Abstract
Microcarriers have been successfully used for many years for growing anchorage-dependent cells and as a means of delivering cells for tissue repair. When cultured on microcarriers, the number of anchorage-dependent cells, including primary cells, can easily be scaled up and controlled to generate the quantities of cells necessary for therapeutic applications. Recently, stem cell technology has been recognized as a powerful tool in regenerative medicine, but adequate numbers of stem cells that retain their differentiation potential are still difficult to obtain. For anchorage-dependent stem cells, however, microcarrier-based suspension culture using various types of microcarriers has proven to be a good alternative for effective ex vivo expansion. In this article, we review studies reporting the expansion, differentiation, or transplantation of functional anchorage-dependent cells that were expanded with the microcarrier culture system. Thus, the implementation of technological advances in biodegradable microcarriers, the bead-to-bead transfer process, and appropriate stem cell media may soon foster the ability to produce the numbers of stem cells necessary for cell-based therapies and/or tissue engineering.
Introduction
Cell transplantation has been widely used in regenerative medicine or tissue engineering applications for many years (32,37,57,100). The most well-established therapeutic applications of cells grown in suspension are blood transfusion and transplantation of hematopoietic stem cells (52,58,66). The key to successful transplantation using cell-based technologies is the preparation of large enough numbers of high-quality cells. Similar to cells grown in suspension, anchorage-dependent cell-based transplantation, such as the use of islet cells for diabetes (6,78), bone marrow cells for myocardial infarct (31,47,79), or critical limb ischemia (3), requires at least 1—2 × 109 cells for an adult patient (50—100 kg). Thus, the development of a practical and scalable bio-process that allows expansion and transplantation of functional cells would potentiate cell-based therapies.
Many culture systems have been designed to expand cells with identical properties (36,87). The key to successful large-scale and long-term cell culture is a well-defined and controlled bioreactor design. Stirred bioreactors (for suspension culture) can overcome several drawbacks of static cultures, including the presence of concentration gradients and difficulties in monitoring and control. Except for hematopoietic cells (38), however, mammalian cells are usually expanded as anchorage-dependent cells, and therefore development of a culture system with suitable support is necessary. The stirred microcarrier culture system can facilitate the expansion of cells that require attachment, and provides an environment that can easily be controlled and monitored.
In addition, owing to their capacity for self-renewal and differentiation, stem cells such as embryonic stem cells (ESCs), induced pluripotent stem cells (iPSCs), mesenchymal stem cells (MSCs), and other adherent stem/progenitor cells have been recognized for their great potential in regenerative medicine. To realize the potential of stem cell-based therapies, however, technical improvements in the large-scale expansion of stem cells and their differentiated progeny are required. Microcarriers may be an alternative culture system for producing significant quantities of cells that are required for future clinical product development.
Microcarriers
The microcarrier culture system was introduced by van Wezel in 1967 and has been applied successfully to the growth of anchorage-dependent cells for the production of vaccines or pharmaceuticals, and to expand cell populations (88). This culture system offers the advantage of providing a larger surface area for the growth of anchorage-dependent cells in a suspension culture system, and overcomes several problems with static cultures, including the requirement for large quantities of culture media, space expense, inefficient gas—liquid oxygen transfer, presence of concentration gradients, and difficulties in monitoring and control.
Although the microcarrier culture system has many advantages, the limitation of cell attachment to the microcarrier surface depends on the carrier diameter (typically in the range of 100—400 μm), density (typically between 1.02 and 1.10 g/ml), and chemical composition (48). Today, microcarriers are commonly composed of a dextran matrix, with diethylaminoethyl (DEAE) anion exchange groups being widely used. This group of microcarriers includes Cytodex™ 1, Cytodex 2, and Hillex™. In addition, Cytodex 3 is also a member of the DEAE-dextran family, but the Cytodex 3 microcarriers are coated with a thin layer of collagen (48). The other materials that are used for microcarriers include plastic, glass, and cellulose. However, the aforementioned microcarriers are less amenable in tissue engineering or direct transplantation because they cannot be decomposed by the human body. Trypsin, collagenase, and dispase have been used to retrieve viable cells from nonbiodegradable microcarriers for use in cell-based transplantation studies. However, perfect separation of the cells from nonbiodegradable microcarriers after enzyme treatment is still difficult. Therefore, development of microcarriers composed of biodegradable chemicals may be an ideal answer.
