Abstract
High levels of donor-derived CCL2 have been associated with poor islet allograft outcome in patients with type 1 diabetes. The aim of our work was to determine whether CCL2 secreted by the islet has independent proinflammatory effects that influence engraftment and graft acceptance. Both in mice and humans CCL2 is significantly positively associated with other cytokines/chemokines, in particular with the highly released “proinflammatory” IL-6 and CXCL8 or CXCL1. Transplantation of CCL2-/- islets into syngenic recipients did not improve the transplant function. Transplantation of islets into CCL2-/- syngenic recipients led to a significant improvement of transplant function and partial abrogation of local hepatic inflammation. When evaluated in human islets CCL2 release was strongly related to the immediate local inflammatory response in the liver and impacted short-term human islet function dependently by the induced inflammatory response and independently by the immunosuppressive therapy. The data showed that islet CCL2 release is a sign of “inflamed” islets without having a direct role in graft failure. On the other hand, a causal effect for developing detrimental proinflammatory conditions after transplant was proved for recipient CCL2. Strategies to selectively decrease recipient, but not donor, CCL2 release may increase the success of islet transplantation.
Introduction
CCL2/MCP-1 is a member of the CC chemokine family and is produced by multiple cell types (i.e., endothelial cells, vascular smooth muscle cells, keratinocytes, fibroblasts, mesangial cells, tubular epithelial cells, lymphocytes, and monocyte/macrophages) in response to proinflammatory stimuli, including tumor necrosis factor-α, γ-interferon, lipopolysaccharide, interleukin-1β, platelet-derived growth factor, and oxidized LDL (5,32,35,45,47,52). In vitro, subnanomolar concentrations of CCL2/MCP-1 are able to induce chemotaxis of monocytes (27,48) and recruit a subset of T cells (9) and IL-2-activated natural killer cells (1). In monocytes, CCL2/MCP-1 induces not only chemotaxis, but also respiratory burst, rapid induction of arachidonic acid release, and changes in Ca2+ concentration (34). Because of its target cell specificity, CCL2/MCP-1 is postulated to play a role in a variety of processes related to organ transplantation, such as acute and chronic rejection (10 23,36,43), ischemia/reperfusion injury (7,12,14,42), and graft vasculopathy (37,46,49).
We have previously shown that human islets prepared for transplant constitutively produce CCL2/MCP-1 in the absence of detectable infections or endotoxin contamination (31). A high amount of CCL2/MCP-1 released by islets was associated with poor clinical outcome of islet transplant in patients with type 1 diabetes. These observations raise the question whether elevated local level of CCL2/MCP-1 exerts a negative influence on engraftment, inflammation, or immune response causing graft failure or, alternatively, whether islet CCL2/MCP-1 release is a sign of “inflamed” islets without having a direct role in graft failure. Establishing a causative role for islet CCL2/MCP-1 production on engraftment outcome should provide a rationale for using specific inhibitors of CCL2/MCP-1 production in novel combination therapies in islet transplantation.
The aim of our work was to determine whether CCL2/MCP-1 has independent proinflammatory effects that influence engraftment and graft acceptance. This was addressed by studying transplant outcome and engraftment in a mouse models of intraportal islet transplantation in which CCL2 knockout mice (CCL2-/-; C57BL/6 background) or litter-matched CCL2+/+ mice were used alternatively as islet donors or recipients and by correlating human islet CCL2/MCP-1 secretion with islet function and early posttransplant events in a cohort of 30 type 1 diabetic receiving first islet infusion from single donor.
Materials and Methods
Islet Transplants in Mice
Control C57BL/6 and BALB/C mice were purchased from Charles River (Calco, Italy). C57BL/6 CCL2-/-, and litter-matched CCL2+/+ mice were obtained from Prof. R. Barrett, who constructed CCL2/MCP-1-deficient mice by targeted gene disruption (25) and have been rederived in our animal house facility. Islet isolation, transplantation, and graft follow-up were performed as previously described (29). Briefly, the recipient mice were anesthetized with isoflurane and then prepared for abdominal surgery. A 2-cm midline incision was made slightly below xyphoid. The intestines were gently reflected leftward from the peritoneal cavity onto dampened gauze and the portal vein was exposed. The islets were suspended in 0.3–0.5 ml RPMI media aspirated from a 1.5-ml plastic tube into a heparinized plastic 1-ml syringe with a 29-gauge 12.5-mm needle. The needle was then inserted into the exposed portal vein and the plunger was gently advanced to expel the islets into the portal vein. A small piece of gelfoam was placed directly over the hole as the needle was being removed from the portal vein and then pressure was applied until all bleeding stopped. The peritoneal cavity was closed.
Blood sugar levels were measured 15, 30, and 60 min after the end of the surgical procedure, daily for the first week and then every second day posttransplantation. Surgical death was defined as death within the first 7 days after transplantation. In the allogenic model, primary non function was defined as the inability to reach nonfasting blood glucose levels under 250 mg/dl for two consecutive measurements after islet transplantation, and rejection was defined as two successive measurements >250 mg/dl in mice where primary function was achieved. In syngeneic models, transplants were classified as full function (FF; nonfasting blood glucose concentration <50% pretransplant concentration for at least 1 week), partial function (PF, nonfasting blood glucose concentration between 50% and 80% of pretransplant concentration for at least 1 week), or no function (NF; nonfasting blood glucose concentration >80% pretransplant concentration for at least 1 week). The local animal ethics committee approved all experiments.
