Abstract
Despite advances in our understanding and research of induced pluripotent stem cells (iPSCs), their use in clinical practice is still limited due to lack of preclinical experiments. Neural precursors (NPs) derived from a clone of human iPSCs (IMR90) were used to treat a rat spinal cord lesion 1 week after induction. Functional recovery was evaluated using the BBB, beam walking, rotarod, and plantar tests. Lesion morphology, endogenous axonal sprouting, graft survival, and iPSC-NP differentiation were analyzed immunohistochemically. Quantitative polymerase chain reaction (qPCR) was used to evaluate the effect of transplanted iPSC-NPs on endogenous regenerative processes and also to monitor their behavior after transplantation. Human iPSC-NPs robustly survived in the lesion, migrated, and partially filled the lesion cavity during the entire period of observation. Transplanted animals displayed significant motor improvement already from the second week after the transplantation of iPSC-NPs. qPCR revealed the increased expression of human neurotrophins 8 weeks after transplantation. Simultaneously, the white and gray matter were spared in the host tissue. The grafted cells were immunohistochemically positive for doublecortin, MAP2, bIII-tubulin, GFAP, and CNPase 8 weeks after transplantation. Human iPSC-NPs further matured, and 17 weeks after transplantation differentiated toward interneurons, dopaminergic neurons, serotoninergic neurons, and ChAT-positive motoneurons. Human iPSC-NPs possess neurotrophic properties that are associated with significant early functional improvement and the sparing of spinal cord tissue. Their ability to differentiate into tissue-specific neurons leads to the long-term restoration of the lesioned tissue, making the cells a promising candidate for future cell-based therapy of SCI.
Keywords
Introduction
Stem cell-based therapy is a promising approach for the treatment of various central nervous system disorders including spinal cord injury (SCI). Neural stem and progenitor cells (NSCs) have been shown to be particularly useful for transplantation therapy for SCI due to their ability to provide an unlimited source of nerve cells for cell replacement (6,14,46,50), as well as trophic support for endogenous neuroregeneration (15,38,49). Previous studies have reported improved functional recovery after the transplantation of several types of human NSCs, derived from either fetal tissue or from embryonic stem cells (ESCs), into the injured spinal cord of rodents and nonhuman primates (7,18,27). Despite promising preclinical results, the use of human ESC-based therapies is controversial due to ethical concerns, the risk of tumorigenesis, and the need for immunosuppression. Induced pluripotent stem cells (iPSCs) offer new prospects for regenerative medicine. Human iPSCs share many properties with human ESCs, including their stemness profile, self-renewal ability, and differentiation potential (26,48). Moreover, iPSCs have the advantage of being patient specific. Therefore, neural precursors derived from human iPSCs (iPSC-NPs) appear to be an ideal source for transplantation therapy in SCI. However, the efficacy of iPSC-NP transplantation in SCI treatment and the mechanisms responsible for functional improvement remain to be fully elucidated.
Here we examined the therapeutic potential of iPSC-NPs by transplanting them into a rat model of SCI. We used neural precursors derived from a clone of human iPSCs (IMR90) that was established from fetal human lung fibroblasts by the lentiviral transduction of four reprogramming factors (33). We report that transplanted human iPSC-NPs survive, engraft, migrate, differentiate, and communicate with the host tissue after traumatic SCI in rats. We further show that human iPSC-NPs promote axonal regrowth, bridge the lesion cavity, and provide trophic support to the spared axons in the injured tissue without tumor formation. These effects promote the recovery of hindlimb motor function. Our results demonstrate that human iPSC-NPs have a therapeutic potential for SCI comparable to that of NSCs from other sources (1,6,7,13,23,46,50) and thus represent a promising cell source for SCI therapy.
Materials and Methods
Human Induced Pluripotent Stem Cell-Derived Neural Precursors
The human iPSC line was derived from female (IMR90) human fetal lung fibroblasts (ATCC, Manassas, VA, USA) transduced according to a previous study (52) with a lentivirus-mediated combination of octamer-binding transcription factor 4 (OCT4), sex-determing region Y box 2 (SOX2), NANOG, and LIN28 human cDNA. Clone selection, validation of the iPSC line, and derivation of NPs are described in detail in Polentes et al. (33). Briefly, early NPs were produced in low-attachment culture in the presence of Noggin (500 ng/ml) (R&D Systems, Minneapolis, MN, USA), the transforming growth factor-β pathway inhibitor SB 431542 (10 nM) (Sigma-Aldrich, St. Louis, MO, USA), basic fibroblast growth factor (bFGF, 10 μg/ml), and brain-derived neurotrophic factor (BDNF, 20 μg/ml) (both from Pepro Tech, London, UK). Human iPSC-NPs were routinely cultured in tissue culture flasks coated with poly-l-ornithine (0.002% in distilled water) and laminin (10 μg/ml in DMEM:F12), both from Sigma-Aldrich. Growth media comprising DMEM:F12 and Neurobasal medium (1:1), B27 supplement (1:50), N2 supplement (1:100) (Gibco, Life Technologies, Grand Island, NY, USA), l-glutamine (2 mM) (Sigma-Aldrich), penicillin and streptomycin (50 U/ml) (Gibco), fibroblast growth factor (FGF, 10 ng/ml), epidermal growth factor (EGF, 10 ng/ml), and BDNF (20 ng/ml) (PeproTech) was changed three times per week. Prior to transplantation the iPSC-NPs were predifferentiated during 7 days in the same medium except for the omission of FGF and EGF.
Fluorescence-Activated Cell Sorting Analysis
iPSC-NPs were dissociated from monolayers by trypsin (Gibco, Paisley, Scotland) for 1–2 min. After rinsing with PBS (Gibco), the cell suspension (107 cells/ml and no less than 3 × 105 cells per sample, 100 μl) was used for flow cytometric analysis using a Becton Dickinson FACSAria flow cytometer (BD Bioscience, San Diego, CA, USA). Data analysis was performed using BD FACSDiVa software. To analyze the human iPSC-NPs, conjugated antibodies against CD29 APC-Cy7 (20 μl/100 μl; BioLegend, San Diego, CA, USA), CD56 PE or APC (NCAM) (20 μl/100 μl), CD24 FITC (20 μl/100 μl), CD15 FITC (SSEA-1) (20 μl/100 μl), HLA-DQ, DR FITC (20 μl/100 μl), CD271 PE (NGFR) (10 μl/100 μl) (Exbio Antibodies, Vestec u Prahy, Czech Republic), HLA-ABC FITC (20 μl/100 μl), SOX2 APC (5 μl/100 μl), β-III-tubulin (20 μl/100 μl) (BD Pharmingen, San Diego, CA, USA), CD133 PE or APC (10 μl/100 μl), PSA-NCAM PE (10 μl/100 μl), A2B5 APC (10 μl/100 μl) (Miltenyi Biotec, Bergisch Gladbach, Germany), NANOG FITC (20 μl/100 μl), TRA-1-60 PE (5 μl/100 μl), CD184 PE (CXCR4) (20 μl/100 μl), SSEA-4 PE or AF488 (20 μl/100 μl) (eBioscience, San Diego, CA, USA), OCT3/4 APC (10 μl/200 μl) (R&D Systems, Minneapolis, MN, USA) were used, along with unconjugated primary antibodies against nestin (1:400; Abcam, Cambridge, UK) with secondary rat anti-mouse IgG conjugated with FITC (1 μl/100 μl; eBioscience) and NF70 (Abcam) with secondary donkey anti-rabbit IgG conjugated with APC (1:50; Jackson Immunoresearch Laboratories, West Grove, PA, USA). As negative controls, unlabeled cells or mouse IgG1 isotype conjugated with FITC or RPE and IgG2a isotype conjugated with RPE (all 10 μl/100 μl; Dako Cytomation, Glostrup, Denmark) were used, as well as rat anti-mouse IgG conjugated with FITC and donkey anti-rabbit IgG conjugated with APC without the addition of the primary antibody.