Recently, microcarriers made of biodegradable substances including gelatin (CultiSpher™ family) (49), collagen (Cellagen™) (30), polylactic-co-glycolic acid (PLGA) (85), poly-l-lactic acid (PLLA) (19), and hydroxyapatite (26) have raised great interest and been extensively studied for their clinical application in the field of regenerative medicine. In addition, cell-seeded microcarriers can be used to transport cells to the tissue(s) that needs to be repaired.
In this article, the microcarrier culture systems used for primary cells, adult stem cells, and embryonic stem cells are reviewed as follows.
Primary Cells
Microcarriers have the benefit of delivering cells to damaged or degenerated tissue for repair and restoration of function. Several studies have shown that primary cells cultured on microcarriers and implanted into animal models can cure several disorders for many years. Table 1 summarizes a number of examples of microcarriers that have been used in animal models. These studies have provided crucial information about liver insufficiency (20—22), cartilage repair (35,49,85), skin repair (44,89,90), and Parkinson's disease (12,14,73).
Microcarrier-Expanded Primary Cells for Transplantation

PLLA, poly-L-lactic acid; PLGA, poly-lactic-co-glycolic acid; hRPE, human retinal pigment epithelium.
In 1986, Demetriou et al. first reported a new method for expansion of primary hepatocytes with Cytodex 3 (dextran matrix with a layer of collagen) and the subsequent hepatocyte transplantation that results in prolonged hepatocyte viability and function in vivo (20,22). In addition, transplantation of microcarrier-attached hepatocytes had a curative effect and provided sufficient metabolic support, detoxification of ammonia, and an increase in the survival rates of liver-damaged rats (54,98). Recently, Xu et al. described the positive effects of intraperitoneal transplantation of Cytodex 3 microcarrier-attached hepatocytes in rats with acute liver failure induced by D-galactosamine (98). Additionally, a microcarrier-attached hepatocyte-based bioartificial liver also facilitated metabolic support and modified the humoral response in an experimental therapy for liver failure (81,97).
For skin repair, Voigt et al. first reported that Cytodex 3 microcarrier-attached keratinocytes provide a new multilayered and keratinized epithelium in a full-thickness wound healing model, but the microspheres could not be degraded and induced an inflammatory response (90). Therefore, this group investigated another biodegradable substance, PLGA microcarriers, which were coated with gelatin and recombinant human epidermal growth factor. At 14 days posttransplantation, a new stratified epithelium was detected in the full-thickness wound healing model (89). Recently, Liu et al. produced autologous melanocytes, keratinocytes, and fibroblasts on a large scale using the CultiSpher-G microcarrier culture system (41—44), and showed a decrease in the size of recalcitrant venous leg ulcers using autologous keratinocytes (44). Using the novel bioreactor microcarrier cell culture system (Bio-MCCS) of Liu et al., autologous human keratinocytes can be rapidly expanded on one specific type of modified CultiSpher-G microcarriers, which are first cultured with autologous dermal fibroblasts that are killed when they reach confluency (43). For cartilage repair, Malda et al. reported that human nasal chondrocytes could be expanded on CultiSpher-G microcarriers, and that the collagen content of microcarrier-expanded chondrocytes 14 days after implantation was slightly higher than T-flask-expanded cells (49). In addition to expansion on gelatin-based microcarriers, expansion of chondrocytes on PLGA microcarriers demonstrated the feasibility of articular cartilage repair in a mouse model (35,85). In addition, gelatin microcarrier-based cell delivery has shown positive clinical results in Parkinson's disease using human retinal pigment epithelial cells (14,27,93).
