Abstract
Naturally occurring CD4+CD25+ Treg cells (nTregs) can be exploited to establish an immunologic tolerance to non-self-antigens. The in vivo administration of a single superagonistic CD28-specific monoclonal antibody (supCD28mAb) to naive rat preferentially expanded the nTregs, which induced a potent inhibition of lethality of the graft-versus-host (GvH) diseases. The appearance of increased Foxp3 molecules was accompanied with a polarization towards a Th2 cytokine profile with a decreased production of IFN-γ and increased production of IL-4 and IL-10 in the serum of the antibody-treated rat. The peripheral Foxp3 nTregs are decreased in acute GvHD, while supCD28mAb administration showed that nTregs were preferentially proliferating in vivo, thus resulting in the significant prevention of the GvH disease. Furthermore, antigen-specific nTregs could suppress conventional T-cell proliferation stimulated with alloantigen in vitro. Taken together, our findings demonstrate that the potent regulatory functions of the Tregs for the treatment of GvHD are antigen specific. These data also provide evidence that GvHD is associated with decrease of Tregs in the periphery of the host. The determination of the Foxp3 Tregs can be a helpful tool to discriminate GvHD severity and lethality after allogeneic stem cell transplantation.
Introduction
Naturally occurring CD4+CD25+ Treg cells (nTregs) function as key regulatory effectors in mice and have provided important information regarding a specific cellular population that performs immune regulation through the suppression of self-responses (1). A thymectomy on neonatal day 3 that resulted in multiorgan autoimmune disease (gastritis, thyroiditis, or insulitis) was associated with the loss of this nTreg population (9, 14, 20). The outcome of the alloimmune response following transplantation is, in part, dependent on the balance between the stimulatory and inhibitory signals that accompany TCR engagement by an alloantigen (13), and there is little doubt that nTregs is one of the inhibitor cells that suppresses excessive immune responses and maintains immune homeostasis (16).
Accumulating evidence indicates that nTregs play a crucial role in the maintenance of immunologic self-tolerance and the negative control of immune responses (10, 16, 17). For example, the elimination of nTregs leads to the spontaneous development of various autoimmune diseases in normal animals and provokes effective tumor immunity in otherwise nonresponding animals (8, 15, 18, 20, 21). The elimination also enhances immune responses to non-self-antigens, including the allogeneic transplantation antigens (10). A crucial area for future study is the identification of the cytokine drugs or costimulatory molecules that reverse energy and enhance growth, while preserving the suppressor function of the nTregs population. The optimal stimulus for the expansion of the nTregs in vitro is the combination of TCR triggering and high concentrations of IL-2. Once the specific antigens that are recognized by nTregs in organ-specific autoimmunity have been defined, the antigen can then be administered in combination with IL-2, in order to expand the nTreg cell population that is specific for the target organ. The administration of the target antigen on iDCs together with IL-2 may be a particularly effective method for the expansion of nTregs suppressors in vivo.
Our previous studies (2, 7, 8) showed that the in vivo application of the unique supCD28mAb to Lewis rat caused a preferential expansion of Foxp3-expressing nTregs over conventional T cells, and maintained not only their phenotype but also their potent regulatory functions against T-cell proliferation via the mitogens, antibodies, and allostimulation in vitro. In addition, the in vivo application also suppressed the alloreactive T-cell response in a model of GvHD and indicated that this therapeutic effect was mediated by in vivo expansion of nTregs (7, 12). Furthermore, using a full MHC-mismatch transplantation model, we found that supCD28mAb preferentially expanded nTregs in vivo and recruited to the grafts, thus resulting in significantly prolonged cardiac and kidney graft survival (7, 8).
