Abstract
Stem cell (SC) therapy represents a promising approach to treat a wide variety of injuries, inherited diseases, or acquired SC deficiencies. One of the major problems associated with SC therapy remains the absence of a suitable matrix for SC growth and transfer. We describe here the growth and metabolic characteristics of mouse limbal stem cells (LSCs) and mesenchymal stem cells (MSCs) growing on 3D nanofiber scaffolds fabricated from polyamide 6/12 (PA6/12). The nanofibers were prepared by the original needleless electrospun Nanospider technology, which enables to create nanofibers of defined diameter, porosity, and a basis weight. Copolymer PA6/12 was selected on the basis of the stability of its nanofibers in aqueous solutions, its biocompatibility, and its superior properties as a matrix for the growth of LSCs, MSCs, and corneal epithelial and endothelial cell lines. The morphology, growth properties, and viability of cells grown on PA6/12 nanofibers were comparable with those grown on plastic. LSCs labeled with the fluorescent dye PKH26 and grown on PA6/12 nanofibers were transferred onto the damaged ocular surface, where their seeding and survival were monitored. Cotransfer of LSCs with MSCs, which have immunosuppressive properties, significantly inhibited local inflammatory reactions and supported the healing process. The results thus show that nanofibers prepared from copolymer PA6/12 represent a convenient scaffold for growth of LSCs and MSCs and transfer to treat SC deficiencies and various ocular surface injuries.
Keywords
Introduction
Stem cell (SC) therapy represents a promising approach to treating various inherited diseases or tissue injuries associated with SC deficiency. Adult (tissue-specific) SCs benefit from the ability to differentiate into the cell type for which they are committed and even from their ability to differentiate into other cell types (9,12). In addition, a population of SCs derived from bone marrow, called mesenchymal stem cells (MSCs), has immunosuppressive properties and thus can contribute to the healing process by inhibiting local inflammatory reactions (3,16,21).
One of the major problems associated with SC therapy remains the absence of a suitable carrier for the transfer of SCs to precise tissue locations. So far, various materials and scaffolds have been tested for the transportation of SCs. For example, macroporous hydrogels have been used to deliver MSCs for spinal cord injury repair (29) or self-assembling peptide nanofibers have been tested for myoblast transplantation in infarcted myocardium (7). To treat severe ocular surface damage and a deficiency in limbal SCs (LSCs), which are irreplaceable for corneal healing, various carriers for the culturing of LSCs and for their transplantation onto the recipient eye have been tested. They include fibrin glue (24), polymers or collagen sponges (26), and human amniotic membrane (30).
In the last years, promising scaffolds for the growth and transfer of various types of SCs have been offered by nanotechnology. Electrospinning processes can fabricate nanofibers with a diameter ranging from a few tens to hundreds of nanometers and with a defined porosity. The three-dimensional structure of nanofibrous materials has an extremely large surface area, and nanofibers can mimic the structure of extracellular matrix proteins, which provide support for cell growth and function. Nanofiber scaffolds can create specific niches where SCs can reside and maintain their unique properties. It has been shown that embryonic SCs or MSCs grow and differentiate on nanofibers comparably or even better than on plastic surfaces (10,20,27,33,34). We sought to determine whether adult tissue-specific SCs can also be grown on nanofiber scaffolds and whether these scaffolds can be used as carriers for cell transplantation in tissue regeneration.
Using the original Nanospider electrospinning technology we prepared nanofiber scaffolds from a panel of natural and synthetic polymers and tested them for their biocompatibility and their ability to support the growth of various cell types (S. Kubinova et al., manuscript submitted for publication). On the basis of the stability of its nanofibrous architecture in aqueous solutions and its optimal biocomptibility, we selected copolymer polyamide 6/12 (PA6/12) for further studies. We characterized the growth properties of LSCs and MSCs on these nanofibers and used PA6/12 scaffolds for the transfer of LSCs and MSCs to treat ocular surface injuries in an experimental mouse model.
Materials and Methods
Mice
Mice of the inbred strains BALB/c and C57BL/10Sn of both sexes at the age of 2–4 months were used in the experiments. The animals were obtained from the breeding unit of the Institute of Molecular Genetics, Prague. The use of animals was approved by the local Animal Ethics Committee of the Institute of Molecular Genetics. The animals were treated in accordance with the Principles of Laboratory Animal Care.
