Abstract
Postisolation islet survival is a critical step for achieving successful and efficient islet transplantation. This involves the optimization of islet culture in order to prolong survival and functionality in vitro. Many studies have focused on different strategies to culture pancreatic islets in vitro through manipulation of culture media, surface modified substrates, and the use of various techniques such as encapsulation, embedding, scaffold, and bioreactor culture strategies. This review aims to present and discuss the different methodologies employed to optimize pancreatic islet culture in vitro as well as address their respective advantages and drawbacks.
Introduction
One of the most challenging and promising fields of regenerative medicine is the treatment of insulin-dependent diabetes mellitus. Islets secrete a variety of hormones, most importantly insulin, which is responsible for the regulation of blood glucose levels. Loss of β-cells through an autoimmune disease process results in type 1 diabetes. It is estimated that perhaps only about 500 thousand to 1 million islets would be needed to treat a patient with type I diabetes (97). With an islet averaging about 150 μm in diameter, this translates into a volume of 1–2 cm3; a volume considerably smaller than that of a whole pancreas. Transplanting just the islets would therefore result in a far simpler surgical procedure (77).
The currently accepted procedure for transplanting islets is referred to as the Edmonton protocol (104), which first involves the isolation of pancreatic islets from cadaveric donors, followed by their respective infusion into the patient's portal vein. A major shortcoming of this procedure includes the requirement of more than one (up to three) donors for each recipient. Also, following transplantation, the administration of various immunosuppressants is required. Furthermore, this procedure requires a few crucial steps, including successful isolation of the human islets from donors followed by the in vitro culturing of the islets in order to maintain functionality until transplantation can be performed. However, the development of a viable method for in vitro human islet culture that successfully mimics the islet microenvironment is hindered by the complexity of the multicellular islet. Moreover, the native islet microenvironment consists of large amounts of surrounding acinar tissue that provides an adapted protective milieu. This review addresses the challenges with postisolation pancreatic islet survival as well as the strategies involved in islet preservation, culture, and regeneration techniques along with their respective advantages and shortcomings.
Challenges
It has been shown that following human islet isolation, apoptosis occurs involving mainly the β-cells (89). This is caused by several factors, including loss of vasculature and eventual hypoxia, because islets are highly innervated and vascularized (75,78,83). Further studies have shown that islet isolation exposes the islet to a variety of cellular stresses and disrupts the cell–matrix relationship, leading to apoptosis (99). Results have shown that immediately after isolation, the peri-insular basement membrane was absent, giving rise to pykontic nuclei and apoptotic bodies, accompanied by elevated proapoptotic p38 and C-Jun N-terminal kinase (JNK) activity relative to prosurvival extracellular signal-regulated kinase (ERK1/2) activity (99). Therefore, it is evident that in order to arrive at a successful strategy to maintain islet survival and functionality, the islet–matrix relationship must be reestablished.
As a starting point it is important to understand which factors are implicated in the apoptosis process that occurs shortly following islet isolation and whether they can be controlled to avoid cell death. Some studies have confirmed the increased activity of JNK and p38 following isolation, which can be stimulated by cytokines such as tumor necrosis factor-α (TNF-α) and interleukin-1β (IL1β); these proapoptotic activities can be reduced by inhibiting p38 activation as well as the addition of insulin to the culture medium (87). Further studies have focused on therapeutic activities aimed at the inhibition of these specific cell death mechanisms. Aikin et al. have demonstrated that activation of phosphoinositide 3-kinase/Akt (PI3K/Akt) suppresses the JNK pathway in islets, thus introducing a cross-talk mechanism that can possess a very important antiapoptotic consequence (1). For further preservation strategies leading to improved culture conditions, both short and long term, whether for prolonged tissue culture or transplantation purposes, it is vital to understand the complex nature of islet function, vasculature, and physical support requirements that arise due to the highly differentiated state of islets.