Furthermore, other biodegradable natural polysaccharides and FDA-approved hydrogel-based microcarriers have been shown to facilitate adhesion, proliferation, and differentiation of anchorage-dependent cells (e.g., fibroblasts, osteoblasts, and MSCs), and may be useful in clinical regenerative medicine in the musculoskeletal or dermatological fields (91,92).
Adult Stem Cells
The technologies described above have encouraged the use of the microcarrier culture system as a powerful tool for expansion of stem cells for clinical use. Moreover, the use of microcarriers in expansion, differentiation, and transplantation has only been explored to a limited extent. Tables 2 and 3 list currently available information about stem cells cultured on microcarriers. Moreover, this system also makes possible the use of continuous bioprocesses to expand and recover human stem cells for subsequent use in clinical cellular therapies or tissue engineering. As illustrated in Figure 1, there are three suitable bioreactors for stem cell expansion or differentiation on microcarriers and three prospective methods of stem cell recovery as well as direct injection for the repair of tissues such as brain, skin, liver, bone, and cartilage.
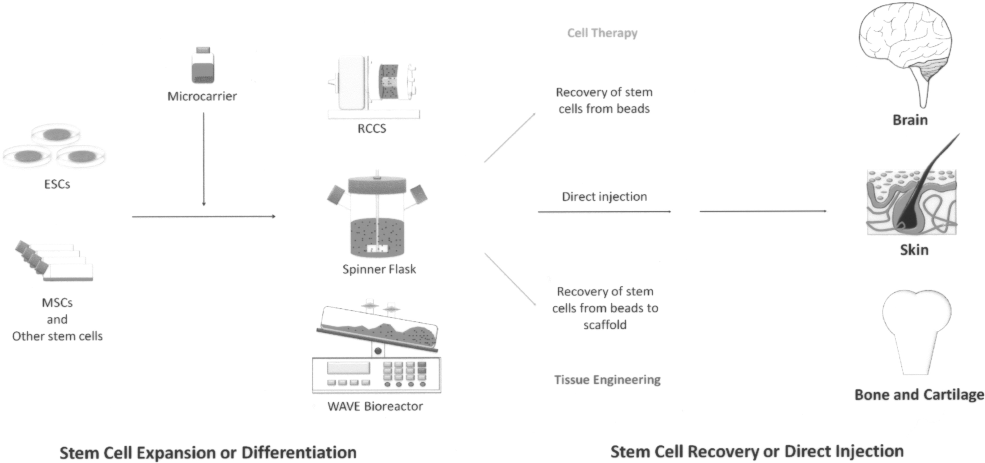
Schematic of stem cells grown in different microcarrier culture systems and the use of stem cell-seeded microcarriers. After detachment from culture dishes or T-flasks, stem cells can be cultured on microcarriers in spinner flasks, a WAVE bioreactor, or a microgravity system such as a rotary cell culture system (RCCS). After expansion or differentiation, cell-seeded microcarriers can be directly injected into the human body, or cells can be enzymatically recovered from microcarriers for use in cell therapy or tissue engineering. ESCs, embryonic stem cells; MSCs, mesenchymal stem cells.