Islet Transplants in Patients with Type 1 Diabetes
From January 1988 to December 2007, 86 patients with type 1 diabetes received islet transplantation at San Raffaele Scientific Institute (SIK = 9, IAK = 45, ITA = 32). Transplantations were considered adequate for the study according to the following criteria: 1) absence of simultaneous kidney transplant; 2) first islet infusion obtained from single donor; 3) availability of pretransplant CCL2/MCP-1 release by the islets; 4) if IAK, patients already under immunosuppression therapy with cyclosporin and treated at the time of transplant with anti-lymphocyte globulin (125 mg/day for 10 days; IMTIX, Marseille, France) as induction therapy, then cyclosporin (7.5 mg kg1 day1), mycophenolate mofetil (2 g day1), and methylprednisone (500 mg immediately before surgery, 0.25 mg kg1 day1 for 2 months after surgery, then lowered to 5 mg/day); 5) if ITA, treated at the time of transplant with daclizumab (1 mg kg1 intravenously given every 2 weeks for a total of five doses) as induction therapy, then FK506 (given orally at an initial dose of 1 mg kg1 day1, then to maintain serum levels of 3–6 ng/ml) and rapamycin (given orally at a loading dose of 0.2 mg kg1 day1, followed by a dose of 0.1 mg kg1 day1 to achieve a range level of 12–15 ng/ml for the first 90 days, and 7–10 ng/ml thereafter). Thirty patients matched these criteria. Sixteen recipients were already immunosuppressed for a previous kidney graft (IAK) while 14 recipients received ex novo the immunosuppressive therapy (ITA). Of the 16 IAK recipients, 7 was already treated with steroid at the time of islet transplantation (methylprednisone 0.13 ± 0.05 mg kg−1 day−1). The characteristics of islet preparations, donors, and recipients are reported in Table 1. All patients gave informed consent for the investigations. The ethical committee of the Istituto Scientifico Ospedale San Raffaele approved the protocols.
Characterization of Islet Preparations, Donors, and Recipients

Assays
Basal Release of Chemokines and Cytokines in Freshly Isolated Mouse and Human Islets
Aliquots of pancreatic islet preparations (500 islets/ml) were cultured in complete medium (mouse islets: CMRL, Seromed Biochrom, Berlin, Germany, supplemented with 10% fetal calf serum, 1% l-glutamine, 100 U/ml penicillin, and 100 μg/ml streptomycin; human islets: final wash culture medium, Mediatech, Inc., Manassas, VA) in a 24-well plate. Culture supernatants were harvested after 24 h and stored at −80°C. Human and mouse cytochines/chemokines were detected using multiplex bead-based assays based on xMAP technology (Bio-Plex Human Cytokine 27-Plex Panel; Bio-Plex Human Group II Cytokine 23-Plex Panel; Bio-Plex Mouse Cytokine 23-Plex Panel; Biorad Laboratories, Hercules, CA, USA). Limits of detection and the coefficient of variability (intra-assay % CV and interassay % CV) were in human and mouse assay: IL-1α o.5 pg/ml (4,4) and 2 pg/ml (3.4, 4); IL-1β 0.6 pg/ml (6, 8)) and 7 pg/ml (43.3, 4.4); IL-2: 1.6 pg/ml (7, 9)) and 3 pg/ml (3.3, 4.8); IL-3: 4.8 pg/ml (7, 4)) and 2 pg/ml (2.4, 4.2); IL-4: 0.7 pg/ml (9, 8)) and 3 pg/ml (3.2, 12.7); IL-5: 0.6 pg/ml (8, 10)) and 2 pg/ml (2.3, 11.6); IL-6: 2.6 pg/ml (7, 11)) and 2 pg/ml (4.2, 8.5); IL-9: 2.5 pg/ml (8, 9)) and 15 pg/ml (6.7, 4.4); IL10: 0.3 pg/ml (5, 6)) and 2 pg/ml (2.8, 3.8); IL-12p40: 23.3 pg/ml (5, 8)) and 2 pg/ml (2.7, 3.3); IL-12p70: 3.5 pg/ml (6, 6)) and 4 pg/ml (2.7, 5.7); IL-13: 0.7 pg/ml (8, 7)) and 9 pg/ml (2.2, 8.7); IL-17: 3.3 pg/ml (8, 6)) and 1 pg/ml (3.2, 7.7); INF-γ: 6.4 pg/ml (15, 9)) and 6 pg/ml (5.4, 3.5); TNF-α: 6 pg/ml (8, 6)) and 6 pg/ml (5.1, 3.9); GM-CSF: 2.2 pg/ml (12, 6)) and 7 pg/ml (5, 2).9); G-CSF: 1.7 pg/ml (10, 5)) and 1 pg/ml (2.3, 4.4); CCL2/MCP-1: 1.1 pg/ml (9, 7)) and 14 pg/ml (4.5, 5.2); CCL3/MIP-1a: 1.6 pg/ml (7, 8)) and 24 pg/ml (3.2, 5.8); CCL4/MIP-1β 2.4 pg/ml (8, 8)) and 2 pg/ml (3.8, 4.9); CCL5/Rantes: 1.8 pg/ml (9, 6)) and 5 pg/ml (3.6, 2.9); CCL11/Eotaxin: 5.5 pg/ml (8, 11)) and 148 pg/ml (3.6, 15.7); VEGF (human only): 3.1 pg/ml (9, 7)); CXCL8/IL-8 (human only): 1.0 pg/ml (9, 4)); CXCL1/KC (mouse only): 3 pg/ml (2.7, 4.7).