The data are expressed as mean ± SEM. Student's t-test was used to evaluate the differences between groups. A value of p < 0.05 was considered statistically significant.
RNA Extraction and Quantitative PCR Analysis of Gene Expression
Total RNA was extracted from rat spinal cord tissue and human iPSC-NPs using the RNeasy Lipid Tissue Mini Kit (Cat. No. 74804) and RNeasy Plus Mini Kit (Cat. No. 74134), both from QIAGEN GmbH (Hilden, Germany), according to the manufacturer's instructions. Spinal cord tissue was dissected from animals of three different groups: intact animals (n = 4), control animals with SCI only (n = 8), and animals grafted with iPSC-NPs (n = 8) sacrificed 8 weeks after transplantation.
The expressions of rat (Rattus norvegicus) target genes brain-derived neurotrophic factor (Bdnf), vascular endothelial growth factor A (Vegfa), nerve growth factor (Ngf), neurotrophin 3 (Nt3) or sortilin (Sort1), fibroblast growth factor 8 (Fgf8) and glial cell-derived neurotrophic factor (Gdnf) and human (Homo sapiens) target genes BDNF, VEGFA, NGF, NT3 (SORT1), FGF8, GDNF, nestin (NES), β III tubulin (TUBB3), microtubule-associated protein τ (MAPT), synaptophysin (SYP), homeobox gene 9 (HB9) or motor neuron and pancreas homeobox 1 (MNX1), ISL LIM homeobox 2 (ISL2), choline acetyltransferase (CHAT), glial fibrillary acidic protein (GFAP), and S100 calcium-binding protein B (S100B) were determined by quantitative real-time reverse transcription qPCR in a 7500 Real Time PCR System (Applied Biosystems, Foster City, CA, USA) using TaqMan Gene Expression Master Mix (catalog No. 4392938) and TaqMan Gene Expression Assays 4331182 (Rn02531967_s1/Bdnf/, Rn01511601_m1/Vegfa/, Rn01533872_m1/Ngf/, Rn01521847_m1/Sort1/, Rn0059 0996_m1/Fgf8/, Rn00569510_m1/Gdnf/, Hs01010223_m1/ BDNF-AS1/, Hs00900055_m1/VEGFA/, Hs00171458_m1/ NGF/, Hs00361760_m1/SORT1/, Hs00171832_m1/FGF8/, Hs01931883_s1/GDNF/, Hs00707120_s1/NES/, Hs00801 390_s1/TUBB3/, Hs00902194_m1/MAPT/, Hs00300531_m1/SYP/, Hs00907365_m1/MNX1/, Hs00377575_m1/IS L2/, Hs00252848_m1/CHAT/, Hs00909233_m1/GFAP/, Hs00902901_m1/S100B/). The qPCR was carried out in a final volume of 20 μl containing 500 ng of extracted RNA. The following thermal profile was used: a single cycle of reverse transcription for 30 min at 50°C and 15 min at 95°C for reverse transcriptase inactivation and DNA polymerase activation, followed by 40 cycles of denaturation at 95°C for 15 s and annealing and extension at 60°C for 1 min. The results were analyzed using the integrated 7500 System SDS Software (version 1.3.1; Life Technologies). Each data set was normalized with an appropriate TaqMan Endogenous control selected by NormFinder (MOMA, Aarhus, Denmark) (2). As housekeeping genes, Actb (Rn00667869_m1) and GAPDH (Hs99999905_m1) were chosen for rat and human target genes, respectively. All qPCR reagents were provided by Applied Biosystems. Finally, the data were recalculated to relative quantities and transformed to a log2 scale using the Relative Expression Software Tool (REST) (Qiagen, Hilden, Germany) (32). In the case of the unexpressed NGF gene in iPSC-NPs before differentiation, the undetermined cycle threshold (Ct) value was set to a maximum (e.g., 40) for calculating the relative expression ratio. All numerical data are presented as mean ± standard deviation and were analyzed statistically using REST.
Animals
To complete this study, 73 10-week-old male Wistar rats were used. Their body weights ranged between 270 and 300 g to minimize differences in body size in order to achieve standardized spinal cord lesions. The numbers of animals used for all parts of the study are summarized in Table 1. The study was demanding in terms of the number of animals and the length of animal survival. Therefore, we optimized the number of animals to create groups that were large enough to obtain statistically relevant numbers in each group. We started our experiments with the Brasso, Beattie, and Bresnahan (BBB) and the plantar test as standard tests (n = 16 for controls-SCI and n = 13 for iPSC-NPs). At the end of the behavioral experiments, the iPSC-NP grafted animals had BBB scores over 10 and were able to move and gain weight support, so we created another group of animals that were used for additional tests requiring better coordination and stepping abilities (n = 6 for control and n = 8 for iPSC-NPs). The total number of animals used in the behavioral experiments in our 8-week study was 22 and 21, respectively, since the second group was also tested using the BBB and plantar test to assure the comparability of both groups of animals. From the 21 iPSC-NP grafted animals, eight were left to survive for 4 months to follow the maturation of the grafted cells, and these were tested using the BBB and plantar test every second week. Seven animals were immunohistochemically evaluated 8 weeks after transplantation. Tissue from eight animals in the control and six in the grafted group was processed for paraffin-embedded sections for morphometrical assessment. Additional groups of animals (iPSC-NPs grafted and control) were created for PCR analysis (n = 8); these animals were checked for their overall health status, but were not tested in detail as were the animals in the behavioral study. Another two groups of five animals each (control and iPSC-NPs) were established for a 2-week morphometrical study. These animals were not included in the behavioral experiments. All experiments were performed in accordance with the European Communities Council Directive of the 24th of November 1986 (86/609/EEC) regarding the use of animals in research and were approved by the Ethics Committee of the Institute of Experimental Medicine ASCR, Prague, Czech Republic.
The Numbers of Animals Used in All Parts of the Study

Total number of animals used in the study is 73. The animals listed in bold were also used for behavioral tests; therefore, they are not included in total number. SCI, spinal cord injury; iPSC-NPs, neural precursors derived from induced pluripotent stem cells; BBB, Basso, Beattie, and Bresnahan.