Materials and Nanofiber Preparation
The copolymer PA6/12 was purchased from Chemopharma (Wien, Austria). This material (10 wt%) was dissolved in 85 wt% formic acid (Penta Company, Fair-field, NJ) and heated at 50°C for 6 h. After reducing the temperature to room temperature, the material was used for electrospinning. A modified needleless Nanospider™ technology (U.S. patent No. WO205024101.2005), in which polymeric jets are spontaneously formed from liquid surfaces on a rotating spinning electrode, was used for the preparation of the nanofibers. This Nanospider technology flexibly enables the formation of fibers tens of nanometers to tens of micrometers in diameter. All nanofibrous samples used during this study were prepared at a basis weight of 3–5 g/m2 and had nanofiber diameter ranging from 290 to 539 nm.
To test the stability of nanofibers in aqueuos solutions, the nanofibrous samples were cut into small pieces and soaked in deionized water in petri dishes. The water was exchanged every day. After a 7- or 14-day period of soaking the samples were dried at room temperature, and their nanofibrous architecture was analyzed using scanning electron microscopy (SEM).
LSCs, MSCs, Corneal Epithelial and Endothelial Cell Lines
LSCs were obtained by enzyme digestion from limbal tissues as we have recently described (13). In brief, limbal tissues from 10–12 BALB/c mice were cut with scissors and subjected to 10 short (10 min each) trypsinization cycles. The released cells were harvested after each cycle, centrifuged (8 min at 250 x g) and resuspended in RPMI-1640 medium (Sigma, St. Louis, MO) containing 10% fetal calf serum (FCS, Sigma), antibiotics (100 U/ml of penicillin, 100 μg/ml of streptomycin), 10 mM HEPES buffer, and 2 × 10-5 M 2-mercaptoethanol. The cells were seeded into 12-well tissue culture plates (Nunc, Roskilde, Netherlands) and after 1 week expanded in 25-cm2 tissue culture flasks (Corning, Schipol-Rijk, Netherlands). For the growth on nanofibers, cells growing in vitro for 2–3 weeks were used.
MSCs were isolated from femurs and tibias of BALB/c mice. The bone marrow was flushed out, a single-cell suspension was prepared by homogenization, and the cells were seeded at a concentration of 4 × 106 cells/ml in complete RPMI-1640 medium in 25-cm2 tissue culture flasks (Corning). On the following day the nonadherent cells were washed out and the adherent cells were cultured with a regular exchange of the medium and passaging of the cells to maintain their optimal concentration. After 3 weeks of culturing, the cells were characterized phenotypically by flow cytometry (over 90% of them were MHC class II-, CD86-, and CD11b-, but the majority was CD105+) and for their ability to differentiate into adipocytes (data not shown).
Mouse corneal epithelial and endothelial cell lines, prepared by the immortalization of mouse corneal epithelial and endothelial cells (11), were also tested for their growth on nanofiber scaffolds.
Demonstration of the Immunosuppressive Properties of MSC In Vitro
Spleen cells (0.5 × 106/ml) from BALB/c mice were cultured in 200 μl of RPMI-1640 medium containing 10% FCS in 96-well tissue culture plates (Nunc), either unstimulated or were stimulated with 1.0 μg/ml of concanavalin A (Con A, Sigma). MSC were added to these cultures at a ratio of MSCs to spleen cells of 1:2, 1:4, or 1:8. Cell proliferation was determined by incorporation of [3H]thymidine (1 μCi/well, Nuclear Research Institute, Rez, Czech Republic) added to the cultures for the last 6 h of a 72-h incubation period. The cells were harvested using an Automash 2000 cell harvestor (Dynex, Chantilly, VA) and the radioactivity was determined. The presence of IFN-γ in the supernatants was assessed by an enzyme-linked immunosorbent assay (ELISA) using capture and detection anti-cytokine antibodies purchased from PharMingen (San Diego, CA) and following the instructions of the manufacturer.
