Abstract
Tissue factor (TF) and monocyte chemoattractant protein-1 (MCP-1) expressed on the islets have been identified as the main trigger of the instant blood-mediated inflammatory reaction (IBMIR) in islet transplantation. Because the key steps that directly induce TF and MCP-1 remain to be determined, we focused on the influence of brain death (BD) on TF and MCP-1 expression in the pancreatic tissues and isolated islets using a rodent model. TF and MCP-1 mRNA levels in the pancreatic tissues were similar between the BD and the control group. However, TF and MCP-1 mRNA in the fresh islets of the BD group were significantly higher than that of the control group (p < 0.01). BD may thus be suggested to be of great importance as an initiator of TF and MCP-1 induction in the isolated islets. Furthermore, the upregulation of crucial inflammatory mediators induced by BD could be exacerbated by warm ischemic damage during digestion procedures. In the present study, the islet yield and purity were affected by BD. However, almost no influences were observed with respect to islet viability, indicating that the expression of inflammatory mediators rather than islet viability is more susceptible to BD. According to the change in time course of TF and MCP-1 expression in the isolated islets, the selected time point for islet infusion in current clinical islet transplantation was thus shown to be at its worst level, at least with respect to the damage caused by BD and ischemic stress. In conclusion, BD in combination with warm ischemic stress during isolation procedures induces a high expression of TF and MCP-1 in the isolated islets. In order to reduce the expression of crucial inflammatory mediators in the islet grafts, the management of the pancreas from brain-dead donors with early anti-inflammatory treatments is thus warranted.
Keywords
Introduction
Islet transplantation is now becoming a viable option for the clinical treatment of type 1 diabetic patients (26, 27, 32). Although the Edmonton protocol introduced various suggestions for the improvement of islet transplantation, one of the most crucial messages was undoubtedly the necessity for multiple donor organs to render diabetic patient insulin independent. In other words, the Edmonton protocol could be regarded as a refined dose-finding study for the amount of islets needed to cure diabetes. Therefore, in order for islet transplantation to become a widespread standard therapy, diabetes reversal must be achieved with a single donor to reduce the risks and costs, and to increase the availability of transplantation.
However, it is well known that a large part of the transplanted islets tend to be destroyed immediately after transplantation. One of the possible explanations for the poor outcome is the instant blood-mediated inflammatory reaction (IBMIR), which is an innate immune response during islet engraftment (4). Our group, as well as others, have showed that tissue factor (TF) and monocyte chemoattractant protein-1 (MCP-1) expressed on the grafted islets elicit an injurious IBMIR when the islets come into direct contact with the blood stream (5, 11, 17, 21, 23). Therefore, in order to improve the outcome of clinical islet transplantation, the expression of these crucial inflammatory mediators in the isolated islets should be reduced prior to transplantation.
It is well known that the result of organ transplantation is highly influenced by brain death (BD) (31). In the field of islet transplantation, it was reported by Contreras et al. that BD upregulated the proinflammatory cytokines, such as tumor necrosis factor-α (TNF-α, interleukin-1β (IL-1β), and IL-6 in the serum and pancreatic tissues (6). Toyama et al. also demonstrated that TNF-α, IL-1β, IL-6, and MCP-1 were activated in the isolated islets from rodent BD donors (33). However, the influence of BD on the TF expression in the isolated islets still remains uncertain.
Furthermore, in islet transplantation, unlike other organ transplantation, the islet grafts are placed under hypoxic condition at 37°C during the whole digestion procedure. This period is theoretically considered as one kind of severe warm ischemia (2, 12, 28).
In the present study, we therefore analyzed the influence of BD on the expression of TF and MCP-1 in both the pancreatic tissues and the isolated islets, in order to understand the key steps that induce crucial inflammatory mediators in the islet grafts.