Spinal Cord Injury
A balloon-induced compression lesion was performed in a total of 77 male Wistar rats (270–300 g) as described by Urdzikova et al. (45). Briefly, the animals were anesthetized with 2% isoflorane (Forane®; Abbott Laboratories, Queenborough, UK) and shaved on the back from C7 to L1. Under sterile conditions, the skin was cut in the midline from Th7 to Th12. The soft tissue was removed, as well as the spinous processes of vertebrae Th10–Th11. A 2-french Fogarty catheter (Baxter, Irvine, CA, USA) was inserted into the epidural space and advanced cranially for 1 cm, so that the center of the balloon rested at the Th8–Th9 level of the spinal cord. The balloon was rapidly inflated with 15 μl of saline for 5 min. The diameter of the inflated catheter reached about 2 mm at maximum, but the shape of the inflated catheter was deformed by the narrow space between the vertebra and the spinal cord. Irrespective of this deformation, the same pressure was transmitted equally over the spinal cord in contact with the inflated catheter.
The catheter was then deflated and removed. The soft tissue and skin were sutured with unresorbable thread (EP1-USP 5/0; CHIRMAX Ltd., Trebišov, Slovakia), and the animals were allowed access to food and drink ad libitum. During the surgical procedure, the body temperature of the animal was maintained at 37°C with a heating pad, and 3% isoflurane in air was administered at a flow rate of 0.3 L/min. After being returned to their cages, the rats were assisted in feeding and urination until they had recovered sufficiently to perform these functions on their own. The animals received gentamicin sulfate (5 mg/kg) (SANDOZ, Prague, Czech Republic) for 3 days to prevent postoperative infections and butorphanol (2 mg/kg) (Pfizer Inc., Gerona, Spain) for pain relief.
Transplantation
The animals were transplanted 1 week after SCI. The animals were secured in a stereotaxic apparatus (Stoelting Co., Wood Dale, IL, USA) with a rat-specific vertebra holder (Cunningham spinal adaptor; Stoelting Co.). The spinal cord was exposed at T8, and a cell or saline injection was made in the midline of the spinal cord (without damaging the dorsal blood vessel) at a depth of 1 mm below the dorsal surface. A total of 5 × 105 predifferentiated iPSC-NPs/5 μl were injected through a glass pipette at a rate of 1 μl/min using a Nano-Injector (Stoelting Co.). The number of cells and the injection volume were based on the study by Amemori et al. (1), which gave excellent results on cell survival and also in order to make the studies comparable. The glass pipette was kept in place after injection for a further 5 min to prevent leakage of the cell suspension. The control group of animals with SCI only received an injection of 5 μl of saline. Triple drug immunosuppression was used to prevent graft rejection (30). Cyclosporine A (10 mg/kg) (Novartis, East Hanover, NJ, USA), azathioprine sodium (2 mg/kg) (Aspen Pharma Trading Ltd., Dublin, Ireland), methylprednisolone (2 mg/kg, tapered to 0.5 mg/kg) (Pfizer Inc., New York, NY, USA), and ampicillin (50–100 mg/kg) (Biotika, Slovenská Lupča, Slovakia) were administered 1 day before transplantation and throughout the experiment (for 8 or 17 weeks). After surgery, transplanted as well as control animals quickly recovered from the isoflurane anesthesia and did not suffer from any serious complications, such as intolerable pain, nausea, or aphagia. Autophagia of their tail, testes, and the skin around the anus was occasionally seen (in 4 animals out of 77); the animals were immediately euthanized and were not included in the calculations presented in Table 1. We did not perform any active targeted rehabilitation except for behavioral testing during the observation period.
Functional Analysis
For motor testing, hindlimb locomotor activity after SCI was assessed by the BBB test (3) (SCI and iPSC-NPs n = 21; controls n = 22). The rats were placed on a floor within a circular enclosure. Their hindlimb joint movement, paw placement, weight support, forelimb– hindlimb coordination, etc., were evaluated using a 0–21-point scale.
Motor coordination was examined on a balance beam. The apparatus consisted of a 140-cm-long and 3-cm-wide wooden flat beam elevated 30 cm above the floor. Lines that were marked 20 cm from each end of the beam indicated the walking distance defined as a 1-m-long section, where the animals were evaluated. A 60-W lamp was positioned at the start of the beam as an aversive stimulus. The animals' own home cage was placed at the other end of the beam. Animals were placed on the beam and allowed 60 s for assessment; in case they were not able to start any movement (after SCI), they were returned to their cage. Before the experiment, the animals were pretrained for 2 weeks with three trials per day for 5 consecutive days. During the first trial, the rat was placed 20 cm away from its home cage. For the second trial, it was placed at the midpoint of the beam, while for the third trial it was placed at the start of the beam. When the animals learned the task, they were placed only at the start of the beam. After this pretraining, the rats were tested twice a day for three consecutive days. The latency to traverse the beam was measured, and the locomotor performance was assessed using a scale modified from Metz et al. (24) as the scores of the flat beam walking test.
Motor coordination was also evaluated by the rotarod test before surgery at a speed of 10 rpm and every 2 weeks after the transplantation of human iPSC-NPs or the injection of saline at a speed of 5 rpm. To learn the task, the animals were pretrained 2 weeks before the SCI. For 3 consecutive days, the animals were placed on the rod, which accelerated from 5 rpm to 10 rpm over a period of 5 min.
For sensitivity testing, hindpaw withdrawal latency to noxious thermal stimuli was assessed by a plantar test apparatus (Ugo Basile, Comerio, Italy) (SCI and iPSC-NPs n = 17; controls n = 19). The animals were placed in a clear plastic chamber and acclimated for 10 min until becoming quiescent. The hindpaw received a heat stimulus through a glass plate. The withdrawal latencies were measured five times for each hindpaw at 5-min intervals. The average of five values was used for statistical evaluation. From the plantar test, we excluded those animals that were not cooperative and with whom five measurements were not obtained. This means the animals were repeatedly urinating and wetting the chamber floor or lying on the chamber floor, so their paws were either wet or not in full contact with the floor.
The data are expressed as mean ± SEM. All data were compared between the control group and the transplanted group by a two-sample t-test for independent samples, if the two samples had equal variances. If they had unequal variances, the Mann–Whitney test was used for evaluation. A value of p < 0.05 was considered statistically significant. All behavioral tests were performed by two independent and blinded observers.
Histological Analysis
To analyze the volume of the spared white and gray matter and the extent of axonal sprouting, control animals with SCI only (n = 13) and animals grafted with iPSC-NPs (n = 11) were sacrificed 2 weeks (controls n = 5; SCI and iPSC-NPs n = 5) or 8 weeks (controls n = 6; SCI and iPSC-NPs n = 6) after transplantation. After perfusion with 4% paraformaldehyde (Sigma-Aldrich), a 2-cm-long segment of the spinal cord was dissected between 1 cm cranial and 1 cm caudal to the injury epicenter. The whole segment was serially transversely cut, and two series of 20 sections (thickness 5 μm) were collected (1 mm distance between individual sections). Serial cross sections were made after paraffin embedding and were stained with either Luxol fast blue or Cresyl violet (both from Sigma-Aldrich) to distinguish the white and gray matter or by antibodies against growth-associated protein (GAP43) to evaluate axonal sprouting. For volumetric measurements, six sections were selected at 1-mm intervals along the craniocaudal axis, and whole images of the spinal cord were taken with an Axioskop 2 plus microscope (Carl Zeiss AG, Oberkochen, Germany) and analyzed by ImageJ software (National Institutes of Health, Bethesda, MD, USA) (45). Grafted iPSC-NPs incorporated nicely into the surrounding host tissues. This made it very difficult to identify transplanted cells in cross-sections stained with Luxol fast blue and Cresyl violet, which were used to distinguish the white and gray matter. The volumes of the white and gray matter were therefore analyzed and calculated from the section area including transplanted cells. Axonal sprouting was expressed as the mean number of GAP43-positive axons per section (4). High magnification images of transversal sections, separated by a 1-mm distance and stained for GAP43, were taken in all animal groups, and GAP43-positive axons were manually counted. Each counted axon was marked with red color to avoid double counting.
