Abstract
Cell transplantation therapies for central nervous system (CNS) deficits such as spinal cord injury (SCI) have been shown to be effective in several animal models. One cell type that has been transplanted is neural precursor cells (NPCs), for which there are several possible sources. We have studied NPCs derived from human embryonic stem cells (hESCs) and human fetal CNS tissue (hfNPCs), cultured as neurospheres, and the expression of pluripotency and neural genes during neural induction and in vitro differentiation. mRNA for the pluripotency markers Nanog, Oct-4, Gdf3, and DNMT3b were downregulated during neural differentiation of hESCs. mRNA for these markers was found in nonpluripotent hfNPC at higher levels compared to hESC-NPCs. However, Oct-4 protein was found in hESC-NPCs after 8 weeks of culture, but not in hfNPCs. Similarly, SSEA-4 and CD326 were only found in hESC-NPCs. NPCs from both sources differentiated as expected to cells with typical features of neurons and astrocytes. The expressions of neuronal markers in hESC-NPCs were affected by the composition of cell culture medium, while this did not affect hfNPCs. Transplantation of hESC-NPC or hfNPC neurospheres into immunodeficient mouse testis or subcutaneous tissue did not result in tumor formation. In contrast, typical teratomas appeared in all animals after transplantation of hESC-NPCs to injured or noninjured spinal cords of immunodeficient rats. Our data show that transplantation to the subcutaneous tissue or the testes of immunodeficient mice is not a reliable method for evaluation of the tumor risk of remaining pluripotent cells in grafts.
Keywords
Introduction
Central nervous system (CNS) injuries often lead to severe functional deficits with no curative treatment available. Much effort is spent on developing therapies for CNS injuries using cell replacement strategies (13, 31). Stem cell therapy could be beneficial by 1) replacing damaged or dead cells, 2) reconnecting injured neurons, 3) secreting neurotrophic factors to support survival and regeneration of remaining cells, 4) remyelinating damaged axons. Neural precursor cells (NPCs) for transplantation therapies can be derived from embryonic stem cells, embryonic or fetal CNS, or adult CNS tissue, and several methods have been developed for neural differentiation and long-term culture of these cells (9,39).
Due to their pluripotency and potential to differentiate into any cell type, human embryonic stem cells (hESCs) are a unique source for various types of cell therapies, including treatment of CNS insults and neurodegenerative diseases (13,23,26). Human ESCs are usually derived from the inner cell mass of blastocyst stage embryos. They can be maintained in pluripotent state in culture and differentiated into tissues of all three germ layers (28,44). Following established differentiation protocols, hESCs can be differentiated into neuroectodermal cells, and into mature neuronal and glial cells (8,47). The ability of these neural cells to regenerate damaged areas of the CNS has been studied intensively. For example, hESC-derived oligodendrocyte precursors cells have been shown to remyelinate axons in spinal cord injury (SCI) animal model (23). Still, further optimization is needed for controlled differentiation and purification of hESC-derived neural cell populations for transplantation purposes (15,34).
A crucial and largely unresolved problem is the necessary elimination of remaining pluripotent cells, because even a few pluripotent cells may be sufficient to give rise to tumors (16). Efficient protocols for phenotype induction, as well as reliable markers of pluripotency to be used in elimination of undifferentiated hESCs, are needed in order to achieve clinically acceptable methods for safe cell therapy. A number of markers for pluripotent cells such as Nanog, Oct-4, DNMT3b, SSEA-4, Tra1-81, and CD326 (1,3,41) have been suggested, but their specificity and selectivity need to be further evaluated.
Human NPCs can also be isolated from the human embryonic or fetal CNS, or the adult CNS, and expanded in vitro for prolonged periods (36,45). Although these cells may have more limited differentiation potential compared to hESC-derived neural precursors, they can give rise to neurons, oligodendrocytes, and astrocytes in vitro (33,45). NPCs derived from the human embryonic and fetal CNS (hfNPCs) have been transplanted in SCI animals (21). These cells survive for months after transplantation to injured rat spinal cord, and differentiate into neurons and astrocytes (2). Importantly, whatever the cell type used for future clinical applications, large amounts of cells have to be produced under Good Manufacturing Practice (GMP) quality standards, which can be accomplished by modifying previously published protocols (7,36,42). However, the main concern of hESC-derived cells in cell therapy is the risk of tumor formation due to remaining pluripotent cells.
Here, we studied the differences between hESC-NPCs and hfNPCs cultured under equivalent conditions. We compared the expression of pluripotency, neural and glial markers, and the propensity for tumor formation after transplantation to immunodeficient animals. Our results show that there are important differences between the two cell types with respect to expression of pluripotency markers, that these differences are associated with the presence of pluripotent cells among hESC-NPCs, but that this cannot be reliably assessed in standard teratoma tests.
