Abstract
Microcarriers are used to expand anchorage-dependent cells in large-scale suspension bioreactors. Proteolytic enzyme treatment is necessary to detach cells cultured on microcarriers for cell harvest or scale-up, but the enzyme treatment damages the cells and extracellular matrices and complicates the culture process. Here, we fabricated thermosensitive microcarriers from which cells can be detached by temperature change without proteolytic enzyme treatment. A thermosensitive polymer, poly-N-isopropylacrylamide (pNIPAAm), was incorporated on the surface of Cytodex-3® microcarriers. pNIPAAm-grafted microcarriers allowed human bone marrow-derived mesenchymal stem cells (hBMMSCs) to adhere, spread, and grow successfully on the microcarriers as nongrafted microcarriers did. By dropping temperature below 32°C, more than 82.5% of hBMMSCs were detached from pNIPAAm-grafted microcarriers. The trypsin treatment for cell detachment induced apoptosis and death of some of the detached cells, but cell detachment from pNIPAAm-grafted microcarriers by temperature change significantly reduced the apoptosis and cell death. pNIPAAm-grafted microcarriers can significantly reduce cell extracellular matrix damage in the cell detachment process and simplify the cell detachment process by avoiding proteolytic enzyme treatment. pNIPAAm-grafted microcarriers would be valuable to a variety of potential fields demanding a large amount of cells without cell damage, such as cell therapy, tissue engineering, and other biological and clinical applications.
Introduction
Microcarriers are widely used to expand various types of anchorage-dependent cells in suspension bioreactors (4,8,12). Microcarriers are spherical units with various substrate matrices that are kept suspended in culture medium in stirred bioreactors and provide a large surface area for cell adhesion and growth (7,10,11). Therefore, use of microcarriers in stirred bioreactors allows large-scale expansion of cells in a homogeneous and controlled environment. Suspension culture technology using microcarriers has been industrially applied in the production of many biologicals, such as vaccines, enzymes, hormones, and cytokines (1,11,15). For cell harvest or scale-up, it is necessary to detach cultured cells from microcarriers by treatment with a proteolytic enzyme such as trypsin; however, this method of cell detachment leads to irreversible damage to cells and extracellular matrix (ECM) proteins (5). For example, use of trypsin for cell detachment inevitably degrades proteins in the ECM between cells and may even damage cell membrane proteins and receptors, resulting in alteration of the structure and function of the cell surface (17). Thus, it is critical to develop a new technique for cell detachment from microcarriers without impairing the cells and their surrounding environment, which includes the ECM.
In this study, we constructed thermosensitive microcarriers from which cells can be detached without proteolytic enzyme treatment. The thermosensitive microcarriers were made by conjugating a thermosensitive polymer, poly-N-isopropylacrylamide (pNIPAAm), onto the surface of commercially available microcarriers, Cytodex-3®. Cytodex-3® is a microcarrier containing denatured collagen bound covalently to the surface layer of a cross-linked dextran matrix. Due to its high cellular interaction, Cytodex-3® microcarrier has been extensively used in large-scale culture of anchorage-depend-ant primary and established cells. Previous studies showed that cell sheets cultured on pNIPAAm-grafted culture dishes can be detached by simply reducing the culture temperature without enzyme treatment (3). Here, we investigated whether pNIPAAm-grafted microcarriers would allow effective detachment of human bone marrow-derived mesenchymal stem cells (hBMMSCs) cultured in suspension bioreactors by dropping the temperature below the lower critical solution temperature (LCST; 32°C) of pNIPAAm. We also investigated whether pNIPAAm grafting on the microcarrier surface alters cell adhesion and growth and whether cell detachment by temperature change causes less damage to the cells and ECMs than proteolytic enzyme treatment.