Suspension Culture
The simplest and most investigated approach to islet preservation and culture in vitro is through alteration in culture conditions, including temperature (58) and media composition (18,23,35,39). Several studies have also involved the administration of various growth factors and compounds in order to enhance the suspension culture of islets such as glutamine compounds (14), human albumin (65), insulin (22), and sericin (85). These investigations have been the focus of many laboratories for the purpose of short-term culture for transplantation as well as long-term culture in vitro (103). There has been much progress and thoroughly investigated reviews on the subject matter (38,79). A summary of various media/supplements for human islet culture are presented in Table 1.
Various Media Supplements for In Vitro Islet Culture

Adapted from Murdoch et al. (79).
Studies performed with nonhuman islets.
Although the general premise behind suspension culture techniques is the sustenance of the islet three dimensional structure, which is lost if subjected to traditional attachment culture, it still does not compensate for the loss of islet basement membrane support and the subsequent loss of functionality and viability that ensues. The ultimate result is diminished integrin expression followed by decrease in phenotypic characteristics leading to apoptosis (88). Therefore, it is important to not only improve the culture conditions in suspension culture, but to also couple these strategies to others that permit the reestablishment of their extracellular matrix (ECM) support as well as their natural physiological needs.
Culture on Ecm-Coated Surfaces
In terms of postisolation islet survival, several studies have focused on restoring the ECM environment and determine the effect of cell–matrix and cell–cell interaction on survival. Some studies investigated the expression of integrins in order to better understand cell–matrix relations (112). It was found that human islets express integrins α3, α5, αV, and β1 with decreased density following the destruction of the peri-insular basement membrane following isolation (121). It has been found that insulin-like growth factor II (IGF-II), found within duct-conditioned medium (DCM), increases the survival of hamster islets suspended in collagen I matrices (52). Similar results have been found with rat islets cultured on plates treated with small intestinal submucosa (SIS), a natural ECM (127). Other studies have demonstrated positive rat islet response to collagen I, collagen IV, laminin, and treatment with anti-β1 antibodies or arginin-glycin-aspartic acid (RGD) peptides—all of which contribute to mimicking the ECM ligation (41,82,93). More specifically, integrins αvβ3 and αvβ5 have been shown to regulate adhesion and differentiation, while α3β1 and α6β1 are involved in the regulation of insulin secretion (21,56,96). This gives insight into the appropriate ECM proteins that are needed to bind these integrins and induce the appropriate signaling. Recent reviews have outlined the various studies investigating islet–ECM interactions and their possible effect on scaffold surface modification for posttransplant engraftment (84,112).
A recent study has investigated the effects of various ECM protein substrates on two-dimensional human islet culture, yielding results that demonstrated better adhesion on fibronectin collagen I, IV, and fibronectin with the latter maintaining structural, three-dimensional islet integrity (25). However, human islet phenotype during two-dimensional culture was eventually lost, although relative insulin gene expression was highest in islets cultured on collagen I and IV. Also, insulin release was highest on fibronectin, along with a decrease in SUR1 expression, while glucose metabolism, along with GLUT2 and GCK expression, was highest on collagen I and IV surfaces (25).
Another interesting approach involved the bioengineering of functional sheets of rat islets following culture on laminin-5 surfaces for transplantation (105). This novel cell-based therapy proved capable of maintaining in vivo islet functionality for more than 7 days posttransplantation into rats. Other strategies that involve islet culture on ECM substrates include the redifferentiation of insulin-secreting cells through the monolayer culture of cells expanded from pancreatic islets. Islets cultured on a flat, two-dimensional surface often spread and form a monolayer of hormone-negative cells. These cells may be differentiated into insulin-secreting, β-like, cell clusters through culture on Matrigel and supplemented with differentiation media containing insulin, transferrin, selenium, and linoleic acid BSA (64). Although promising, these insulin-secreting cells are only able to produce insulin at 34% of the native islet capability (64).