Materials and Methods
Human ESC Cultures
Human ESC lines (HS181 passage 65, HS360 passage 12) were derived from the inner cell mass of supernumerary blastocyst-stage embryos at the Karolinska Institute, Karolinska University Hospital Huddinge, Sweden. Approval for these procedures was obtained from the Regional Ethics Vetting Board in Stockholm. The ethics committee of the Hospital District of Pirkanmaa, Finland (Hovatta, R05051 and Skottman, R05116) has given approval to perform research utilizing the hESC lines derived at the Karolinska Institute. Human ESCs were cultured as previously described (37). Briefly, hESCs were cultured on top of mitotically inactivated human foreskin fibroblasts (CRL-2429, ATCC, Manassas, CA) in hESC medium containing knockout Dulbecco's modified Eagle's medium (DMEM) (Gibco Invitrogen, Carlsbad, CA), 20% knockout-SR (Gibco Invitrogen), 2 mM GlutaMax (Gibco Invitrogen), 0.1 mM minimal essential medium nonessential amino acids (Cambrex Bio Science, Karlskoga, Sweden), 0.1 mM β-mercaptoethanol (Gibco Invitrogen), 50 U/ml penicillin/streptomycin (Cambrex Bio Science), and 8 ng/ml human basic fibroblast growth factor (bFGF, R&D Systems, Minneapolis, MN). Human ESC colonies were passaged by mechanical splitting at 5–7-day intervals and replated on fresh fibroblasts. Karyotypic normality was confirmed after every 25–30 passages.
Neural Induction of hESCs in Suspension Culture
HS181 and HS360 colonies were mechanically dissected and differentiated into neural cells in suspension culture (29,41). The neural differentiation was performed in two different media: neural differentiation medium (NDM) and neural stem cell medium (NSM). NDM contained DMEM/F-12 and neurobasal medium (1:1) supplemented with 1 × B27, 1 × N2, 2 mM GlutaMax (Gibco Invitrogen), 25 U/ml penicillin/streptomycin (Cambrex Bio Science), and 20 ng/ml bFGF. The NSM contained DMEM/F-12 supplemented with 1 × N2, 2 mM GlutaMax (Gibco Invitrogen), 0.6% glucose, 25 U/ml penicillin/streptomycin supplemented (Cambrex Bio Science) with 20 ng/ml human epithelial growth factor (EGF, R&D Systems), 20 ng/ml bFGF (R&D Systems), and 10 ng/ml human ciliary neurotrophic factor (CNTF, R&D Systems). The neurospheres were mechanically split once a week and cultured up to 20 weeks. All cultures were maintained in a humidified atmosphere at 37°C and 5% CO2. Unless otherwise specified, analyses of hESC-NPCs were carried out in intact neurospheres.
Fetal Neural Precursor Cells
Cultures of hfNPCs derived from human first trimester CNS tissue (6–10 weeks of gestation) were established as previously described (2,33). The procedure was approved by the Regional Ethics Vetting Board, Stockholm, Sweden. Briefly, fetal tissue was retrieved from clinical first trimester routine abortions with informed consent by the woman undergoing termination of pregnancy. Identified CNS tissue dissected from the spinal cord and forebrain was homogenized with a glass-Teflon homogenizer and cultured at 100,000–200,000 cells/ml in NSM or NDM. The cultures of free-floating neurospheres were passaged every 7–14 days by enzymatical dissociation with TrypLE Express (Invitrogen) and gentle trituration, and fresh medium added twice a week. All cultures were maintained in a humidified atmosphere at 37°C and 5% CO2. Unless otherwise specified, analyses of hfNPCs were carried out in intact neurospheres.
RNA Extraction and cDNA Synthesis
Samples were collected from hESC-NPCs and hfNPCs. RNA was extracted using RNeasy® Micro kit (Qiagen, Hilden, Germany). A total of 50 ng of RNA was used for cDNA synthesis (Sensiscript RT kit, Qiagen) according to the manufacturer's instructions.
PCR
Each PCR reaction contained 1 μl of cDNA, 0.25 μM of forward and reverse primers, 1× Taq-buffer (–MgCl, +KCl) (Fermentas, Leon-Rot, Germany), 2.5 mM dNTP (Fermentas), 25 mM MgCl2 (Fermentas), dH2O, and 0.6 U Taq-DNA-polymerase enzyme (Fermentas). The PCR program included: denaturation in 95°C for 3 min followed with 35 cycles of 95°C for 30 s, 55°C for 30 s, 72°C for 1 min, and final extension for 5 min at 72°C. The following primers were used for a) pluripotent cells: Nanog, Oct-4, DNMT3b, Activin A receptor; b) mesodermal cell lineages: Brachyury, endodermal cell lineage: alpha-fetoprotein (AFP); c) NPCs: Sox2, nestin, Pax-6, Musashi, Mash1, neural cell adhesion molecule (NCAM); d) radial glial cells: brain lipid binding protein (BLBP); e) neuronal cells: Doublecortin, MAP-2, β-tubulin III; f) astrocytes: glial fibrillary acidic protein (GFAP); g) oligodendrocytes: Olig1, Olig2; h) antiapoptosis: Birc5; and i) housekeeping gene: GAPDH. See Table 1 for primer sequences. Time points for PCR sample collections were 2, 4, 6, 8, and 12 weeks in culture.