Materials and Methods
Preparation of Thermosensitive Microcarriers
N-Isopropylacrylaminde (NIPAAm, Aldrich, St. Louis, MO, USA) was purified by recrystallization from n-hexane followed by filtration and drying. pNIPAAm terminated with a carboxyl group at one end was prepared by chain transfer polymerization using 3-mercaptopropionic acid (MPA; Aldrich) as a chain transfer agent (Fig. 1a). pNIPAAm (0.01 mol), MPA (0.2 μmol), and ammonium persulfate (AMP, 0.025 μmol) were dissolved in 50 ml deionized distilled water. The solution was purged with dry nitrogen gas for 15 min to remove the dissolved oxygen, then tetramethylenediamine (TEMED, Sigma) was added as a catalyst. After the reaction was completed at 25°C for 3 h under nitrogen atmosphere, the resultant carboxyl acid-terminated pNIPAAm was precipitated in ethanol. To obtain pNIPAAm with a narrow molecular weight distribution, the precipitated pNIPAAm was dissolved in acetone and reprecipitated with n-hexane. This procedure was repeated three times and then the pNIPAAm polymer product was dried in a vacuum oven. The carboxyl acid terminal group of pNIPAAm was activated by N-hydroxysuccinimide (NHS, 0.06 M) and 1-ethyl-3-[3-dimethylaminopro-pyl]carbodiimide hydrochloride (EDC, 0.12 M) in 0.05 M morpholinoethanesulphonic acid (MES, 0.05 M) buffer solution (pH 6) for 3 h at room temperature. Cytodex-3® microcarriers (diameter 170–380 μm; Sigma, St. Louis, MO, USA) were added to the activated pNIPAAm solution and gently stirred at 4°C for 12 h. The solution containing pNIPAAm-grafted microcarriers was centrifuged at 1500 rpm for 10 min, washed five times with deionized distilled water, and lyophilized for 3 days. Incorporation of pNIPAAm into Cytodex-3® microcarriers was confirmed by Attenuated Total Reflection-Fourier Transform Infrared (ATR-FTIR, IFS 66 spectrometer, Bruker, Germany) spectra. The morphologies of Cytodex-3® and pNIPAAm-grafted Cytodex-3® were examined with a scanning electron microscope (SEM, S-4800 UHR; Hitachi, Tokyo, Japan).

Surface modification of microcarrier (Cytodex-3®). (a) Synthesis of pNIPAAm and conjugation of pNIPAAm to microcarrier surface. (b) SEM images of nongrafted microcarrier and pNIPAAm-grafted microcarrier. Scale bars: 30 μm. (c) Confirmation of pNIPAAm conjugation to microcarrier (Cytodex-3®) by ATR-FTIR analysis.
Suspension Cell Culture Using Microcarriers
Both Cytodex-3® microcarriers and pNIPAAm-grafted Cytodex-3® microcarriers were used for suspension cell cultures. First, the two types of microcarriers were rehydrated in Dulbecco's phosphate-buffered saline (PBS, Sigma) for 5 h at room temperature, sterilized by autoclaving (0.15 MPa, 120°C, 30 min), and washed with Dulbecco's modified Eagle's medium (DMEM; Gibco BRL, Gaithersburg, MD, USA) supplemented with 10% (v/v) fetal bovine serum (FBS; Gibco BRL) and 1% (v/v) penicillin-streptomycin (PS; Gibco BRL). Spinner flasks (50 ml; Wheaton, Millville, NJ, USA) were siliconized prior to use to prevent cells adhering to the flask walls. Thirty milliliters of culture medium [DMEM containing 10% FBS, 1% (v/v) PS] containing hBMMSCs (2 × 105 cells/ml; Lonza, Walkersville, MD, USA) and microcarriers (5 mg/ml) was placed in spinner flasks with a suspended magnetic impeller and incubated at 37°C in a humidified 5% CO2 atmosphere. The culture began with intermittent agitation (agitation at 40 rpm for 5 min and static for 30 min) for 3 h and continued with gentle agitation at 40 rpm for 5 days. For medium exchange, every 2 days the agitation was temporarily stopped for 5 min to allow the microcarriers to settle and 70% of the culture supernatant was replaced with fresh medium. Cells present in the supernatant were collected by centrifugation and replaced in the spinner flasks.