For histological evaluation of the grafts, animals transplanted with iPSC-NPs were sacrificed 8 weeks (n = 7) or 17 weeks (n = 8) after transplantation. To monitor the survival, migration, and differentiation of the transplanted cells, serial cryostat longitudinal sections of the spinal cord (14 μm) were cut through the areas of interest. To identify human stem cells transplanted into the rat spinal cord, antibodies directed against human nuclei (HuNu, 1:40) (Chemicon, Temecula, CA, USA), KU80 (1:250), and human mitochondria (MTC02, 1:125) (both Abcam) were used. To follow the fate of the transplanted iPSC-NPs and their communication with the host tissue, antibodies directed against nestin (1:200), chondroitin sulfate proteoglycan (NG2, 1:400), calcitonin gene-related peptide (CGRP, 1:1,000), GAP43 (1:2,500), neurofilament 70 (NF70, 1:200), calbindin (1:400), tyrosine hydroxylase (TH-5, 1:1,000) (all Millipore, Billerica, MA, USA); neurofilament 200 (NF200, 1:400), glial fibrillary acidic protein (GFAP, 1:200), βIII-tubulin (1:100) (all Sigma-Aldrich); choline acetyltransferase (ChAT, 1:100), Ki-67 (1:50), microtubule-associated protein 2 (MAP2, 1:500), serotonin (SERO, 1:100), dopamine (1:1,000)- and 2′,3′-cyclic nucleotide 3′-phosphodiesterase (CNPase, 1:100) (all Abcam); Islet2 (1:20), Nkx6.1 (1:20) (DSHB, Iowa City, IA, USA) and doublecortin (DCX, 1:500) (Santa Cruz, Heidelberg, Germany) were used. To visualize primary antibody reactivity, appropriate secondary antibodies were used: goat anti-mouse IgG conjugated with Alexa-Fluor 488 (1:200) or 594 (1:500) and goat anti-rabbit IgG conjugated with Alexa-Fluor 594 (1:500) (Molecular Probes, Eugene, OR, USA). Confocal images were taken with a Zeiss LSM 5 Duo confocal microscope (Carl Zeiss AG).
To estimate the number of human cells present 8 weeks after transplantation, every sixth longitudinal frozen section (14 μm thickness) of the spinal cord sections was chosen for imaging of the whole graft area. Images were taken with an Observer D1 microscope (Carl Zeiss AG). The surviving human cells, which were recognized as HuNu-positive cells, were counted in each section using ImageJ software. The total number of cells was estimated according to the volume of the section in which the cells were found. The percentage of surviving transplanted cells was calculated by dividing the estimated total number of surviving cells by the total number of transplanted cells (5 × 105). The index of cell mitotic activity was calculated as the ratio of Ki-67-positive cells to the total number of surviving cells positive for HuNu.
Results
Characteristics of the Human iPSC-NPs Before Transplantation
In order to prepare iPSC-NPs for transplantation, cells were predifferentiated during 7 days in medium containing BDNF, but omitting FGF and EGF. To characterize the expression of pluripotent and neural markers in human iPSC-NPs before and after their predifferentiation, a series of FACS analyses were performed (Fig. 1A). The cytometry results revealed a significant decrease in the expression of the pluripotent marker OCT3/4, the early neural progenitor markers SOX2, CD133, CD29, PSA-NCAM, and NF70, the early glial marker A2B5, and the neural proliferative marker nestin. At the same time a highly significant increase in the expression of CXCR4 was detected (p < 0.001).

Characteristics of the human iPSC-NPs, neural precursors derived from induced pluripotent stem cells (iPSC-NPs) before transplantation into the lesioned spinal cord. FACS profiles of pluripotent and neural markers in iPSC-NPs before (n = 5) and after predifferentiation (n = 5) (A) (*p < 0.05; **p < 0.01). The changes in the expression of neural growth factors (B) and neural-specific markers (C) in human iPSC-NPs after predifferentiation evaluated by qPCR.
Quantitive PCR analysis revealed that the predifferentiation of iPSC-NPs led to significant changes in the expression of genes for neural growth factors and neuronal/glial markers: an increase of BDNF and NT3, a decrease of VEGF, an increase of TUBB3, MAPT, SYP, HB9, and ISL2, and a decrease of S100B (Fig. 1B, C).
On the whole, the high level of expression of such markers as SSEA-1, CD133, CD24, CD29, CD56, and NF70 confirm the neural and, to some extent, the neuronal profile of the iPSC-NPs. Cell predifferentiation led to a shift in the cell population from glial to NPs—decreasing A2B5 expression according to FACS analysis, as well as decreasing GFAP and S100B gene expression and the increasing expression of neuronal genes (TUBB3, MAPT, SYP, HB9) according to qPCR. However, the qPCR results do not allow us to estimate the absolute number of neuronal precursors in the cell population.
BBB Test
The motor function of the hindlimbs was examined weekly after SCI using the open-field BBB test. The BBB test revealed 1 week after SCI that the injured rats had either no functional movement or only slight movement in their hindlimb joints (Fig. 2A). The balloon-induced compression lesion allowed spontaneous recovery afterward; however, control animals receiving saline scored only 6.16 ± 0.38 at 9 weeks after SCI, while unambiguous and rapid functional recovery was observed after the transplantation of iPSC-NPs. A statistically significant difference from the lesioned control group was already apparent 2 weeks after treatment with iPSC-NPs (p < 0.0001). In the grafted group, functional improvement was observed 2 weeks after transplantation and continued until the end of the 9-week observation period; the final BBB score was 10.93 ± 0.41. Eight grafted animals were left to survive for 17 weeks, and to assure that their BBB score remained stable they were tested every second week.