Primer Sequences

Quantitative RT-PCR
Quantitative RT-PCR (qPCR) was performed with FastStart Universal SYBR Green Master Mix (ROX, Roche) for the genes Oct-4, DNMT3b, Gdf3, GAPDH and with a TaqMan assay (Applied Biosystems, Foster City, USA) for Nanog. Primers for SYBR Green chemistry were designed specifically for qPCR and ordered from Thermo Electron GmbH (Germany), and primers and probes for Nanog were custom designed by Applied Biosystems. Primers were designed to span exon–exon boundaries and primer sequence specificity was confirmed with NCBI BLAST to eliminate the risk of genomic contamination. Primers were analyzed with PCR; amplified products were run on agarose gels, cut out, and sequenced. qPCR was done using the 7500 Fast Real-Time PCR System (Applied Biosystems) with the following profile: 1 cycle of 95°C for 10 min, 40 cycles of alternating 95°C for 15 s, and 60°C for 30 s followed by a melting curve analysis for specificity control. Quantification was done using the ΔΔCt-method as described previously (27). Undifferentiated hESCs (line HS181) were used as a reference sample and GAPDH as reference gene. The Ct values were calculated using the 7500 Fast System software, version 1.3.1. Data analysis was done with Microsoft Excel.
Immunohistochemistry
Neurospheres derived from hESCs and human fetal CNS tissue were collected at different time points, fixed in 4% paraformaldehyde (PFA), treated with 30% sucrose over night at 4°C, mounted in Tissue-Tek mounting medium (Sakura Finetek, Zoeterwoude Netherlands), and frozen on dry ice. The neurospheres were cryostat sectioned at a thickness of 5 μm. Tissue sections were blocked with 1.5% goat/donkey serum in PBS at 30 min in room temperature (RT), and primary antibodies were diluted in 0.3% Triton X-100 in PBS and incubated overnight at 4°C. After rinsing in PBS the sections were incubated with secondary antibody in 0.3% Triton X-100 PBS for 1 h at RT. Primary antibodies used were towards β-tubulin type III (1:800, Sigma), BLPB (1:500, Chemicon, Temecula, CA), GFAP (1:500, DAKO), MAP-2 (1:50, Chemicon), nestin (1:200 Chemicon). Secondary antibodies used were Cy3-conjugated AffiniPure goat anti-mouse IgG (1:2400, Jackson Immuno Research Laboratories, Baltimore, MD, USA), Alexa Fluor 488-conjugated goat anti-mouse IgG, and Alexa Fluor 488-conjugated goat anti-rabbit IgG (1:1200, both from Molecular Probes, Leiden, the Netherlands). For nuclear counterstaining Hoechst 33342 (1:200, Sigma) was used. For negative control, samples were incubated with secondary antibody only. At least five sections/sample were analyzed. Analysis was performed with hESC-NPCs cultured in NSM and NDM (HS360 and HS181), and hfNPCs cultured in NSM (spinal cord: 566sc and 569sc, forebrain: 575for and 558for) at time points of 2, 4, 8, 12, and 20 weeks. Samples were evaluated semiquantitatively using the fluorescence microscope (Zeiss, Axiophot).
Flow Cytometry
Human ESC- and fetal-derived neurospheres were dissociated into single cells with TrypLe Express (Invitrogen) and labeled with antibodies diluted according to the manufacturer's instructions: mouse IgM anti-A2B5 (R&D Systems), mouse IgG1 anti-CD133-PE (Miltenyi Biotech, Bisley, Surrey, UK), mouse IgM CD15-PerCP (Beckman Coulter, Fullerton, CA), mouse IgM anti-PSA-NCAM (Chemicon), mouse IgG1 anti-CD44-PE (BD Pharmingen, San Diego, CA), mouse IgG anti-CD326 (EP-CAM, Chemicon), anti-CD56-PE-Cy7™ (NCAM, BD Pharmingen, San Diego, CA), and mouse IgG anti-SSEA4 (Millipore, St Charles, MO). Unconjugated antibodies were stained with the following secondary antibodies: FITC-conjugated goat anti-mouse IgM (Jackson ImmunoResearch Laboratories) and Alexa Fluor 488-conjugated goat anti-mouse IgG (Invitrogen). Cells were incubated with primary antibodies for 30 min in the dark on ice and washed once with PBS followed by incubation with the appropriate secondary antibody for 30 min in the dark on ice and washed twice with PBS. Flow cytometry was run on a FACsort flow cytometer (Beckton Dickinson) using the software Cell Quest. Isotype controls were used to determine nonspecific staining and gates were set using directly conjugated isotype controls and/or unconjugated isotype controls and the corresponding secondary antibodies as negative control. Human ESC-NPCs and hfNPCs were cultured in vitro for 8 weeks prior to flow cytometry analysis. For analysis of hfNPCs, cells from five different cases were used and the average presented.