Expansion of Adult Stem Cells Using Microcarrier Culture

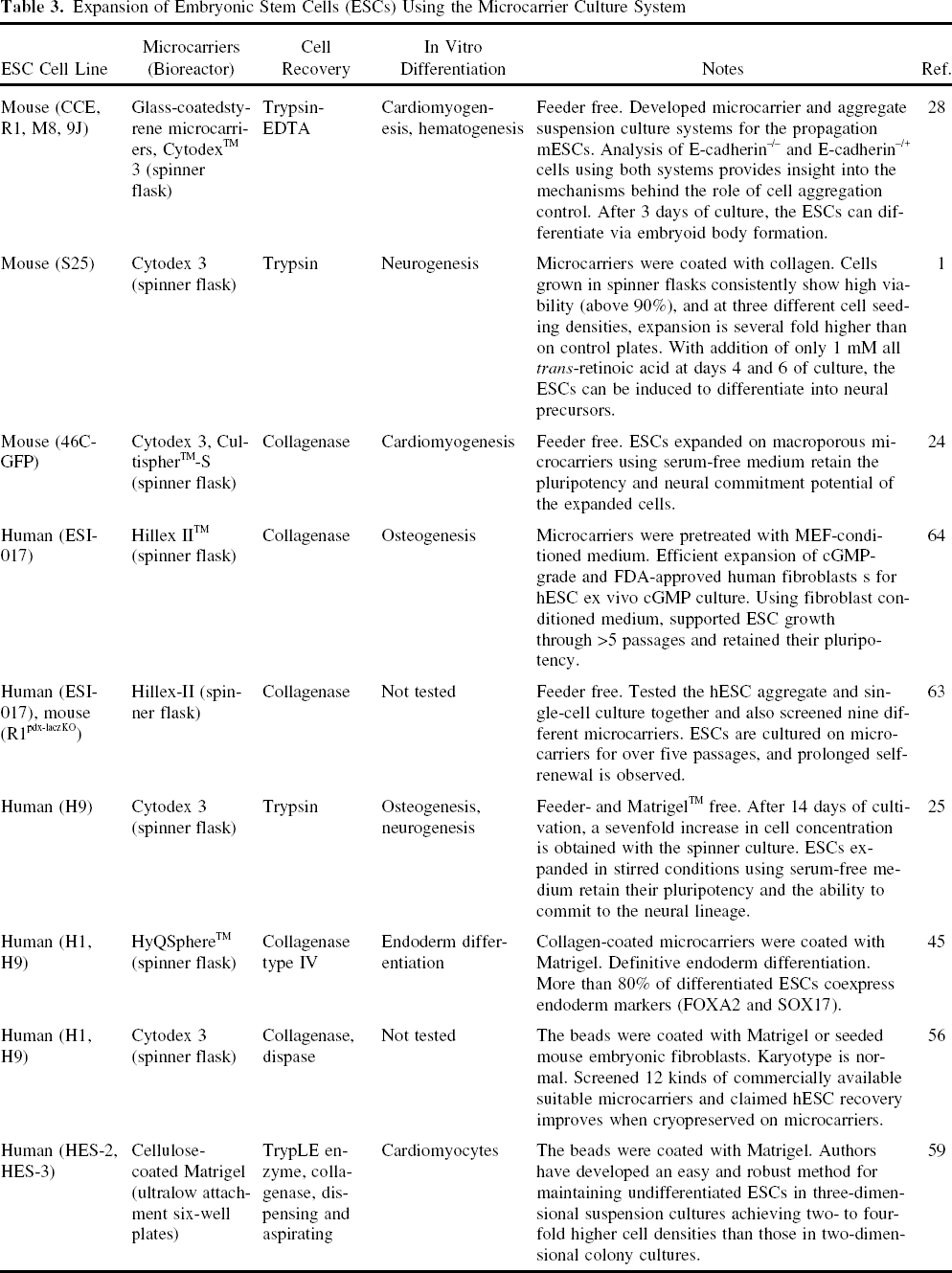
Expansion of Embryonic Stem Cells (ESCs) Using the Microcarrier Culture System
Mesenchymal Stem Cells
Owing to their high plasticity (8,15,61,65,68,83), high expansion capacity (65,82,83), and ability to avoid rejection after transplantation (72), these adult stem cells are likely to play a prominent role in regenerative medicine. Although some clinical studies have demonstrated successful tissue engineering with MSCs (8,61,68), technological problems remain, such as a time- and money-consuming expansion phase due to a long expansion period and a labor-intensive process. Therefore, the development of bioreactors that allow monitoring, control, and automation of the cell culture process will be decisively important.