Islet Embedding
Several studies have focused on increasing the survival of islets in vitro through embedding in a solid matrix. Culturing in collagen I gels obtained from rat tail (71,122,131) and fibrin gels (5) have shown promising results in terms of prolonging survival. Islet embedding has also provided valuable insight into the regenerative properties of pancreatic islets, which can contribute to the efficacy of islet culture in vitro and in turn the survival and preservation of postisolated islets. A recent study has developed a novel technique in which canine islets were embedded in autologous plasma, supplemented with VEGF (43). The resulting coagulum was transplanted into the greater omentum, resulting in accelerated revascularization and modulation of the immune response.
This new approach to human islet culture in vitro has also been investigated for islet regeneration through de-differentiation to duct like structures, which are highly proliferative, followed by the redifferentiation back to functional islets, thus undergoing a process of islet neogenesis. This is the most attractive process to date with respect to islet regeneration. When isolated islets are suspended in type I collagen matrix or Matrigel with appropriate media that induces cyclic adenosine mono-phosphate (cAMP), the inner β-cells undergo apoptosis and give rise to a highly proliferative population of duct-like cells that grow as duct-like epithelial cysts (DEC) in a process that takes approximately 10 days (120). This differentiation is associated by a balance in prosurvival ERK1/2&Akt versus proapoptotic JNK and caspase 3 pathways (53). These DECs undergo differentiation to islet-like clusters when stimulated by the proneogenesis factor islet neogenesis associated protein (INGAP), but not glucagon-like peptide-1 (GLP-1) or exendin-4 secreatogue (67,68); further analysis revealed that these islet-like clusters are indistinguishable from freshly isolated islets and are glucose responsive (54).
Another recent model involving acinar transdifferentiation has successfully demonstrated that acinar tissue can be differentiated into DECs through collagen I embedding and supplementation with epidermal growth factor (EGF) and insulin. The resulting DECs could then be differentiated into functional islet-like clusters though INGAP stimulation (69) as previously described. However, this model must be further investigated in order to arrive at more conclusive information regarding feasibility and functionality of the generated islets.
Encapsulation
Islet encapsulation is a well-researched strategy that provides a means of culturing and delivering islets for transplantation purposes (6,59,78). This method involves creating a semipermeable environment around a group of islets, which provides appropriate mass and oxygen transfer as well as immunoprotection. This is accomplished by allowing the regulation of blood glucose levels through insulin while being impermeable to larger proteins and cells of the immune system (Fig. 1).
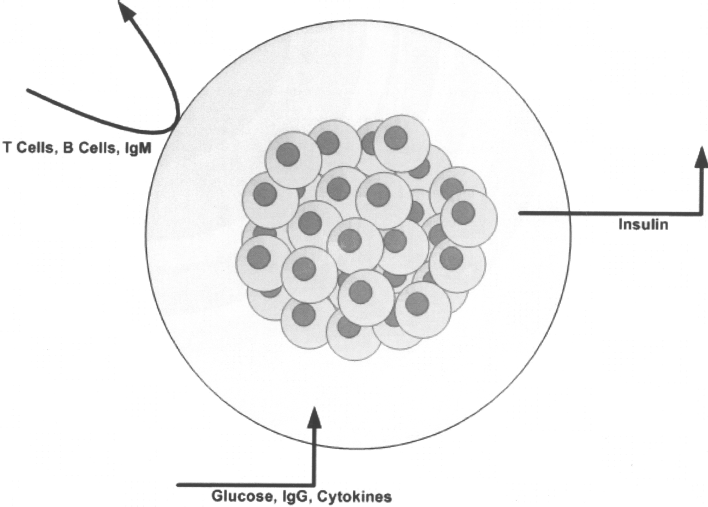
Principle behind islet encapsulation.