Teratoma Formation Analysis
To study teratoma formation, 10–12 intact neurospheres (in total ~100,000 cells) derived from hESCs and human fetal CNS were transplanted into the right testis of severe combined immunodeficiency (SCID) mice (n = 10), as previously described (19,20). Equivalent subcutaneous transplantations of 10–12 neurospheres (~100,000 cells) in the left groin were also performed in the same animals. Undifferentiated hESC of the same cell lines (100,000 cells/injection) were used as positive controls. After the cell injections, the development of tumors in the testes and subcutis in transplanted animals were followed by manual palpation during 12 weeks. Animals were sacrificed by a lethal dose of intravenous barbiturates before transcardiac perfusion with 4% PFA in 0.1 M PBS. Testes were dissected out and a 1-cm2 piece of the skin and superficial layer of underlying skeletal muscle at the location of the subcutaneous transplantation was cut out, the tissue was postfixed for 4 h in PFA, and then transferred to 10% sucrose for at least 24 h. Sections (10 μm) were cut on a cryostat (Micron) and stained with hematoxylin-eosin (Sigma) for histological analysis. Immunohistochemistry was performed as described above. Human cells were detected using human nuclear marker HuNu (1:250, Chemicon) and hESC-NPCs with β-tubulin III (1:800, Sigma).
Spinal Cord Injury In Vivo Model
A total of 24 adult immunodeficient female rats (170&–200 g, HsdHan:RNU-rnu, Harlan, UK) were included in the study. After a laminectomy of thoracic vertebra 9, a spinal cord contusion injury was produced using the IH Spinal Cord Impactor (Precision Systems & Instrumentation) set at a force of 150 kilodynes, with no dwell time. The lesioned spinal cords were covered with one layer of meningeal substitute (Lyoplant, B/Braun Aesculap) prior to suture of the wound as previously described (2). Eight days later the animals were reanaes-thetized and 10–12 hESC-derived neurospheres (cultured for 8 weeks to allow neural induction) selected by size to contain a total of approximately 100,000 cells were transplanted to the lesion area. Three groups of injured rats (6 animals/group) were included and transplanted with: a) hESC-NPC cultured in NDM (HS360), b) hESC-NPC cultured in NSM (HS360), c) hESC-NPC cultured in NDM (HS181). One group of rats with a laminectomy but without contusion injury (6 animals/group = sham rats) was injected with hESC-NPC cultured in NDM (HS360). Animals were followed up to 12 weeks after injury and testing of hind limb motor function was performed 1 day before injury and 1, 3, 6, 10, and 12 weeks after injury. Hind limb function was assessed using the Basso, Beattie, and Bresnahan (BBB) motor performance scale (4), with 21 as the score for normal hind limb function, and 0 for complete paralysis. Functional testing was performed by an experienced investigator blinded to the experimental design. After the motor assessment at 12 weeks, the rats were sacrificed, and perfusion fixed with 400 ml of 4% PFA in phosphate buffer, pH 7.4. The dissected spinal cords were postfixed in 4% PFA for 90 min and thereafter rinsed and kept in 30% sucrose until they were cut in 10-μm sections and processed for histological analysis as described above.
Results
Gene Expression Profiles of hESC-NPCs and hfNPCs
Gene expression analysis revealed that the expression of pluripotency markers Oct-4, Nanog, and DNMT3b was downregulated already 2 weeks after initiation of neural induction in hESC-NPCs while quite surprisingly these markers were found at comparable levels in hfNPCs during the 12 week follow-up time (Fig. 1). Expression of neural markers sox2, nestin, Pax-6, double-cortin, Musashi, Mash1, β-tubulin III, MAP-2, and NCAM increased in hESC-NPCs during differentiation while hfNPCs expressed these markers constantly during 2–12 weeks follow-up. The expression levels of the astrocyte marker GFAP increased in hESC-NPCs and forebrain-derived hfNPCs during later time points of culture, while GFAP expression in spinal cord-derived hfNPCs remained constant from 2 weeks (Fig. 1). In hESC-NPCs Olig1 was expressed weakly, and Olig2 expression varied, while both of them were expressed at constant levels in hfNPCs. In addition, the expression of antiapoptosis marker BIRC5 was detected in varying levels in both cell types (Fig. 1).

Gene expression profiles of hESC-NPCs and hfNPCs. (A) Representative images of agarose gels of RT-PCR products from hESC-NPC derived from hESC-lines HS181 and HS360. (B) Representative images of agarose gels of RT-PCR products from hfNPC derived from spinal cord (SC) or forebrain (Fbr). Cells were harvested at 2, 4, 6, 8, and 12 weeks of in vitro culture. NPCs were cultured in NSM or NDM.
We also compared cells cultured in NSM and NDM with respect to the gene expression levels. hESC-NPCs cultured in NDM expressed neuronal markers MAP-2 and NCAM, and radial glial marker BLPB, already after 2 weeks of culturing, whereas hESC-NPCs cultured with NSM expressed these markers at lower levels and after longer time in culture. In contrast, hfNPCs expressed all of these markers at similar levels regardless of medium used (NSM vs. NDM).
Quantitative RT-PCR Analysis of Pluripotency Genes
The results of the semiquantitative RT-PCR with pluripotency genes were confirmed using quantitative PCR. The mRNA levels for Nanog, Oct-4, DNMT3b, and Gdf3 were significantly lower in hESC-NPCs after 2 weeks of neural induction compared to undifferentiated hESCs, and decreased during further neural differentiation (Fig. 2A). In hfNPCs the levels of Nanog, DNMT3b, and Gdf3 mRNAs were higher at 2 weeks after the initial derivation, compared to hESC-derived NPCs (Fig. 2B) and fluctuated during the 12 weeks of study. Two weeks after differentiation, hESC-NPCs cultured with NDM had higher mRNA levels of Nanog and Oct-4 than hESC-NPCs cultured at NSM (Fig. 2A). In hfNPCs, the mRNA levels of the pluripotency markers did not show any consistent differences with respect to the region of origin or cell culture medium used (Fig. 2B).