Human Blood
Serum aspartate (AST) and alanine aminotransferase (ALT) activities and C-reactive protein (CRP) were determined with the ADVIA 2400 Chemistry System (Bayer Healthcare, Tarrytown, NY). Cross-linked fibrin degradation products (XDPs) were determined with Immuno-Turbimetric method (STA-Liatest, Diagnostica Stago, France). Serum C-peptide levels (intra-assay CV 3.0%; interassay CV 3.0%) were assayed by RIA using commercial kits (Medical System, Genoa, Italy). Serum cytochines/chemokines were detected using multiplex bead-based assays based on xMAP technology (Biorad Laboratories).
Mouse Blood
Whole blood was collected from the retro-orbital plexus or alternatively from tail vein. Blood glucose measurements were performed using a Glucometer Elite (Bayer Canada, Toronto, Ontario, Canada). ALT activity was measured in a Paramax chemical analyzer (Baxter Diagnostics Inc., McGaw Park, IL, USA). Platelets, white blood cells, red blood cells, hemoglobin, and hematocrit values were counted with an automated cell counter (System 9000; Serono-Baker Diagnostics, Allentown, PA, USA).
Mouse Liver
The RNase protection assay (RPA) for quantitation of mRNA was performed as previously described (19). Total liver RNA isolated from all the lobes was analyzed for cytokine and chemokine by RPA, exactly as previously described (41).
Statistical Analysis
Data were generally expressed as mean ± SE or median (Min-Max). Differences between parameters were evaluated using Student's t-test when parameters were normally distributed, Mann-Whitney U-test when parameters were not normally distributed, and a chi-square test for categorical variables. Multivariate tests of repeated measures ANOVA were used to evaluate the relationships between islet CCL2/MCP-1 release and early posttransplant events. Correlations were assessed with a Spearman rank correlation coefficient. Univariate General Linear Model was used to evaluate the relationships between islet CCL2/MCP-1 release, the area under the curve for CRP during the 5 days post-Tx and the Islet Estimated Function 1 and 3 months after islet infusion. Kaplan-Meier analysis was used to compare survival. A value of p < 0.05 was considered an indicator of statistical significance. Analysis of data was done using the SPSS statistical package for Windows (SPSS Inc., Chicago, IL, USA).
Results
Recipient CCL2/MCP-1 Plays a Direct Role in Islet Function
To study whether islet CCL2/MCP-1 has independent proinflammatory effects that influence engraftment we used a syngenic mouse model of intraportal marginal islet mass transplantation (29). CCL2 knockout mice (CCL2-/-) or litter-matched CCL2+/+ mice were used alternatively as islet donors or recipients. CCL2-/- mice were first compared to CCL2+/+ mice with respect to glucose metabolism, islet size and number, basal and IL-1β-stimulated islet cytokines secretion. With the exception of CCL2/MCP-1 release both in basal and IL-1β-stimulated condition, no differences were evident between CCL2-/- and CCL2+/+ mice for all the parameters tested (Figs. 1, 2, and 3).

The absence of CCL2 did not significantly affect glucose tolerance in mice. Not fasting and fasting glycemia (A), weight (B), and glucose tolerance after IVGTT (KG30) (C) in CCL2+/+ (open symbols; n = 8) and CCL2-/- (gray symbols; n = 8) C57BL/6 mice starting from 12 weeks to 42 weeks of life. Data are expressed as mean ± SD for glycemia and weight, as boxplots for glucose tolerance. Statistical analysis was performed by tests of repeated measures ANOVA.

The absence of CCL2 did not significantly affect islet number, morphology, and size. Fifty milligrams of dithizone was dissolved in a mixture of 5 ml of dimethyl sulfoxide and 20 ml of absolute ethanol. Following isolation and dithizone staining in a petri dish, pictures of fresh islet from CCL2-/- or CCL2+/+ were taken with a digital camera (Leica DFC Cameras, Cambridge, UK) attached to an inverted microscope (Leica) and linked to a computer program (Leica IM50 image archiving software) for determination of the number and size of particles. (A) Islet appearance at inverted microscope and maximum diameter frequency distribution in a representative experiment of 14 performed. Scale bar: 300 μm. (B) After saving the pictures, the images were evaluated for size and numbers of the islet cells. Reported are the maximum diameter, the minimum diameter, and the islet number after isolation. Data are expressed as median (min-max).