Some popular materials used for encapsulating islets include alginate and polysulphone (PS), which have been widely employed with relative success in protecting islets from host immune systems (28,31,57,73). Other materials include poly(ethylene glycol) (PEG) and poly-l-lysine (PLL) incorporated into alginate, which have been shown to enhance mass and oxygen transfer as well as islet viability in animal models. In order to further address the issue of islet culture and functionality in vitro, for later transplantation purposes, strategies have focused on incorporating factors such as VEGF (62,91,106,107,111), GLP-1 (48), and HGF (50) into the encapsulation biomaterial for reducing hypoxic stress (114). Other strategies aim at decreasing capsule size in order to increase surface area to volume ratio and in turn oxygen diffusion (6). Furthermore, ECM proteins have been incorporated as well for the maintenance of islet functionality both in vitro and in vivo such as passive adsorption and layer-by-layer deposition of collagen IV onto PS membranes (6). This potentially serves to exploit integrin/ECM interactions to better create a viable environment for the encapsulated islet populations.
Simple alginate capsules were shown to increase canine islet survival in tissue culture up to 3 weeks postisolation (61). Also, when these encapsulated islets were transplanted into diabetic mice, they exhibited improved survival and function, thus demonstrating positive effect of alginate both in vitro and in vivo. Another interesting procedure involved the encapsulation of individual rat islets by sol-gel SiO2, thus allowing for homogeneous distribution of encapsulation material on islet surface and control over the layer thickness (11). Transplantation into diabetic rats showed a prolonged restoration of normal glycemia levels as well as protection from immunological attack.
Another study has investigated the feasibility of alginate-polylysine spherical-bead microcapsules, which provide improved biocompatibility, mechanical strength, and chemical stability (110). In addition, they possess a large surface area, enhanced nutrition and oxygen supply, and controlled porosity, which is able to distinguish nutrients from immunoglobulins. These microcapsules also provide maximum protection from membrane failure, and direct injectability into the peritoneal cavity. Encapsulated islets were then injected into an insulin-dependent human male directly into the peritoneal cavity. Results have shown that insulin secretion from the transplanted cells was detected as early as 24 h after injection for up to 58 months. Furthermore, the patient displayed an increased energy level accompanied with no adverse effects.
Although islet encapsulation appears to provide an attractive solution to islet transplantation dilemmas, it does possess crucial drawbacks. Despite major improvements in the biomaterials used for encapsulation as well as the incorporation of various factors that enhance islet functionality, some issues need to be addressed. Most importantly, the limitations of long-term oxygen diffusion and nutrient transport as well as biocompatibility, viability and eventual hypoxia (31,117).
Scaffolds
Pancreatic islets in vitro require fixation within a solid matrix in order to promote survival and compensate for the absence of a peri-insular basement membrane; thus, culturing on a biodegradable scaffold provides a suitable alternative. However, not many studies have investigated this avenue of an islet culture platform for transplantation purposes. Scaffolds provide many distinct features including a high surface area to volume ratio, a rigid support of adhesion, high interconnectivity and pore geometries, as well as biodegradability to allow for tissue regeneration over time (Fig. 2).

Islet-seeded polymer scaffold.
The main problem with fabricating such a scaffold is that the islet is approximately 100–200 μm, which means that the scaffold pore sizes must be 5–10 times that diameter (63); thus, it is appropriate to investigate other fabrication methods such as rapid prototyping techniques that use computer-aided design (CAD) models to create the desired scaffold with controlled geometries (63,129). Conventional scaffold fabrication techniques, such as freeze-drying, do not allow for controlled pore structure; moreover, larger pore sizes required for islet culture would render the resulting scaffold unstable. Also, the use of harsh solvents and conditions limits the use and incorporation of growth factors (128). Another crucial point is the oxygen demand of islets; in the human body they make up 2% of the pancreatic tissue yet they need over 10% of the pancreatic blood flow (55). This stresses even more the importance of interconnectivity and the need for a well-defined pore structure that enhances porosity and interconnectivity.