In the present study, the in vivo administration of a single supCD28mAb to naive rat preferentially expanded nTregs, which in turn induced a potent inhibition of the lethality of GvHD. The peripheral Foxp3 nTregs are decreased in acute GvHD, while supCD28mAb administration showed that nTregs were preferentially proliferated in vivo, and resulted in a significant prevention of GvHD. Taken together, our findings demonstrate that the potent regulatory functions of Tregs for the treatment GvHD are antigen specific. These data also provide evidence that GvHD is associated with a decrease of Tregs in the periphery of the host. The determination of the Foxp3 Tregs can be a useful tool for discriminating the severity and lethality of GvHD after an allogeneic stem cell transplantation.
Materials and Methods
Animals
Adult male Lewis (RT1l), DA (RT1a), and (Lewis x DA) F1 rats were purchased from Shizuoka Laboratory Animal Center (Shizuoka, Japan) and used at 4–6 weeks of age. The DA tolerant of (Lewis x DA) F1 rats were made by injection of 1 × 108/150 μl (Lewis x DA) F1 bone morrow cells to the neonatal DA rats (11). The animals were maintained under standard conditions and fed rodent food and water according to the laboratory animal care principles and the guide for the care and use of laboratory animals established by our institution.
Total Spleen Cell Purification and Assay of Systemic GvH Reactivity
For the preparation of purified spleen cells, naive Lewis rat spleens were harvested and gently ground with frosted objective slides in PBS. They were filtered through 200-μm nylon mesh for single-cell suspension preparation and then separated by Lympholyte-Rat (Cedarlane Laboratories, Ontario, Canada). Total spleen single-cell suspensions (2.5 × 108) were injected IV into 4-week-old (Lewis x DA) F1 or DA tolerant of (Lewis x DA) F1 rats. Antibodies (supCD28mAb, JJ316) were administered to the host rat via tail veins at the various points (day -3, 0, 3, 7, and 10 after T-cell infusion). The rats were weighed on alternate days during the active phase of the GvHD. Animals undergoing typical GvHD showed rapid weight loss and commonly described signs of the disease, including ruffled fur, reddening of the skin, a hunched posture, and ultimately death.
CD4+CD25+ and CD4+CD25- T-Cell Purification
For the isolation of purified CD4+CD25+ and CD4+CD25- T cells, splenocytes were first stained with FITC-conjugated anti-rat CD8, CD11b/c, and NK1.1 (BD Pharmingen, San Diego, CA) followed by anti-FITC MicroBeads (Miltenyi Biotec, K.K. Tokyo, Japan) and anti-rat Kappa MicroBeads (Miltenyi Biotec) and then sorted on an AutoMACS system (Miltenyi Biotec). To isolate CD4+CD25+ T cells, the negative fraction of the enriched CD4+CD8- cells was stained with biotinylated anti-rat CD25 (BD Pharmingen) followed by antibiotin MicroBeads (Miltenyi Biotec) and sorted on an Auto MACS. The negative fraction, enriched CD4+CD25- cells, was labeled with anti-rat CD4 MicroBeads (BD Pharmingen) sorted on an Auto MACS. The enriched T-cell populations were evaluated by FACScan (Becton Dickinson) and more than 92% purity of the CD4+CD25+ and 95% purity of the CD4+CD25- cells population were routinely obtained.
T-Cell Proliferation Assays
CD4+CD25+ T cells from naive Lewis or DA rat spleens were prepared and mixed (1:1) with Lewis CD4+CD25- T cell (1 × 105) and cultured with DA spleen CD11b/c+ cells (APCs, 1 × 104) in a U-bottom 96-well plate (Iwaki, Tokyo, Japan) at a final volume of 200 μl/well in a humidified atmosphere at 37°C for 5 days. The proliferation of T cells was measured with cell proliferation ELISA kits (Roche Diagnostics Gmbh, Penzberg, Germany) (4). Briefly, the cells were labeled with 5-bromo-2-deoxyuridine (BrdU) solution at 10 μl/well and incubated for an additional 2 h at 37°C. After centrifugation, the supernatant was removed, 200 μl/well of fixation and DNA denaturation (FixDenat) solution was added to the cells, and they were reincubated for 30 min at 15–25°C. The cells were cultured for 90 min with anti-BrdU-peroxidase solution and subsequently washed three times. After adding substrate solution at 100 μl/well, the BrdU incorporation was measured with a chemiluminescence reader (Wallac ARVO™ SX; Perkin Elmer, Inc., Wellesley, MA) and the data was processed using Wallac1420 manager software (Perkin Elmer).
