Abstract
Islet isolation and purification using a continuous density gradient may reduce the volume of tissue necessary for implantation into patients, therefore minimizing the risks associated with intraportal infusion in islet transplantation. On the other hand, the purification procedure might result in a decreased number of islets recovered due to various stresses such as exposure to cytokine/chemokine. While a Ficoll-based density gradient has been widely used in purification for clinical trials, purification with iodixanol (OptiPrep) has been recently reported in islet transplant series with successful clinical outcomes. The aim of the current study was to compare the effects of the purification method using OptiPrep-based and Ficoll-based density gradients. Human islet isolations were performed using a modified automated method. After the digestion phase, prepurification digests were divided into two groups and purified using a semiautomated cell processor with either a continuous Ficoll- or OptiPrep-based density gradient. The quantity, purity, viability, and cellular composition of islet preparations from each group were assessed. Cytokine/chemokine and tissue factor production from islet preparations after 48-h culture were also measured. Although islet purity, postpurification IEQ, islet recovery rate, FDA/PI, and fractional β-cell viability were comparable, β-cell mass after 48-h culture significantly improved in the OptiPrep group when compared to the Ficoll group. The production of cytokine/chemokine including IL-1β, TNF-α, IFN-γ, IL-6, IL-8, MIP-1β, MCP-1, and RANTES but not tissue factor from the OptiPrep group was significantly lower during 48-h culture after isolation. Each preparation contained the similar number of ductal cells and macrophages. Endotoxin level in both gradient medium was also comparable. The purification method using OptiPrep gradient media significantly reduced cytokine/chemokine production but not tissue factor from human islet preparations and improved β-cell survival during pretransplant culture. Our results suggest that the purification method using OptiPrep gradient media may be of assistance in increasing successful islet transplantation.
Introduction
The islet isolation procedure consists of a mechanically enhanced enzymatic digestion of the pancreas, which allows dissociation of the islets from the surrounding acinar tissue (39). The separation of islets from other pancreatic tissue is performed by density gradient purification. This procedure reduces the volume of tissue implanted into the patients, therefore minimizing the risks associated with intraportal islet infusion, such as increased portal pressure and thrombosis (11). On the other hand, the purification procedure might result in decreased numbers of islets recovered. Islets are exposed to various stresses during the purification procedure that may cause cellular damage and functional impairment and eventually lead to an overall reduction of the viable islet mass engrafting and to poor clinical outcomes (1,2,8,14,27,36,40,42).
Continuous Ficoll-based density gradient purification method (FPM) with top loading using a semiautomated computerized COBE-2991 cell processor is considered the gold standard method at the present time (15–17,29,31,35,45). It takes advantage of the differential densities of acinar and islet tissues, which are separated by centrifugation on a density gradient. However, the density gradient itself is likely harmful for islets (43,44,49). Exposure to the sucrose-based Ficoll density gradient as well as enzyme and endotoxin during isolation may cause release of inflammatory mediators in vitro (10,25). This could contribute to the generation of inflammation, apoptosis, and immunologic attack after islet implantation in vivo. Therefore, minimizing the stress to islet cells during purification may be important for improving islet yield and quality.
The OptiPrep-based density gradient purification method (OPM) has been used for clinical islet transplantation at a limited number of centers (19,20,30) and was associated with high rate of success evaluated based on clinical outcomes (30). Ficoll is a neutral, highly branched, high mass, hydrophilic polysaccharide, which dissolves readily in aqueous solutions. OptiPrep is a 60% iodixanol in water solution. Iodixanol is a nonionic, iso-osmolar contrast medium used in patients for intravenous administration. There is currently no consensus on which density gradient is better suited for human islet purification. Insufficient data are currently available regarding the characteristics of islet preparations purified by these different gradients.