Materials and Methods
Rodent Brain Death Model
All the animals in this study were handled in accordance with the Guide for the Care and Use of Laboratory Animals published by the National Institutes of Health (3) and the guidelines for animal experiment and related activities at Tohoku University. BD was induced in male Lewis rats weighing 250–300 g by inflation of a No. 3 Fogarty catheter (Edwards Lifesciences Corporation, Irvine, CA, USA) placed intracranially, as previously described (6, 24). Briefly, anesthesia was induced with diethleter and maintained by the IP administration of pentobarbital sodium, Nembutal (Abbott Laboratories, Abbott Park, IL, USA) at a dose of 30 mg/kg. A No. 3 Fogarty catheter was inserted through a 1-mm hole drilled through the skull at 3 mm lateral to the sagittal suture. For the gradual rise in the intracranial pressure, the balloon was inflated with 40 μl/min of distilled water until respiration ceased. The absence of refluxes, apnea, and the maximally dilated and fixed pupils confirmed the condition. The average balloon volume for making BD was 210 μl. The rats were tracheotomized for intubation and mechanically respirated (respiratory rate: 60/min, tidal volume: 10 ml/kg) with SAR-830 Ventilator (CWE, Inc., Ardmore, PA, USA) for 6 h until the removal of the pancreas. The arterial blood pressure was monitored continuously via 24G SURFLO I.V. Catheters (TERUMO, Tokyo, Japan) placed into the right femoral artery and attached to a Dynascope (Fukuda Denshi, Tokyo, Japan). In order to avoid the ischemic effects, the mean arterial pressure (MAP) was maintained over 80 mmHg. When the MAP fell under 80 mmHg during the maintenance of BD, the balloon volume was reduced by 10 μl/min until the animal became normotensive. During the 6-h period, 6 ml/kg/h of normal saline solution was administered IV. The control rats were anesthetized and tracheotomized using the same method. Thereafter, a Fogarty catheter was inserted without ballooning. The control rats were not mechanically respirated because sustained anesthesia was needed for the 6-h ventilation.
Islet Isolation and Culture
Before the removal of the pancreas, the cannulated bile duct was injected with 10 ml of cold Hanks' balanced salt solutions (HBSS) containing 1 g/L collagenase (Sigma type V; Sigma Chemicals, St. Louis, MO, USA). After the addition of 10 ml HBSS, the pancreas was digested at 37°C for 14 min. Thereafter, density-gradient centrifugation was performed using Histopaque-1119 (Sigma Diagnostics, St. Louis, MO, USA) and Lymphoprep™ (Nycomed Pharma AS, Oslo, Norway) to isolate the pancreatic islets. The islet count was performed as islet equivalents (IEQs) under a scaled microscope using diphenylthiocarbazone (Wako, Osaka, Japan) staining (BD, n = 8; control, n = 7). One IEQ was the islet mass equivalent to a spherical islet of 150 μm in diameter. The islets were cultured in RPMI-1640 containing 5.5 mmol/L glucose and 10% fetal bovine serum at 37°C in 5% CO2 and humidified air before examination.
Islet Viability and Function
The adenosine triphosphate (ATP)/deoxyribonucleic acid (DNA) ratio was measured to evaluate both the energy status of the isolated islets after 3 h (BD, n = 4; control, n = 4) and the overnight culture (BD, n = 6; control, n = 5). A total of 80 IEQs of the islets were used in both groups. The ApoGlow™ kit (Lonza Rockland Inc., Rockland, ME, USA) was used for the ATP measurements as described previously (10). Using the same sample, the DNA content was measured using the DNA Quantify kit (Primary cell, Sapporo, Japan) as described previously (34). We have evaluated the respiratory activity of the isolated islets after 3 h (BD, n = 4; control, n = 4) and of the overnight culture (BD, n = 6; control, n = 5) using scanning electrochemical microscopy. The stimulation index of the respiratory activity, defined as the ratio of the respiratory activity in the high glucose (16.7 mmol/L) to that in the basal glucose (1.67 mmol/L), is a novel marker that could be applied as a rapid and potent predictor for the outcome of clinical islet transplantation (8).