Morphology of Cells Growing on Nanofibers and Plastic
LSCs or MSCs were cultured at various cell concentrations on nanofibers fixed in the inserts or on plastic surfaces. Nanofiber scaffolds were cut into squares (approximately 1.5 × 1.5 cm) and fixed into CellCrownTM24 inserts (Scaffdex, Tampere, Finland). The inserts with nanofibers were sterilized by UV light, soaked in sterile destilled water, washed in culture medium, and transferred into 24-well tissue culture plates (Corning). Fifty thousand cells in a volume of 700 μl of culture RPMI-1640 medium with 10% of FCS was transferred into each well. One or 2 days after seeding, the cells were fixed for 15 min in 4% paraformaldehyde, washed with phosphate-buffered saline (PBS), and treated with Chemiblocker (1:20, Chemicon, Temecula, CA) and Triton X-100 (0.2%, Sigma). To label F-actin, the cells were incubated with Alexa fluor 568 Phalloidin (Molecular Probes, Invitrogen, Paisley, UK) diluted 1:300 in PBS containing 1% bovine serum albumin (Sigma) and 0.5% Triton X-100 (Sigma) overnight at room temperature. The nuclei were vizualized by using 4′,6-diamidino-2-phenylindole (DAPI) fluorescent dye (Invitrogen). Images were taken by a laser scanning confocal microscope (Zeiss, Jena, Germany).
Determination of Cell Proliferation
The proliferation of cells growing on plastic or nanofibers was determined according to [3H]thymidine incorporation. The cells (50 × 103/well/700 μl of culture medium) were seeded into the wells of 24-well tissue culture plates (Corning) with or without inserts containing nanofibers. The plates were incubated for 24 or 48 h and cell proliferation was determined by adding [3H]thymidine (3 μCi/well, Nuclear Research Institute) for the last 6 h of the incubation period. The radioactivity incorporated in cells growing on plastic or nanofibers was measured using a Tri-Carb 2900TR scintilation counter (Packard, Meridien, CT).
Determination of Metabolic Cell Activity
The metabolic activity of living cells was determined by the WST assay. The assay is based on the ability of living cells to cleave by mitochondrial dehydrogenases tetrazolium salts into water soluble formazan, which is then measured by spectrophotometry. Fifty thousand cells in 700 μl of RPMI-1640 culture medium were cultured in the wells of 24-well tissue culture plate (Corning) with or without inserts containing nanofibers for 24 h at 37°C in an atmosphere of 5% CO2. WST-1 reagent (Roche, Mannheim, Germany) (10 μl/100 μl of the medium) was added to each well, and the plates were incubated for another 4 h to form formazan. Formazancontaining medium (100 μl) was transferred from each well into the wells of a 96-well tissue culture plate (Corning) and the absorbance was measured using a Sunrise Remote ELISA Reader (Grödig, Austria) at a wavelength of 450 nm.
A Model of the Damaged Ocular Surface and Cell Transfers
The recipient BALB/c mice were deeply anesthetized by an intramuscular injection of a mixture of xylazine and ketamine (Rometar, Spofa, Prague, Czech Republic). The surface (corneal region) of the right eye was damaged by epithelial debridement with a sharp needle (G23) and the limbus was cut out with Vannas scissors (Duckworth and Kent, Baldock, UK). To induce a stronger immune reaction in the anterior segment of the eye, an allogeneic limbus from C57BL/6 donors was grafted orthotopically to the recipients with a removed limbus according to the technique of Maruyama et al. (17). A 4-mm-diameter nanofiber circle (with or without SCs) was used to cover the limbal and corneal region and was sutured with four interrupted sutures using 11.0 Ethilon (Ethicon, Johnson & Johnson, Livingston, England) on the damaged ocular surface. The nanofibers with growing cells were transferred with the cell side facing down towards the ocular surface. For the cell transfer, equal numbers of LSCs and MSCs growing on the nanofiber scaffold were transferred, approximately 4 × 104 cells of each type. The eyelids were closed by tarsorhapy using one suture of Resolon 7.0 (Resorba, Nuremberg, Germany) for 72 h. An ophthalmic ointment compound containing bacitracin and neomycin (Ophthalmo-Framykoin, Zentiva, Prague, Czech Republic) was applied on the ocular surface for 3 days. The nanofiber scaffolds were removed from the ocular surface on day 3 after the operation.