The ability of MSCs to attach, proliferate, and differentiate on macroporous microcarriers in spinner culture has been confirmed (71,95,99) (Fig. 2). The culture of MSCs on microcarriers was first explored by Wu et al. in 2003 (95); the results showed that 7 days of cultivation yielded a 10-fold increase in viable cells in spinner culture, and that the average lactate yield from glucose consumption in the spinner culture was only 1.63, which is lower than that in stationary culture (2.44). Additionally, after growth in the spinner culture for 12 days, the MSCs maintained the characteristics of stem cells (95). Recently, the effect of porcine bone marrow mesenchymal stem cell (BMMSC) proliferation using three types of commonly available Cytodex microcarriers was examined by Frauenschuh et al. The results showed that Cytodex 1 microcarriers promoted a higher level of cell attachment and proliferation compared with Cytodex 2 and 3 (29). In addition, this group also demonstrated increased colonization of microcarriers after carrier addition in a 1:1 ratio after almost all microcarriers had become confluent (bead-to-bead transfer) (29). Recently, the microcarrier culture scalability of ear MSCs was expanded with bead-to-bead transfer, and cells maintained their potential for differentiation (75). Also, addition of 30% fresh medium containing microcarriers every 3 days provided decreased production of lactate from glucose and continuous proliferation of goat MSCs on microcarriers (76). In addition, a more homogenous cell distribution on the microcarriers was obtained as a result of bead-to-bead transfer (76). Based on the use of bead-to-bead transfer, the available area can be extended and the culture of anchorage-dependent cell can be prolonged by free cells colonizing from old beads to fresh beads (60).

The location and proliferation of BMMSCs on CultiSpher-G microcarriers after 7 days with an initial density of 5 × 104 cells/m and a microcarrier concentration of 3 g/L. Samples from (A, D) 5 h, (B, E) 1 day, and (C, F) 7 days stained with Hoechst 33342 dye (D, E, F). Starting on day 3, 50% medium changes were performed every 3 days. Scale bar: 500 μM. The alkaline phosphatase (ALP) activity of bone marrow mesenchymal stem cells (BMMSCs) undergoing direct osteo-differentiation in stirred microcarrier cultures under osteogenic conditions is shown by ALP staining (G). The lipid spheres in BMMSCs undergoing direct adipo-differentiation in stirred microcarrier cultures under adipogenic conditions is shown with Oil Red O staining (H). Scale bar: 200 μM.
Recently, adult MSCs have been proposed as attractive candidates for regenerative therapy of connective tissues. MSC-seeded microcarriers may simplify and enhance the performance of tissue engineering approaches for cartilage and bone repair. Rubin et al. reported a strategy for the proliferation and differentiation of adipose-derived stem cells on CultiSpher-G microcarriers that would allow induction of differentiation ex vivo, precise placement of cells, and correct tissue architecture (71). In addition, Yang et al. reported that short-term direct subcutaneous transplantation of BMMSCs expanded on CultiSpher-S microcarriers was associated with significantly less apoptosis than trypsinized control cells, and that induction of de novo trabecular bone formation in vivo occurred in the long term (99).
In addition to the CultiSpher family, hydroxyapatite microparticles—another type of biodegradable microcarrier that varies in size and microporosity—were evaluated in vitro and in vivo for their suitability for use as a carrier in an injectable tissue-engineered bone filler. Fischer et al. showed that both the size and microporosity of nonporous hydroxyapatite microparticles affect in vivo bone formation by cultured mesenchymal progenitor cells. The diameter particle size range of 212—300 mm is optimal for use as carriers of cultured goat mesenchymal progenitor cells for the in vivo production of bone tissue (26).