In this study, we compared FPM and OPM in terms of the efficiency of purification, islet yield, islet quality, cellular composition, and anti-inflammatory aspects of purified islet preparations. The results show that OPM significantly reduced the cytokine/chemokine; interleukin-1β (IL-1β), tumor necrosis factor-α (TNF-α), interferon-γ (IFN-γ), IL-6, IL-8, macrophage inflammatory protein 1β (MIP-1β), monocyte chemoattractant-1 (MCP-1), and regulated upon activation, normal T cell expressed and secreted (RANTES), but not tissue factor (TF) production from islet preparations, and it was associated with improved β-cell survival during pretransplant culture.
Materials and Methods
Human Islet Isolation and Purification
Five pancreata were recovered from deceased donors and preserved in University of Wisconsin solution (UW) (VioSpan; Bristol-Myers, Country Dublin, Ireland). Islets were isolated using a modified automated method at the Human Cell Processing Facility of the Diabetes Research Institute, University of Miami School of Medicine (39). Pancreata were digested using Collagenase NB1 with Neutral Protease NB (SERVA Electrophoresis, Islandia, NY) in three preparations, Collagenase V (Sigma-Aldrich, St. Louis, MO) in one, and Liberase MTF C/T (Roche, Indianapolis, IN) in one (Table 1). After enzymatic digestion, the pancreatic digest was equally divided into two groups. Prepurification yield (in islet equivalents, IEQ) was calculated by normalizing the islets to a standard islet diameter of 150 μm using the dithizone-stained islets method in both fractions (38). Islets were purified using either continuous Ficoll-based (Biocoll Separating Solution, density 1.108 g/ml, osmolality 320–373 mOsm/kg/H2O; Biochrom KG, Berlin, Germany) or OptiPrep-based (density 1.320 g/ml, osmolality 170 mOsm/kg/H2O; Axis-Shield, Oslo, Norway) density gradients, with a semiautomated cell processor (Cobe 2991; COBE Laboratories, Inc., Lakewood, CO) as described previously with modifications (4,6). Density gradients were made by mixing UW (osmolality 320 mOsm/kg/H2O) and either Ficoll or OptiPrep, and the density measured with a densitometry. After obtaining 110 ml of base medium (1.100 g/ml), a linear density gradient medium at 1.063–1.074 g/ml was created by mixing 140 ml of a light medium with 130 ml of a heavy medium during load to the Cobe 2991 spinning at 3,500 rpm. Up to 50 ml of digested tissue were suspended in 150 ml UW and top loaded onto the gradient medium. After 5 min of centrifugation, fractions were collected from the gradient. The first 200 ml or less was discarded, and the remainder was collected in 11 fractions of 30 ml each. Purified islets appeared between the 2nd and 10th fractions. After washing the purified islets, the purity was determined using the dithizone stain and postpurification IEQ was calculated for both groups. The recovery rate was calculated using the following formula: recovery rate = postpurification IEQ/prepurification IEQ x 100 (%).
Characteristics of Donors

BMI, body mass index; CIT, cold ischemic time.
Islet Cell Culture
Islet preparations from both experimental groups were cultured at a density of 3,000 IEQ/ml in Miami-defined Medium 1 (MM1, Mediatech Inc., Herndon, VA) (23) supplemented with 20 μg/ml ciprofloxacin (Cipro® I.V., Schering Co., Kenilworth, NJ) and 10 μg/ml heparin (Pharmaceutical Products, Schaumburg, IL) in a 37°C humidified incubator, 5% CO2 for the first day, followed by incubation at 227°C for anotherday.
Fluorescein Diacetate-Propidium Iodide (FDA/PI) Viability Staining
Islet preparations were assessed for islet cell viability after 48-h culture using cell membrane exclusion dyes as described previously (21). Briefly, a small aliquot of islets was obtained and transferred in phosphate-buffered saline (PBS) to a 10 × 35-mm counting petri dish. Fluorescein diacetate (FDA) and propidium iodide (PI) stock solutions were added to the sample at a final concentration of 0.67 and 75 μM, respectively. Living cells convert the nonfluorescent FDA into the green fluorescent compound “fluorescin,” a sign of viability, while nucleus of membrane-compromised cells will fluoresce red by PI, a sign of cell death. Using a fluorescent microscope, 50 islets were then assessed for cell viability by estimating the percentage of viable cells (FDA positive in green) versus the percentage of nonviable cells (PI positive in red) within each islet (FDA/PI). The mean values and standard deviation of viable cells were then calculated for each sample.