The absence of CCL2 did not significantly affect secretion of cytokines and chemokines by unstimulated and IL-1β-stimulated mouse pancreatic islets. Freshly isolated islets were cultured for 24 h in the absence (upper panel; CCL2-/-: n = 19; CCL2+/+: n = 15) or in the presence (lower panel; CCL2-/-: n = 20; CCL2+/+: n = 14) of 10 ng/ml IL-1β, and the chemokine/cytokine concentration in the supernatants was measured by a multiplex sandwich enzyme-linked immunosorbent assay (Bio-Plex Mouse Cytokine 23-Plex Panel; Biorad Laboratories, Hercules, CA, USA). Data are expressed as box plots. The exposure to IL-1β significantly increased the release of IL-1α, IL-6, CCL11/Eotaxin, G-CSF, CXCL1/KC, CCL3/MIP-1α CCL5/Rantes in islet derived from both CCL2+/+ and CCL2-/- mice, while CCL2/MCP-1 was increased in CCL2+/+ mouse but not in CCL2-/-. Statistical analysis was performed by Mann-Whitney U-test.
A total of 109 STZ-induced severely diabetic mice (mean nonfasting glycemia before transplantation: 520 ± 71 mg/dl) were transplanted with 350 syngenic islets through the portal vein. Four donor/recipient combination were tested: CCL2+/+ mice as both donors and recipients (Tx-Ctrl, N = 28); CCL2-/- mice as both donors and recipients (Tx-CCL2-/-, N = 19); CCL-/- mice as donors and CCL+/+ mice as recipients (Tx-donor CCL-/-, N = 45); CCL-/- mice as recipients and CCL+/+ mice as donor (Tx-recipient CCL-/-, N= 17). Fourteen of 109 mice (12.8%) died within 7 days of transplant due to surgical complications (3/28, 3/19, 7/45, and 1/17 in the four groups; p = 0.7) and were excluded from subsequent analyses.
Islet function significantly differed between the four different donor/recipient combinations (Fig. 4). For all the parameters tested, transplant outcome was superior when CCL2-/- mice were used as recipients. The prob-ability and median time to reach euglycemia (<200 mg/dl) were: 53% and 42 days for Tx-Ctrl compared to 87.5% and 1 day when CCL2+/+ islets were transplanted into CCL2-/- recipients (p < 0.001) (Fig. 4A). The use of CCL2-/- donor islets did not improve transplant outcome (36.8% and >70 days in CCL2+/+ recipients and 87.5% and 8 days in CCL2-/- recipients; p > 0.1). A Multivariate Cox Regression Analysis was performed in which donor CCL2-/-, recipient CCL2-/- and recipient pretransplant glycemia (28) were included as covariates. The analysis confirmed that outcome was improved by the use of CCL2-/- mice as recipients (odds ratio: 3.0; 95% CI: 1.7–5.2; p < 0.0001), and further suggested that outcome was negatively affected when CCL2-/- islets were used as donors (odds ratio: 0.6; 95% CI: 0.3–1; p = 0.05). As shown previously (28), pretransplant glycemia also affected outcome (odds ratio: 0.995; 95% CI: 0.991–0.999; p = 0.007). Graft function classification (full function, partial function, no function) and glucose tolerance (KG60) after IVGTT 2 weeks after transplantation were consistent with the data of euglycemia (Fig. 4B, C). Taken together these observations demonstrate that recipient CCL2/MCP-1 is a major determinant of islet graft function.

CCL2 knockout mice (CCL2-/-; C57BL/6 background) or litter-matched CCL2+/+ mice transplanted with syngenic islet and used alternatively as islet donors or recipients. CCL2-/- and CCL2+/+ recipients were transplanted with 350 autologous islets alternatively from CCL2-/- or CCL2+/+ mice through the portal vein. Four donor/recipient combination were tested: CCL2+/+ mice as both donors and recipients (Tx-Ctrl, N = 28); CCL2-/- mice as both donors and recipients (Tx-CCL2-/-, N = 19); CCL-/-mice as donors and CCL+/+ mice as recipients (Tx-donor CCL-/-, N = 45); CCL-/- mice as recipients and CCL+/+ mice as donor (Tx-recipient CCL-/-, N = 17). (A) Kaplan-Meier survival analysis and Kaplan-Meier analysis for the gain of normoglycaemia (<200 mg/dl) between the four different donor/recipient combinations. Differences were tested using the log rank statistic. (B) The percentages of full function (FF), partial function (PF), no function (NF), and surgical death (SD) in the four different donor/recipient combinations. Significance of difference between combinations was tested using the Pearson chi-square tests. ∗p < 0.05; ∗∗p < 0.001; ns: no statistical difference. (C) Glucose tolerance after IVGTT (KG60) in the four different donor/recipient combinations 14 days after islet infusion. ∗p < 0.05; ∗∗p < 0.001; ns: no statistical difference. Data are expressed as box plots. Statistical analysis was performed by Mann-Whitney U-test.
