Abstract
The isolation of cells with stem-like properties from prostate tumors suggests the presence of a cancer stem cell (CSC) population, which may account for the initiation, progression, and metastasis as well as drug resistance of the disease. We hypothesized that containing, or at least immobilizing, the CSCs in a nano-self-assembling material might help prevent prostate tumor progression or metastasis. CSCs were plated in three conditions: 1) placed in 1% concentration self-assembled peptide (SAP) preequilibrate with culture medium; 2) placed in 3% concentration SAP preequilibrate with culture medium; and 3) in nonadherent culture. All were grown for 14 days, after which the cells in both 1% and 3% concentrations were washed out of the SAP and grown for an additional 14 days. Here we report that CSCs from prostate cancer cell lines remained quiescent for more than 28 days when embedded in SAP. When the prostate CSCs were embedded in 1% and 3% SAP, most of the CSCs remained single cells 14 days after plating in a nonadherent plate; no prostaspheres could be detected 14 days after plating, suggesting that self-renewal was significantly suppressed. In the controls, prostate CSCs began to divide 1 day after plating in a nonadherent plate and prostaspheres were visible at day 10, indicating the active self-renewal property of the prostate CSCs. Our findings suggest that SAP can completely inhibit a prostate CSC from self-renewal while preserving its viability and CSC property. Therefore, SAP may be an effective nanomaterial for inhibiting cancer progression and metastasis to stop the progression during treatment and removal.
Introduction
Prostate cancer (PCa) is responsible for the largest number of cancer deaths, with the exception of lung cancer. In the early stage, where the tumor is still localized, the disease can be cured by surgery or radioprostatectomy. However, due to the slow growing nature of the tumor, many PCa patients have already developed metastatic disease upon diagnosis and will inevitably enter the hormone refractory stage after hormone ablation therapy. There is currently no cure for hormone refractory prostate cancer (HRPC). The most effective treatment regime for HRPC patients, docetaxel-based chemotherapy, can only improve the median survival time for 3 months (12,14). Therefore, effective treatment strategies against metastatic HRPC are urgently needed.
The reason that current therapies for metastatic HRPC fail is not completely understood. However, the isolation of cancer cells with stem-like characteristics provides solid evidence that prostate cancer stem cells (CSCs) may exist within the tumor. As in other types of cancer, prostate CSCs may account for the treatment failure; increasing evidence shows that current therapies are only successful in targeting the more differentiated tumor cells, sparing the putative cancer stem/progenitor cells (11). Like normal stem cells, CSCs are thought to be quiescent compared to mature cancer cells (6,7,13). This property makes CSCs resistance to chemotherapeutic drugs, which mainly target the actively replicating cells. The expression of multiple drug resistant gene (MDR1) (1) and ABC transporter (15) also helps to protect the CSCs from cytotoxic drugs. In addition, prostate CSCs do not express androgen receptor; thus, they do not respond to hormone ablation in the same way as mature tumor cells. Therefore, elimination of the bulk of frequently replicating tumor cells, as well as the rare subset of slow dividing stem-like cells that are responsible for tumor regeneration, may represent a better therapeutic strategy in the treatment of PCa.
Due to their ability to self-renew and differentiate, prostate CSCs are capable of regenerating the heterogeneous tumor population (including both androgen-dependent and −independent cells) and it is believed that CSCs may be responsible for the metastatic growth of primary prostate tumors (9). CSCs isolated from PCa cell lines have been found to be more invasive than the non-CSCs; likewise, PCa cells, which are more invasive, were found to possess stem cell characteristics. These results suggest that prostate CSCs may be the origin of prostate tumor metastasis and may be an ideal target for inhibiting disease metastasis. By limiting replication of CSCs the progression of metastasis can be slowed. If the cells could be prevented from migrating away from the treatment area, it would allow for additional local targeting.
Previously, we demonstrated that by manipulating the cell density and concentration of the self-assembling peptide (SAP) material we were able to control the proliferation, elongation, differentiation, and maturation of cells in vitro. This included the nanoenvironment surrounding PC12 cells, Schwann cells, and neural precursor cells, as well as implants in the brain and spinal cord with and without cells (3). Here we show that prostate CSCs can be placed into stasis for an extended period of time without causing them to differentiate.