Fractional β-Cell and Ductal Cell Viability Assay
The fractional β-cell and ductal cell viabilities were determined as reported previously (21,22). Briefly, single cell suspensions were incubated for 30 min at 37°C in PBS without Ca2+ and Mg2+ with Newport Green PDX acetoxymethylether (NG; 1 mM) and tetramethylrhoda-mineethylester (TMRE; 100 ng/ml; both from Molecular Probes, Eugene, OR). Cells were incubated with mouse human anti-CA19-9 antibody (1:100; Novocastra, UK) for 15 min followed by goat anti-mouse antibody (Alexa-647). After washing, cells were stained with 7-aminoactinomycin D (7-AAD; Molecular Probes), a marker of cell death. Cell suspensions were analyzed (minimum 3.0 × 104 events) using a FACScalibur cytometer with the CellQuest-pro software (Becton Dickinson, Mountain View, CA). We have evaluated apoptosis selectively in the NG bright (mainly β-cells) and CA19-9+ (mainly pancreatic ductal cells) cell populations, respectively. After counting, dead cells (7-AAD-) were excluded from further analysis, live β-cells (NGbright) and PDC (CA19-9+) were analyzed for mitochondrial membrane potential (TMRE), which allows discriminating between viable (TMRE+) and apoptotic (TMRE) cells. The percentage of TMRE+ for each cell subset was determined based on the gate of TMRE unstained samples.
Cellular Composition Assessment of Islet Preparation
The content of macrophages, β-cells, and ductal cells was determined using laser scanning cytometey (LSC/iCys) (CompuCyte, Cambrige, MA) as described previously (21,22). Dispersed cells were fixed on glass slides with 2.5% paraformaldehyde (Electron Microscopy Sciences, Washington, PA). After incubation with Protein Block (BioGenex, San Ramon, CA) at room temperature for 30 min to reduce nonspecific binding, cells were incubated at room temperature for 2 h with the following primary antibodies: mouse monoclonal antibody to c-peptide (1:100 dilution, Abcam Inc., Cambridge, MA), cytokeratin 19 (1:50; Dako North America, Inc., Carpinteria, CA), and CD68 (KP1, 1:100; Dako North America, Inc.). After washing in Optimax Wash Buffer (Bio-Genex, San Ramon, CA), cells were incubated at room temperature for 1 h with Alexa Fluor 488 goat anti-mouse IgG (1:200 dilution, Molecular Probes), Alexa Fluor 647 goat anti-mouse IgG (1:200 dilution, Molecular Probes) antibodies and 4′,6-diamidino-2-phenylindole (DAPI; 1:500 dilution, Molecular Probes). The percentage of macrophages, β-cells, and ductal cells was automatically evaluated with iCys software.
Calculation of Absolute β-Cell and Ductal Cell Mass
Absolute β-cell and ductal cell mass was calculated using the following formulas: absolute β-cell mass = β-cell content (%) x protein content (μg)x10−2 and absolute ductal cell mass = ductal cell content (%) x protein content (μg) x 10−2.