Quantitative mRNA analysis of Nanog, Oct-4, DNMT3b, and Gdf3. (A) hESC-NPCs (HS181, HS360) at 2, 8, or 20 weeks of in vitro culture. NPCs were cultured with NSM or NDM. The results are presented as relative fold-induction compared to undifferentiated hESCs. ND: mRNA expression was not detected. (B) hfNPCs (SC and Fbr) at 2, 8, 12, or 20 weeks of in vitro culture. NPCs were cultured with NSM or NDM. The results are presented as relative fold-induction compared to undifferentiated hESCs. ND: mRNA expression was not detected.
Protein Expression Analysis
According to the mRNA analysis, expression of pluripotency markers was almost completely suppressed in hESC-NPCs after 8 weeks of neural differentiation. Protein expression in NPCs at this time point was analyzed using flow cytometry and immunocytochemistry. A small proportion of hESC-NPCs expressed the pluripotency markers SSEA4 and CD326 after 8 weeks of differentiation in vitro, while hfNPCs were negative for these proteins. The other early stem/progenitor cell markers CD133 and CD15 (SSEA1) was found in hfNPCs although at lower frequency than hESC-NPCs cultured in NDM. Cells expressing markers for neural stem/progenitor cells, neuronal and glial cells were common among hESC-NPCs cultured in NDM while less so among hfNPCs and hESC-NPCs cultured in NSM (Fig. 3). The most pronounced difference concerned the neuronal marker PSA-NCAM which occurred on twice as many hESC-NPCs as hfNPCs.

Flow cytometry analysis of hESC-NPCs and hfNPCS. Protein expression of SSEA-4, CD326, CD133, CD15, CD44, CD56, PSA-NCAM, and A2B5 in hESC-NPCs cultured with NDM or NSM (HS181 and HS360) and hfNPCs (SC) cultured in NSM for 8 weeks.
Neurospheres were also analyzed with immunohistochemistry. In accordance with flow cytometry (CD326) and PCR (Oct-4) analysis, occasional cells expressing these pluripotency markers were found in hESC-NPCs. No one of these markers could be detected in hfNPCs (Fig. 4). Neuronal markers β-tubulin III and MAP2, glial marker GFAP, and radial glia marker BLBP all increased in sectioned neurospheres of hESC-NPCs during the neural differentiation in NDM. The increase in β-tubulin III did not appear during differentiation in NSM. In hfNPCs the immunoreactivity for these markers remained the same during the study period except for GFAP, which increased in spinal cord-derived hfNPCs, while a progressive decrease was seen in forebrain-derived hfNPCs (data not shown).

Immunocytochemical analysis of hESC-NPCs and hfNPCS. (A) Expression of pluripotency markers. Embryoid bodies (2 DIV) were used as a positive staining control for pluripotency markers Oct-4 (green) and CD326 (red). A proportion of cells in hESC-NPCs, differentiated for 8 weeks, were immunoreactive for Oct-4 and for CD326. hfNPCs did not show any immunoreactivity for these markers. (B) Expression of neural markers. Both hESC-NPCs and hfNPCs were positive for neural markers nestin (red), MAP-2 (green), and GFAP (green). Same magnification in all pictures. Scale bar: 100 μm.
hESC-NPC Transplantation to SCID Mice Testis and Subcutaneous Tissue
Initially, a pilot experiment was performed in which dissociated hESC-NPCs, derived from HS181 and HS360, and differentiated for 8 weeks in vitro, were injected into the testis of 8 SCID mice. As these cells did not give rise to any macroscopical teratomas, we transplanted hESC-NPC neurospheres grown in NDM into the lesioned spinal cord of 10 immunodeficient rats. Unexpectedly, the majority of these rats developed tumors, and histological analysis of a few of these animals verified that the tumors were teratomas.
For the analysis of pluripotent cells and teratoma formation, we reasoned that the lack of teratomas in the first testis transplantation experiment may have been due to the injection of dissociated cells, while intact hESC-NPC neurospheres were transplanted to the spinal cord, according to the procedures we previously used for hfNPCs. We therefore transplanted intact hESC-NPC neurospheres into the testis and the subcutaneous tissue of the groin of a total of 10 immunodeficient SCID mice. However, again the hESC-NPCs did not give rise to any macroscopical tumor formation after 12 weeks of in vivo observation, whereas undifferentiated hESC used as positive control did (data not shown). On microscopic examination, the hESC-NPC testis grafts consisted of homogenous tissue with a histological appearance resembling neuropil. There was no invasive growth into the seminiferous tubuli. No cartilage, muscle fibers, pigmented cells, fat, or glands, typical of the teratomas found after transplanting undifferentiated hESCs, could be seen in the grafts (Fig. 5B). hfNPCs transplanted to SCID mice testes also survived and developed into a tissue with neural appearance (Fig. 5A).