To trace the fate and survival of LSCs after their transfer onto the ocular surface, the cells were labeled with the fluorescent vital dye PKH26 (PKH26 Red Fluorescent Cell Linker Kit, Sigma) according to the instructions of the manufacturer, cultured for 24 h on a nanofiber scaffold, and transferred on the damaged eye surface as described above. The recipients were killed 2, 7, or 14 days after cell transfer and the whole globes were dissected and placed for 1 h into 4% paraformaldehyde. Then the globes were transferred into a 15% sucrose solution in PBS for 24-h fixation; subsequently, cryosections at a thickness of 7 μm were prepared using a Leica CM 3050 S cryostat (Leica, Wetzlar, Germany). The nuclei were stained with DAPI. The presence of stained cells was analyzed using a fluorescent microscope.
Determination of Inflammatory Reaction by Real-Time PCR
The expression of genes for IL-2 and IFN-γ and for inducible nitric oxide synthase (iNOS) in cells from the ocular surface was detected by real-time PCR. The whole ocular surface (including the cornea and limbal region) was removed using Vannas scissors on day 7 after the operation and transferred into Eppendorf tubes containing 200 μl of TRI Reagent (Molecular Research Center, Cincinnati, OH). Total RNA was extracted using TRI Reagent according to the manufacturer's instructions. Total RNA (2 μg) was treated using deoxyribonuclease I (Sigma) and used for subsequent reverse transcription. The first-strand cDNA was synthesized using random hexamers (Promega, Madison, WI) in a total reaction volume of 25 μl using M-MLV Reverse Transcriptase (Promega). Quantitative real-time PCR was performed in an iCycler (BioRad, Hercules, CA) using the primers described in Table 1. iQ SYBR Green Supermix (BioRad) was used in all experiments. The PCR parameters included denaturation at 95°C for 3 min, then 40 cycles at 95°C for 20 s, annealing at 60°C for 30 s, and elongation at 72°C for 30 s. Fluorescence data were collected at each cycle after an elongation step at 80°C for 5 s and were analyzed on the iCycler Detection system, Version 3.1. Each single experiment was done in triplicate. The relative quantification model was applied to calculate the expression of the target gene in comparison to GAPDH used as an endogenous control.
Mouse Primer Sequences Used for Real-Time PCR

Statistical Analysis
Analysis of data showed normal distribution and the results are expressed as mean ± SE. Comparisons between two groups were analyzed by Student t-test, and multiple comparisons were analyzed by ANOVA followed by Bonferroni post hoc test. A value of p < 0.05 was considered statistically significant.
Results
Comparison of Metabolic Activity and Growth Properties of Cells Growing on PA6/12 Nanofibers or on Plastic Surfaces
LSCs, MSCs, and corneal epithelial or corneal endothelial cells were grown for 24 or 48 h on nanofibers (fixed in inserts) or on a plastic surface in 24-well tissue culture plates and their metabolic and proliferative activities were determined. As demonstrated in Figure 1, all four cell types had comparable metabolic activities and proliferative capacities irrespective of whether they grew on the plastic surface or on nanofibers. The growth of LSCs and MSCs on nanofibers was confirmed when the metabolic activity that corresponds to the number of living cells was determined at different time intervals. As shown in Figure 2, the metabolic activity of SCs gradually increased during the 48-h incubation period.

Metabolic and proliferative activities of LSCs, MSCs, and corneal epithelial and corneal endothelial cells growing on plastic or nanofibers. The same number of cells was seeded on plastic (24-well tissue culture plate) or PA6/12 nanofibers fixed in inserts. (A) The metabolic activity was determined by adding WST-1 reagent to the cultures for the last 4 h of a 24-h incubation period. (B) The proliferative activity was determined by adding [3H]thymidine into the culture medium for the last 6 h of a 24-h incubation period. Each bar represents the mean ± SE from three to four determinations.

Comparison of the growth of LSCs and MSCs on plastic or nanofibers. Equal numbers of MSCs (A) or LSCs (B) were seeded into the wells of a 24-well tissue culture plate or onto PA6/12 nanofibers fixed in inserts and the metabolic activity of the living cells was determined at the beginning of culture (0 h) and after 24- or 48-h incubation. Each bar represents the mean ± SE from three determinations.