Materials and Methods
Preparation of the SAP Solution
The SAP solution was prepared by using RADA16-I dry powder (made in the Ellis-Behnke lab) and mixed in an Eppendorf tube. The solution contained 10 or 30 mg of RADA16-I powder in 1 ml of Milli-Q water (Millipore Corp., Billerica MA), mixed, then sonicated for 30 s and filtered; this produced 1% and 3% SAP, respectively.
Culture Medium Preparation
To prepare medium for spheroid assay, we mixed 50 ml SFM (DMEM-F12 high glucose with 0.25% methyl-cellulose) and added 1 ml b27 per 50 ml SFM (50x stock). The following growth factors were used: basic fibroblast growth factor (bFGF) (Sigma cat. No. F0291), 25 μg dissolved in 1.0 ml of 5 mM Tris (pH 7.6) and sterilized with a 0.22 μm pore size filter, to make 20 μl aliquots (stored at −20°C) and add 10 μl per 50 ml of SFM; epidermal growth factor (EGF) (Sigma cat. No. E4127), 0.1 mg dissolved in 1.0 ml of phosphate-buffered saline containing 0.1% bovine serum albumin (Sigma cat. No. A7511), to make 20 μl aliquots (stored at −20°C) and add 10 μl per 50 ml of SFM; insulin 10 mg/ ml and add 20 μl per 50 ml.
Cell Preparation
To remove prostate cancer cells (DU145, ATCC, Rockville, MD) from adherent culture, trypsin was used to liberate and then resuspend 400 cells per 1 μl plated in each well (10). Before plating the cells, the plate was coated with polyhema (Sigma, St. Louis, MO).
SAP Preparation
Untreated SAP has a very low pH (about 3—4). We neutralized the SAP in culture medium before implanting cells in the material. Briefly, for both 1% and 3% SAP were placed in a dry dish and then 500 μl of culture medium was added at the edge of the dish. This is to ensure that the culture medium will not wash the SAP away. The self-assembly occurred as soon as the material made contact with the medium. The medium was changed once at 30 min, and then removed. The 400 cells were placed either in the assembled SAP or in a control well. Culture medium was then added at the edge of the dish in both SAP cases, and topped up every 2 days to compensate for evaporation. On day 14 the cells were vigorously washed out of the SAP using culture medium. In the 1% case, group 1 (n = 4), the cells and SAP were moved to a new well and contents were washed multiple times to separate them from the SAP. In the 3% case, group 2 (n = 4), the wells were also vigorously washed to remove the cells from the SAP. However, in both the 1% and 3% SAP concentration not all of the cells were liberated. The culture continued to grow for 28 days at which point the wells were fixed with 4% paraformaldahyde.
Imaging
The cultures were observed with an inverted micro-scope and pictures were taken.
Results
Metastatic prostate cancer cells (DU145), when placed in 1% concentration SAP (group 1) and 3% concentration SAP (group 2), saw delayed division and formation of colonies for the duration of the cells' encapsulation in the material. In the controls all of the cells that were floating had formed colonies by the end of 14 days (Fig. 1). However, if the cells were completely surrounded by the material no colonies were seen. In some cases, if part of the cell was exposed, a colony of reduced size was seen. We were able to suspend the proliferation of cells out to 28 days, and then the experiment was stopped.
