Abstract
Worldwide, colorectal cancer is the third most common type of cancer affecting both sexes. It has been proposed that a small subset of cancer cells (cancer stem cells) within each tumor is able to initiate tumor growth. In 2007, two research groups simultaneously identified a colon cancer stem cell population in human tumors by the use of CD133 expression. In the present study, we used a human colon cancer cell line, SW620, to analyze the cancer stem cell-like characteristics of CD133+ cells in vitro and in vivo. In vitro, CD133+ SW620 cells had a higher proliferative capacity, were more irradiation- and chemotherapy-resistant, and had a higher expression of β-catenin compared with CD133- cells. Injections of either CD133+ or CD133- cells into the skin or rectal mucosa of NOD/SCID mice led to tumors; however, injection of CD133+ cells resulted in the formation of larger tumors. Tumors derived from injections of CD133- cells did not contain any CD133+ cells, whereas tumors derived from injections of CD133+ cells did contain CD133+ cells, suggesting self-renewing capability. However, the proportion of CD133+ cells in the newly formed tumors in vivo was lower than the proportion of CD133+ cells in vitro. In conclusion, the human colon cancer cell line, SW620, contains both CD133+ and CD133- phenotypes, and the CD133+ phenotype has characteristics consistent with those of cancer stem cells.
Keywords
Introduction
Colorectal cancer (CRC) is the third most common form of cancer and the second leading cause of cancer-related death in the Western world, causing 655,000 deaths worldwide per year (12). Knowledge related to the occurrence, development, and treatment of CRC has been accumulating, and this has been accompanied by remarkable developments in medical treatment. However, radical cures for metastasis and recurrent CRC remain difficult, and the establishment of strategies for new treatments is expected. In recent years, it has become clear that only a small proportion of cells in cancer tissues have the ability to proliferate. These cells are called cancer stem cells (CSCs) or cancer-initiating cells and are characterized by having a self-renewing ability, differentiation potential that gives configurationality to cancer tissues, and tumorigenic potential (13, 16). The assumption that CSCs are responsible for tumor growth was first proven in acute myeloid leukemia by John Dick's group in 1997 (3). Subsequently, CSCs have been isolated in solid cancers by their self-renewing activity and daughter cell-producing ability. Al-Hajj's group first identified CSCs in solid tumors; they demonstrated that only a small fraction of breast cancer cells expressing CD44+CD24-/low were able to form tumor xenografts and to give rise to both tumorigenic and non-tumorigenic cells (1).
In human CRC, two research groups identified a CSC population by using a cell surface marker, CD133 (15, 17). CD133 is an antigen that is a 120-kDa five-transmembrane domain glycoprotein expressed on normal primitive hematopoietic, endothelial, neural, and epithelial cells (18, 22, 23). In addition, CD133 is a marker of CSCs in brain tumors, pancreatic cancer, and lung cancer (8, 9, 19).
It is now thought that these CSCs are involved not only in the formation of tumors, but also in metastasis and, moreover, they have resistance against existing chemical and irradiation therapies (2, 6, 9, 10).
Based on such knowledge, CSCs have gained attention as targets for new cancer treatments. In this study, we investigated the cancer stem cell-like properties of CD133+ cells by evaluating differences from CD133-cells with respect to irradiation resistance, chemotherapy resistance, and tumorigenic potential using a human CRC cell line, SW620.
Materials and Methods
Cell Culture
A human colon cancer cell line, SW620, was used in the present study. SW620 cells were maintained in high-glucose DMEM medium (Invitrogen) containing 10% fetal bovine serum (Invitrogen), 100 U/ml penicillin G, and 100 μg/ml of streptomycin (Invitrogen) at 37°C in a 5% humidified CO2 atmosphere.