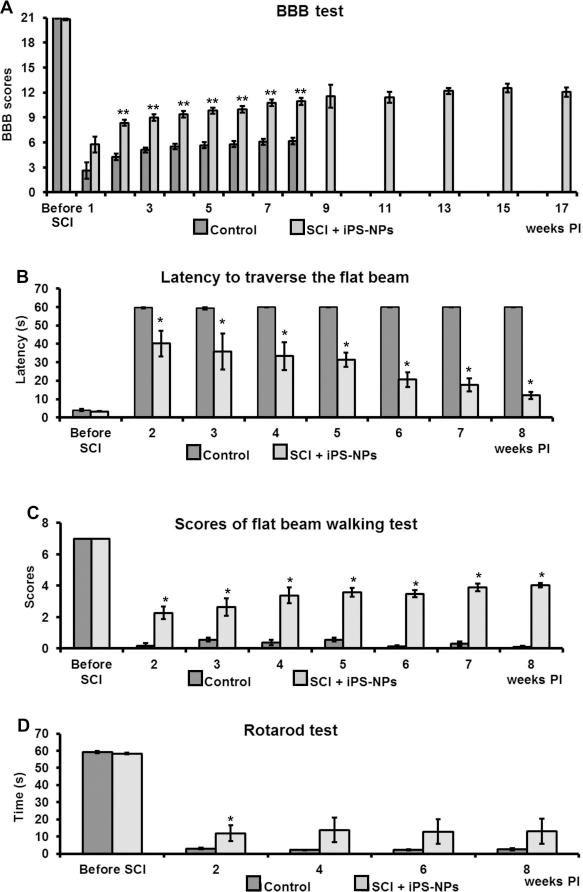
Locomotor function after iPSC-NP transplantation was assessed during 17 weeks. The motor function of the hindlimbs was examined using the open-field Basso, Beattie, and Bresnahan (BBB) test (A). A significant improvement of the BBB scores in the transplanted group (n = 21 until the eighth week and n = 8 after the eighth week) compared to controls (n = 22) appeared already from 2 weeks postinjection (PI) (**p < 0.0001). Significant differences between the control (n = 6) and the transplanted animals (n = 8) in their latency to traverse a beam with a flat surface (B) and in the scores of the flat beam walking test (C) were found at 2 weeks PI (*p < 0.05). Motor coordination was also evaluated with the rotarod test (n = 6 for controls, n = 8 for transplanted animals) (D). Significant improvement in the rotarod test was found in the iPSC-NP-treated animals only 2 weeks PI (*p < 0.05).
Beam Walking Test
The latency to traverse a beam with a flat surface was measured before surgery and every week from the second week after the injection of iPSC-NPs or saline (Fig. 2B). After surgery, all animals were unable to initiate any movement; the latency reached almost 60 s. The lesioned control animals never improved their ability to traverse the beam during the 9-week observation period. Some of them recovered their ability to maintain their bodies on the flat beam, but none of them could use their hindlimbs to walk on the beam. The transplanted animals, however, shortened the latency week by week after the transplantation of iPSC-NPs. A statistically significant difference was already observed 2 weeks after transplantation compared to the lesioned control group (40.2 ± 0.7 s in the transplanted group, 59.7 ± 0.3 s in the lesioned control group, p < 0.05). All transplanted animals succeeded in traversing the beam from 5 weeks after transplantation. The latency continuously decreased to 12.1 ± 0.1 s at 8 weeks after transplantation. All transplanted animals could use their hindlimbs partially or fully to traverse the beam at the end of the experiments.
When the locomotor performance was assessed using a scale modified from Metz et al. (24) as the scores of the flat beam walking test, a similar tendency of improving after cell transplantation was observed (Fig. 2C). The final score for animals transplanted with human iPSC-NPs was 4.0 ± 0.1 out of 7 (the maximum for healthy animals), which corresponded to the ability of the rats to traverse the whole length of the beam.
Rotarod Test
Motor coordination was evaluated in the rotarod test before surgery at a speed of 10 rpm and every 2 weeks after the transplantation of iPSC-NPs or the injection of saline at a speed of 5 rpm. The latency (i.e., the time spent on the rod) at 2 weeks after treatment was 11.9 ± 4.6 s in the transplanted group and 3.1 ± 0.6 s in the lesioned control group (p < 0.05) (Fig. 2D). The latency did not change to any marked extent in either group during 8 weeks observation after treatment.
Plantar Test
For sensitivity testing, the withdrawal latency of the hindpaws from a thermal stimulus was examined weekly up to 8 weeks after transplantation and determined as the average from five repeated measurements. Before surgery, the withdrawal latency in the hindpaws was 6.07 ± 0.32 s. The withdrawal latency increased 1 week after SCI up to 7.05 ± 0.28 s in the control animals and to 7.36 ± 0.29 seconds in the transplanted animals and then gradually returned to the normal range. After 8 weeks posttransplantation, eight grafted animals were left to survive for 17 weeks and were tested every second week. None of the transplanted animals show any hypersensitivity or allodynia, since their score did not differ from that of the control animals.
Morphometric Evaluation of the Spared White and Gray Matter
The area of the white and gray matter was calculated between 1 cm cranial and 1 cm caudal to the injury epicenter. The cross-sectional areas (mm2) of the white and gray matter were calculated at each cross-section and plotted at 1-mm increments from the injury epicenter, which was recognized as the smallest area of the spinal cord (Fig. 3A). The areas of spared white matter were significantly larger in the transplanted group than in the lesioned control group except 1–2 mm caudal to the injury epicenter (p < 0.05). The gray matter was spared around the transplanted sites corresponding to the injury epicenter. A significant difference was observed between 3 mm cranial and 4 mm caudal to the injury epicenter when compared to the lesioned control group (p < 0.05). When the volume (mm3) of the white and gray matter was compared in five different parts of the spinal cord (front: 7–10 mm cranial; middle front: 3–6 mm cranial; center: between 2 mm cranial and caudal to the injury epicenter; middle back: 3–6 mm caudal; back: 7–10 mm caudal) (Fig. 3B), statistically significant differences were found in all parts of a 2-cm long spinal cord segment in terms of white matter sparing (p < 0.05), while significant sparing of the gray matter was seen in the center and middle back parts of the examined spinal cord (p < 0.05). The total volume of the white and gray matter was estimated from a 2-cm-long spinal cord segment and compared at 2 weeks and 2 months after SCI. There were no significant differences between the lesioned control and transplanted groups in terms of white or gray matter sparing at 2 weeks after SCI (p > 0.05), but remarkable differences were found at 2 months after SCI (p < 0.01) (Fig. 3C).
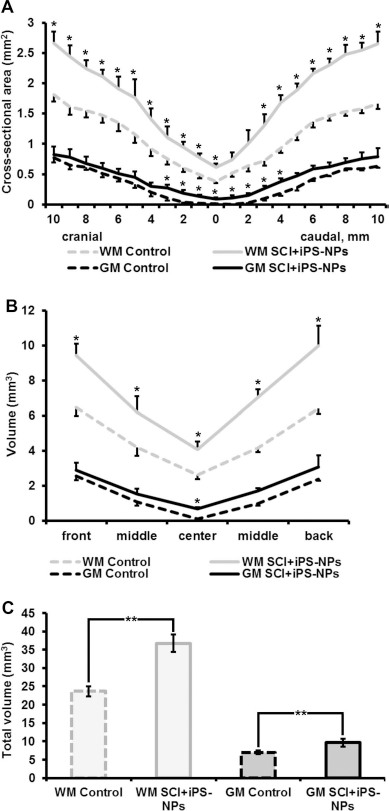
Morphometric evaluation of the spared white and gray matter 8 weeks PI. Whereas the gray matter was significantly spared only in the central part of the spinal cord cross-section, the white matter was significantly spared in the whole cross sectional flat area of the spinal cords of grafted animals compared to control animals (*p < 0.05) (A). This was confirmed by measuring the volume (mm3) of the white matter in five different parts of the spinal cord (B) and estimating the total volume of the white and gray matter (C) (**p < 0.001).