In some specific cases, MSCs can be cultured on microcarriers in a microgravity environment modeled by a rotary cell culture system (51,102). Zayzafoon et al. first indicated that microgravity modeled by a rotating wall vessel bioreactor inhibits osteoblastic differentiation of human MSCs on plastic microcarriers and induces the development of an adipocytic lineage phenotype (102). After 1 year, Meyers et al. confirmed that reduced osteoblastogenesis and enhanced adipogenesis of human MSCs on plastic microcarriers in modeled microgravity were mediated by RhoA and cytoskeletal disruption (51). These reports suggest a possible reason for the reduced bone mass of the human body after prolonged time in space, but also imply that this culture system cannot maintain the differentiation potential of MSCs.
Neural Stem Cells
Kallos and Behie first reported the growth and inoculation conditions for the large-scale expansion of mammalian neural stem cells in suspension bioreactors (33). These investigators showed that optimization of pH of the medium and inoculation conditions are critical for robust cell proliferation. The best growth characteristics for mammalian neural stem cells were obtained with a pH range of 7.1—7.5, a high oxygen level (20%), and low osmolarity of the medium (below 400 mOsm/kg). Kallos and coworkers also proposed optimal serial passaging protocols that would allow mammalian neural stem cells to be grown effectively in suspension culture (34). For the first time, this group was able to grow mammalian neural stem cells on a larger scale in suspension culture and maintain their stem cell characteristics.
Pancreatic Stem Cells
Adult pancreatic stem cells can be grown for more than 140 passages while maintaining the expression of typical stem cell markers (39). Serra et al. evaluated two different culture strategies, including cell aggregated culture and the microcarrier culture system. The use of a microcarrier support (Cytodex 1 or Cytodex 3) promoted the expansion of cell populations that retained their self-renewal ability, cell markers, and differentiation potential, allowing them to differentiate into adipocytes (77). In addition, although both Cytodex 1 and Cytodex 3 could sustain cell expansion, Cytodex 3 was a better substrate for the promotion of cell adherence and growth.
Progenitor Cells
Melero-Martin et al. reported that the use of Culti Spher-G microcarriers resulted in a 17-fold expansion of chondro-progenitor cells grown in batch cultures (50). In addition, chondro-progenitor cells were capable of undergoing bead-to-bead migration, which allowed sub-cultivation without a harvesting step. In addition to chondro-progenitor cells, Sugo and Ogawa also reported that rat bone marrow cells grown on hydroxyapatite microcarriers maintained the potential to differentiate into osteoblasts, and the cell proliferation was more rapidly in the case of the hydroxyapatite microcarrier than in the case of hydroxyapatite disk or T75 flasks (80). Barrias et al. also reported that bone marrow stromal cells could grow and differentiate into osteoblastic lineage cells when expanded on calcium titanium phosphate microspheres (7).
Hematopoietic Stem/Progenitor Cells
Hematopoietic stem/progenitor cells have been expanded or differentiated with or without coculture with bone marrow cells grown in microcarrier cultures (23, 74,101). Recently, Chiu et al. demonstrated that CD34+ human umbilical cord blood progenitors are capable of transdifferentiation to yield a vascular endothelial cell phenotype and that they could assemble into three-dimensional tissue structures in rotary wall vessels when expanded with or without microcarrier beads (17).
Embryonic Stem Cells
Recently, owing to the pluripotent differentiation potential of human ESCs that are derived from the inner cell mass of blastocysts (86), many groups have suggested that human ESCs have the potential to treat many diseases, including myocardial infarct (53), diabetes (62), and Parkinson's disease (10,18,70). However, both mouse and human ESCs are adherent cells and uniquely grow as colonies. In addition, ESCs are typically grown on a layer of inactivated mouse embryonic fibroblasts (MEFs) that function as feeder cells and maintain the ESCs in an undifferentiated state, so feeder cells are commonly realized as necessary for ESC culture (86). Replacement of MEFs with nonxenogeneic human fibroblasts was shown to support human ESC growth as well (67). In addition, recent studies have described the successful use of feeder-free (with feeder-conditioned medium but without feeder cell coculture) and feeder-independent systems (without conditioned medium or feeder coculture) with appropriate extracellular matrix molecules, such as Matrigel™, laminin, fibronectin, collagen IV, and vitronectin, for human ESC culture (4,5,46,69,96). Thus, the current methods for scaling up ESC culture using microcarriers have focused on the extracellular matrix molecules present on the surface of microcarriers.