Measurement of Proinflammatory Cytokine and Chemokine Production
Two aliquots of islets (350 IEQ/ml) from both experimental groups were cultured in MM1 for 48 h. Concentrations of proinflammatory cytokines/chemokines in the supernatants were determined using Multi-Plex cytokine kits following the manufacturer's protocol (Bio-Plex; Bio-Rad Laboratories, Hercules, CA). These included IL-1β, TNF-α, IFN-γ, IL-6, IL-8, MIP-1β, MCP-1, and RANTES. Aliquots of islets were lysed with lysating buffer (Bio-Plex; Bio-Rad Laboratories) in order to determine total protein. Total protein was measured with an assay kit (BCA Protein Assay Kit, PERBIO, Rock-ford, IL). The amount of cytokines/chemokines was normalized by total protein content (mg) (22,32). The concentrations of proinflammatory cytokines/chemokines in the supernatants were measured in duplicate for each islet aliquot and the average values were used for statistical analysis.
Measurement of Tissue Factor Production
Islet aliquots (350 IEQ/mL) from each group were cultured in MM1 for 48 h. Recovered islets were then homogenized and TF was measured by a conventional colorimetric ELISA kit (Imubind Tissue Factor, American Diagnostica, Greenwich, CT). The TF measurements were normalized by total protein of islet aliquots (24).
Endotoxin Assay in Density Gradients
To evaluate endotoxin contamination in density gradients, endotoxin in four lots of Ficoll and two lots of OptiPrep, which were used for the experiments, was measured using Endosafe®-PTS Portable Test System following the manufacturer's protocol (Charles River Laboratories, Kingston, NY).
In Vivo Assessment of Islet Potency in the Diabetic Nude Mouse Model
Animal protocols were approved by IACUC. Athymic nu/nu mice (Harlan Laboratories, Indianapolis, IN) were rendered diabetic with streptozotocin (200 mg/kg, single IV administration; Sigma). Diabetes occurrence (nonfasting glycemia >300 mg/dl) was monitored on peripheral blood (OneTouch Ultra2; Lifescan, Milpitas, CA). Human islets (1000 IEQ) were transplanted under the left kidney capsule of diabetic immunodeficient mice as described previously (39). After transplantation, nonfasting blood glucose values were assessed to determine reversal of diabetes (defined as stable nonfasting blood glucose <200 mg/dl). Nephrectomy of the graft-bearing kidney was performed to exclude residual function of the native pancreas (39).
Statistical Analysis
Data are expressed as mean values ± SEM for parametric samples and as median (quartiles) for paired nonparametric samples. Data were analyzed using SAS 9.1.3 software and Prism software for descriptive statistics and data plotting. Comparisons were assessed using Student's t-test for parametric samples and Wilcoxon signed-rank test for paired nonparametric samples. Statistical significance was considered for one-sided values of p < 0.05. This one-sided significance level requires that any outcome variables that are found to be significantly different between OptiPrep and Ficoll purified islets demonstrate improvement in each of the paired samples obtained from the five pancreata.
Results
Characteristics of Donors
The comparisons of purification methods with different density gradients were performed using five independent human pancreata. The donor characteristics of pancreata used in this experiment are shown in Table 1. Average age was 41.2 ± 7.2 years, body mass index (BMI) was 24.8 ± 2.1 kg/m2, pancreas size was 86.4 ± 14.5 g, and cold ischemic time (CIT) was 744 ± 127 min.
Purification Outcomes Using Ficoll or OptiPrep-Based Density Gradients
There was no significant difference in postpurification islet yields between the OPM and FPM (120,268 ± 18,320 vs. 132,560 ± 22,745 IEQ, respectively, p = N.S.) (Table 2). The recovery rate, which was calculated by the formula postpurification IEQ/prepurification IEQ, was also comparable within groups (OptiPrep vs. Ficoll: 52.9 ± 12.0% vs. 49.4 ± 4.5%, respectively, p = N.S.). The islet purity in OPM was lower than FPM, but this variable did not achieve statistical significance (OptiPrep vs. Ficoll: 76.3 ± 11.1% vs. 81.9 ± 9.3%, p = 0.060). Collectively, these results indicate that both methods had a similar efficiency in terms of islet purification.
Purification Outcome by OptiPrep- or Ficoll-Based Density Gradient

Statistical method utilized Student's t-test.