Determination of TF and MCP-1 mRNA in the Pancreatic Tissues
The pieces of the pancreatic tissues from the BD and the control groups were snap-frozen in liquid nitrogen and stored at −80°C until further use (n = 6 and n = 4, respectively). The total RNA was extracted using the RNeasy Mini Kit (Qiagen, Tokyo, Japan) according to the manufacturer's protocol. The RNA concentration was estimated from the absorbance at 260 nm. The first-strand complementary DNA (cDNA) was synthesized from 2500 ng of total RNA using the Transcriptor First Strand cDNA Synthesis Kit (Roche Diagnostics, Indianapolis, IN, USA). The cDNAs were amplified by the polymerase chain reaction (PCR) using a rat TF primer and a probe, rat MCP-1 primer probe set (Nihon Gene Research Laboratories Inc., Sendai, Japan), and rat glyceraldehydes-3-phosphate dehydrogenase (GAPDH) primer probe set (Nihon Gene Research Laboratories Inc.) with a Lightcycler (Roche Diagnostics). The primer sequences of the rat TF from the 5′ to 3′ direction were as follows: forward, AGC TAC TGC TTC TTC GTA CA; reverse, AAA GAC AGT GAC CAG GAA CA. The hybridization (FRET) probe sequences from 5′ to 3′ direction were as follows: TCC CAG GAC ACT CTT CCA TTG CTC AGT G-Fluorescein; LC Red 640-ACT TGG TGA TGC TTT CTG GGC TCT TGT G-phosphorylation. In order to perform the PCR for TF and GAPDH, an initial denaturation step of 10 min at 95°C was followed by 40 cycles of 10 s at 95°C, an annealing of 15 s at 60°C, and extension of 7 s at 72°C. For MCP-1, an initial denaturation step of 10 min at 95°C was followed by 40 cycles of 10 s at 95°C, an annealing of 15 s at 62°C, and extension of 6 s at 72°C.
Determination of TF and MCP-1 mRNA in the Fresh Isolated Islets
The total RNA extracted from 300 IEQs of islets with a 3-h culture was prepared using the RNeasy Micro Kit (Qiagen) according to the manufacturer's protocol (BD, n = 3; control, n = 3). The RNA concentration was estimated from the absorbance at 260 nm. The first-strand cDNA was synthesized from 100 ng of total RNA using the Transcriptor First Strand cDNA Synthesis Kit (Roche Diagnostics). The cDNAs were amplified by PCR using the rat TF primer and a probe, rat MCP-1 primer probe set, and rat GAPDH primer probe set with a Lightcycler. The primer and probe sequences and the PCR conditions were the same as above.
Time Course Change of TF and MCP-1 mRNA Expression in the Isolated Islets
The isolated islets from one donor were divided equally into seven groups (BD, n = 3; control, n = 4). Each group was cultured for 3, 6, 12, 18, 24, 48, and 72 h, respectively. Just before examination, the cultured islets were handpicked for preparation. The TF and MCP-1 mRNA in each group were analyzed using the same procedure as above.
Statistical Analyses
All the data are expressed as the mean ± SD. The comparisons between two groups were performed by using the Student t-test. One-factor ANOVA with Bonferroni-Dunn post hoc test was used to determine the time course effect of the TF and MCP-1 expression in the isolated islets. Statistical significance was established at p < 0.05.
Results
Islet Recovery and Purity After Isolation
The islet yield was considerably lower (BD, 2110 ± 231 IEQs; control, 2390 ± 528 IEQs; p = 0.19), and the purity was significantly lower in the BD group in comparison to the control group (BD 87.7 ± 7.5%; control 97.0 ± 2.6%; p = 0.002) (Fig. 1).

Islet recovery and purity after isolation. The appearance of the isolated islets without brain death (A) and with brain death (B). Scale bar: 100 μm. (C) The isolated islet yield from brain-dead donors (black bar) and control donors (white bar).