Previous studies have successfully utilized microporous, biocompatible, and biodegradable poly(lactide-co-glycolide) (PLG) scaffolds in mouse models (10) as well as chitosan/poly(lactic-co-glycolic acid) (PLGA) scaffolds for islet culture and transplantation (33). Another recent study has demonstrated that rat islets can successfully grow and survive on polyglycolic acid (PGA) scaffolds that were constructed with 15-μm-diameter fibers, and pore sizes of ~100–150 μm (20). The seeded islets demonstrated enhanced viability, survival and insulin secretion. Further, PLGA scaffolds seeded with islet like cells, derived from stem cells, were shown to reverse hyperglycemia in diabetic mice (74).
Scaffolds also possess the ability to be modified successfully with ECM proteins, those that are lost during basement membrane destruction during isolation. These ECM components are crucial for mediating cellular interactions required for cell adhesion and growth in a scaffold setting (72). An interesting study has investigated the potential of nonwoven/sponge fabrics in a hyaluronan-based (namely benzyl ester of hyaluronan) scaffold, modified with fibroblast secreted ECM, for the culture of rat pancreatic islets (132). The ECM-modified hyaluronan scaffolds displayed improved biological properties and represented a suitable platform for the promotion of islet organization and proliferation. Another study has demonstrated the successful use of PLG scaffolds adsorbed with collagen IV, fibronectin, and laminin for culture of mouse islets (102). This demonstrated the efficacy of surface-modified scaffolds, which create a well-defined microenvironment suitable as a platform for extrahepatic transplantation. Furthermore, a recent study assessed the efficiency of fibroblastcontaining clotted blood plasma, referred to as a plasma–fibroblast gel, as a scaffold for subcutaneous islet transplantation in mouse models (90). Mouse islets embedded in the plasma–fibroblast gel displayed restored vascularization and successfully reestablished normoglycemia, as well as body weight in transplanted mice over a period of 60 days. Some shortcomings include the inability to effectively control scaffold geometries, such as pore sizes, coupled with the nonuniform distribution of islets within the scaffold construct during cell seeding. Moreover, most cell-seeded scaffolds have been cultured under batch conditions, as opposed to continuous culture configurations, thus not allowing for the continuous perfusion of media as well as effective waste removal and oxygen/mass transfer.
One of the problems with scaffold use for islet culture is the seeding efficiency. Cell seeding on scaffold in general has long been a factor in need of improvement. Optimally, cells must be immobilized in a uniform manner throughout the three-dimensional scaffold support. The seeding efficiency of a given method is a measure of the ability to immobilize cells in suspension within the scaffold pores, conventionally reported as the percentage of cells effectively seeded onto a scaffold. Traditionally accepted islet seeding methods have focused on using passive diffusion of islets into the scaffold (116); however, these methods are fairly unsuccessful in achieving the goal of efficient seeding to give a uniform islet density within the scaffold. Other approaches have been examined to enhance seeding efficiency, such as the use of spinner flasks for seeding chondrocytes onto PGA scaffolds (118). However, not many techniques have focused on pancreatic islets. This is partly attributed to the wide variance in islet size as well as the fragility of islets. An attractive alternative to passive diffusion is the use of centrifugation forces, developed by Yang et al. (130), coined as centrifugational cell immobilization (CCI). High seeding efficiencies of 40% were attained for hepatocytes seeded onto hydrophilic porous poly(vinyl formal) cubes. Further developments have been made to enhance efficiency and a more controlled approach was applied to cardiomyocytes seeded onto a hydrophilic alginate scaffold, achieving seeding efficiencies of up to 80–90% (26). Recently, a novel method, based on CCI principles, has been successfully applied to pancreatic islet by seeding cells onto a variety of PGA/PLA scaffolds, using centrifugal forces of 800–2000 rpm while maintaining islet survival (Rezania, A.; Xu, J.; Ghabrial, R. Seeding Pancreatic Cells on Porous Matrices. Patent No. WO/2005/005607; 2005).