In Vivo Cell Survival and Differentiation
Human iPSC-NPs were evaluated in the lesioned rat spinal cord in terms of their survival and differentiation into neuronal and glial phenotypes. A total of 5 × 105 cells were implanted into the center of a spinal cord lesion. The engrafted cells were identified by immunohistochemical staining for the human-specific markers HuNu or MTCO2 (Fig. 4A). The animals were sacrificed 8 or 17 weeks after transplantation, and cell survival was evaluated by examining serial longitudinal sections of the lesioned spinal cord at both time points. The results showed that the cells robustly survived in the lesion; during the entire experiment, we found grafts in 28 out of 29 animals. Grafted cells also did not form tumors during the entire period of observation. Quantification of the mitotic activity with Ki-67 immunoreactivity identified 2.84 ± 0.42% dividing cells in grafts at 17 weeks, which was similar (2.34 ± 0.3%) to the index shown for these cells after transplantation into a rat stroke model (32).
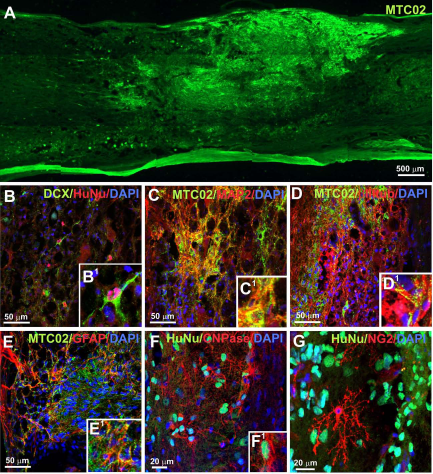
The robust engraftment and migration (MTC02, green) of iPSC-NPs in a lesioned rat spinal cord was observed 8 weeks after transplantation. (A). Transplanted cells were positive for doublecortin (DCX) (B and B1—higher magnification of B), microtubule-associated protein 2 (MAP2) (C), βIII-tubulin (D), glial fibrillary acidic protein (GFAP) (E), and 2′,3′-cyclic nucleotide 3′-phosphodiesterase (CNPase) (F). The grafts were infiltrated by microglial cells (NG2 positive) that were not positive for the human-specific marker HuNu (G).
The survival of iPSC-NPs 8 weeks after transplantation was estimated from serial longitudinal sections. Transplanted cells were distinguished from host cells as HuNu- or MTCO2-positive cells. The total volume of the transplanted cell mass was 0.27 ± 0.09 mm3, and the percentage of surviving iPSC-NPs was 9.8 ± 2.93% (2.32~19.84%), which was calculated by dividing the total number of surviving cells by the number of transplanted cells (5 × 105 cells).
Eight weeks after implantation, grafted iPSC-NPs expressed neural markers such as doublecortin (Fig. 4B, B1), MAP2 (Fig. 4C), and βIII-tubulin (Fig. 4D) and the astroglial marker GFAP (Fig. 4E). A small fraction of cells was positive for CNPase (Fig. 4F), a marker of oligodendrocytes. Staining for NG2 showed that the grafts were infiltrated with NG2-positive endogenous glial cells (they were not positive for HuNu or MTC02) (Fig. 4G).
To evaluate the effect of transplanted iPSC-NPs on endogenous regenerative processes, we examined the gene expression of several neurotrophic factors: Bdnf, Vegf, Ngf, Nt3, Fgf8, and Gdnf in control and transplanted animals 8 weeks after cell grafting using qPCR. Our results demonstrate that the transplantation of iPSC-NPs did not cause changes in the gene expression of rat neurotrophic factors. However, qPCR analysis of the expression of the same human genes in iPSC-NPs 8 weeks after their transplantation into the lesioned rat spinal cord revealed a statistically significant upregulation of NGF (p < 0.05), FGF8 (p < 0.001), and GDNF (p < 0.05) and the downregulation of VEGF and NT3 (p < 0.001), compared to the mRNA levels before transplantation (i.e., in predifferentiated cell cultures) (Fig. 5A).

The results of qPCR analysis demonstrate significant changes in neural growth factor gene expression (A) and in the expression of the human genes NES, TUBB3, MAPT, SYP, HB9, ISL2, CHAT, GFAP, and S100B (B) in iPSC-NPs 8 weeks after transplantation into a lesioned rat spinal cord, compared to the same cells before transplantation (*p < 0.05; **p < 0.001).
The ability of the transplanted iPSC-NPs to differentiate toward neurons/glia cells was also assessed 8 weeks after grafting, using qPCR analysis. At this time point, the gene expression of neural-specific markers in iPSC-NPs transplanted into the site of a SCI also significantly changed (Fig. 5B). Whereas the expression of the NES and TUBB3 human genes was downregulated and the expression of the MAPT, SYN, and CHAT human genes was upregulated only about two- to fourfold (p < 0.001) compared to the expression detected in cells before transplantation, the expression of the human glial genes GFAP and S100B was upregulated about 7- to 12-fold (p < 0.05 and p < 0.001, correspondingly), compared to the cells in culture prior to transplantation. These results suggest that 8 weeks after SCI, the hostile lesion environment facilitated the expression of glial markers more than the expression of neuronal markers.
To evaluate the influence of the transplanted iPSC-NPs on axonal sprouting during regeneration, staining for GAP43 was performed in cross-sections from both control and grafted spinal cords. The number of GAP43-positive fibers reflected axonal sprouting in the lesion (Fig. 6). More intense staining for GAP43 in the spinal cord cross-sections was detected in the tissue of transplanted rats (Fig. 6A–C). The number of GAP43-positive axonal fibers per section in the grafted animals was 25.61 ± 6.63, while in control animals only 8.41 ± 3.35 (Fig. 6D).

Staining for GAP43 demonstrates intensive sprouting in the injured spinal cord tissue of rats transplanted with iPSC-NPs (A) compared to control injured spinal cord tissue (C). (B) Higher magnification views of the transplant area in (A). (D) Quantitative analysis of GAP43-positive fibers in the spinal cord tissue of control and transplanted animals (*p < 0.05).
The further maturation of grafted iPSC-NPs was investigated 17 weeks after their transplantation into the site of a SCI (Fig. 7). Histochemical staining for NF200 (heavy chain neurofilament) demonstrated that the whole volume of the grafts was penetrated by endogenous neurofilaments (Fig. 7A). However, individual cells in the graft were also positive for NF70 and NF200—an advanced marker of neuronal differentiation (Fig. 7B, C, and C1). Transplanted cells were also able to differentiate toward interneurons— cells with a neuron-like morphology positive for calbindin (Fig. 7D–F, F1), dopaminegric neurons—cells positive for tyrosine hydroxylase (TH-5) (Fig. 7G–I, H1, and I1), serotoninergic neurons—serotonin-positive cells (Fig. 7J), and motor neurons positive for cholineacetyltransferase (ChAT) (Fig. 7K), all in colocalization with MTC02. Newly forming motor neurons were found in the ventral part of the spinal cord. At this time point, a large volume of the graft was positive for GFAP. Costaining for GFAP together with a marker of human cells, MTC02, revealed newly forming glial cells (Fig. 7L). Nevertheless, cells that appeared to be newly forming neurons were found in very close vicinity to newly forming astrocytes, as shown in Figure 7L1 (a newly forming neuron is marked by a white dashed line). In addition to the results of NF200/ MTC02 staining, costaining for CGRP together with MTC02 revealed that the transplants were also infiltrated with endogenous sensory fibers (Fig. 7M, M1).