Islet Viability and Function After Isolation
Unexpectedly, the ATP/DNA ratio and the respiratory activity were comparable between the groups, irrespective of the time point (ATP/DNA 3-h BD 54.7 ± 10.7; control 54.0 ± 13.3, p = 0.94; ATP/DNA overnight BD 51.6 ± 12.8; control 59.1 ± 3.47, p = 0.20; respiratory activity 3-h BD 2.50 ± 0.41; control 2.42 ± 0.21, p = 0.74; respiratory activity overnight BD 2.39 ± 0.55; control 2.58 ± 0.19, p = 0.45) (Fig. 2).

Islet viability and function after isolation. The ATP/DNA ratio in the fresh isolated islets (A) and in the overnight-cultured islets (C). The stimulation index of the respiratory activity in the fresh isolated islets using scanning electrochemical microscopy (B) and in the overnight-cultured islets (D). The black bar represents the brain-dead donors and the white bar represents the control donors.
mRNA Expression of TF and MCP-1 in the Pancreatic Tissues
The TF and MCP-1 mRNA levels in the pancreatic tissues prior to the isolation procedures were similar between the BD and control groups (TF/GAPDH BD 0.059 ± 0.015; control 0.066 ± 0.038, p = 0.67, MCP-1/GAPDH BD 0.018 ± 0.0098; control 0.012 ± 0.0040, p = 0.40) (Fig. 3).

The mRNA expression of TF and MCP-1 in the pancreatic tissues. The mRNA expression of TF (A) and MCP-1 (B) in the pancreatic tissues from the donors with/without brain death was analyzed using a real-time PCR assay. The black bar represents the brain-dead donors and the white bar represents the control donors.
mRNA Expression of TF and MCP-1 in the Fresh Isolated Islets
The TF mRNA levels in the fresh isolated islets of the BD group was significantly higher in comparison to the control group (TF/GAPDH BD 0.148 ± 0.010; control 0.061 ± 0.0096, p = 0.0004). The MCP-1 mRNA levels in the fresh isolated islets of the BD group was also significantly higher in comparison to the control group (MCP-1/GAPDH BD 0.240 ± 0.035; control 0.140 ± 0.0070, p = 0.008) (Fig. 4).

The mRNA expression of TF and MCP-1 in the fresh isolated islets. The mRNA expression of TF (A) and MCP-1 (B) in the fresh isolated islets from the donors with/without brain death was analyzed using a real-time PCR assay (∗∗p < 0.01 vs. control). The black bar represents the brain-dead donors and the white bar represents the control donors.
Time Course Change of TF and MCP-1 mRNA Expression in the Isolated Islets
In the fresh islets, a sharp difference was observed between the BD and the control groups with respect to the TF and MCP-1 mRNA expression. In both groups, the TF and MCP-1 mRNA levels decreased time dependently during the culture period. Between the fresh and the 48-h culture, a significant difference was seen in the TF expression in the islets from the BD donors (p < 0.05) (Fig. 5).
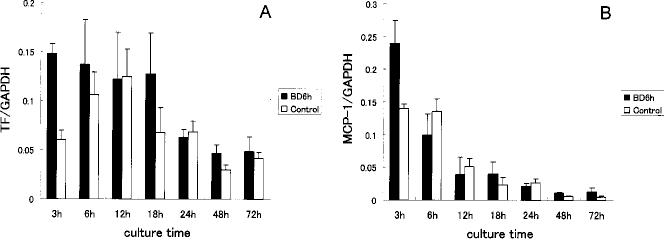
The change in time course of the TF and MCP-1 mRNA expression in the isolated islets. The change in time course of the TF (A) and MCP-1 (B) mRNA expression in the isolated islets from the donors with/without brain death. The black bar represents the brain-dead donors and the white bar represents the control donors. A significant difference was observed in the TF expression in the islets from the brain-dead donors between the 3- and 48-h cultures (p < 0.05).