Viability Assessment of Islets Purified Using OptiPrep- or Ficoll-Based Density Gradients
The viability of islet preparations from each experimental group was assessed by FDA/PI, which is currently used as one of the product release criteria for clinical islet transplantation. There was no significant difference between the two groups (OptiPrep vs. Ficoll: 91.9 ± 3.1% vs. 93.5 ± 2.3%, p = N.S.). Moreover, the fractional β-cell viability assessed by flow cytometery also showed no significant differences between the experimental groups (OptiPrep vs. Ficoll: 60.6 ± 5.3% vs. 67.3 ± 4.1%, p = N.S.).
Cytokine/Chemokine and TF Production From Islet Preparations Purified Using OptiPrep- or Ficoll-Based Density Gradients
To investigate the anti-inflammatory effects of the density gradients on human islet preparation, cytokine/chemokine production in the supernatant of islet preparations cultured for 48 h was evaluated. The production of IFN-γ, TNF-α, IL-1β, IL-6, IL-8, RANTES, MCP-1, and MIP-1β in the OPM group was significantly decreased when compared with the FPM group (Fig. 1, Table 3). Tissue factor levels in islet preparations were not significantly lower with OptiPrep vs. Ficoll [median (quartiles): 1,879.5 (1,037.3–5,728.0) vs. 5,181.3 (3,241.5–8,842.5) pg/ml, p = N.S.].
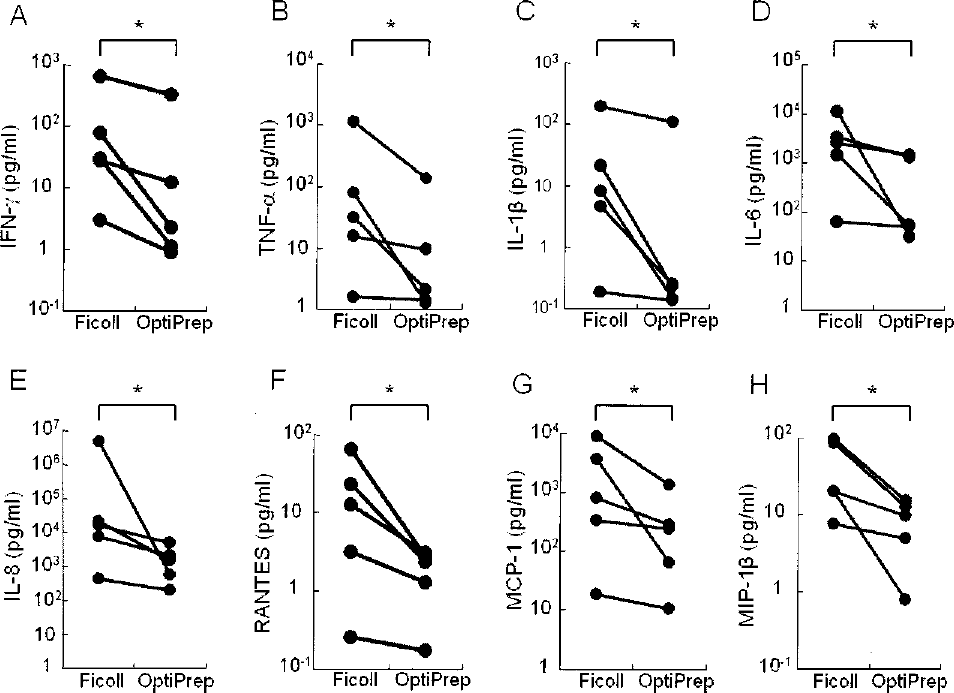
Cytokine/chemokine production from human islet preparations purified uisng Ficoll- or OptiPrep-based density gradients. Prepurification digests were equally divided into two fractions. Islets were purified using either Ficoll-based or OptiPrep-based density gradients. After 48-h culture, cytokines/chemokines in the supernatant were determined using Bio-Plex in five independent experiments. The concentrations of IFN-γ (A), TNF-β (B), IL-1β (C), IL-6 (D), IL-8 (E), RANTES (F), MCP-1 (G), and MIP-1β (H) were shown. The values were normalized by the protein content of islet preparation in each group. *p < 0.05.