Analysis of Cell Proliferation
Cell-proliferating capacity was routinely examined during the culture of SW620 cells. Isolated cells were seeded at 1 × 103 cells/well in a 96-well microtiter plate (Microtest Tissue Culture Plate, Becton Dickinson, Tokyo, Japan) in a final volume of 100 μl of culture medium per well. After incubation, cell proliferation was analyzed by an MTS assay (CellTiter-Glo Luminescent Cell Viability Assay, Promega, Madison, WI), and the absorbance at 492 nm was determined by a plate reader (Multiskan JX; Thermo Labsystems).
CD133 Expression Analysis in SW620 Cells
Dissociated SW620 cells were stained with anti-CD133/2-APC antibody (Miltenyi Biotec, Tokyo, Japan), incubated for 10 min at 4°C, and analyzed by a FACSAria cell-sorting system (Becton Dickinson). Mouse IgG2b-APC (Miltenyi Biotec) was the isotype control antibody. Dead cells were eliminated with propidium iodide.
Magnetic Cell Separation of SW620 Cells by CD133 Expression
SW620 cells were separated by immunomagnetic selection on the basis of CD133 expression. Cells were labeled with CD133/1 microbeads using a Miltenyi Biotec CD133 cell isolation kit according to the manufacturer's protocol and separated by autoMACS (Miltenyi Biotec). After magnetic sorting, cell purity was evaluated by flow cytometry as described above.
Analysis of Resistance to a Chemotherapeutic Agent
5-Fluorouracil (5-FU) was used as a chemotherapeutic agent to analyze the chemotherapy resistance of SW620 cells. Isolated cells were seeded at 1 × 104 cells/well in a microtiter plate (Becton Dickinson) in a final volume of 100 μl of culture medium with either 0, 1, or 10 μg/ml 5-FU per well. After incubation for 48 h, cell proliferation was analyzed by an MTS assay as described above.
Analysis of Resistance to Irradiation
In order to examine the irradiation resistance of SW620 cells, isolated cells were seeded at 1 × 104 cells/well in a microtiter plate (Becton Dickinson) in a final volume of 100 μl of culture medium per well. After incubation for 24 h, cells were irradiated at 4 or 6 Gy. After further incubation for 48 h, cell proliferation was analyzed by an MTS assay.
Analysis of the Expression of a Self-Renewal Gene, β-Catenin
Reverse transcriptase-polymerase chain reaction (RT-PCR) analysis was conducted in SW620 cells to examine the difference in the expression of a self-renewal gene, β-catenin, between CD133+ and CD133- SW620 cells. Total RNA was extracted from CD133+ and CD133- cells using TRIzol (Invitrogen) reagent according to the manufacturer's instructions. Reverse transcription was performed at 22°C for 10 min, followed by 42°C for 20 min, using specific primers with 2.0 μg of RNA per reaction. Primers used were β-catenin, sense 5′-ACTGGCAGCAACAGTCTTACC-3′ and antisense 5′-TTTGAAGGCAGTCTGTCGTAAT-3′, and human glyceraldehyde-3-phosphate dehydrogenase (GAPDH) as an internal control, sense 5′- TCCCCATCACCATC TTCCAG-3′ and antisense 5′-GAGTCCTTCCACGAT ACCAA-3′. Amplification was performed for 27 cycles (25 cycles for GAPDH) for 1 min at 95°C, 1 min at 59.3°C (60°C for GAPDH), and 1 min at 72°C. PCR products were resolved on 1% agarose gels and visualized by ethidium bromide staining.
Animal Experiments
All animal procedures were approved by the Okayama University Institutional Animal Care and Use Committee, and thus within the guidelines for human care of laboratory animals. Six- to 8-week-old nonobese/severe combined immunodeficiency (NOD/SCID) mice (Charles River Laboratories, Tokyo, Japan) were used in the following transplantation experiments. The mice were maintained in a central animal facility at Okayama University on a 12-h light/dark cycle, receiving water and nutrition ad libitum.