Recipient CCL2/MCP-1 Affects Islet Function by Inducing a Local Inflammatory Response
Because recipient CCL2/MCP-1 is a mediator of proinflammatory response, we asked whether CCL2-/- recipients differed in their inflammatory response to intrahepatic islet transplantation and potentially explain the improved graft survival. Eleven CCL2+/+ and 11 CCL2-/-diabetic mice were transplanted with 500 syngenic CCL2+/+ C57BL/6 islets into the liver through the portal vein and monitored with respect to hematopoietic cell numbers, liver function, and serum cytokines (Table 1). Serum ALT concentrations rose abruptly in CCL2+/+ recipient mice and this rise was significantly less in the CCL2-/- recipients (Fig. 5). No other differences were observed (Table 2). We subsequently performed a second set of experiments on 18 diabetic mice (CCL2+/+ n = 9; CCL2-/- n = 9) transplanted with 500 syngenic CCL2+/+ C57BL/6 islets, and examined local liver inflammatory status before and 4, 24, and 48 h after intrahepatic islet infusion using RNAse protection (RPA)-based assays for detecting cytokine and chemokine transcripts (Fig. 6). In CCL2+/+ recipients, the intrahepatic messages for CXCL1/KC, IL-1β, CCL2/MCP-1, and CXCL9/MIG were strongly induced immediately after islet infusion (100-fold for CXCL1/KC, sixfold for IL-1β, fivefold for CCL2/MCP-1, threefold for CXCL9/MIG at 4 h), and message for CCL6/C10 and CCL4/MIP-β were induced late after islet infusion (sixfold for CCL6/C10 and fourfold for CCL4/MIP-1β after 48 h). The rise in CXCL1/KC, CCL2/MCP-1, CCL4/MIP-1β, CCL6/C10, and CXCL9/MIG RNA was completely abrogated in CCL2-/- recipient mice; whereas IL-β RNA expression was unaffected. These findings indicate that the presence of CCL2/MCP1 is crucial for the hepatic inflammatory response seen post islet transplantation.

Serum alanine aminotransferase (ALT) concentrations in mice transplanted with syngenic islet. CCL2-/- (n = 11) and CCL2+/+ (n = 11) recipients were transplanted with 500 autologous islets from CCL2+/+ mice through the portal vein. Serum ALT concentration was determined at different time after islet injection (4, 24), and 48 h). Data are expressed as mean ± SD. The p-value was calculated by tests of repeated measures ANOVA.

Local liver inflammatory status before and 4, 24, and 48 h after intrahepatic islet infusion evaluated using RNAse protection (RPA)-based assays for detecting cytokine and chemokine transcripts. CCL2-/- (n = 9) and CCL2+/+ (n = 9) recipients were transplanted with 500 syngenic islets from CCL2+/+ mice through the portal vein. Mice were sacrificed at different times after islet injection (4, 24), and 48 h) and the liver was harvested. Total hepatic RNA was analyzed by RPA for the expression of various chemokines, as indicated. The housekeeping mRNA encoding the ribosomal protein L32 was used to normalize the amount of RNA loaded and a quantitative phosphor imaging analysis of the RNAs was performed. The indicated numbers in the CCL2-/- and CCL2+/+ groups are displayed as fold induction over NaCl-injected diabetic recipient controls (set as 1). The numbers were obtained by dividing each cytokines/chemokines value by the amount of the corresponding housekeeping gene L32 RNA. Data are expressed as boxplot of three mice per time. ∗p < 0.05: statistical analysis was performed by Mann-Whitney U-test.
Blood Analysis Before and After Islet Transplantation in CCL2+/+ and CCL2-/- Recipients

Data are expressed as mean ± SD or median (min-max).
Tests of repeated measures ANOVA.
Mann-Whitney U-test.
Human Islet CCL2/MCP-1 Release Correlates with Hepatic Inflammatory Response
Our previous data in humans show a correlation between islet CCL2/MCP-1 release and transplant islet function, where high levels of CCL2/MCP-1 release by donor islets were associated with failed islet transplantations. The current data from the CCL2-/- mice, however, suggested that the recipient (and not donor) CCL2/MCP-1 status was a stronger predictor of transplant outcome. Because the recipient inflammatory response was markedly higher in CCL2+/+ mice, we hypothesized that the previously observed relationship between donor islet CCL2/MCP-1 release and transplant outcome in human islet transplants was due to an indirect effect of islet CCL2/MCP-1 on hepatic inflammation. We examined data from 30 single donor human islet preparations transplanted into 30 patients with type 1 diabetes (see Table 1 for islet and patient characteristics). As previously reported (31), detectable heterogeneous levels of CCL2/MCP-1 were found in all the 30 islet preparations during 24 h of culture (12.04 ± 20.8 pg islet−1 24 h−1, ranging from 0.49 to 104.4 pg islet−1 24 h−1). Consistent with our hypothesis, high islet CCL2/MCP-1 release was associated wih increased recipient biochemical parameters of coagulation, liver damage, and inflammation postislet transplant (Fig. 7).