Cytokine/Chemokine Concentration After 48-h Culture

IFN-γ, interferon-γ TNF-α, tumor necrosis factor-α; IL-1β, interleukin-1β; IL-6, interleukin-6; IL-8, interleukin-8; RANTES, regulated upon activation, normal T cell expressed and secreted; MCP-1, monocyte chemoattractant-1; MIP-1β, macrophage inflammatory protein 1β. Statistical method utilized Wilcoxon's signed-rank test.
Endotoxin Contamination in Gradients
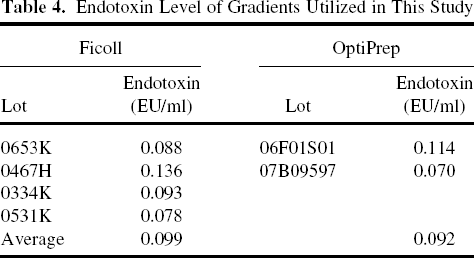
It is known that endotoxin contaminant in gradient medium could increase proinflammatory reactions in the islet preparation. To examine endotoxin contamination in each density gradient, endotoxin levels were measured in four lots of Ficoll and two lots of OptiPrep, which were used for these experiments. The level of endotoxin in all lots was low (Table 4). Thus, it was unlikely that endotoxin contamination in gradient medium could have caused the significant differences in cytokine/ckemokine production between the experimental groups in our studies.
Endotoxin Level of Gradients Utilized in This Study
Effects of Different Gradients on Ductal Cell Viability and Content
It is also known that pancreatic ductal cells contained in human islet preparations can produce higher amounts of cytokines/chemokines (22). Ductal cell viability and content after purification showed no statistically significant differences between experimental groups (OptiPrep vs. Ficoll: viability, 73.9±17.1 vs. 79.6±12.7, p = N.S.; content, 18.5 ± 12.7% vs. 11.5 ± 6.1%, p = N.S.). The results indicated that the significant reduction of cytokine /chemokine production in the OPM group was not caused by the difference of viable ductal cell numbers contained in the islet preparations.
Macrophage Population in Islet Preparation
Although macrophage (CD68+) numbers contained in human islet preparations are small, they could produce a substantial amount of cytokine/chemokine (32). Therefore, they were also evaluated by immunocytofluorescence analysis using LSC/iCys. The population of CD68+ cells within islet preparations was comparable between OPM and FPM groups (0.87 ± 0.28% and 0.78 ± 0.04%, respectively, p = N.S.).
Assessment of Islets Purified by OptiPrep- or Ficoll-Based Density Gradients After 48-h Culture
In the clinical settings, islet preparations are usually cultured for 48–72 h before transplantation. Islet cell loss during pretransplant culture in the clinical settings was evaluated. Although fractional β-cell viability after 48-h culture showed no statistically significant differences between the experimental groups (OptiPrep vs. Ficoll: 57.6 ± 7.4% vs. 64.5 ± 5.1%, p = N.S.), β-cell survival during culture, calculated from cellular composition and protein content, significantly improved in the OPM group when compared to the FPM group [median (quartiles): 188.0 (49.5–436.7) vs. 142.8 (39.4–235.6) pg/ml, p < 0.05] (Fig. 2A). Ductal cell survival in the OPM group also significantly increased when compared to the FPM group [median (quartiles): 155.5 (29.7–262.0) vs. 62.2 (19.7–221.7) pg/ml, p < 0.05] (Fig. 2B). Islet function in vitro was compared with static glucose-stimulated insulin release. As previously reported (30), no significant difference between groups was observed (data not shown). To examine islet function in vivo, islet aliquots from each experimental group were recounted and transplanted under the kidney capsule of diabetic immunodeficient mice. There was no significant difference between the two groups in terms of the proportions of diabetes reversal (Ficoll vs. OptiPrep: 3 out of 4 vs. 2 out of 4, p = N.S.). The results indicated that OPM significantly improved β-cell and ductal cell survival during pretransplant culture without deteriorating islet function.

