's trichrome method, of a bACL cellularized with skin fibroblasts initially. The bACLs contained functional blood vessels as shown by the presence of erythrocytes in red (a), particularly between the fasciculations in formation (b), like in a native goat ACL (c). Scale bars: 35 μm.
Innervation of the Grafts Ex Vivo
Interestingly, some nerve fibers were observed in the grafts initially seeded with autologous DFs (Fig. 2a) and LFs (Fig. 2b). This is a good sign of integration of these grafts in situ postimplantation.

Light microscope histology of paraffin-embedded sections of nerve endings immunolabeled with an antineurofilament-H + M phosphorylated form antibody revealed by peroxidase (arrow heads) (a, b). In the regenerated synovial membranes attached to a bACL initially seeded with autologous DFs at 6 months postgrafting (a), like in a native goat ACL (b). Scale bars: 60 μm.
Bovine Collagen Scaffold Remodeling and its Progressive Replacement
Before implantation, the bilayered scaffold of the bACLs contained 100% bovine collagen as demonstrated with a polyclonal antibody specifically raised against bovine type I collagen (Fig. 3a, b). In vivo, the bovine scaffolds of collagen fibers were denatured and progressively replaced by caprine collagen fibers after colonization by the goat's cells during the 6-month period of implantation. Successive tissue sections stained with the Masson's trichrome method showed a dense collagen matrix in all bACLs grafted for 6 months (Fig. 3c, e), which was not labeled with our anti-bovine type I collagen antibody (Fig. 3d, f). As expected, tissue sections of native goat's ACLs did not show any positive staining with this antibody either (Fig. 3g, h). No antibody specifically raised against caprine type I collagen is currently available commercially and, thus, our polyclonal antibody directed against bovine type I collagen is a very useful tool to monitor bovine collagen fiber degradation on tissue sections of bACLs ex vivo.

Immunohistological labeling using the anti-bovine type I collagen antibody (a, c, e, g) and light microscope histology of the corresponding tissue sections stained with Masson' trichrome method (b, d, f, h). The cell nuclei were visualized by Hoescht staining. Before implantation, the bACLs contained 100% bovine collagen (a, b). The bovine collagen was replaced by goat's matrix in all 6-month-old grafts, as shown on sections of the femoral insertion of the bACLs initially populated with skin (c, d) and ACL (e, f) autologous goat fibroblasts. No bovine collagen labeling was detected on native goat ACLs histologcal sections (g, h). Negative controls were exposed to the second antibody alone (not shown). Scale bar: 40 μm.
Sharpey's Fibers and Fibrocartilage Formation
Sharpey's fibers were also present between the cell-seeded bACLs and their femoral attachment site (not shown). Well-structured fibrocartilage was also observed at the interface bone–ligament (Fig. 4). These observations also suggest that the bone plugs grafted with each bACL were integrated to the bones of the host's knee joint.
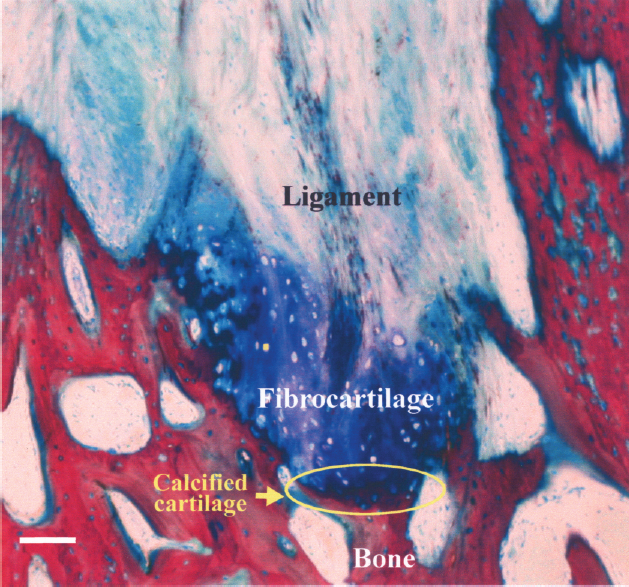
Direct graft bone insertion at the tibial insertion site. Note the four distinct zones: ligament, fibrocartilage, calcified fibrocartilage, and bone. Scale bar: 100 μm.