To measure the efficiency of cell attachment onto microcarriers within 5 h of culture, the microcarrier-free supernatant was carefully removed and the number of cells in the supernatant was determined with a hemocytometer. The number of cells attached to microcarriers was calculated by subtracting the number of cells in the supernatant from the total cell number at inoculation. The attachment yield was calculated as follows: attachment yield (%) = (number of cells attached to microcarriers/total number of cells number at inoculation) × 100.
The morphology of hBMMSCs cultured on microcarriers was observed using SEM. Samples were fixed in 2.5% glutaraldehyde for 1 h, washed with 0.1 M phosphate buffer, and then fixed again in 1% osmium tetra-oxide solution. Next, the samples were dehydrated in ascending grades of ethanol, dried, and mounted on an aluminum stub using double-sided carbon tape. The specimens were coated with platinum using an Ion Sputter Coater (E-1030; Hitachi, Tokyo, Japan) and examined at an acceleration voltage of 15 kV.
In addition, cell growth and mitochondrial metabolic activity were determined by nucleus counting and 3-(4, 5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide (MTT) assay, respectively. For nucleus counting, 1 ml cell culture suspension containing cell-adherent microcarriers was placed in a centrifuge tube. After removing supernatant, the microcarriers were washed with warm PBS (37°C) and 0.1% (w/v) crystal violet (Sigma) in 0.1 M citric acid (Sigma) was added to give a final volume of 1 ml. After incubating the samples for 1 h at 37°C, the number of nuclei was determined with a hemocytometer. For the MTT assay, 1 ml cell culture suspension containing cell-adherent microcarriers was placed in a 24-well plate, and 100 μl MTT stock solution in PBS (pH 7.4) was added. After incubation at 37°C in 5% CO2 for 4 h, the medium was removed and 100 μl dimethyl sulfoxide was added to dissolve the formazan crystals. The absorbance at 405 nm was spectrophoto-metrically detected with an enzyme-linked immunoscorption assay plate reader (PowerWave X340, Bio-Tek Instruments, Inc., Winooski, VT, USA).
Detachment of Cultured Cells From Microcarriers
After 5 days of suspension culture, the temperature of the culture medium was reduced from 37°C to 32°C by adding a calculated volume of fresh medium at 4°C and hBMMSCs cultured on two types of microcarriers were incubated for 10 min at 32°C. During the cell detachment step, microcarriers containing a high density of cells settled much faster than detached cells. The number of detached cells in the microcarrier-free supernatant was counted with a hemocytometer. As a control, cells were detached from microcarriers by trypsin treatment. After detachment of cells from the microcarriers, 1 ml of the microcarrier-free supernatant containing detached cells was carefully retrieved and subcultured in new culture dishes for 1 day. Cell viability was determined by a live/dead assay using fluorescein diacetate (FDA; Sigma) and ethidium bromide (EB; Sigma). FDA stains the cytoplasm of viable cells green, while EB stains nuclei of nonviable cells red. Apoptotic activity of the detached cells was determined by the terminal deoxynucleotidyl transferase-mediated uridine triphosphate nick end labeling (TUNEL) method using a commercially available apoptosis detection kit (ApopTaq®, Chemicon, Chandlers Ford, Hampshire, UK).
hBMMSCs detached from two types of microcarriers by either trypsin treatment or temperature change were allowed to attach to slide chambers for 30 min and fixed with 4% (w/v) paraformaldehyde for 20 min at 20°C. To detect the fibronectin and laminin on the cells, the proteins were immunofluorescently stained by using anti-fibronectin (Abcam, Cambridge, UK) or anti-laminin (Abcam) antibodies. The staining signals for fibronectin and laminin were visualized with TRITC- and FITC-conjugated secondary antibodies (Jackson Immuno Research Laboratories, West Grove, PA, USA), respectively. The sections were counterstained with DAPI and examined using a fluorescence microscope (TE2000, Nikon, Tokyo, Japan).