Discussion
This study demonstrated that BD influences the TF and MCP-1 expressions in the isolated islets but not in the pancreatic tissues prior to the digestion procedure. It is possible that the difference may be attributed to the warm ischemic stress during the digestion procedures. However, TF and MCP-1 were not upregulated in the isolated islets from the donors without BD, thus suggesting that the warm ischemic damage per se during the digestion procedure was not sufficient to induce crucial inflammatory mediators in the islet grafts. We therefore believe that upregulation of the crucial inflammatory mediators induced by BD could be further exacerbated by warm ischemic damage during the digestion procedure.
In the present study, the islet yield and purity were certainly affected by BD. However, the difference was extremely low in comparison to the previous report (6). Moreover, almost no influences were observed in terms of the islet viability. One of the possible explanations for this discrepancy is the difference of the isolation procedure. In our methods, the pancreatic tissues were kept on ice during the whole procedure, with the exception of the digestion phase. Furthermore, at the density-gradient centrifugation phase, we applied Histopaque-1119 and Lymphoprep™. However, a dextran gradient separation was performed in the previous report (6). Therefore, the important message from our present study is that the expression of the inflammatory mediators, rather than the islet viability, is more susceptible to BD. In other words, it seems more likely that the current standard methods for the islet quality assessment are not suitable tools for detecting graft damage in the early phase of islet transplantation.
Although the islet culture modulated the inflammatory status of the human pancreatic islets (19, 20), the effect remains controversial. We therefore investigated the change in time course of the TF and MCP-1 expressions in the isolated islets with/without BD. As shown in Figure 5, the influence of BD and ischemic stress during the isolation procedure was most pronounced after a 3-h culture. Clinical islet transplantation is currently being performed in most institutions, using fresh islets according to the Edmonton protocol (26, 27). Notably, in most clinical cases, the islet grafts with a 3-h culture are used in fresh islet transplantation because several quality tests and preparation for graft injections are needed. Therefore, the present study clearly showed that the worst time point was selected for islet infusion in current clinical islet transplantation, at least with respect to the damage due to BD and ischemic stress during the isolation procedure.
One way to avoid the adverse effect of BD and ischemic stress is by performing short-term culture of the islet grafts. This may be one of the possible explanations for the outstanding result of the clinical islet transplantation reported by Froud et al. (7) and by Hering et al. (14). In support of our previous findings (30), the present study also implied that the TF expression in the islets without BD was substantially upregulated during overnight culturing. The reason for this is uncertain but the hypoxic condition during culturing may at least be partially responsible for the TF induction (1, 13, 25). Taking into account this finding, it is most likely that the 48-h culture introduced in the previous reports (7, 14) was reasonable. However, it was also reported that the number and function of the isolated islets may decreased after a short-term culture (9, 15, 16, 18, 22). This is consistent with our previous findings that isolated islets without culture are more beneficial to the transplant outcome under a current style of culture (29). Moreover, it may be difficult to maintain an adequate number of islets from the marginal donors after substantial periods of culturing, especially in Japan, where only non-heart-beating donors are available for islet transplantation. Therefore, further improvements are required to maintain the number and function of the cultured islets. Another way to avoid the adverse effect of BD and ischemic stress is to establish effective anti-inflammatory treatments through whole steps from the intensive care unit to the digestion procedure.
In conclusion, BD in combination with warm ischemic stress during the isolation procedures induces high expression of TF and MCP-1 in the isolated islets. In order to reduce the expression of the crucial inflammatory mediators in the islet grafts, the management of the pancreas from brain-dead donors with early anti-inflammatory treatments is warranted.
Footnotes
Acknowledgments
We thank Takehiro Imura and Megumi Goto for their excellent technical assistance. This study has been supported by grants from Innovation Plaza Miyagi of JST (Japan Science and Technology Agency), the Japanese Grant-in-Aid for Scientific Research (B), the Ministry of Health, Labour, and Welfare, Japan, the Nakajima Foundation, and Takeda Foundation.