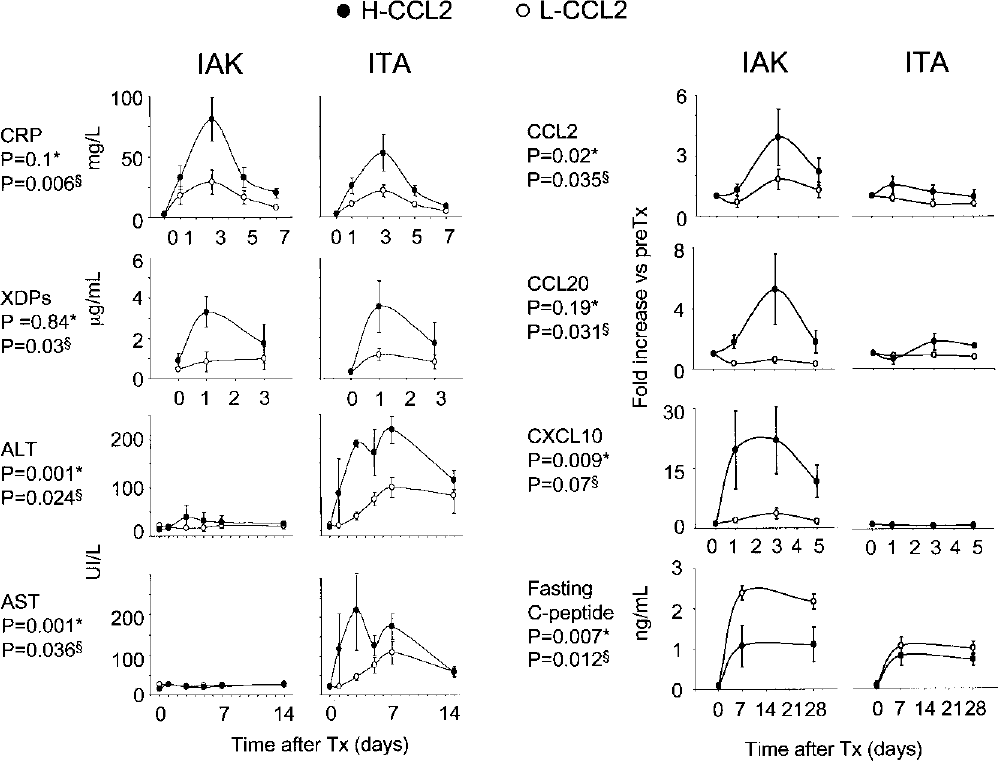
Relationships between pre transplant islet CCL2/MCP-1 release and early posttransplant events. Posttransplantation levels of C-reactive protein (CRP), cross-linked fibrin degradation products (XDPs), aspartate and alanine aminotransferases (AST and ALT), inducible inflammatory chemokines (CCL2, CXCL10, CCL20), and fasting C-peptide were evaluated in patients with type 1 diabetes that received islet allografts. Patients were divided into two groups according to values of CCL2/MCP-1 released by islet preparations, during a 24-h culture (50th percentile = 3.64 pg islet−1 24 h−1). Low-CCL2 group (L-CCL2) received islet preparations below the 50th percentile (mean CCL2 value 1.58 ± 0.77 pg islet−1 24 h−1); high-CCL2 group (H-CCL2) received islet preparations above the 50th percentile (mean CCL2 value, 22.5 ± 25 pg islet−1 24 h−1). Values are mean ± SE. Statistical analysis was performed by a multivariate tests of repeated measures ANOVA including H-CCL2 versus L-CCL2 (§) and IAK versus ITA (∗).
Independent of the immunosuppressive regimen, patients who received islets with high CCL2/MCP-1 release (>50th percentile, 3.64 pg islet−1 24 h−1) had significantly higher posttransplant concentrations of cross-linked fibrin degradation products (XDPs), liver damage markers (AST and ALT), and inflammatory markers (CRP, CCL2, CXCL10, and CCL20) than recipients of islets with low CCL2/MCP-1 release (<50th percentile) (Fig. 7).
CCL2/MCP-1 Is a Marker of Proinflammatory Islets
In order to determine whether islet CCL2/MCP-1 per se was the contributor to posttransplant inflammation or whether it was a marker of a more generalized inflammatory state of the islets, we tested basal release of chemokines and cytokines in freshly isolated mouse and human islets (Table 3).
Basal Release of Chemokines and Cytokines in Freshly Isolated Mouse and Human Islets

NS, not statistically different; NT, not tested.
Spearman correlation coefficients.
In humans, copious amounts of CCL2/MCP-1, CXCL8/IL-8, IL-6, and VEGF, as well as appreciable levels of CCL11/Eotaxin, CCL4/MIP-1β, IL-12p40, and G-CSF were secreted in the supernatant during a 24-h culture. In mice (C57BL/6 background), copious amounts of CCL2/MCP-1, CXCL1/KC, and IL-6, as well as appreciable levels of CCL5/Rantes, CCL11/Eotaxin, G-CSF, IL-12p40, IL-12p70, and CCL3/MIP-1α were secreted in the supernatant during a 24-h culture. In both species, CCL2/MCP-1 was significantly positively associated with the other cytokines/chemokines, in particular with the highly released “proinflammatory” IL-6 and CXCL8/IL-8 or CXCL1/KC. We conclude, therefore, that islet CCL2/MCP-1 is a marker of islet inflammatory state and that it is likely to be the combination of cytokines and chemokines released by islets will contribute to posttransplant inflammation.
Islet Function Posttransplant in Humans Is Related to Both Recipient Inflammatory Response and Islet Proinflammatory State
The combined findings in mice and humans suggested that the inflammatory response to islet transplantation and the islet inflammatory state could both contribute to islet transplant outcome. Islet function in the patients who received a single donor islet infusion was evaluated 1 month (n = 25) and 3 months (n = 19) after transplantation as islet estimated function (IEF), an index estimating the average insulin secretion of the single transplanted islet (11) and correlated to islet and recipient inflammation (Fig. 8). IEF 1 and 3 months after transplantation was negatively associated both with posttransplant inflammation, expressed as the area under the curve for recipient CRP during the 5 days after islet infusion [CRPAUC(0–5); p = 0.006 and p = 0.06, respectively, 1 and 3 months after transplantation), and islet CCL2/MCP-1 release (p = 0.02 and p = 0.023, respectively, 1 and 3 months after transplantation) when both were included as covariates in a General Linear Model. Taken together these observations suggest that islet cytokine/chemokine release affects the immediate local inflammatory response in the liver and that both affect islet function.