The Morphology of Cells Growing on Nanofibers
The shape of the cells and the organization of the actin cytoskeleton of LSCs growing on PA6/12 nanofibers were compared with those of cells growing on plastic surfaces. Figure 3 shows that the shape of the cells and the organization and thickness of the actin filaments formed in adherent cells were comparable between cells growing on nanofibers and plastic surfaces. Confocal and electron microscopy showed the penetration of LSCs into the nanofibrous structure and the growth of pseudopodia among the nanofibers (data not shown).
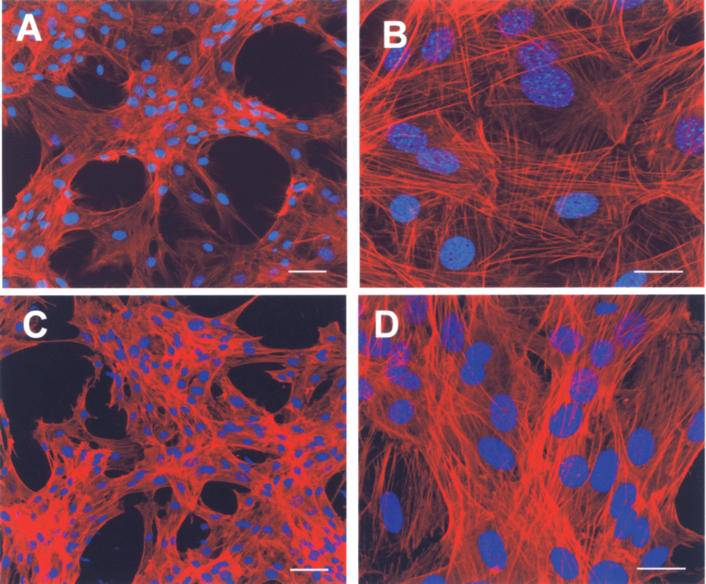
The morphology of LSCs growing on a glass surface or on PA6/12 nanofibers. The cells were cultured for 24 h on poly-l-lysine-coated glass inserts in 24-well tissue culture plates or on nanofibers fixed in inserts and were stained for F-actin with phalloidin (red filaments). The nuclei are blue (DAPI staining). (A, B) LSCs growing on the glass surface at two different magnifications. (C, D) LSCs growing on nanofiber scaffolds. Scale bars: (A, C) 50 μm; (B, D) 20 μm.
Transfer of LSCs and MSCs Using Nanofiber Scaffolds Onto the Damaged Ocular Surface
To prove that LSCs can be transferred using a nanofiber scaffold onto the ocular surface and that they can subsequently migrate from the scaffold onto the damaged ocular surface, we labeled LSCs with the fluorescent dye PKH26, cultured them on PA6/12 nanofibers, and transferred them onto the damaged ocular surface. The globes were harvested at different time intervals after cell transfer and cryosections were prepared. As demonstrated in Figure 4, PKH26-labeled cells were clearly detected on the ocular surface on days 2, 7, and 14 after cell transfer.

Detection of PKH26-labeled LSCs on the damaged ocular surface after their transfer on a PA6/12 nanofiber scaffold. LSCs were labeled with PKH26 and cultured for 24 h on nanofibers fixed in inserts. The nanofibers with cells were transferred and fixed (with the cell side facing down) for 3 days on the damaged ocular surface. The globes were removed 2, 7, or 14 days after the operation and 7-μm cryosections were prepared. The nuclei were vizualized with DAPI. The cryosections were prepared (A) from the control undamaged eye (without labeled cells), (B) from the eye 2 days after operation (the nanofiber scaffold with labeled LSCs is seen as a red lane, corneal epitheliun is removed), and from the eyes 7 (C) and 14 (D) days after the cell transfer (red stained cells are still present, the corneal epithelium is regenerated). Scale bars: 50 μm.
Suppression of a Local Inflammatory Reaction by LSCs and MSCs
First, we demonstrated the immunosuppressive properties of MSC in vitro. Mouse spleen cells were stimulated with the T-cell mitogen Con A in the presence or absence of MSCs. As demonstrated in Figure 5, MSCs inhibited cell proliferation (Fig. 5A) and IFN-γ production (Fig. 5B) in a dose-dependent manner. No immunosuppression was observed if LSCs were used instead of MSCs, and the suppression by MSCs was preserved if MSCs were tested in a mixture with LSCs (data not shown).