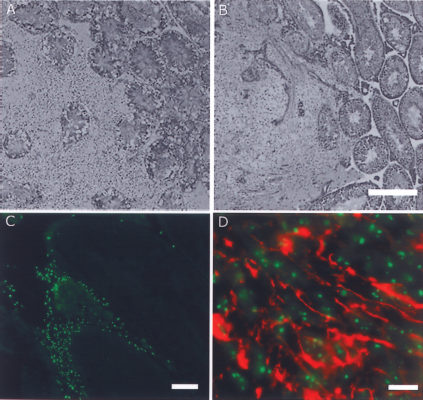
Testis transplantations. (A) Hematoxylin-eosin staining shows the hfNPC graft (left) displacing the normal testicular tissue (right). Scale bar: 500 μm. (B) Hematoxylin-eosin staining shows the hESC-NPC graft (left) displacing the normal testicular tissue (right). Scale bar: 500 μm. (C) HuNu (green) expression in testis confirms the human origin of cells in the graft (hESC-NPCs). Scale bars: 100 μm. (D) Double staining with HuNu (green) and β-tubulin III (red) demonstrates neuronal differentiation of grafted hESC-NPC. Scale bar: 20 μm.
To verify that hESC-NPC neurospheres were indeed successfully transplanted to the mice testes, immunohistochemical evaluation using human-specific antibody (HuNu) was applied to cryosections of the tissue. The specificity of the HuNu antibody allowed an efficient screening of the serially sectioned testes in fluorescence microscope, with reliable detection also of small groups of human cells. In 9 out of 10 testes, a human graft could be identified in the testis parenchyma (Fig. 5c). Some transplanted cells were positive for both HuNu and β-tubulin III, with a neuron-like morphology indicating neuronal maturation of the transplanted hESC-NPCs (Fig. 5d).
hESC-NPC Transplantation to Rat Spinal Cord
In parallel with the testis and subcutis transplantation experiments, we transplanted the same hESC-NPCs as neurospheres into sham or injured spinal cords of immunodeficient rats. In sham rats, the median BBB score was 21 up to 10 weeks postsurgery (Fig. 6A). Experimental groups with an SCI initially had a reduction in the median BBB score from a prelesion score of 21 to 7–12 at 1 week postlesion. The hind limb function thereafter improved up to 6 weeks after injury, reaching BBB scores around 15. However, between 6 and 12 weeks postinjury, hind limb function of all rats gradually declined, including the sham-operated rats transplanted with hESC-NPCs (Fig. 6A).

Outcome of hESC-NPCs to rat model of spinal cord injury. (A) Hind limb motor function after hESC-NPC transplantation to the spinal cord. hESC-NPCs were injected into intact or traumatized spinal cords (Tp) and rats were followed for 12 weeks thereafter. BBB locomotor scores of hind limb motor function show that during first 6 weeks the rats' hind limb function either remained stable or improved. However, thereafter the function gradually was reduced in all the transplanted experimental groups. (B) Macroscopically, expanding tumor masses could be seen in the spinal cords 12 weeks after transplantation (1), while no tumors were seen in the shrunken, non-transplanted injured spinal cord (2).
At postmortem analysis, the spinal cords were swollen and semitranslucent at the site of the injury, suggestive of tumor masses with extensive cyst formation (Fig. 6B). Microscopically, hematoxylin-eosin-stained sections showed the typical appearance of teratomas, dominated by large cystic cavities surrounded by tissue representing all three germ layers (Fig. 7).

Intraspinal teratoma after hESC-NPCs transplanted to the injured rat spinal cord. Bright-field micrographs of hematoxylin-eosin-stained cross sections of the spinal cord. Scale bars: 500 μm. (A) Normal thoraco-lumbar spinal cord of a noninjured rat. (B) In the traumatized spinal cord, the degenerated gray matter is replaced by a large central lesion cavity. (C) In animals transplanted with hESC-NPC, several millimeters of the injured spinal cord consists of expanding teratomas, displaying examples of the different germ layers. In this section, connective tissue and fat cells (short arrow) and typical exocrine gland tissue (long arrow) are seen. The large cystic cavity (arrowhead) is most likely the pathologically widened central canal.
Discussion
In the present study, we have investigated the differences between hESC-derived NPCs and fetal CNS-derived hfNPCs—two types of cells with potential for future clinical use—in terms of neural differentiation, pluripotency, and risk of teratoma formation. Both hESC-NPCs and hfNPCs were cultured at similar culture conditions and their gene and protein expression were compared. Thereafter, the teratoma formation capacity of NPCs was tested in testis and subcutaneous tissue of immunodeficient animals. Finally, we performed a long-term safety study of hESC-NPCs a rat SCI model.