Flow Cytometric Analysis
Host T cells from supCD28mAb-treated or IgG-treated control (Lewis x DA) F1 rat spleens and peripheral blood were collected and suspended in PBS and incubated at 4°C for 30 min with an optimal concentration of Cy-Chrome-conjugated anti-rat CD4 antibody (OX-35, BD Pharmingen) in combination with FITC-conjugated anti-rat CD25 antibody (OX-39, BD Pharmingen) diluted with PBS containing 1% fetal calf serum. The membranes were stained and intracellular staining was then performed using a BD Cytofix/Cytoperm™ Kit (BD Pharmingen). The cells were fixed with 100 μl of BD Cytofix/Cytoperm™ (BD Pharmingen) at 4°C for 20 min. The cells were next washed twice with BD Perm/Wash™ buffer (BD Pharmingen) and then resuspended in 50 ml of the same buffer containing the optimal concentration of PE-conjugated anti-Foxp3 antibody (FJK-16s, eBioscience, San Diego, CA) for permeabilization. The cells were gently mixed and incubated for 30 min in the dark at 4°C. They were then washed twice with BD Perm/Wash™ buffer and resuspended in a staining buffer. Thereafter, the cells were analyzed by flow cytometry (FACScan, Becton Dickinson). The characterization of the DA tolerant of (Lewis x DA) F1 rat was performed by FCM analysis using a specific antibody (clone I1–69) recognized the Lewis MHC class I molecular.
Cytokine Assay
Serum were collected and quantified by BD Cytometric Bead Array rat IL-4, IL-10 Flex Kit (BD Biosciences) or IL-2, IL-4, IL-10, and IFN-γ rat ELISA kit (BioSource, Camarillo, CA) according to the manufacturer's protocol.
Statistical Analysis
Student's t-test was used to compare the paired and unpaired variables. A statistical evaluation for graft survival was performed using the Kaplan-Meier's test. Values of p < 0.05 were considered to be statistically significant. All in vitro experiment data were representative of three independent experiments and expressed as the mean ± SD.
Results
Cytokine Concentrations in the Serum From supCD28mAb-Treated Rats
We showed previously that a single injection of supCD28mAb led to a significant increase in the CD4+CD25+ population in the spleen, the peripheral blood, and the lymph nodes, and the expanded CD4+CD25+ T cells expressed much higher levels of IL-2, IL-4, and IL-10 mRNA in comparison to CD4+CD25-T cells and to nTreg cells (7, 8). In the present study, we investigated the concentration of the cytokines in the serum from the Lewis rats that received a supCD28mAb treatment at the various time points (Fig. 1). Consistent with the mRNA expression data, we found that the IL-2 and IFN-γ production exhibited a high level on day 7 and day 3, respectively. In addition, IL-10 and IL-4 showed significantly high concentrations on both day 3 and day 7, whereas the other kinds of cytokines such as IL-6 and TNF-α showed no change (data not shown).

The cytokine profiles analysis after supCD28mAb treatment. Serum from the Lewis rats received supCD28mAb treatment at the various point. IL-2 and IFN-γ production exhibited a high level on day 7 and day 3, respectively. IL-10 and IL-4 showed significantly high concentrations on both day 3 and day 7. Data are representative of three independent rats and indicate the mean ratio of triplicate results in each experiment.