Assessment of absolute β-cell and ductal cell mass in human islet preparations purified with Ficoll- or OptiPrep-based density gradients. After 48-h culture, β-cell and ductal cell content in islet preparation was determined using laser scan cytometer (LSC/iCys). Absolute β-cell (A) and ductal cell (B) mass were calculated. *p < 0.05.
Discussion
The current modified automated islet isolation method creates substantial stress to the islet cells, which not only triggers apoptosis and necrosis, but also induces proinflammatory cytokine/chemokine production. It is known that cytokine/chemokine production from human islet preparations varies widely. Many cytokines/chemokines may be detrimental to islet engraftment. In fact, IL-1β, IFN-γ, and TNF-α induce apoptosis in human islet cells (13,18), and the elevation of those cytokines is confirmed in the mouse islet transplantation model (9). Blocking MCP-1 prevents allograft rejection in islet transplantation using the mouse model (28) and a low level of MCP-1 production from human islet preparations is associated with a better clinical outcome in islet transplantation (37). The β-cell death associated with a nonspecific inflammation seems to be mediated by TNF-α and IL-1β in the grafted islets (7). Moreover, IL-1β has been reported to injure islets in patients with type 1 diabetes mellitus (5). Administration of drugs that inhibit cytokine action results in improved islet graft function after transplantation (47). Thus, regulating cytokine/chemokine production may favor islet transplantation outcomes. Our current data extend previous pilot studies performed on a smaller sample size (33) demonstrating that OPM significantly reduced proinflammatory cytokine/chemokine production from human islet preparations without deteriorating the islet cell viability and potency or altering cellular composition. In addition, OPM significantly improved the viable β-cell survival during pretransplant culture.
Even though improved islet transplantation protocols are employed, successful islet transplantation is achieved using islets generally obtained from multiple donors (16,29,45,46). Hering et al. have reported successful islet transplantation outcomes using islets isolated from a single donor (19). All eight recipients achieved insulin independence, and five of these patients remained insulin free for more than 1 year. In this protocol, islet preparations were purified using the OptiPrep-based gradients. Moreover, Matsumoto et al. have reported successful islet transplantation in Japan from pancreata of donors after cardiac death using OptiPrep-based gradient purification combined with ET-Kyoto solution (30).
In order to achieve reproducibly an ideal donor to recipient ratio of 1:1 in clinical islet transplantation, major efforts are currently being focused toward the standardization of islet cell processing among centers, including those participating in the National Institute of Health sponsored Clinical Islet Transplant (CIT) consortium, which is currently involved in the first Phase III trials of islet transplantation. OPM is currently being utilized in all islet cell processing centers in the CIT consortium.
There has been a steady progress in the area of islet cell purification in recent years thanks to the introduction of the semiautomated computerized purification method in the early 1990s (4). Density gradient centrifugation using the semiautomated computerized COBE-2991 cell processor is the most effective method to purify human islets because it allows the processing of a large amount of human pancreatic digest in a relatively short time (4,6,12). Continuous gradient purification with top loading of the pancreatic digest is currently considered the gold standard method for clinical and research preparations at the present time. The Ficoll-based density gradient purification method has been widely used for human islet purification in most islet processing centers (16,17,29,31,35,45). The OptiPrep-based density gradient purification method is more recently being used in a limited number of centers. Optiprep is a 60% iodixanol in water solution. Iodixanol is a nonionic, iso-osmolar contrast medium used in patients for intravenous administration. Iodixanol has a lower viscosity than Ficoll so the islets should suffer less force. In terms of the efficiency of islet purification, comparable results were obtained regarding purity, postpurification number, and recovery rate in both FPM and OPM in our study. We performed purification using OPM and FPM in parallel using aliquots of the pancreatic digest obtained from the same human islet preparations, an experimental design that allows for side-by-side comparisons and reducing the confounding elements of inter-experimental variability. Notably, our results did not concur with a previous report concluding that a higher amount of postpurification islet yields are obtained in OPM when compared with FPM (30). However, in that report, ET-Kyoto solution instead of UW was used for making the OptiPrep-based gradient. Further study will be needed to determine which solution combined with OptiPrep is more suitable for making the gradients for human islet purification.