hESC-NPCs and hfNPCs were cultured for prolonged time (over 12 weeks) in neural differentiation medium (NDM) and neural stem cell medium (NSM) and their gene expression profile for pluripotency and neural markers were studied. As expected, we found a rapid downregulation of the pluripotency genes Nanog, Oct-4, DNMT3b, and Gdf3 (25,29,41) in hESC-NPCs compared to undifferentiated hESCs. Surprisingly, hfNPCs expressed mRNA for Nanog, Oct-4, DNMT3b, and Gdf3 at levels that were even higher than in hESC-NPCs, according to quantitative PCR analysis. To eliminate false-positive results, primers for the qPCR analyses of Nanog, Oct-4, DNMT3b, and Gdf3 were designed to span two exons to avoid the risk of contamination with genomic DNA. In addition, the primers used were validated with undifferentiated hESCs. In accordance with our results on hfNPCs, Oct-4 is expressed in nonpluripotent NPCs isolated from adult rhesus macaque brain (10), and Gdf3 has been found in the human cerebral cortex, hippocampus, and cerebellum (17), as well as in other somatic cell types (22). Importantly, in a recent study on dorsal root ganglia-derived neural stem cells, a large number of cells were immunoreactive to Oct-4, as well as to DNMT1 (40). However, we are not aware of any previous reports on expression of Nanog in nonpluripotent cells. Despite the low mRNA levels in hESC-NPCs, we could also detect low protein expression of pluripotency markers Oct-4, SSEA4, and CD326 in these cells. Using a slightly different protocol, Kozubenko and collaborators generated neural precursor cells from hESC which they transplanted as dissociated cells to an ischemic brain injury. Interestingly, according to their flow cytometry data at P5 these cells contained between 2% and 5% cells identified as SSEA-4-, TRA-1-60-, and Nanog-immunoreactive cells, although classified as “negative reaction” by the authors. These P5 cells resulted in teratomas in 5/11 animals while P8 cells did not give rise to a single case. This contrasts to the fraction of cells immunoreactive to the three suggested pluripotency markers, which did not change during this time, and for SSEA-4 increased to 16.5% at P10 (25).
According to our results, in contrast to hESC-NPCs the hfNPCs did not express detectable levels of these proteins. Thus, it seems that even relatively low expression levels of pluripotency mRNAs results in protein translation in hESC-NPCs but not in hfNPCs. It is possible that mRNA for pluripotency markers such as Oct-4 is not translated into proteins in hfNPCs, due to micro RNAs that posttranscriptionally modulate genes, and may be responsible for repressing translation (43). The biological significance of this is not clear, but we hypothesize that the hfNPCs expressing mRNA for these pluripotency genes are the least differentiated precursor cells (i.e., the minor subpopulation of “core” neural stem cells of the fetal neurospheres).
During neural differentiation of hESCs, the expression of the neural markers sox2, nestin, Pax-6, double-cortin, Musashi, Mash1, β-tubulin III, MAP-2, and NCAM as expected increased with time. hfNPCs expressed these markers already soon after establishment as neurospheres, and this expression remained stable during the 12 weeks of culture. This agrees with a previous study showing that hfNPCs have a more pronounced expression of genes such as GAP43 (growth associated protein 43) and NNAT (neuronatin) compared to hESC-NPCs (39). It is important to note, however, that the two cell types studied in this analysis were not cultured under similar conditions, which reduces the accuracy of their comparisons, and also prevents a more conclusive comparison with our data. Here, both NPCs derived from hESCs and human fetal CNS expressed neural markers at protein level after 8 weeks of culture. As we have described previously, expression of neural proteins in hESC-NPC increases over time (41), while their expression is relatively constant in hfNPC s during in vitro culture (33).
When comparing different medium compositions, we found that hESC-NPCs cultured in NDM expressed neuronal markers MAP-2 and NCAM, and radial glial marker BLPB, already after 2 weeks of culturing, whereas these markers appeared later and at lower levels in hESC-NPCs cultured in NSM. Flow cytometry analysis showed that NDM also promoted expression of neural stem cell/precursor markers CD133, CD15 as well as neuronal markers CD56 and PSA-NCAM. In contrast, hfNPCs expressed all of these markers at similar levels regardless of medium used (NSM vs. NDM). Thus, while composition of the medium critically affects the cell fate of hESC-NPCs, the neural commitment of hfNPCs is independent of the cell culture medium used. NDM consists of neurobasal medium with B27 supplement, which are known factors to induce neuronal differentiation (11,29), while NSM contains N2 supplement in DMEM/F12, which are less permissive to neural differentiation (11). Compared to hESC-NPCs cultured in NDM, hfNPCs expressed lower amounts of CD133, CD15, and PSA-NCAM in NSM but higher amounts of CD44. In addition, when fetal CNS tissue is the source of NPCs, the regional differences between forebrain- and spinal cord-derived NPCs can affect the expression levels of neural markers (32,46). Here, the spinal cord-derived NPCs expressed astrocyte marker GFAP constantly and at higher levels compared to forebrain-derived NPCs, as shown previously at the protein level (33). Hence, both the medium composition and the origin of the cells are important factors to take into account when differentiating NPCs either from hESCs or fetal tissue.