supCD28mAb Treatment Suppressed GvH Reactions
A clinically relevant in vivo model was used to investigate the role of the supCD28mAb treatment on the allo responses in vivo (Fig. 2A). The systemic GvH reactivity of the supCD28mAb treatment was tested by injecting an antibody via the tail veins, at the various time points (day -3, 0, 3, 7, and 10 after the spleen cell infusion), of the (Lewis x DA) F1 hybrid recipients. As shown in Table 1 and Figure 2B and C, the rats that received 2.5 × 108 naive Lewis spleen lymphocytes developed host's weight loss and definite lethality (survived to 15±1.3 days). The supCD28mAb treatment, on day 3 before the spleen cell transfer, did not mediate GvHD lethality in any of the recipients in comparison to the control (60 ± 0.0 days). Furthermore, the supCD2 8mAb treatment on day 0 significantly inhibited GvHD mortality (39.9 ± 18.0 days) in comparison to the recipients that received only the naive spleen cells (p = 0.0001) (Fig. 2B, C). Consistent with these observations, the extensive perivascular infiltration of the mononuclear cells and the tissue damage were observed in the histological studies of the skin, tongue, lung, and liver from the naive lymphocytes transferred to the host on day 14 (7). In contrast, relatively few infiltrating mononuclear cells and almost normal tissue architecture were observed in the supCD28mAb group (data not shown). On the other hand, there was no protective effect in the supCD28mAb treatment group on day 7 and day 10.

supCD28mAb treatment suppressed GvH reactions. (A) Experiment design. (B, C) Systemic GvH assay: transfer of the naive Lewis lymphocytes to F1 rat leads to host's weight loss and definite lethality in a nonirradiated rat GvHD model (filled circles). Nonetheless, the group from supCD28mAb-treated host rats on day -3 (filled triangles) completely suppressed the GvH reaction and the rat had slight weight loss and no animal died within the observation times of (over 60 days). In addition, the protective effect of the supCD28mAb treatment on day 0 (filled boxes) and day 3 (X) was still apparent. On the other hand, there were no protective effects in the supCD28mAb treatment group on day 7 (open boxes) and day 10 (open circles).
In Vivo supCD28mAb Treatment Suppressed Systemic GvHD

Systemic GvHD was performed transferring 2.5 × 108 naive Lewis spleen cells to (Lewis x DA)F1 rats without x-ray. supCD28mAb (JJ316, 1 mg/rat) was administered to the host rat via tail veins at the various points after T-cell infusion. The p-value was compared with no treatment control group rat and determined by the Kaplan-Meier test.
Kinetic Changes of the CD4+CD25+ T Cells and Foxp3+ Tregs Population in GvHD Host Spleen and Peripheral Blood (PB)
A kinetic analysis of the Foxp3+ cells in the peripheral blood of the GvHD host was conducted (Fig. 3A). The FCM analysis showed a progressive increase of the CD4+CD25+ T-cell population on day 7 in PB and on day 3 in the spleen, after the transfer of the naive Lewis lymphocytes, accompanied by a decrease of the Foxp3+ cells in CD4+CD25+ T-cell populations on day 7 in PB and on day 3 in spleen (Fig. 3B). The population of the CD4+CD25+ T cells in the total lymphocytes and Foxp3+ cells in CD4+CD25+ T cells was maintained at a fixed level in the spleen and the PB of the nontreated control group.

Kinetic CD4+CD25+ T-cell and Foxp3+ Treg population changes in host spleen and PB. (A) A representative FCM data showed the CD4, CD25, and Foxp3 three-color staining. (B) FCM analysis showed a progressive increase of the CD4+CD25+ T-cell population (open triangles) on day 7 in PB and on day 3 in spleen, after the transfer of naive Lewis lymphocytes, accompanied by a decrease of the Foxp3+ cells in CD4+CD25+ T-cell populations (open circles) on day 7 in PB and on day 3 in spleen. The population of the CD4+CD25+ T cells in total lymphocyte and Foxp3+ cells in CD4+CD25+ T cells was maintained at a fixed level in spleen and PD of the no-treatment control group rats.