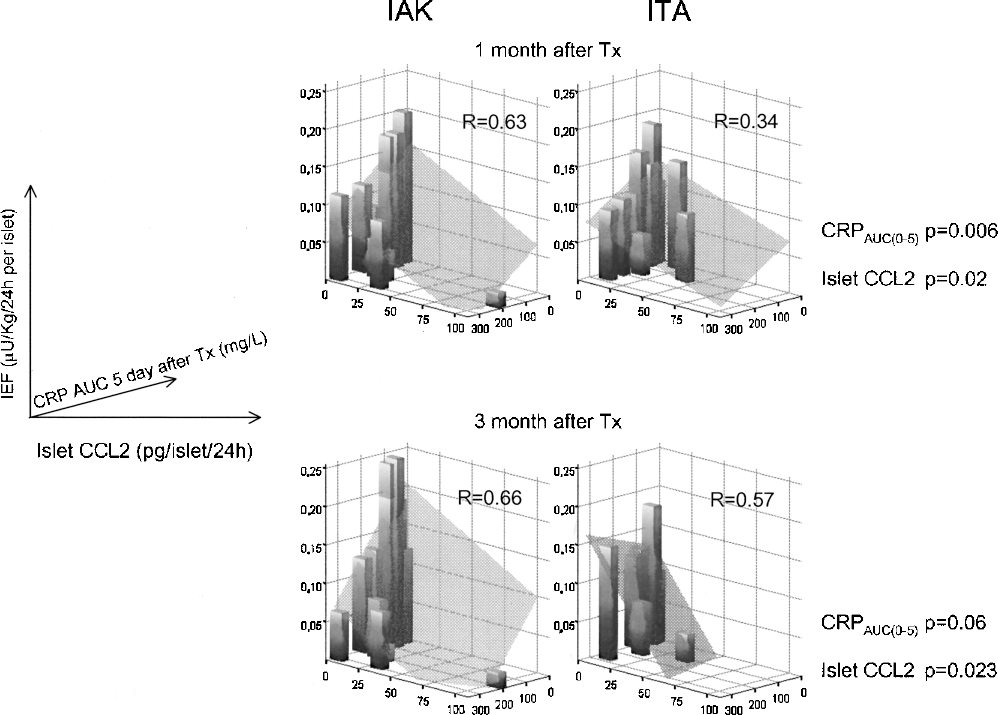
Relationships between pre transplant islet CCL2/MCP-1 release, area under the curve for CRP during the 5 days post-Tx, and posttransplant islet function. Significant relationships were observed between CCL2/MCP-1, area under the curve for CRP during the 5 days post-Tx [CRPAUC(0–5)] and islet estimated function (IEF) 1 and 3 month after transplantation. Represented are 3D bar plots. Gray plan: regression fit plan. Univariate General Linear Model was used to evaluate the statistical significance after including as fixed factor the different immunosuppressive treatments (ITA vs. IAK and steroid vs. not steroid at the time of islet infusion).
Discussion
Despite recent publications on CCL2/MCP-1 secretion by pancreatic islets (4,8,13,15,16,22,51), no mechanistic data outlining the role of donor-derived CCL2/MCP-1 on intrahepatic islet engraftment are available. The objective of our work was to determine whether CCL2/MCP-1 has independent proinflammatory effects that influence islet engraftment and acceptance after transplantation.
The CCL2/MCP-1 release by the islets was significantly positively associated with the release of other proinflammatory factors, in particular with the highly released IL-6 and CXCL8/IL-8 (or CXCL1/KC in mice). This evidence, in agreement with a previous report (6), was not unexpected because the regulation of CCL2/MCP-1 expression in β-cells is under the control of NF-κB (22), a key transcription factor involved in regulating proinflammatory cytokines, chemokines, adhesion molecules, and inflammatory enzymes. The association with other proinflammatory molecules raises justified concerns about the direct role of islet CCL2/MCP-1 on engraftment and postinfusion inflammatory reaction. To address this issue we studied a mouse models of intraportal islet transplantation in which CCL2 knockout mice or litter-matched CCL2+/+ mice were used alternatively as islet donors or recipients. When we used CCL2-/- mice as recipients the transplant results improved while when we used CCL2-/- mice as donors not only the infused islet function did not improve, but also a significant trend to worsen the transplant function was evident. The data show a dual role of CCL2/MCP-1 in islet engraftment and favors a causal effect for developing detrimental proinflammatory conditions after transplant for recipient but not donor CCL2/MCP-1.
Our results suggest that one of the primary causes of transplanted islet dysfunction is antigen-nonspecific inflammation at the graft site mediated by the recipient CCL2/MCP-1 as demonstrated by the fact that its depletion resulted in the complete abrogation of CXCL1/KC, CCL4/MIP-1β, CCL6/C10, and CXCL9/MIG RNAs increase in the liver. On the other hand, no direct causal effect emerged for donor CCL2/MCP-1. The results from donor CCL2-/- mice are not totally unexpected. A dual role of CCL2/MCP-1 and more generally of inflammation is well described for all the active process of tissue remodeling and neo-angiogenesis including for example wound healing, tumor growth and hepatic metastasis (3,20,24). As recently suggested (21), the isolated islets injected into the portal vein should follow the same steps of a tumor metastasis to successfully complete the engraftment process and CCL2/MCP-1, as described for tumor, could be protective (17,38,44) or destructive (18,26,33,50).