Intratesticular injections of cells (38) followed by analysis of teratoma formation are generally considered a reliable assay for identifying the presence of pluripotent cells in cell preparations. Recent studies have shown that there are differences between target organs with regard to the propensity of transplanted hESC to develop teratomas (16), and also between different brain regions (14). In the present study we found even more pronounced differences than previously reported. In repeated experiments with hESC-NPCs transplanted as small neurospheres, a total of 100,000 cells consistently gave rise to teratomas in the rat spinal cord, while we could not detect a single case of tumor growth after intratesticular or subcutaneous transplantations in mice. To ensure that there was sufficient time for the teratomas to develop, the transplanted animals were kept for 12 weeks. During this time, the hind limb motor function of SCI rats recovered to some extent according to the BBB rating, reaching similar levels that are shown in other studies at 6 weeks time point (23). However, during the later part of the 12 weeks follow-up time, functional deterioration and extensive growth of teratomas in spinal cords were seen in 26/26 rats with hESC-NPCs transplants. In contrast, hESC-NPC spheres grafted to testis survived in 9/10 mice and differentiated into neural tissue, with no signs of expansive tumor growth. By using undifferentiated hESC as positive controls, we confirmed that the procedures used were appropriate, as described previously (19). The survival of transplanted hESC-NPCs was also confirmed.
We want to emphasize that we do not claim that cells giving rise to intraspinal tumors are unable to develop into tumors in testis or other locations. The hESC-NPCs may develop into teratomas in testis if sufficient numbers are transplanted. We used no more than 100,000 cells for these grafts, because this is a number of cells that previously have been used in SCI transplantation experiments; 100,000 undifferentiated hESCs were sufficient for generating tumors in the positive controls, as has also been shown previously (24). Intraspinal transplantation of a suspension of 1 million cells or more—typically used in teratoma experiments—is not compatible with the small tissue volume of the rat spinal cord. We conclude that with the number of cells used, there are surprisingly large differences in the permissiveness for tumor formation between the rat spinal cord and SCID mouse testis and subcutis. A consequence of this finding is that there may be similar differences between the human spinal cord and the models commonly used to evaluate tumorigenesis; that is, neural cells derived from pluripotent cells may appear as safe in routine teratoma tests and lead to tumor growth after transplantation to human SCI patients. Whether the spinal cord is a CNS region uniquely permissive to growth of pluripotent cells is to our knowledge not known, because comparisons have only been made between regions of the brain, excluding the spinal cord (14).
There are several examples of protocols for neural differentiation of hESC that apparently eliminate pluripotent cells (30,47) and thus the risk of teratoma growth, also after transplantation to the spinal cord (23). It has been shown that the presence of pluripotent cells and the associated risk of teratoma formation decreases over time under differentiating conditions (5) but it is not a guarantee for complete elimination of all pluripotent cells. We found a large downregulation of pluripotent genes in hESC-NPCs differentiated for 8 weeks before transplantations, but these genes are according to our results not specific for pluripotent cells. The number of pluripotent cells in hESC-derived neurospheres was apparently sufficient for generating teratomas after transplantation to the rat spinal cord, but did not exceed the number needed for growth of teratomas in SCID mouse testis (16).
A factor that may influence the teratoma formation capacity of transplanted cells is the use of intact neurospheres versus dissociated neural cells. Dissociated neural cells survive poorly after transplantation to the rodent CNS (18) while cell death after transplantation of intact neurospheres is low (12). Hence, in transplanted neurospheres pluripotent cells may survive better. In agreement with this, Kishi and colleagues (24) found that pluripotent cells transplanted as cell aggregates more likely survived and generated teratomas than dissociated cells.
Our results illustrate a number of important issues with regard to the use of hESC derivatives for cell therapy, including the use of appropriate protocols for production of differentiated neural populations and the need of cell purification using methods such as magnetic beads (6) or fluorescence-activated cell sorting (35,41).
In summary, this study emphasizes the importance of proper characterization of cell grafts in means of gene and protein expression levels and the importance of using reliable animal models for preclinical transplantation studies to measure the safety and efficacy. First, protein expression of pluripotency genes is a better predictor of tumorigenicity than mRNA, but there are problems with specificity and sensitivity. Second, reliable evaluation of the safety and efficacy of cell therapies should be assessed in the expected target of the clinical application, and include extended observation times. Taken together, many aspects including the graft composition and characterization, transplantation site, and follow-up time are all important factors that influence the reliability of safety transplantation studies. This needs to be taken into account especially in hESC-associated studies.
Footnotes
Acknowledgments
We would like to thank Hanna Koskenaho (Biomed. Lab. Technologist) from skillful assistance with RT-PCR analysis. We would like to thank Virpi Himanen (M.Sc.) and Niina Ikonen (Lab. Technician) for help of stem cell culturing at Regea-Institute for Regenerative Medicine, and Eva-Britt Samuelsson (Lab Technician) for expert NPC cell culturing at Karolinska Institutet. The study was supported by the Academy of Finland, the Alfred Kordelinin Foundation, the Competitive Research Fund of the Pirkanmaa Hospital District, the Finnish Cultural Foundation, the Finnish Funding Agency for Technology and Innovation TEKES, the Orion-Farmos Research Foundation, the Craig Hospital Foundation, the Swedish Research Council, Stiftelsen Stockholms Sjukhem, Knut och Alice Wallenbergs stiftelse, M. Bergvalls Foundation, the Swedish Society of Medicine, and the research funds of Karolinska Institutet.