Transplantation Experiments
NOD/SCID mice (6–8 weeks of age) were anaesthetized with the use of diethyl ether solution. Purified CD133+ or CD133- SW620 cells (5 × 104) were suspended in a 1:1 mixture of culture medium and Matrigel (BD Biosciences, Tokyo, Japan) and injected subcutaneously into the mice. For submucosal transplantation into the rectum, mice were placed in a supine position, the ano-rectal wall was cut at a length of 7 mm to prevent colonic obstruction resulting from rectal tumor progression (21), and cells (5 × 104) were slowly injected into the submucosal space with a 30-gauge needle. Mice were sacrificed at 5 weeks posttransplantation.
Histopathological Analyses
At the time of sacrifice, the developed tumors were removed, fixed in 20% formalin, and embedded in paraffin. Serial tissue sections were stained with hematoxylin and eosin (H&E) or examined immunohistochemically using anti-CD133/1 antibody (Miltenyi Biotec Co., Tokyo, Japan). For immunohistochemical staining, tissue sections were processed with an automated immunostainer (Ventana XT system BENCHMARK XT, Ventana Japan, Japan) using heat-induced epitope retrieval and standard DAB detection kit (Ventana). Endogenous peroxidase activity was quenched by immersion in 3% hydrogen peroxide for 10 min before automated imunohistochemistry. Ten different sections were examined for CD133 expression analysis in each group by third-party examiners.
Statistical Analyses
Mean values are presented with SDs. Differences between groups were estimated by Student's t-test analysis. All differences were deemed significant at the level of p < 0.05.
Results
Identification of CD133 Expression in SW620 Cells
Flow cytometry was used to determine CD133 expression level in SW620 cells. The results showed that 39.62% of SW620 cells were CD133+ (Fig. 1B). After magnetic selection using CD133/1 microbeads, cell purity was evaluated by flow cytometry using CD133/2-APC antibody. The purity was 97.2% in CD133+ cells (Fig. 1C), and 98.8% in CD133- cells (Fig. 1D), indicating that high purity separation was obtained by this procedure.

Analysis of CD133 expression in the colon cancer cell line SW620 by flow cytometry. (A) Isotype control, (B) proportion of CD133+ cells in cultures of SW620 cells, (C) proportion of CD133+ cells in the CD133+ population after magnetic sorting cells, (D) proportion of CD133+ cells in the CD133- population after magnetic sorting.
Proliferation, Chemotherapy Resistance, Irradiation Resistance, and Gene Expression of CD133+ SW620 Cells
We examined the in vitro proliferation of CD133+ and CD133- SW620 cells using a MTS assay. As shown in Figure 2A, CD133+ cells proliferated at a higher rate than did CD133- cells. As well, CD133+ cells showed significantly higher proliferation in the presence of both 1 and 10 μg/ml 5-FU (Fig. 2B) and after 6 Gy irradiation (Fig. 2C). β-Catenin mRNA expression was detected in both CD133+ and CD133- SW620 cells by RT-PCR analysis. CD133+ cells possessed a higher level of β-catenin expression CD133 cells (Fig. 2D), which was confirmed by real-time PCR analysis.

In vitro analysis of CSC-like characteristics of CD133+ cells compared with CD133- cells. (A) Cell proliferation as determined by MTS assay. (B) Proliferation in the presence of various concentrations of 5-FU. (C) Proliferation after irradiation at 6 Gy. (D) Expression of β-catenin mRNA as determined by RT-PCR. GAPDH was used as an internal control. Data are means ± SDs.