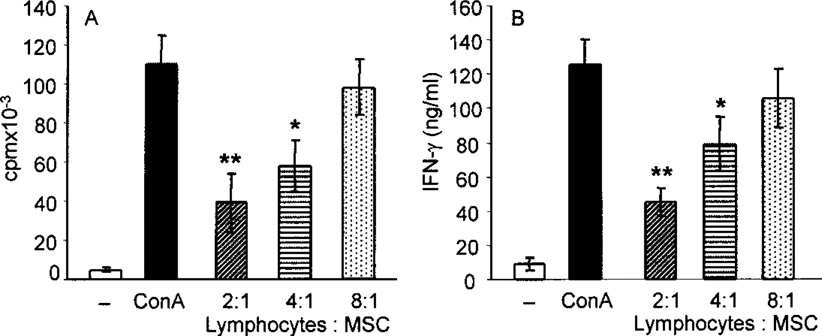
Immunosuppressive properties of MSCs in vitro. Spleen cells from BALB/c mice were cultured either unstimulated or stimulated with Con A (1.0 μg/ml) in the presence or absence of MSCs (at a ratio of 2:1, 4:1, or 8:1). Cell proliferation (A) was determined by [3H]thymidine incorporation after a 72-h incubation period, the production of IFN-γ (B) was measured by ELISA in the supernatants after a 48-h incubation period. Each bar represents the mean ± SE from three experiments. *p < 0.01, **p < 0.001.
To demonstrate the suppression of a local inflammatory reaction by the transfer of LSCs and MSCs in vivo, two models of ocular surface damage and treatment were used. In the first model, the ocular surface was mechanically damaged. The second model combined mechanical damage and the transplantation of allogeneic limbus. In the healthy, nondamaged eye, no detectable expression of IL-2, IFN-γ, or iNOS genes was found. The mechanical injury induced a moderate inflammatory reaction associated with the production of IFN-γ and iNOS. This response was inhibited after the transfer of nanofibers containing LSCs and MSCs (data not shown). The ocular surface damage associated with orthotopic limbal allotransplantation induced a strong inflammatory reaction characterized by the expression of the IL-2, IFN-γ, and iNOS genes (Fig. 6). This reaction was slightly inhibited by covering the eye surface with cell-free nanofibers and was significantly attenuated after the transfer of LSCs and MSCs growing on nanofiber scaffold (Fig. 6).

Suppression of the local inflammatory reaction after the transfer of LSCs and MSCs on a nanofiber scaffold. The inflammatory reaction was induced on ocular surface by epithelial debridement and orthotopic limbal allotransplantation. The ocular surface then remained untreated, covered with nanofibers without cells or covered with nanofibers with LSCs and MSCs. On day 7 after cell transfer the ocular surface was removed and the expression of genes for IL-2, IFN-γ, and iNOS was determined by real-time PCR. The comparative Ct method was used to determine the extent of the targeted gene expression normalized to an internal GAPDH control. Each bar represents the mean ± SE from three experiments (two mice in each experiment). *p < 0.05 in comparison to uncovered damaged eyes, **p < 0.001 in comparison to damaged eyes covered with cell-free (empty) nanofibers.
Discussion
A growing body of recent studies has shown that electrospun nanofibers of various polymers allow the adhesion, proliferation, metabolic activity, morphology, and organized assembly of different cell types in vitro (2,5,18,25,35). The constructs formed by nanofiber scaffolds and specialized cells have been suggested as perspective and promising tools for tissue engineering (14,15). However, the performance and behavior of nanofibrous materials in vivo are not well understood. Only scarce recent data are available to demonstrate the usefulness of nanofiber scaffolds in vivo.