CD4+CD25+ T-Cell and Foxp3+ Treg Populations in Host Spleen and PB on Day 14 After supCD28mAb Treatment
The changes of the CD4+CD25+ T-cell and Foxp3+ Treg populations in host spleen and PB after supCD28 mAb treatment were investigated. An FCM analysis in Figure 4 showed the CD4+CD25+ T-cell population on day 14 in spleen and PB, after the transfer of naive Lewis lymphocytes, increased, accompanied by a decrease of the Foxp3+ cells in the CD4+CD25+ T-cell populations. In contrast, the population of the CD4+CD25+ T cells in total lymphocyte was maintained at a fixed level and Foxp3+ cells dramatically increased in CD4+ CD25+ T cells in supCD28mAb treatment on day 3. Similar to the untreated control group, the CD4+CD25+ T cells increased and the Foxp3+ cells decreased in the antibody-treated noneffective group (day 10).

CD4+CD25+ T-cell and Foxp3+ Treg populations in host spleen and PB on day 14 after supCD28mAb treatment. An FCM analysis showed an increase of the CD4+CD25+ T-cell population on day 14 in spleen and PB after the transfer of naive Lewis lymphocytes, accompanied by a decrease of the Foxp3+ cells in the CD4+CD25+ T-cell populations. The population of the CD4+CD25+ T cells in total lymphocyte was maintained at a fixed level and Foxp3+ cells dramatically increased in CD4+CD25+ T cells in the supCD28mAb treatment group on day 3. Similar to the no-treatment group, the CD4+CD25+ T cells increased and the Foxp3+ cells decreased in the antibody-treated non effective group (day 10).
Prevention of GvHD by supCD28mAb-Expanded nTregs Was Antigen Specific
Lastly, to clarify whether or not the activity of the supCD28mAb-expanded nTregs was antigen specific, we used a neonatal tolerant model to make a unique DA tolerant of (Lewis x DA) F1 rat (Fig. 5A). Briefly, 1 × 108/150 μl (Lewis x DA) F1 bone morrow cells were injected into the neonatal DA rats (6, 11) and, 4 weeks after, the characterization was performed by an FCM analysis using a specific antibody (clone I1–69) that recognized the Lewis MHC class I molecules (Fig. 5B). As shown in Table 2 and Figure 5C, the transfer of the naive Lewis lymphocytes to DA tolerant of (Lewis x DA) F1 rat leads to host's weight loss and a definite lethality. However, there was no protective effect on the group receiving the supCD28mAb treatment on day 3 compared to the same antibody-treated (Lewis x DA) F1 rat group, which did not mediate GvHD lethality in any recipients (Fig. 2).
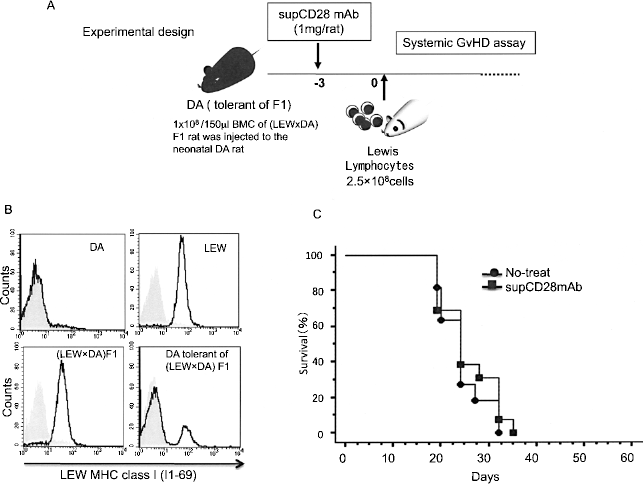
Prevention of GvHD by supCD28mAb-expanded nTregs was antigen specific. (A) Experimental design. (B) Representative FCM data showed the characterization of the DA tolerant of (Lewis x DA) F1 rat by a specific antibody (clone I1–69) recognized the Lewis MHC class I molecules. (C) The transfer of the naive Lewis lymphocytes to DA tolerant of (Lewis x DA) F1 rat leads to host's weight loss and definite lethality (filled circles). There was no protective effect in the supCD28mAb treatment group on day 3 (filled squares).