In 2005, Fok and Zandstra first reported a method in which glass-coated styrene microcarriers and Cytodex 3 were used to establish a feeder-free microcarrier culture system for mouse ESCs, which resulted in shorter doubling times than substrate-free aggregate cultures grown in suspension (28). The effects of inoculation of mouse ESCs and the concentration of Cytodex 3 on cell growth and metabolism were analyzed by Abranches et al. (1). After seeding, the cells typically exhibited a growth curve consisting of a short death or lag phase followed by an exponential phase leading to a maximum cell density of 2.5—3.9 × 106 cells/ml and a maximum specific growth rate of 1.2 day−1 (1). In addition, 46C mouse ESCs cultured on two different microcarriers were compared by Fernandes et al. (24). The maximal fold increase in the population of these mouse ESCs cultured on macroporous CultiSpher-S was higher than that of cells cultured on Cytodex 3, but the maximum specific growth rate was lower when cells were grown in serum-containing conditions (24).
It is possible to establish an economical and c-GMP-compliant microcarrier system to produce human fibroblasts to support clinical-grade human ESC culture. Phillips et al. recently demonstrated the efficient expansion of clinical-grade human fibroblasts on polystyrene-based cationic trimethyl ammonium-coated microcarriers (Hillex II, SoloHill Engineering, Inc.) using c-GMP reagents (64). Notably, while retaining pluripotency, the extended self-renewal and expansion (for more than five passages) of human ESCs were supported by microcarrier-expanded fibroblasts and their conditioned medium. The feeder-free microcarrier system can be used to expand human ESCs as well. Oh et al. demonstrated that Matrigel-coated cellulose microcarriers allow routine passaging and stable propagation of human ESCs without differentiation and maintenance of pluripotent markers such as oct-4, SSEA4, and TRA-1-60 (59). Recently, Fernandes et al. established a feeder- and Matrigel-free microcarrier system for growth of human ESCs with continuous agitation (25). This report describes a promising scale-up system for expansion of human ESCs for use in clinical therapy and research. In addition, Lock et al. established a Matrigel-coated HySphere™ (collagen-coated microcarriers)-based microcarrier bioreactor for generating therapeutically useful quantities of endoderm progeny including pancreatic islet cells and liver cells that were differentiated from human ESCs (45).
Moreover, iPSCs derived from several differentiated cell types via ectopic expression of transcription factors such as Oct4, Sox2, Klf4, and C-Myc have been developed over the past 3 years (84). Generally, these cells exhibit a normal karyotype, are similar to ESCs, and maintain the potential to differentiate into lineages of all three germ layers (2). Recently, mouse iPSCs were used in an animal model of Parkinson's disease (94). Although there is no information regarding iPSCs cultured in the microcarrier system, ex vivo expansion of iPSCs using the microcarrier system for autotransplantation may be possible.
Future Directions and Conclusion
Microcarrier technology can be used to produce large numbers of functional cells that maintain desired properties. Based on the continuing study of biodegradable microcarriers and continuous bioprocesses such as bead-to-bead transfer, further scale-up to produce cells for cell therapy or tissue engineering is possible. Currently, information from in vivo studies is limited and has mostly been focused on adult MSC-seeded microcarriers for regenerative therapy of cartilage or bone defects. Although the in vivo use of ESC-seeded or iPSC-seeded microcarriers has not yet been described, these studies are likely imminent. In conclusion, the microcarrier culture system has great potential as a powerful tool for efficient ex vivo stem cell expansion for future autotransplantation approaches.
Footnotes
Acknowledgment
This work was supported by grants from (GX 99-SC-03-CM) the Gwo Xi Stem Cell Applied Technology, Co., Hsinchu, Taiwan, ROC.