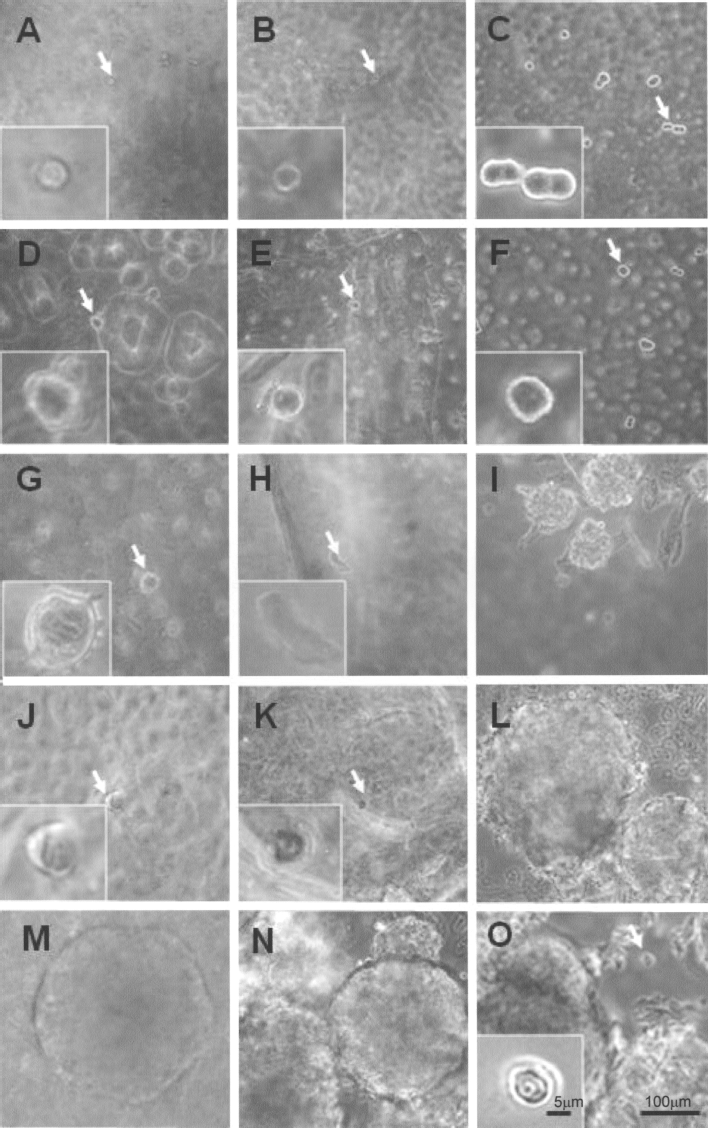
Prostate cancer stem cells in 1% SAP (A), 3% SAP (B), and control (C) on day 1. The insets are magnified images of the cells indicated by the arrows. Cells in 1% SAP (D), 3% SAP (E), and control (F), respectively, at day 4. Note the size of the cells is slightly larger in the 1% and 3% conditions, when compared to day 1 in the insets. Cells in 1% SAP (G), 3% SAP (H), and control (I) at day 10. In (I), the prostaspheres are clearly visible, while the two treatment conditions (G, H) are still single cells. Cells (J), (K), and (L) at day 22, with cells that are still in the SAP (insets), while the control (L) has clear prostasphere formation. Day 22 washed out: (M) and (N) are also at day 22, but are cells that have been washed out of the SAP. (M) cells in 1% SAP and washed out at day 14; (N) cells were washed out of 3%, also at day 14. Both have clear prostasphere formation. Day 28 (O) in 3% SAP with a prostasphere and a single cell that that has remained in the SAP (arrow), still the same size as day 1 (inset). Scale bars: 100 μm and 5 μm (insets).
1% SAP Concentration
In group 1 (n = 4), the cells in the 1% SAP remained quiescent (Fig. 1). When the cells were washed out of the material the colony formation proceeded in a normal fashion. After replating the washed out 1% SAP group of cells into the nonadherent plate, prostaspheres were visible after 8 days (Fig. 1M), with colonization visible beginning at 10 days post-washout, confirming that CSC-embedded cells were viable and capable of self-renewal. However, the remaining cells in each of the four wells of the 1% SAP continued to be quiescent for up to 28 days, showing no evidence of division and colony formation. In the majority of cells that were washed free of the 1% material, colony formation proceeded (Fig. 1M).