Fibronectin and laminin on hBMMSCs before and after detachment from two types of microcarriers by either trypsin treatment or temperature change were analyzed with Western blot analysis. The cells were washed three times with PBS and lysed by adding SDS sample buffer [62.5 mM Tris-HCl (pH 6.8), 2% SDS, 10% glycerol, 50 mM dithiothreitol, 0.1% bromophenol blue]. Proteins were electrophoretically separated on 4–10% SDS polyacrylamide gels and transferred to membranes (Millipore, Bedford, MA, USA). For protein detections, the membranes were incubated with primary antibodies against fibronectin (Abcam) and laminin (Abcam) overnight at 4°C, washed, and incubated with secondary antibodies conjugated to horseradish peroxidase (Sigma) for 50 min at room temperature. Blots were developed using enhanced chemiluminescence (LumiGLO, KPL Europe, Guildford, UK) as recommended by the manufacturer.
Statistical Analysis
All quantitative data were expressed as mean ± SD. Statistical analysis was performed with one-way analysis of variance (ANOVA) with Tukey's honestly significant difference post hoc test using SPSS software (SPSS Inc., Chicago, IL, USA). A value of p < 0.05 was considered statistically significant.
Results
Characterization of pNIPAAm-Grafted Microcarrier
pNIPAAm grafting on microcarriers did not affect the surface morphology of the microcarriers (Fig. 1b). ATR-FTIR analysis showed that the spectrum of pNIPAAm-grafted Cytodex-3® microcarriers exhibited the characteristic absorptions of stretching and vibration of the hydroxyl group of the Cytodex-3® microcarrier (3400 and 1026 cm−1) as well as the characteristic absorptions of the0 C = O stretching (1640 cm−1) and amide bending (1530 cm -1 ) of pNIPAAm (Fig. 1c). This indicates that pNIPAAm was successfully grafted onto the surface of the Cytodex-3® microcarrier.
Cell Attachment and Growth on pNIPAAm-Grafted Microcarrier
hBMMSCs were cultured on Cytodex-3® microcarriers and pNIPAAm-grafted Cytodex-3® microcarriers in stirred bioreactors. Grafting of pNIPAAm on the microcarrier surface did not affect cell attachment to the microcarrier, as the cell attachment kinetics of Cytodex-3® and pNIPAAm-grafted Cytodex-3® were similar (Fig. 2a). Cell attachment increased as the incubation time increased, with a dramatic increase over the first 30 min: approximately 35% of the inoculated cells attached to both types of microcarrier after 30 min of incubation, and more than 99% of cells were attached within 270 min. Grafting of pNIPAAm on the microcarrier surface did not affect adhesion morphology, growth, or metabolic activity of cells cultured on the microcarriers. SEM analysis showed no difference in cell adhesion morphology between the two types of microcarrier (Fig. 2b). Crystal violet staining of cells cultured on microcarriers showed that cells can grow on both types of microcarrier (Fig. 2c), and nuclear counting (Fig. 2d) and MTT assay (Fig. 2e) showed no difference in cell growth and metabolic activity, respectively.

Attachment and growth of hBMMSCs on two types of microcarriers, nongrafted microcarrier, and pNIPAAm-grafted microcarrier, in spinner flask culture. (a) Percentage of hBMMSCs attached to two types of microcarriers monitored over 5 h (filled circles: nongrafted microcarrier; open circles: pNIPAAm-grafted microcarrier). (b) SEM images of hBMMSCs attached to the two types of microcarriers and cultured for 1, 3, or 5 days. Scale bars: 30 μm. (c) Light microscope images of hBMMSCs attached to two types of microcarriers and cultured for 1, 3, or 5 days. Crystal violet-stained cell nuclei. Scale bars: 50 μm. Insets show images at 4x magnification compared to main figure. (d) Growth of hBMMSCs cultured on two types of microcarriers, as determined by nucleus counting (n = 3) (filled circles: nongrafted microcarrier; open circles: pNIPAAm-grafted microcarrier). (e) Mitochondrial metabolic activity of hBMMSCs cultured on two types of microcarriers, as determined by MTT assay (n = 3) (filled circles: nongrafted microcarrier; open circles: pNIPAAm-grafted microcarrier).