Human islet preparations contain not only endocrine cells but also many non-endocrine cells including ductal, acinar (34), and inflammatory cells (32,48). The significant difference in cytokine/chemokine production between islet preparations purified using FPM and OPM might be caused by a different cellular composition of the islet preparations. The data regarding the cellular composition assessed using LSC/iCys clearly showed a similar percentage of ductal cells and macrophages as well as endocrine cells in the human islet preparations utilized in these studies. In addition, the viability of ductal cells showed no differences between the two groups. Our results indicate that the significant reduction of cytokine/chemokine production from islet preparations in the OPM group was not caused by the removal of specific cells that can produce a significant amount of cytokines/chemokines.
Endotoxins are part of the outer membrane of the cell wall of gram-negative bacteria and well known to cause proinflammatory reactions to cells. Jahr et al. reported that variable endotoxin concentrations were detected in the reagents used for islet isolation, including Ficoll, collagenase, and Liberase (25). Endotoxin contaminating Ficoll has been recognized as a potent inducer of IL-1β generation by human peripheral blood mononuclear cells (25). The Ficoll we used in the present study was tested and determined to be endotoxin free and showed comparable levels as that measured in OptiPrep. Thus, we concluded that Ficoll itself might stimulate cytokine/chemokine production from islet preparations.
The degree of islet loss in culture has been associated with the quality of islet preparations and donor/isolation variables (14,23,26). The prevention of islet loss during culture as well as improving islet yield and quality is an urgent issue to solve. In this study, we demonstrated OPM significantly improved β-cell survival during pretransplant culture without deteriorating islet quality. Recently, islet cell culture prior to transplantation is widely used in most islet transplantation centers (16,19,20,23). Islet cell culture offers several advantages ranging from ensuring the quality of islet preparations to possibly decreasing immunogenicity of allograft tissue (3). Islet cell culture also allows additional time for subject preparation and possible pretransplant interventions including immunosuppression (19,41). Induction of immunosuppression using anti-thymogloblin antibody or Campath-1H administered to recipients before transplant improves clinical outcome in islet transplantation (20). Development of efficient culture media formulations and of islet isolation protocols specific for human islets may improve the preservation of β-cell mass and quality during pretransplant culture, leading to the increase in the success rate of islet transplantations in the clinical setting.
In summary, OPM significantly reduce cytokine/chemokine production from islet preparations when compared to FPM. The OptiPrep-based density gradient purification method results in the improvement of β-cell survival during pretransplant islet cell culture. These results indicate that OPM may be of assistance in improving clinical outcomes, and that cytokine/chemokine profiling from islet preparations may be a useful means to evaluate newly developed islet isolation media or procedure for the preservation, purification, digestion, shipment, and pretransplant culture.
Footnotes
Acknowledgments
This work was supported in part by NIH-NCRR, GCRC MO1RR16587, NIDDK RO1-DK55347-IU42RR016603, 5R01 DK25802, ICR 5U42RR016603, and the Diabetes Research Institute Foundation. The authors are grateful to the members of the Human Cell Processing Facility, Preclinical Cell Processing Laboratory of the Cell Transplant Center, Clinical Islet Transplant Program, General Clinical Research Center, Imaging Core at the Diabetes Research Institute, Administrative Offices at the Diabetes Research Institute, and Organ Procurement Organizations for the continuous enthusiasm and support to our program.