Histopathological Examination of Developed Tumors In Vivo
We examined whether there were differences in the tumor-forming ability of CD133+ and CD133- SW620 cells when transplanted into subcutaneous or rectal sites in NOD/SCID mice. Transplantation of both CD133+ and CD133- cells led to the formation of tumors composed of poorly differentiated adenocarcinoma (Figs. 3, 4, and 5). Transplantation of CD133+ cells resulted in faster tumor formation and larger tumors in both sites (Fig. 3). The tumors formed by CD133+ cell transplant exhibited more active mitosis of tumor cells, thus reflecting the high level of tumorigenic potential. Immunochemical analysis of the tumors that developed from transplantation of CD133- cells showed that there were no CD133+ cells within the tumors. On the other hand, in the tumors developed by transplantation of CD133+ cells, CD133+ cells were observed within the tumor at a proportion of 1.48 ± 0.39% for the subcutaneous site and 4.19 ± 0.62% for the rectal mucosa (Figs. 4 and 5). No mice showed metastases at the time of sacrifice.

Tumor-forming ability of CD133+ cells. (A) Representative photograph of a mouse taken 5 weeks after subcutaneous transplantation of CD133+ or CD133- cells. (B) Representative photograph of a mouse and a tumors taken 5 weeks after transplantation of CD133+ or CD133- cells into the rectal mucosa. Scale bar: 1 cm. (C) Table shows the tumor size after transplantation of 50,000 CD133+ or CD133- SW620 cells.
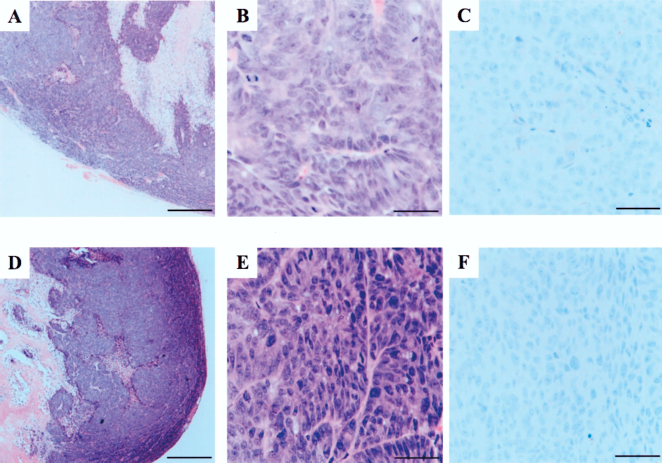
Histopathological analysis of tumors that developed after subcutaneous transplantation. (A, B, C) Sections of tumors that developed after the transplantation of CD133+ cells were stained with (A, B) hematoxylin and eosin or (C) anti-CD133 antibody. Sections of tumors that developed after transplantation of CD133- cells were stained with (D, E) hematoxylin and eosin or (F) anti-CD133 antibody. (A, D) Original magnification: 40x, scale bar: 500 μm. (B,C, E, F) Original magnification: 400x, scale bar: 50 μm.

Histopathological analysis of tumors that developed after transplantation into the rectal mucosa. (A, B, C) Sections of tumors that developed after the transplantation of CD133+ cells were stained with (A, B) hematoxylin and eosin or (C) anti-CD133 antibody. Sections of tumors that developed after transplantation of CD133- cells were stained with (D, E) hematoxylin and eosin or (F) anti-CD133 antibody. (A, D) Original magnification: 40x, scale bar: 500 μm. (B, C, E, F) Original magnification: 400x, scale bar: 50 μm.
Discussion
In 1997, it was reported that only some leukemia cells have tumorigenic potential, and these could be distinguished by using surface markers (3). Cells with such characteristics are called cancer stem cells (CSCs), and many studies on CSCs have been carried out. Moreover, in cases of solid cancer, there have been many reports on the identification and separation of CSCs. Regarding human colorectal cancer (CRC), there were two reports in 2007 on the identification and separation of CSCs using CD133 as a marker. These studies found that all cells capable of initiating colon cancer were positive for CD133 and speculated that CD133 is a marker for CSC in CRC (15, 17). Therefore, in the present study, we used a CRC cell line, SW620, to investigate the CSC-like characteristics of CD133+ cells with regard to in vitro proliferating potential, resistance against 5-FU, resistance against irradiation, and tumor formation in vivo.