Cell Detachment From pNIPAAm-Grafted Microcarrier
Trypsin treatment induced detachment of most of the adherent cells from both types of microcarriers (Fig. 3). In contrast, reduction of the temperature from 37°C to 32°C induced cell detachment from pNIPAAm-grafted Cytodex-3® but not from Cytodex-3® microcarriers (Fig. 3). After the temperature drop, more than 82.5% of cells were detached from pNIPAAm-grafted Cytodex-3®, whereas fewer than 8.5% of cells were detached from Cytodex-3® (Fig. 3b). This demonstrates that cells cultured on pNIPAAm-grafted microcarriers can be detached from the microcarriers by simply dropping the temperature, although the detachment efficiency is slightly lower than that induced by trypsin treatment.

hMBMSC detachment from two types of microcarriers by either trypsin treatment or temperature reduction from 37°C to 32°C. (a) Crystal violet staining of hBMMSCs after culture on two types of microcarrier and cell detachment treatment with either trypsin or temperature drop. Scale bars: 50 μm. (b) Percentage of hBMMSCs detached from microcarriers by trypsin treatment (black bars) or temperature drop (white bars). *p < 0.01 (n = 3).
Viability and Apoptotic Activity of Cells Detached From pNIPAAm-Grafted Microcarrier
Cell detachment from both types of microcarrier by trypsin treatment resulted in death of a few cells (Fig. 4a). In contrast, dead cells were rarely observed when cells were detached from pNIPAAm-grafted microcarriers by temperature reduction. Trypsin treatment caused death in 18.2± 1.1% of cells, whereas the temperature drop significantly reduced the level of cell death to 7.8 ± 1.3% (Fig. 4b). Furthermore, the TUNEL assay revealed that cell detachment by temperature reduction resulted in a significantly lower level of cell apoptosis than that induced by trypsin treatment (Fig. 4c, d).