In Vivo supCD28mAb-Expanded T-Cell-Suppressed Systemic GvHD Is Antigen Specific

Systemic GvHD was performed transferring 2.5 × 108 naive Lewis T cells to DA tolerant of (Lewis x DA) F1 rats without x-ray. supCD28mAb (JJ316, 1 mg/rat) was administered to the host rat via tail veins at day 3 before T-cell infusion. The p-value was compared with control group rat and determined by the Kaplan-Meier test.
nTregs Can Suppress the T-Cell Proliferation Antigen Specifically
In order to examine the antigen-specific nTregs activity, suppression assays were performed with autologous naive Lewis rat and allogeneic DA rat nTregs to test their suppressive activity using a mixed lymphocyte reaction assay. In agreement with this finding in the in vivo study (Figs. 2 and 5), Lewis nTregs inhibited the proliferation of naive Lewis CD4+CD25- T cells stimulated with allogeneic DA rat APCs, at a ratio of 1:1, while the nTregs from DA rat had no effect on the suppression of Lewis CD4+CD25- T-cell proliferation (Fig. 6).
Discussion
The recent attempts to translate the supCD28mAb-mediated nTreg stimulation failed because of an unexpected induction of a life-threatening cytokine release syndrome (19). The transient polyclonal Tregs activation, regardless of the stimulus used for its induction, still represents an attractive therapeutic option in autoimmunity and in transplantation tolerance. In the present study we investigated the concentration of the cytokines in the serum from the Lewis rats that were treated with supCD28mAb at various time points. Consistent with our previous data on the mRNA expression (2, 7), we found that the IL-2 and IFN-γ production exhibited a high level on day 7 and day 3, respectively. In addition, IL-10 and IL-4 showed significantly high concentrations on both day 3 and day 7, whereas the other kinds of cytokines such as IL-6 and TNF-α showed no changes in rats (data not shown). These results suggest that the antibody action in humans and rats is different (19).
In the present study, the systemic GvH reactivity of the supCD28mAb treatment was tested by injecting an antibody via the tail veins of (Lewis x DA) F1 hybrid recipients at various points. In agreement with our previous reports (2, 7, 8), the rats that received the naive Lewis spleen lymphocytes experienced host weight loss and definite lethality (Table 1, Fig. 2). The supCD28mAb treatment on day 3, before the T-cell transfer, did not mediate GvHD lethality in any recipients in comparison to the control. Furthermore, the supCD28mAb treatment on day 0 significantly inhibited the GvHD mortality in comparison to the recipients that received only the naive spleen cells. These findings were accompanied by an extensive perivascular infiltration of the mononuclear cells, and tissue damage was seen in the histological studies of the skin, tongue, lung, and liver from the naive lymphocytes-transferred host on day 14 (7). In comparison, relatively few infiltrating mononuclear cells and almost normal tissue architecture were observed in the supCD28mAb group (data not shown). On the other hand, there were no protective effects in the supCD28 mAb-treated group on day 7 and day 10. These results indicate that supCD28mAb-treated early have potent immunoregulatory activity on the response to allogeneic MHC antigens, even in a more sensitive assay.
Tregs specifically express the transcription factor Foxp3, which appears to act as a master control gene for their development and function (3, 5, 6). The forced expression of the Foxp3 gene can convert the murine naive T cells to nonnatural Tregs, which phenotypically and functionally resemble the natural Tregs (5). Natural Tregs also show homeostatic proliferation in a T-cell-deficient environment, and a fraction of the cells can proliferate in normal animals, presumably by recognizing the self-antigens (5). These findings collectively indicate that Foxp3 is a master control gene for the development and function of natural Tregs.