3% SAP Concentration
Group 2 (n = 4) results were similar to group 1: in the 3% SAP no colony formed while cells were resident in the material (Fig. 1B, E). When the cells were washed free of the material, the percentage of cells that were retained by the SAP was much greater in the 3% material than in the 1%. The cells that remained in the material showed no cell division or colony formation at 28 days. In the cells that were washed out of the SAP, prostaspheres were evident in 8 days post-washout and colony formation proceeded, similar in size and growth rate as the controls (Fig. 1N). Figure 1O shows a colony that was formed when the cell was washed out of the 3% material. Next to the colony there is a cell that is still encased in the material and has not divided (Fig. 1O, inset).
Controls
In the controls (n = 2), the colony formation began as early as day 1 (Fig. 1C) and proceeded such that at the end of 10 days all of the cells that were floating had formed colonies (Fig. 1L). The colony size was similar in the experimental groups at the end of 28 days for all of the floating cells.
Discussion
The use of this material stops the formation of colonies in cancer stem cells, appearing to mimic extracellular matrix (ECM) and cell—cell contact. Unlike Matrigel, the material does not contain any other additives; the only difference between the groups was the concentration of the material.
Stops Cell Division
In each group, cells placed in the SAP stopped both the division of cancer cells in vitro and subsequent colony formation. The implication of this finding is that colonization of stem cells could be delayed in order to stop or slow the metastasis of cancer cells before or during a surgical procedure or treatment.
Cells Remain Alive and in Stasis
The nanomaterials appear to mimic cell—cell interaction, thus causing the cell to go into stasis, stop dividing, and possibly inhibit tumor progression and metastasis by suppressing the self-renewing ability of the prostate CSCs.
Stasis Does Not Change the Phenotype of the Cell
Once the cells are liberated from the material they will grow and multiply, similar to the control cells when first placed into the culture medium.
Cancer Stem Cells Unaltered
The ability to form spheroids under nonadherent culture condition is one of the characteristics of cancer stem cells. Cells that are liberated from the material not only can proliferate, but can also form spheroids again in floating culture, suggesting that the stem cell property of the prostate cancer cells was preserved by the self-assembled nanomaterials.
Conclusions
The contribution of different structures, and how they affect the spread of cancer cells, has just begun to be explored. We know that some types of cancers will not invade certain types of tissue, whereas other cell types invade all tissues. This “invasion mechanism” may be an avenue to stop metastasis; and it may be as simple as the physical interaction of the cells with the local environment.
We have shown that the colony formation of cancer stem cells can be delayed by using a nanostructured material to mimic contact inhibition. This could be a first step towards a type of treatment to stop, or slow, metastasis and allow for better treatment with chemotherapeutic agents.
One area that can be a problem for the spread of cancer is the inadvertent spread of cancer cells through clinical processes (2,5,8), often referred to as malignant seeding. Tumors that are biopsied or otherwise “interfered with” have a higher incidence of metastasis than tumors that were removed in an untouched block with wide margins and good tumor hygiene. Perhaps this nanomaterial could be used to stop the spread and limit the growth of metastatic cells by tricking them into thinking they are next to other cancer cells. This would potentially slow, or stop, the signal that triggers the spread of cancer cells because the material mimics the ECM of the tumor environment, tricking metastasis, thus slowing the tumor growth and allowing for a more thorough and effective treatment.
By injecting the material directly into the tumor it might be possible to stop the spread of metastatic cells before or during resection. In addition, by loading the SAP material with chemotherapeutic agents the efficacy of the localized treatment could potentially be increased due to the increased contact time.
As we have previously shown in the brain (4), SAP allows the regeneration of the central nervous system (CNS) tissue. There are parallels between CNS regeneration and treating cancer: both require the careful manipulation of cells and the ability to divide and migrate. The goal of treating cancer is to reduce the ability of these cells to divide and migrate to other tissues. CNS regenerative therapies try to coax the cells to regrow; in cancer that same type of regeneration is considered aberrant (9). Perhaps the marriage of cancer and neural regeneration will yield many new possibilities for the control and management of metastatic cancer.
Footnotes
Acknowledgment
This work was supported by a grant from the University of Hong Kong Seed Fund Programme for Applied Research.