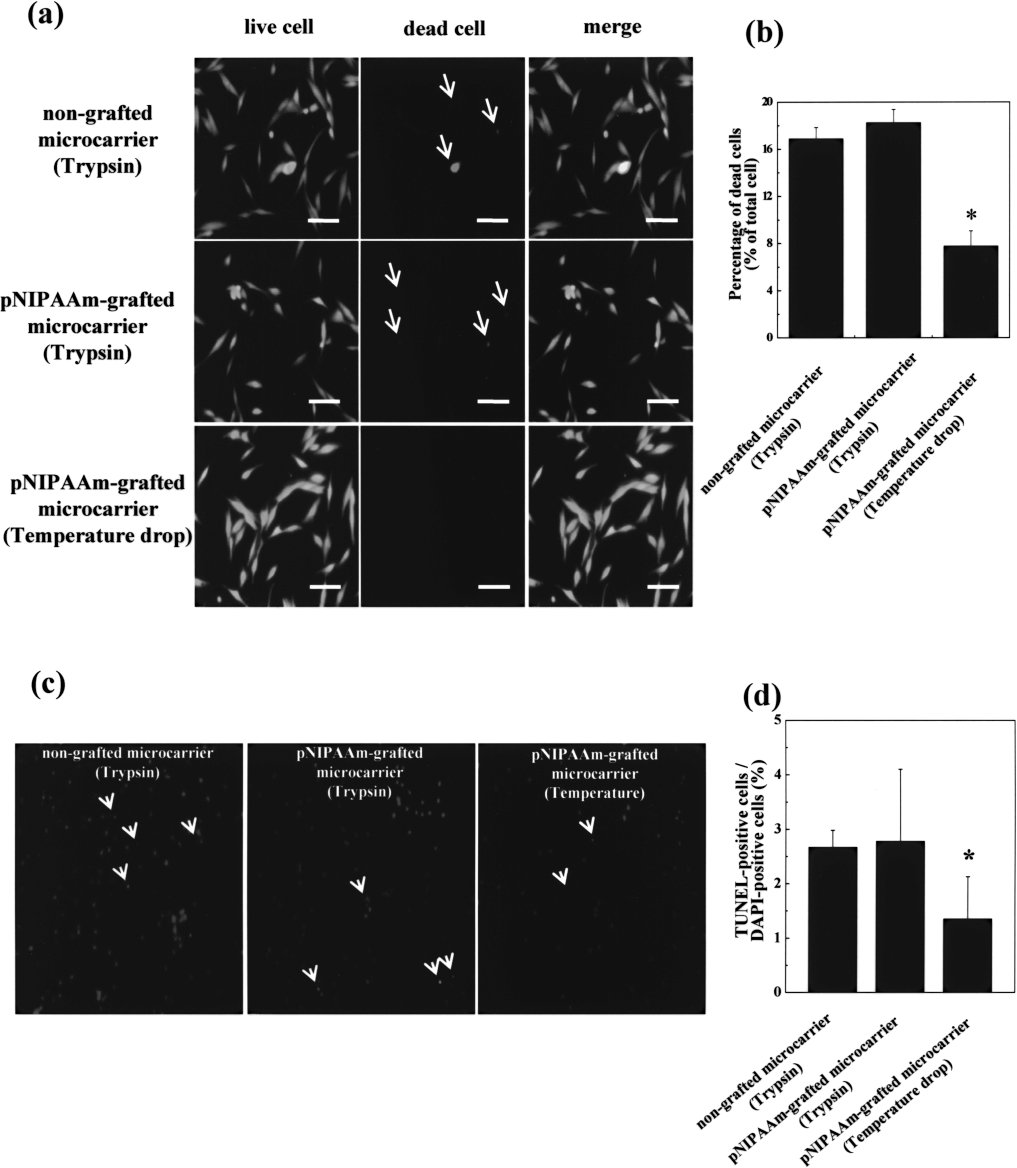
Viability and apoptotic activity of hBMMSCs detached from two types of microcarriers with either trypsin treatment or temperature drop and cultured on tissue culture dishes for 1 day. (a) Live and dead (FDA/EB) cell assay. Live cells are stained green (FDA) and dead cells are stained red (EB, arrows). Scale bars: 100 μm. (b) Percentage of dead cells as determined by FDA/EB assay. *p < 0.05 compared with any other group. (c) Apoptotic activity determined by TUNEL assay. DAPI-stained nuclei are blue, and TUNEL-stained apoptotic cells are red (arrows). (d) Percentage of TUNEL-positive cells. *p < 0.05 compared with any other group.
ECMs on Cells Detached From pNIPAAm-Grafted Microcarrier
To determine whether cell detachment by temperature change is less invasive to ECMs on the detached cells compared to that by trypsin treatment, fibronectin and laminin on cells detached from both types of microcarriers by either trypsin treatment or temperature change were analyzed. Immunocytochemical examinations showed that cells detached by temperature change contained larger amounts of fibronectin and laminin than cells detached by trypsin treatment (Fig. 5). Western blot analysis showed that fibronectin and laminin on cells detached by temperature change remained intact while those on cells detached from both types of microcarriers by trypsin treatment were disrupted (Fig. 5b).