An explicative example of the potential biphasic effect of CCL2/MCP-1 was reported in a nontumorigenic melanoma system: low-level CCL2/MCP-1 secretion, with physiological accumulation of tumor-associated macrophages, promoted tumor formation, whereas high CCL2/MCP-1 secretion resulted in massive macrophage infiltration into the tumor mass and its destruction (30). The reason for the biphasic effect resides in the fact that the target cells of CCL2/MCP-1, the monocyte–macrophage lineage, have complex dual functions in tissue remodeling. Whereas macrophages of M1 phenotype have the potential to kill cells after activation by IL-2 and IFNs, M2 macrophages produce a number of potent angiogenic and lymphangiogenic growth factors, cytokines, and proteases, all mediators that potentiate tissue remodeling (2).
Another possible explanation for the fact that we did not observe significant difference in the murine model when comparing wild-type recipient of wild-type or CCL2/MCP-1-/- islets is the lack of ischemic injury of the pancreas in the mouse experiments that could have introduced a certain degree of inflammation/injury (on top of isolation related) to isolated islets, similar to what is observed in human pancreata, inducing high level of CCL2 secretion by the islets. Schröppel et al. (40) previously reported (5,39) that transplantation of murine islets with high levels of CCL2 into syngeneic recipients led to a significantly greater influx of macrophage cells, to a higher expression of monocyte/macrophage-associated inflammatory cytokines compared with low CCL2-donor islets, and finally to a worse isograft function. Because the authors did not use CCL2-/- mice as donor, they were not able to demonstrate the role of donor CCL2 as independent proinflammatory factor that influences islet engraftment. On the other hand, of great interested is the fact that also in Schröppel et al.'s work (40), any detrimental effect associated with high levels of CCL2 release by the islet disappeared when CCR2-/-mice were used as recipients, in agreement with our data that blocking the CCL2/CCR2 pathway in the recipients increased the success of islet engraftment
By correlating human islet CCL2/MCP-1 secretion with islet function, we extended the previous evidences to a larger cohort of type 1 diabetic patients, including recipients of islet transplant alone treated with Edmonton protocol. IEF, an index estimating the average insulin secretion of the single transplanted islet (11), and C-peptide increase after islet transplantation were strongly inversely influenced by islet CCL2/MCP-1. Moreover, a strong direct association between islet CCL2/MCP-1 release and early biochemical parameters of coagulation, liver damage, and inflammation postislet infusion was evident. Taken together, these observations confirmed that islet CCL2/MCP-1 release is strongly related to the immediate local inflammatory response in the liver and impacted to short term islet function and survival. Of note, the different immunosuppressive regimens in ITA and IAK partially influenced the early posttransplant events (the Edmonton immunosuppressive treatment was associated to a higher liver damage with less release of inflammatory mediators) but did not modify the significant association of islet CCL2/MCP-1 with the immediate local inflammatory response in the liver and the islet function. This observation confirms that the quality of the islets in terms of their proinflammatory state is a major determinant of the posttransplant inflammatory response. It also suggests that the Edmonton immunosuppressive treatment, even if able to partially modulate the inflammatory response of the recipient, do not control the impact of the proinflammatory state of islets on the engraftment.
Taken together, our results have several potential clinical implications. First, they provide evidence that recipient CCL2/MCP-1 should be a major pharmacological target for the control of potentially damaging reactions at the site of transplant. Its selective inhibition should reduce the number of islets required to achieve insulin independence and also the incidence of clinical complications observed early after islet infusion and ultimately make islet transplantation a more attractive clinical option for patients with type 1 diabetes. Second, measurement of pretransplantation CCL2/MCP-1 level, as marker of “inflamed” islets may be a useful tool to preselect islets to increase engraftment success rates. Third, strategies to selectively prevent CCL2/MCP-1 secretion by islets should not be used in clinical islet transplantation. More generally, due to the potential dual role of inflammation on engraftment, every anti-inflammatory treatment for islet or for recipients should be preventively tested in adequate animal model of engraftment (intrahepatic islet infusion) before clinical application.
In conclusion, high pretransplantation islet CCL2/MCP-1 release predicts a negative result for the immediate islet graft function but does not have independent proinflammatory effects that influence islet engraftment and acceptance after transplantation. Therefore, strategies to selectively decrease CCL2/MCP-1 release by islet preparations may not be enough. On the other hand, strategies to block the CCL2/CCR2 pathway in the recipient might increase the success of islet engraftment and long-term insulin independence in human islet transplantation.
Footnotes
Acknowledgments
This work was supported by EU (DIAPREPP Project, HEALTH-F2-2008-202013), EFSD/JDRF/Novo Nordisk Type 1 Diabetes Research Programme, CARIPLO Foundation and Famiglia Ferrè. Valeria Sordi is enrolled as a Ph.D. student at the Ludwig-Maximilians University of Munich, Germany.