Bioreactors
A very attractive method of culturing cells in vitro involves the use of tissue bioreactors for the purpose of emulating native physiological conditions such as temperature, pH, and pressure. Moreover, it allows for the continuous perfusion of fresh media in order to continually supply the tissue under culture. One of the most prominent and innovative approaches has been the NASA-developed bioreactor, or microgravity reactor (Fig. 3), which provides a continuous circular rotation of media and allows for cultured cells to be suspended in continuous free fall at a terminal velocity with the advantages of low hydrodynamic shear stress force and low turbulence coupled with high mass and oxygen transfer (119). This culture technique has been applied to singe-cell suspensions as well as immobilized cells, either encapsulated or within microcarriers, or seeded within scaffolds (42). This reactor configuration has been successful in culturing many cell types, including prostate and skeletal tissue, maintaining their morphology and promoting cell–cell interactions as well as enhanced proliferation and differentiation (19,76). This reactor configuration has also been applied successfully to islets derived from human patients that suffer from hyperinsulinemic hypoglycemia of infancy (PHHI), and has been shown to provide a unique environment that is conductive to the upregulation of endocrine expression (123). An alternate configuration of this system, which encourages continuous perfusion, has proven successful in maintaining human islet structural integrity for over 1 week in suspension (126).
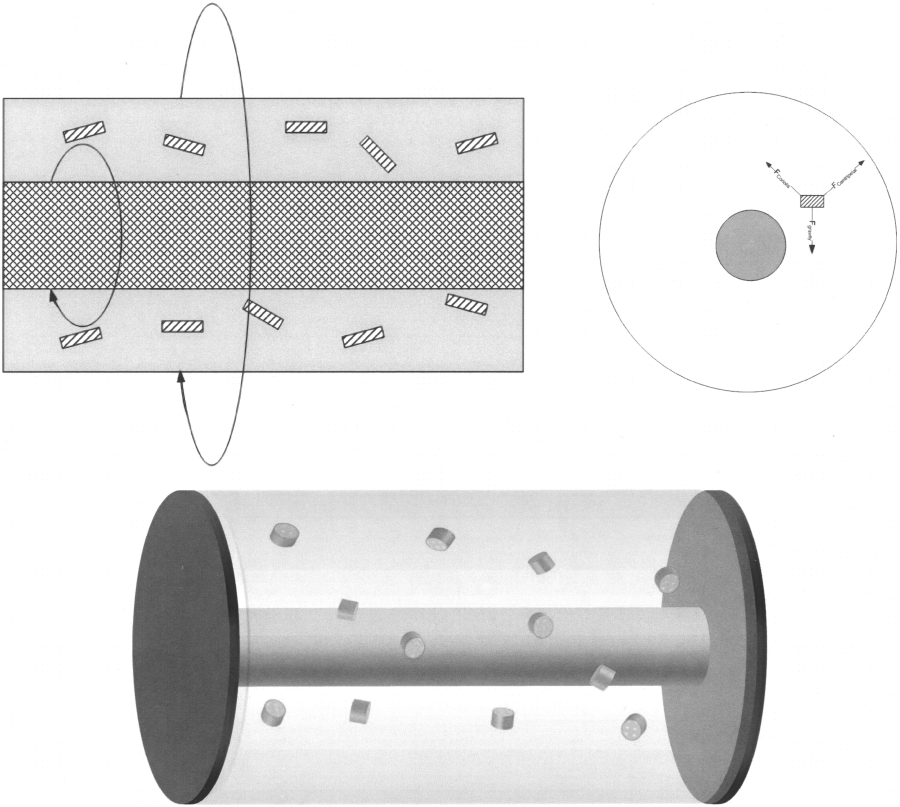
A microgravity, or rotating wall, reactor. There is an outer wall that rotates at the same speed as the inner silicon oxygenator. Cell-seeded scaffolds are suspended. A cross-sectional view shows forces acting on the constructs.