We described here the preparation of electrospun nanofibers and their use as a scaffold to grow and transfer LSCs and MSCs to treat ocular surface injuries and SC deficiencies. The nanofibers were prepared by the original needleless Nanospider technology, which enables the creation of nanofibers from various polymers and of defined fiber diameter, porosity, and a basis weight. From a large panel of polymers tested in our pilot experiments, we selected copolymer PA6/12. This polymer turned out to be sufficiently biocompatible, forming nanofibers stable in aqueous solutions and suitable for the growth of limbal and mesenchymal SCs. In addition, differentiated corneal epithelial and endothelial cells were grown on PA6/12 nanofibers and their metabolic and proliferative activities were comparable with those of the same cells grown on plastic surfaces.
We have made a successful attempt to use the nanofiber scaffolds for the transfer of LSCs to treat ocular surface injuries or LSC deficiencies in an experimental mouse model. The limbus represents the region in the eye where SCs reside that are responsible for corneal renewal and repair (4,31). We have recently shown that the cells obtained by trypsinization of limbal tissue express the markers and characteristics of LSCs and that these cells can be propagated in vitro in tissue culture (13). Here we have shown that LSCs can be cultured on a polyamide nanofiber scaffold and that their proliferation, metabolic activity, and morphology when grown on nanofibers are comparable with those of cells grown on plastic surfaces. In addition, we have shown that LSCs growing on a nanofiber scaffold can be successfully transferred onto the damaged ocular surface. So far, human amniotic membranes have been used most frequently as a matrix for the growth and transfer of LSCs for therapeutic purposes (26,30). In spite of the use of human allogeneic amniotic material as a scaffold of LSCs for the reconstruction of the ocular surface, the beneficial effects of LSC transplantation have been reported (22). The use of biocompatible synthetic polymers for the preparation of nanofiber scaffolds for the growth and transfer of LSC would have apparent advantages.
We are aware that the limbal cell population that we transferred using the nanofiber scaffold was not a pure SC population. The population contained also differentiated cells originating from LSCs and other cell types of the LSC niche. Analysis of the gene expression of the transferred cells revealed that this cell population contained both differentiated epithelial cells (expressing CK12 and connexin 43) and cells expressing the putative LSC markers ABCG2, p63, and Lgr5 [(13), and unpublished data]. However, such a cultured population of limbal epihelial cells is generally referred to as LSCs, and their therapeutic potential in the treatment of LSC deficiencies has been documented (22,32).
In addition to LSCs, we also grew on nanofiber scaffolds MSCs, which can differentiate to various cell types including corneal epithelial cells (8,19,23) and that have the ability to inhibit immunological reactions (1,6). We confirmed the immunosuppressive effects of MSCs in vitro by the inhibition of T-lymphocyte proliferation and by the suppression of cytokine production. It has been shown that the transfer of MSCs onto the damaged eye can support the healing process, but the effects of MSCs were attributed to the ability of MSCs to inhibit inflammatory reactions (15,21). In our experiments we cocultured LSCs and MSCs on nanofiber scaffolds and transferred them onto the damaged ocular surface where a local inflammatory reaction characterized by the expression of genes for IL-2, IFN-γ, and iNOS was induced by epithelial debridement and limbal allotransplantation. Analysis of the gene expression in tissue harvested from the damaged ocular surface 1 week after the injury showed that there was a significant suppression of the gene expression of the inflammatory proteins IL-2, IFN-γ, and iNOS, suggesting that the MSCs suppress the inflammatory reaction. This suggestion is supported by previous data showing that MSCs applied to the ocular surface can inhibit the local inflammatory reaction (15,21).
It has been demonstrated previously that embryonic SCs can be grown, propagated, and differentiated on nanofibers (20,28). Our results suggest that nanofiber scaffolds prepared from PA6/12 by electrospinning technology can serve as a convenient matrix for the growth of adult tissue-specific SCs and for their transfer to treat ocular surface injuries. Studies are in progress to test the survival and propagation of cells in terms of the persistance of SC markers and characteristics during their growth on nanofiber scaffolds and to monitor the presence of LSCs after their transfer onto the ocular surface in experimental LSC deficiency models.
Footnotes
Acknowledgments
This work was supported by grant KAN200520804 from the Grant Agency of the Academy of Sciences, grant 310/08/H077 from the grant Agency of the Czech Republic, projects 1M0506 and MSM0021620858 from the Ministry of Education of the Czech Republic, and project AVOZ50520514 from the Academy of Sciences of the Czech Republic.