Discussion
Production of bACLs to be Implantated in Goat Knee Joints
Tissue engineering offers new strategies to enhance wound repair and to replace injured tissues (18). Torn ACL replacement is a challenge, because the ACL plays an important role as a knee joint stabilizer, being subjected to a lot of stress, notably because of its anatomic location (4,6). Our bACL may offer an option for torn ACL replacement, but several parameters have to be established to optimize our technology. For instance, the bilayered collagen matrix that is the main scaffold of the bACL could be eventually modified and strengthened further. To adapt and update concepts and technologies from basic science to clinic, any tissue engineering approach needs to be relatively simple but efficient. The first choice for torn ACL replacement is the bone–ligament–bone implantation, using the central portion of the patellar tendon (5,8,19). Thus, the combination of the biodegradable scaffold with two bone anchors seems to be an important feature of any potential ACL substitute for torn ACL replacement.
Host's Cell Migration Postimplantation in bACLs Seeded with Autologous LFs and DFs
In order to produce ACL substitutes seeded with autologous cells, a tissue sample has to be collected from the host to isolate these cells. Technically and surgically, it is much simpler and less invasive, faster and safer to collect a skin microbiosy (1 mm2) than a piece of ACL or other tissues to isolate autologous cells from the host. Dermal fibroblasts synthesize types I and III collagens, so these cells could initiate matrix synthesis like ACL fibroblasts. Moreover, the cells that are seeded in ACL substitutes before implantation are not expected to remain forever in the grafts. Nonimmortalized cells have a life span that is limited (about 9 passages in vitro but not determined in vivo) and they will have to be replaced by the host's cells progressively, most likely by mesenchymal stem cells. This is a natural process that is expected to occur in any tissue that is integrated in situ postgrafting. Cell labeling, with carboxyfluorescein diacetate (CFDA) or other fluorescent biomolecules, could be achieved before grafting, to evaluate the fate of the labeled cells populating the grafts after implantation (8,17). However, such cell biomarker cannot be traced after 1–2 months in the grafts postimplantation, because the marker is lost by labeled cells that undergo proliferation. This observation, made in vitro on cells grown in monolayers, limits the information that could be obtained from such experiments. At 6 months postimplantation, the autologous DFs and LFs were capable of synthesizing new caprine collagen fibers that had totally replaced the bovine collagen scaffold initially used to produce the bACLs. The new matrix fibers were oriented properly in the grafts. No difference was observed between the results obtained with bACLs seeded with DFs or LFs. These data suggest that DFs could be used to produce graftable bACLs for torn ACL replacement.
Vascularization and Innervation of the Grafts Ex Vivo
We may postulate that the colonization of the bACLs is dependent on vascularization, because the regions highly populated with cells were vascularized. The regeneration of the synovial sheath, which is destroyed during the surgical replacement of the native ACL by the bACL, may play a crucial role on the faith of any ACL substitute postimplantation, bringing the vascular network up to the graft and promoting its innervation. In fact, the regeneration of the synovial membrane, notably its vascularization, seems to be a prerequisite for bACL's innervation. Vascularization is relatively easy to monitor, because it can be observed directly on histological tissue sections, using the Masson's trichrome staining method. In contrast, the quality of innervation, analyzed by the immunolocalization of peripheral nervous endings in the bACL, is technically more difficult to demonstrate. Both physiological phenomena are reported as closely related in several tissues (16).
Bovine Collagen Scaffold Remodeling and its Progressive Replacement
Bovine type I collagen was used to produce the scaffold of each bACL grafted. The bovine matrix was progressively replaced with caprine collagen. Autologous caprine fibroblasts could initiate such matrix remodeling in vitro and continue this process in situ, in the bACLs cellularized with LFs or DFs. These observations demonstrate that the bilayered bovine scaffold is a good biomatrix for implantation, allowing a balanced ratio between degradation and de novo synthesis of collagen fibers by the cells in vivo.
In conclusion, our technology developed to produce graftable ACL substitutes seems very promising. These data are good indicators of the potential that can be explored to produce and graft bioengineered ACL substitutes permanently, in the near future.
Footnotes
Acknowledgments
F. Goulet is the recipient of a Scholarship from the Arthritis Society of Canada. This work was supported by the Canadian Institutes of Health Research (F. Goulet, FRN-79520), Canada Foundation for Innovation (F. Goulet), The Canadian Orthopaedic Association (R. Cloutier, J. Lamontagne, M. Bouchard), The Foundation of CHA, (F. Goulet), and Club Richelieu de Limoilou (F. Goulet), Quebec, Canada. We thank all the members of the team of the Animal Care Unit of Laval University for their help, Dr. Véronique Moulin for statistical analysis, Claude Marin for photographic assistance, Mr. Flavio Marinelli from Smith & Nephew for lending surgical instrumentation.