Firstly, we showed that a progressive increase of the CD4+CD25+ T-cell population on day 7 in the PB and on day 3 in the spleen, after the transfer of naive Lewis lymphocytes, accompanied by a decrease of the Foxp3+ cells in CD4+CD25+ T-cell populations on day 7 in PB and on day 3 in spleen (Fig. 3B). Secondly, the changes of the CD4+CD25+ T cells and Foxp3+ Tregs populations in the host spleen and the PB after the supCD28mAb treatment were investigated. The population of the CD4+ CD25+ T cells in the total lymphocyte was maintained at a fixed level, whereas the Foxp3+ cells dramatically increased in the CD4+CD25+ T cells in the supCD28 mAb treatment group on day 3. Consistent with the data in Figure 2, the CD4+CD25+ T cells increased and the Foxp3+ cells decreased in the antibody-treated noneffective group (day 10). These data indicate that an analysis of the Foxp3 Tregs may be a helpful tool to discriminate the severity and lethality of GvHD after allogeneic stem cell transplantation.
Thirdly, we used a unique neonatal tolerant model, DA tolerant of (Lewis x DA) F1 rat, to determine whether or not the activity of the supCD28mAb-expanded nTregs was antigen specific (Fig. 5A). We verified that the cells derived from the Lewis rat was present in the DA tolerant of (Lewis x DA) F1 rat, by using a specific antibody (clone I1–69) that recognized the Lewis MHC class I molecules (Fig. 5B). After the antibody treatment, the majority of the expanded nTregs were DA derived. Similar to the (Lewis x DA) F1 host, the transfer of the naive Lewis lymphocytes to the DA tolerant of (Lewis x DA) F1 rat experienced host weight loss and definite lethality. However, there was no protective effect in the supCD28mAb-treated group on day 3 in comparison to the same antibody treatment in the (Lewis x DA) F1 rat group, which did not mediate GvHD lethality in any recipients. Furthermore, we examined the antigen-specific nTregs activity by employing autologous naive Lewis rat and allogeneic DA rat nTregs to test their suppressive activity by performing a mixed lymphocyte reaction assay. In agreement with the finding in the in vivo study (Figs. 2 and 5), Lewis nTregs inhibited the proliferation of naive Lewis CD4+CD25- T cells stimulated with allogeneic DA rat APCs, whereas the nTregs from the DA rats had no effect on suppression of the Lewis CD4+CD25- T-cell proliferation. These results indicate that nTregs can suppress the T-cell proliferation antigen specifically and the supCD28mAb-expanded nTregs have potent antigen-specific immunoregulatory activity in response to the allogeneic MHC antigens.
In conclusion, the current results confirm that supCD28mAb can preferentially expand nTregs, which induced a potent inhibition of the lethality of GvHD via an antigen-specific manner. These data also provide evidence that GvHD is associated with a decrease of Foxp3 nTregs in the periphery of the host. The determination of the Foxp3 Tregs can be a helpful tool to discriminate the severity and lethality of GvHD after allogeneic stem cell transplantation.

Lewis lymphocyte proliferation was suppressed by Lewis nTregs, not DAs, in the MLR assay. Lewis CD4+CD25+ nTregs can potentially inhibit the Lewis CD4+CD25- T-cell proliferation stimulated by DA rat splenocytes (rlu/s: relative light units/second) whereas the nTregs from DA rat have no effect on the suppression of Lewis CD4+CD25- T-cell proliferation. Data are representative of three independent rats and indicate the mean ratio of triplicate results in each experiment.
Footnotes
Acknowledgments
The authors acknowledge Drs. Megumu Miyamoto and Hiromitsu Kimura for their scientific comments and discussion. This study was supported by research grants from the Ministry of Education, Culture, Sports, Science and Technology of Japan (a Grant-in-Aid 17390355, 20390349), and Ministry of Health, Labour and Welfare of Japan (HS KHC1025).