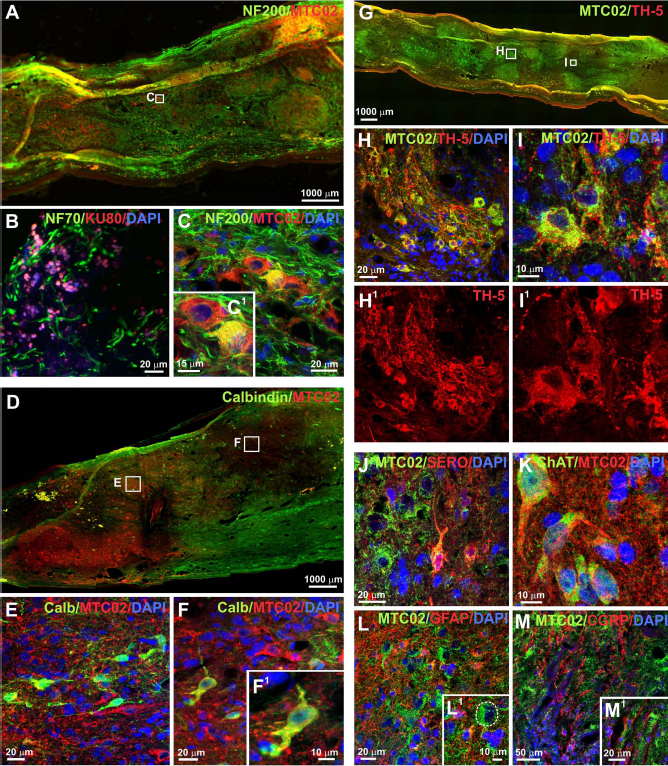
iPSC-NP survival and migration 17 weeks after transplantation was similar to that seen at 8 weeks. The grafts were infiltrated by endogenous cells and cell elements positive for NF200 (A, C), GFAP (L and L1 – higher magnification of L) and calcitonin gene-related peptide (CGRP) (M and M1 – higher magnification of M). At the same time, the transplanted cells were able to differentiate into GFAP-positive astrocytes (L1, the white dashed line on L1 shows a human cell that is not positive for GFAP and that has a neuron-like morphology), NF70- and NF200-positive neurons (B and C, respectively) and could form populations of mature neurons positive for calbindin (D–F and F1—higher magnification of F), tyrosine hydroxylase (TH-5) (G–I and H1, I1), serotonin (J), and choline acetyltransferase (ChAT) (K).
Discussion
The development of cell reprogramming technology offers new opportunities for stem cell-based therapy for various pathologies (10,11,39,40,47). Moreover, iPSCs enable autologous transplantation, while avoiding ethical obstacles and immunological rejection of the grafts. A large number of ESCs or iPSCs and their differentiated derivatives have already been obtained and successfully used in experimental models of various disorders (18,20,28,29,33,41,42), among them different models of SCI (7,18,23,27,28,43). Our current study adds valuable data into the mosaic of understanding the mechanisms of action of NPs derived from human pluripotent cells in a model of SCI. The majority of studies injected oligodendrocyte progenitors (OPCs) derived from ESCs or iPSCs (7,23,27,43), showing that OPCs can myelinate the remaining axons in the lesion and improve locomotor recovery. In our study, we used iPSC-NPs, which were able to differentiate in vitro into all three neuronal phenotypes (neurons, astrocytes, and oligodendrocytes). Our results are in agreement with the findings of Nori et al. (28), in which neurospheres containing a mixture of progenitor cells were used to treat a contusive SCI in mice. Similarly, Erceg et al. (7) injected two different populations of cells derived from human ESCs, OPCs, and motoneuron progenitor cells (MNPs) into a spinal cord transection. The authors reported the cells' differentiation into neurons, astrocytes, and oligodendrocytes in animals grafted with either of the cell populations; however, there was a synergistic effect on functional outcome in rats grafted with both types of progenitor cells.
We have previously demonstrated that iPSC-NPs (IMR90) can successfully alleviate stroke-induced dysfunction in a rat middle cerebral artery occlusion (MCAO) model. The transplantation of iPSC-NPs reversed stroke-induced somatosensory and motor deficits and also dramatically protected the host substantia nigra from atrophy (33). In the present study, we investigated the therapeutic potential of this cell source for the treatment of a SCI in a rat balloon-induced compression lesion model and found that iPSC-NPs significantly support the recovery of locomotor function already from the second week after transplantation. To assure that the lesions in both groups were comparable and that the iPSC-NPs had a neuroprotective effect, we performed a morphometric evaluation 2 weeks after transplantation. We did not find any significant difference in either the lesion volume or in the volume of the spared tissue. We therefore suggest that such a rapid effect on functional improvement is caused by the trophic effect of the grafted iPSC-NPs, not by cell replacement in the lesioned tissue. Our results are in agreement with our findings from a stroke model (33), in which the same iPSC-NP line reversed the stroke-induced somatosensory deficit in a similar time course—functional recovery appeared already 2 weeks after cell transplantation, also mainly because of a paracrine effect, accompanied by the slow maturation of the grafted cells, their interaction with the host tissue, and the regrowth of human fibers into the host substantia nigra during the next 4 months. Previous studies in which NSCs were used for SCI treatment have shown functional recovery in animal models (6,30,43); however, not so early after grafting and usually in experiments with a less severe lesion (BBB scores for lesioned control animals of ~10) than in our experiments (BBB scores for lesioned control animals of 5–6). In addition, functional improvement was demonstrated not only by the BBB test, but was also confirmed by sensory motor tests, such as the beam walking and rotarod tests, which are more sensitive and objective compared to the BBB test and require better coordination in movements. The quantitative assessment of beam walking performance is a very sensitive tool to monitor even discrete deficits in foot placement and body balance, including tail movements (36). We also showed that IMR90-derived iPSC-NPs did not cause hypersensitivity or allodynia—two of the possible adverse effects of cell transplantation into a spinal cord lesion (13). Unlike Erceg et al. (7), we did not perform any active rehabilitation of the injured rats, which can further improve recovery via the increase of neurotrophins in the lesion (9,51); therefore, our observed locomotor improvement in grafted animals is based only on graft–host tissue interactions.
Any considerable effect of transplanted iPSC-NPs on the restoration of locomotor function after SCI is based on good cell survival in the grafts. Parr and colleagues reported less than 5% survival of adult NSCs derived from the adult spinal cord after transplantation into the rat spinal cord (30). On the other hand, Fujimoto and colleagues found approximately 20% survival of transplanted human iPSC-derived neuroepithelial-like stem cells in the injured mouse spinal cord at 4 weeks after transplantation (8). In our study, a single injection of 0.5 million cells into the spinal cord resulted in almost 10% surviving cells and facilitated host tissue regeneration 8 weeks after transplantation. In our previous study with the spinal progenitor cell line SPC-01 (1), we also observed robust graft survival (17%), also when injecting 0.5 million cells into the lesion epicenter, though the functional improvement was less prominent than with iPSC-NPs.