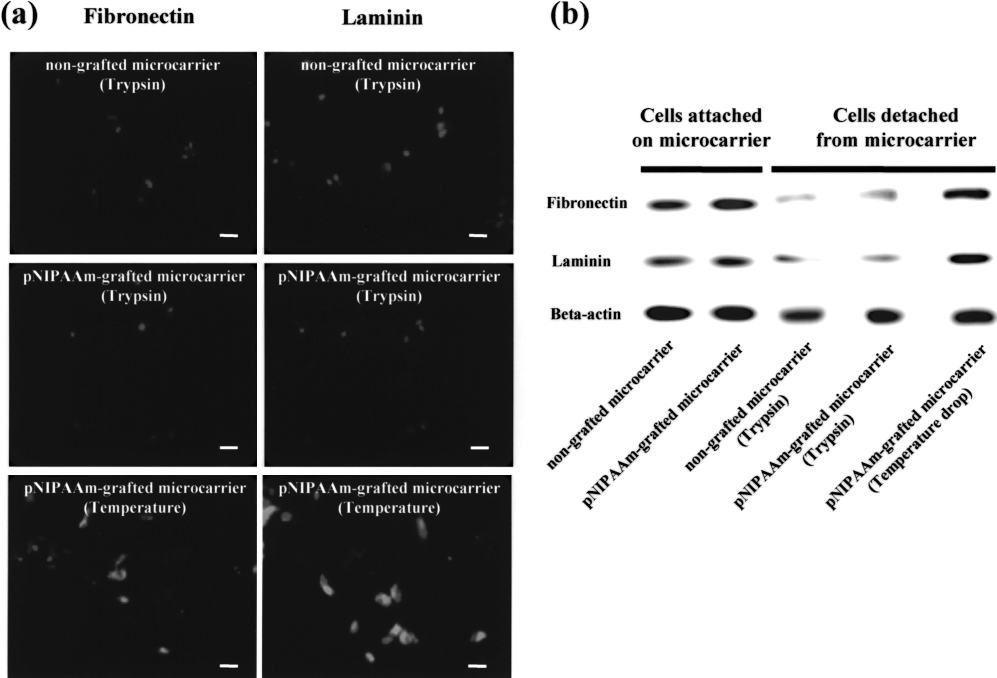
Fibronectin and laminin on cells detached from two types of microcarriers by either trypsin treatment or temperature change after 5 days of suspension culture. (a) Immunocytochemical staining for fibronectin (red) and laminin (green) on hBMMSCs detached from two types of microcarriers by either trypsin treatment or temperature change and allowed to attach to slide chambers for 30 min. Nuclei were counterstained with DAPI (blue). (b) Western blot analysis for fibronectin and laminin on hBMMSCs before and after detachment from two types of microcarriers by either trypsin treatment or temperature change.
Disscussion
Large numbers of cells (e.g., stem cells for cell therapy and transformed mammalian cells for industrial recombinant protein production) are essential for therapeutic applications, for example. Use of bioreactors combined with microcarriers is one of the most powerful cell expansion techniques developed to date, and can enormously increase cell numbers within a short period of culture time. However, cell harvest or scale-up using conventional microcarriers requires treatment with proteolytic enzyme followed by centrifugation and resuspension of cells to remove remaining enzyme. Throughout these steps, cells are damaged directly by proteolytic enzymes and by physical stress from centrifugation and resuspension (13). The pNIPAAm-grafted microcarrier developed in the present study does not require enzyme treatment; therefore, it simplifies the cell harvest or scale-up step and cell detachment from the microcarrier is much less detrimental to cells.
Cells cultured on pNIPAAm-grafted microcarriers detach from the surface due to a change in the hydrophobicity of pNIPAAm at the LCST (32°C). At temperatures higher than the LCST, thermosensitive pNIPAAm is hydrophobic and efficiently adsorbs cell adhesive proteins such as fibronectin so that cells can adhere on the proteins (16). At temperatures below the LCST, pNIPAAm becomes hydrophilic and hydrated. Cell adhesion proteins and ECMs that have been secreted from the cultured cells and adsorbed onto pNIPAAm may de-sorb from the hydrated pNIPAAm surface, leading to cell detachment.