The microgravity reactor configuration has also proved successful in culturing mouse islets, preserving their functionality and morphology (100). Furthermore, rat and mouse islets cultured in microgravity also showed reduced immunogenicity and enhanced vasculature (100,109). Rutsky et al. compared islets cultured using a Rotary Cell Culture System (RCCS) and conventional culture dishes. While fresh islets survived approximately 15 days when transplanted into streptozotocin diabetic mice, their RCCS cultured counterparts survived over 100 days posttransplantation. Also, when compared to the conventional cultured islets, a significantly smaller number of RCCS islets were required to maintain euglycemia in the diabetic mice (115). The main drawback of islet culture within these bioreactors lies in the tendency of cultured islets to aggregate when cultured at higher densities required for viable human islet transplantation, thus increasing the risk of anoxic necrosis. However, a study has shown that human islets cultured under a rotational cell culture system, with a configuration known as the High Aspect Ratio Vessel (HARV) at high densities of 500–1500 islets/ml, exhibit decreased central necrosis, due to highly efficient gas exchange, and preserve their structural integrity as well as glucose-stimulated insulin release over a 10-day culture period (81). Moreover, human islet culture under rotary conditions was shown to be supported by the coculture of epithelial duct cells (80). More recently, Hou et al. have successfully demonstrated the use of the RCCS coupled with PGA scaffolds, which also addresses clumping concerns, for the culture of islets and their subsequent transplantation into streptozotocintreated diabetic Wistar rats. The islet seeded and rotary cultured scaffolds displayed healthy islet adhesion, induced normalized insulin production and blood glucose concentration as well as preserved islet functionality and morphology (49). Although not widely explored, this technology has proved to be very promising in the culture and preservation of islet function for transplantation purposes. Further improvements and experimentation into this technology, such as incorporating the use of solid supports for islet culture, namely microcapsules and scaffolds, are required in order to further enhance islet culturing techniques in vitro.
Implantable Chambers
An interesting alternative strategy to achieve islet preservation is through vascularized tissue-engineered chambers that promote survival and function of transplanted islets (51). The conventional method of islet injection into the portal vein (101,104), as well as microencapsulated islet transplantation (29), through the Edmonton protocol is associated with poor islet viability posttransplantation that are caused by hypoxia, susceptibility to ischemia-reperfusion injury within 48 h after transplantation (12,66), as well as vascular thrombosis (44), and metabolic changes such as liver steatosis (9). In addition, microencapsulation hinders the longevity of encapsulation islets in vivo through cellular overgrowth, despite various attempts to maintain long-term membrane porosity (30). A recent study has shown the viability of implanting tissue-engineered prevascularized pancreatic encapsulating devices (PPEDs) (45). The devices comprised of two layers of vacularized collagen gels sandwiching a central gel containing embedded islets, which were then implanted subcutaneously into immunodeficient mice. The implanted PPEDs enhanced islet survival and maintained intraislet endothelial cell structures following 28 days of culture (45). Another proposed strategy involves the development of a three dimensional silicone chamber that is inserted into the splenic, groin, or renal capsule vascular beds of mice. Matrigel-suspended mouse islets are then seeded into the chambers in order to quickly reestablish the ECM and vascularization. Recent studies have shown that hyperglycemia in mice can be treated through the seeding of islets into vascularized chambers, maintaining both long-term viability and functionality (51).
Alternative Strategies
Although many strategies have focused on islet preservation and culture techniques for the eventual purpose of transplantation, shortage in donors (24) has encouraged the investigation of an alternative route, namely islet regeneration from stem cells and other precursor cells (37). More regeneration strategies are being investigated that are focused on endocrine progenitor cells that have the capabilities of being induced into human adult pancreatic islets (40). Mesenchymal stem cells (MSC) have been investigated as potential therapy tool to improve long-term allogenic islet graft survival and decrease the requirement for immunosuppression due to the posttransplantation suppression of T helper cell (Th1) activation (108). Moreover, a greatly viable alternative transplantation site to the liver has been demonstrated through bone marrow engrafting, which promotes better euglycemia and induces compact morphology with minimal effects on bone structure (17). Acinar tissue is also being investigated for its ability to differentiate into duct-like structures, which are in turn differentiated into islet-like structures displaying similar functionality to freshly isolated islets (69). However, these alternative strategies discussed have not been shown to yield structures that fully resemble freshly isolated islets and are thus not very effective for the ultimate goal of transplantation, although they provide potential through further exploration and the advantage of regenerating islet-like structures from tissue that is readily available, unlike human pancreatic islets that are arduously isolated.