We found that CD133+ cells existed at a ratio of 39.6% in the human colon cancer cell line, SW620. In other studies, the ratios of CD133+ cells in CRC cells were 2.5 ± 1.4% and 8.9–15.9% (15, 17). According to the previously reported literature on human CRC cell lines, the proportion of CD133+ cells ranged from 99.0% to less than 1%, and therefore the frequency is not constant at all in the studies using cell lines (11). This variability may be explained by the fact that in established cell lines, changes in the characteristics of the primarily isolated parental cells occur to some degree due to the accumulation of DNA methylation and unexpected mutations during culture. The major advantage of the use of well-established cell lines allows us to repeat the experiments and share the data with researchers all over the world.
In vitro, we found that CD133+ SW620 cells exhibited a higher proliferating potential and showed a higher resistance to 5-FU and irradiation compared with CD133- cells, which is consistent with the characteristics of CSCs. Other studies have reported mechanisms for the drug and irradiation resistance of CD133+ cells. Drug-resistance mechanisms include a higher mRNA expression of the antiapoptotic molecules. Breast cancer resistance protein 1 (BCRP 1) and O6-methylguanine-DNA methyltransferase (MGMT) in CD133+ glioblastoma cells (14) and the autocrine effect of interleukin-4, which protects from apoptosis, in freshly obtained human CRC specimens (20). Irradiation-resistance mechanisms include a stronger DNA repair ability in CD133+ glioblastoma cells compared with CD133- cells (2).
Next, we examined the in vivo characteristics of CD133+ cells by transplanting them into subcutaneous tissue and rectal submucosa of NOD/SCID mice. Formation of tumors was observed after transplantation of both CD133+ and CD133- cells in both sites; however, CD133+ cells produced larger tumors, indicating a higher tumorigenic potential. This result is in contrast with those of previous studies in which CD133- cells isolated from fresh human CRC specimens did not have tumorigenic potential (15, 17). However, our results are consistent with those of Ieta et al., who also reported that CD133- cells in a CRC cell line formed tumors and that CD133+ cells had a higher tumorigenic potential than CD133- cells (11). Moreover, there is also a report that CD133- cells from a glioma cell line (C6) exhibited tumorigenic potential and had characteristics similar to CSCs, even though CD133 is considered to be a CSC marker in brain cancer (24).
In both sites, tumors that developed from transplantation of CD133+ cells contained CD133+ cells, whereas no CD133+ cells were observed in the tumors induced by transplantation of CD133- cells. In the tumors formed by transplantation of CD133+ SW620 cells, the frequency of occurrence of CD133+ cells was higher in the rectum compared with the subcutaneous site, suggesting that the rectal mucosa provided a better niche for CSCs. However, the frequency of CD133+ cells in vivo (1.48–4.19%) was lower than the 39.6% observed in vitro. Under in vitro culture conditions, cells receive many stresses such as cell passage, which might favor those cells with higher proliferation capacity, resulting in high ratios of CD133+ cells. As well, environmental factors in vivo may affect the behavior of CD133+ cells. Some of this difference may be due to the different assay methods used: flow cytometry for in vitro experiments and immunochemical staining for in vivo experiments.
Ideally, experiments characterizing CSCs in CRC would be best performed using human tumor tissue as a source. As surgeons, we have access to such tissue and have recently begun a project on isolation and identification of CSCs in human tumors. Unfortunately, the success rate of the primary culture of CRC cells is low at less than 20%. Xenograftment of the freshly isolated CRC cells into immunodeficient mice and spheroidal culture of the cells in serum-free medium are now under investigation to maintain the properties of the original cancer cells.
In conclusion, we found that a human colon cancer cell line, SW620, has both CD133+ and CD133- subsets and that the CD133+ subset exhibited more CSC-like characteristics compared with the CD133- subset. More refined studies are required to narrow down CSC identity by combining CD133 with other markers, such as EpCAMhigh/CD44 (4, 5, 7), and by applying in vivomimicking 3D culture systems.