The pNIPAAm conjugated on the surface of microcarriers did not affect cell adhesion and growth on the microcarriers (Fig. 2). This indicates that the affinity of the pNIPAAm polymer is equivalent to that of the denatured collagen coat on Cytodex microcarriers, and is in accordance with a previous study showing that pNIPAAm did not significantly affect cell adhesion. Furthermore, another study showed that primary bovine aortic endothelial cells adhere, spread, and grow well on a pNIPAAm-grafted surface (9), indicating that other cell types can be directly cultured on pNIPAAm without specific modifications. pNIPAAm coating on microcarrier may not affect the multipotency of BMMSCs cultured on microcarriers, because BMMSCs cultured on pNIPAAm-coated microcarriers had similar cell growth and better viability and ECM preservation as compared to BMMSCs cultured on plain microcarriers. A study reported that BMMSCs undergo progressive replicative aging and osteogenic differentiation during culture at subconfluency (2). However, pNIPAAm coating on culture surface may not affect these phenomena.
Cell detachment by temperature change significantly improved viability and decreased apoptotic activity of the detached and reattached cells compared with detachment by trypsin treatment (Fig. 4). This is probably because the temperature change causes less disruption to cell–cell interactions and deposited ECM than trypsin treatment. A previous study showed that detachment of cell sheets cultured on pNIPAAm-grafted culture dishes by temperature change preserved cell–cell interactions and deposited laminin much better than detachment by proteolytic enzyme treatment (13). The inevitable disruption of deposited ECMs in cells detached by proteolytic enzyme treatment often results in decreased grafting efficiency when the cells are transplanted for therapeutic applications (13). To improve grafting efficiency, rat BMMSCs were cultured on gelatin microcarriers and the cell-adherent microcarriers were directly transplanted into subcutaneous sites of rats without cell detachment (4). Direct transplantation without proteolytic enzyme treatment resulted in higher cell viability and lower apoptosis than transplantation of cells detached by trypsin treatment due to reduced damage to the cells and ECM; however, the transplanted microcarriers might cause inflammation, immune responses, or other unknown side effects. pNIPAAm dose not degrade in vitro and is considered as a nontoxic, biocompatible material (6). In contrast, pNIPAAm can degrade in vivo to monomers by the action of hepatic glutathione S-transferase in the cytosol, and the main metabolite is identified as acrylamide in the body (14). The acrylamide was shown to produce neurotoxicity and testicular toxicity after administration to mice (14). In the present study, pNIPAAm-coated microcarriers are not implanted into the body and used in vitro as a cell culture substrate, avoiding possible toxicity problem related to pNIPAAm.
The pNIPAAm-grafted microcarrier also simplifies the processes of cell harvest and scale-up. Cell harvest by proteolytic enzyme treatment is complicated and labor intensive, because it includes steps of enzyme treatment, enzyme deactivation, and centrifugation. Furthermore, extensive washing of harvested cells to remove residual enzyme activity is required for clinical use of the harvested cells. Cell harvest by temperature change avoids these complications and has great advantages for rapid, large-scale cell expansion. Large quantities of cells are required for clinical and industrial applications such as cell therapy and recombinant protein production. Because cell detachment from microcarriers by temperature change is less harmful to cells than enzyme treatment, cells can grow exponentially at a higher rate after passage or scale-up, and therefore a shorter time is required to expand cells. Furthermore, used pNIPAAm-grafted microcarriers can be recycled continuously, because the gel-sol translation of pNIPAAm involves a reversible thermosensitive mechanism.
Conclusion
A simple temperature change successfully induced cell detachment from pNIPAAm-grafted microcarriers, and was less harmful to the detached cells than trypsin treatment. pNIPAAm grafting on the microcarrier surface did not affect cell adhesion and growth on the microcarriers. pNIPAAm-grafted microcarriers could be an advantageous culture system for in vitro large-scale cell expansion, because this system can significantly save labor and simplify the culture process. Furthermore, this system allows cellular microenvironments to be kept intact after cell harvest, which would significantly enhance viability and therapeutic efficacy of the cells upon implantation compared with cell harvest by proteolytic enzyme treatment. Thus, this system has potential value in a variety of biological and clinical applications that demand a large amount of cells without damage to cells and associated proteins, including cell therapy and tissue engineering.
Footnotes
Acknowledgment
This work was supported by the Korea Research Foundation Grant funded by the Korean Government (MOEHRD) (KRF-2007-013D00029).