Discussion and Conclusions
Pancreatic islet transplantation is currently a reality that is being implemented through the Edmonton protocol. However, the shortcomings of this procedure include a poor viability of isolated islets as well as the need for immunosuppression therapy. Following isolation, the islet mass is greatly affected by apoptosis due to the harshness of the isolation procedure. Nearly half of the islet mass is lost during donor surgery, preservation, transport, and isolation; this leaves many patients with the necessity of a second islet cell infusion in order to achieve insulin independence (24). Furthermore, the cocktail of administered immunosuppressants give rise to many complications, in addition of those complications encountered when performing the transplantation procedure itself (86). Therefore, it is necessary to investigate avenues of postisolation islet culture, for as many as 2 months, in order to improve islet transplantation by allowing such procedures as donor matching as well as immunologic precondition of the recipients. Moreover, culture strategies aimed at preserving and improving islet functionality may serve to limit the requirement for many donor pancreases and even reach goals such as the use of a single pancreas for two recipients (86).
In order to arrive at a viable islet culture method, it is vital to develop a proper understanding of islet biology as well as emulate the basement membrane (BM) of native islet tissue through proper matrix support, adhesion proteins, and growth factors. There a few factors that hinder the successful regeneration of pancreatic islets in vitro for transplantation purposes. Firstly, in the process of culturing islets in vitro, whether for prolonging survival or inducing differentiation and regeneration, the culture media contains a cocktail of growth factors and proteins and thus it is not clear which factor induces the desired effects. Also, we are still unable to reemulate the ECM conditions of the pancreatic islet and restore the BM that is lost during isolation. As a result, the adult phenotype is lost and the islets eventually undergo apoptosis and eventually lose their functionality in vitro.
Early studies have focused on developing a suitable media for suspension culture of islets. However, this does not address the issue of restoring ECM support that is lost during isolation and its success is limited. Other studies based on the two-dimensional culture of islets, on various surfaces modified with ECM, eventually lead to the loss of islet structural integrity and in turn phenotype. This has lead to the development of solid support matrices that embed islets and maintain their structural integrity, thus allowing for longer preservation of their functionality. Unfortunately, such systems do not provide islets with a suitable environment for nutrient and oxygen transfer; in addition, there is a need to again suspend such islets after culturing and thus subjecting them to additional trauma prior to transplantation. An alternative investigated route is the microencapsulation of islets, which provides immunoprotection and thus circumvents the need for immunosuppressants following transplantation. However, the long-term membrane integrity of such capsules is compromised.
The progression of islet culture investigation has lead to the understanding that there is a need to maintain islet structural integrity, as well as to provide a viable environment for which islet preservation can be maintained. Furthermore, such culture must be preformed and maintained within a graft in order to provide a favorable structure for subsequent implantation. These requirements are embodied within the development of a three-dimensional scaffold, with controllable pore geometries and surface modified characteristics, which cater to the islet microenvironment. Moreover, these structures must be maintained within a tissue bioreactor setting, such as the rotating wall bioreactor, which provides minimal shear stress and has already been shown to promote islet culture in suspension.
In order to achieve all the requirements prior to transplantation, it is necessary to combine all aspects of past research strategies. For successful bioreactor culture of islet-seeded scaffolds, the perfused media must be optimized in accordance with investigated suspension culture techniques. Furthermore, using microencapsulated islets within scaffolds may produce a graft that does not require postsurgical immunosuppression. It is also important to investigate the other avenues of islet regeneration, since there is an overwhelming shortage of donors. Many research strides have been accomplished in order to arrive at favorable conditions and strategies for islet culture in vitro and the ultimate goal of viable transplantation remains in the foreseeable future.