Rapid functional recovery is most likely due to the production of neurotrophic factors by the transplanted cells. Neurotrophin levels after injury play an important role in the normal regenerative processes that follow SCI (12). A large number of studies have demonstrated the upregulation (5,10,16,17,25,44), downregulation (16,17,25), or no change (42) of neurotrophin expression in the spinal cord tissue after lesion. However, only a few studies have examined neurotrophin levels more than 1 week after injury (23,42). In the present study, we showed that 9 weeks after SCI, only the expression of the rat glial-derived neurotrophic factor (Gdnf) gene was downregulated while the expression of Bdnf, Vegf, Ngf, Nt3, and Fgf8 did not undergo significant changes. We also showed that the expression of the same rat genes was not affected by iPSC-NP transplantation. However, the expression of human neurotrophins significantly changed 8 weeks after cell grafting into the injured spinal cord. We found the upregulation of the NGF, FGF8, and GDNF genes and the downregulation of the VEGFA and NT3 genes. Similar changes of neurotrophin gene expression in grafted cells were reported by Lu et al. and Yan et al. (22,49). In their studies, NSC transplantation supported extensive endogenous axonal regeneration. Similarly, the study of Lukovic et al. (23) suggested that neural protective factors secreted from transplanted cells in synergy with reactive astrocytes could create a neuroprotective environment for the neurogenesis observed in transplanted cells. The same fact was demonstrated in our experiments using immunostaining for GAP43. Taken together, our data suggest that iPSC-NP transplantation does not influence the expression of host neurotrophins in the injured rat spinal cord, but rather alters the expression of exogenous factors that can support axonal sprouting and functional recovery after SCI.
Morphometric measurements revealed that the white matter was markedly spared at sites remote from the injury epicenter (i.e., not around the injury epicenter), while the gray matter was spared around the injury center, where the transplants were injected. The total volumes of spared white and gray matter in a 20-mm-long spinal cord segment of the transplanted group were 55.6% and 37.4% greater, respectively, compared to those in the control group 8 weeks after transplantation. In our stroke experiments (33), we have seen that iPSC-NPs grafted into the striatum have rescued remote structures, such as the substantia nigra, from degeneration. We speculate that iPSC-NPs transplanted into SCI might not only spare tissue in the lesion, but might positively influence the apoptotic cells located all along the degenerating ascending and descending tracts and subcortical structures impaired by SCI. However, this hypothesis requires further investigation.
The greatest potential drawback of iPSC-based therapy is its risk of tumorigenicity. Therefore, we observed transplanted animals for an extended period up to 17 weeks. No signs of tumor formation or hyperproliferation were observed in 40 histochemically analyzed animals during this period of time. These results can be supplemented with data from our previous stroke experiments, in which no non-neural structures and no overgrowth or hyperproliferation were observed in 28 iPSC-NP-transplanted animals during the 4 months of the study (33).
In the present study, we also demonstrate by qPCR analysis a fourfold decreased expression of nestin (NES human gene) in the iPSC-NPs 8 weeks after transplantation. Consequently, we found only 2.84 ± 0.42% dividing cells in the grafts 17 weeks after transplantation. We still do not know how many cells are necessary for long-term tumor formation and if the dividing cells reflect the steady state due to dying cells or a net continuous increase in cell numbers. However, we did not notice any excessive increase in the size of the cell implant between 8 and 17 weeks after transplantation. Taken together, all these data support the possibility that iPSC-derived cell-based therapy might be safe in humans.
Another significant contribution of neural cell therapy after SCI is cell replacement. A large number of studies have confirmed the capability of transplanted NSCs of different origin to differentiate toward all three neural lineages (6,44,49), and similar results were also found in experiments with human iPSC-derived NPs (8,31). Here we demonstrate that 17 weeks after their transplantation into SCI, iPSC-NPs (IMR90 line) were able to form astrocytes, oligodendrocytes, and specific neurons (serotoninergic neurons, dopaminergic neurons, interneurons, and motor neurons). We also found that these main types of neural cells (astrocytes and neurons) closely communicated with each other, making a contribution to tissue restoration. Our findings are in agreement with our previous study on a stroke model in which the same cells slowly matured (over 17 weeks) into a striatal neuron phenotype (D2 receptor-, DARPP32-, and calretinin-positive cells), showing that the cells are to a certain extent able to adopt a tissue-specific phenotype. Interestingly, graft-derived calbindin-positive cells were localized in the middle central part of the spinal cord, while ChAT-positive neurons were found in the ventral part of the spinal cord tissue.
The fate of grafted cells of pluripotent origin and related adverse effects, such as tumor formation or hyperproliferative activity in vivo, often depends on the relative role of intrinsic and environmental factors in the host tissue (19,37). The low level of pluripotent marker expression and also the decrease of OCT3/4 and SOX2 expression as the result of in vitro predifferentiation suggest that iPSC-NPs are safe for use in transplantation experiments, whereas the low level of HLA-I and -II expression allows us to presume a weak response of the host immune system after transplantation.
The decrease of nestin expression after cell predifferentiation suggests a reduction of nonspecific proliferation. At the same time, the increase in CXCR4 expression after predifferentiation points to the active migration of iPSC-NPs after transplantation, since CXCR4, a receptor for SDF-1 factor, has been shown to be an important component for NSC migration (34,35).
However, qPCR analysis demonstrated that 8 weeks after transplantation iPSC-NPs were more prone toward astroglial differentiation than to neuronal. Interestingly, a comparison of these data with the characteristics of in vitro predifferentiated iPSC-NPs reveals different tendencies in their differentiation ability and neurotrophin expression during artificial in vitro predifferentiation compared to their in vivo differentiation in the lesioned host tissue. Only 7 days of predifferentiation in vitro turned iPSC-NPs toward neuronal differentiation to a greater extent than did 8 weeks of differentiation in the host environment. These data confirm the finding of Kumamaru et al. concerning the vulnerability of engrafted NSCs to environmental cues and the importance of in situ analysis in advancing the efficacy and safety of stem cell-based therapies (21).
Conclusions
To conclude, iPSC-NPs can be expanded in large quantities and, when implanted into an animal model of SCI, they robustly survive in the lesion, express neurotrophins, and facilitate the sprouting of endogenous GAP43-positive axons. All of these actions lead to rapid improvements in locomotor functions and to the sparing of the white and gray matter in a relatively short time. In the long term, transplanted iPSC-NPs can slowly mature into region-specific and mature neurons and can participate to some extent in tissue reconstruction.
Footnotes
Acknowledgments
This study was supported by the grants LH 12024, GACR 13-00939S, P304/12/G069, MEYS NPU I LO1309, and Biocev CZ.1.05/1.1.00/02.0109. We thank James Dutt for critical reading of the manuscript. The authors declare no conflicts of interest.
