Abstract
Islet transplantation is an attractive therapy for type 1 diabetes, although some issues remain. One of them is the severe donor shortage in some countries. In this study, we investigated the possibility of international islet shipping beyond 10,000 km to supply islets to countries with donor shortages. Human islets were isolated from six cadaver donors and cultured until shipment. Islets were packed in either gas-permeable bags or in non-gas-permeable bags and shipped from Baylor Research Institute (Dallas, TX, USA) to Fukuoka University (Fukuoka, Japan). Pre- and postshipment islet number, purity, viability, and stimulation index (by glucose stimulation test) were assessed. Shipped 1,500 IE islets were transplanted into streptozotocin-induced diabetic nude mice for in vivo assay. The distance of our shipment was 11,148.4 km, and the mean duration of the shipments was 48.2 ± 8.2 h. The islet number recovery rate (postshipment/preshipment) was significantly higher in gas-permeable bags (56.4 ± 10.1% vs. 20.5 ± 20.6%, p < 0.01). Islet purity was significantly reduced during shipment in non-gas-permeable bags (from 47.7 ± 18.6% to 40.2 ± 28.2 in gas-permeable bags vs. from 50.4 ± 6.4% to 25.9 ± 15.6% in non-gas-permeable bags, p < 0.05). Islet viability and stimulation index did not change significantly between pre- and postshipping, in either gas-permeable bags or in non-gas-permeable bags. One of three diabetic nude mice (33.3%) converted to normoglycemia. It is feasible to ship human islet cells internationally in gas-permeable bags. This strategy would promote basic and preclinical research for countries with donor shortages, even though the research centers are remote (over 10,000 km from the islet isolation center).
Introduction
Islet cell transplantation (ICTx) is clearly a promising therapy for type 1 diabetes mellitus (DM); however, several issues have limited its widespread use (6, 14, 22). Among these issues is a severe shortage of cadaveric donors in some countries, such as Japan (15). Even in the US, difficulties arise when the islet isolation institute is far from the ICTx candidate (17). Moreover, ICTx for type 1 DM is recommended by the American Diabetes Association only within the setting of controlled research studies by a small number of institutes (20).
Recently, we reported that islets could be transported safely within the US: from Miami to Texas (5). Langer et al. showed the possibility of safe islet isolation, delivery of islets, and ICTx up to 1,000 km (8). However, the safety of islet shipping is still unclear and challenging when the distance and duration are over 10,000 km and over 48 h, respectively.
From the point of view that shipped islets might be useful for research purposes and ultimately ICTx, maintaining their function is important. As many have reported, the current system, in which surgeons will send a procured pancreas to a remote islet isolation center and the center sends back the isolated islets to the transplant surgeons, is safe and efficient within 2,500 km (5, 8, 17). Small charter jets may be used in these cases. Thus, the changes in temperature and pressure are likely to be milder than for international flights by large cargo jets.
The viability and function of islets that are shipped over 10,000 km and for longer than 48 h are still unclear. It is still unknown whether or not the shipping of islets over such long distances can be used for research and preclinical purposes.
Here we show islet quality following shipment from Texas in the US to Fukuoka, Japan, a distance of over 10,000 km and duration of more than 48 h.
Materials and Methods
Islet Isolation
Pancreata were procured from cadaveric donors in Texas (USA), through LifeGift (Fort Worth, TX) and Southwest Transplant Alliance (Dallas, TX), from January to July 2008. These pancreata were transferred by us to Baylor Institute for Immunology Research (BIIR) immediately after procurement. After ductal injection (11, 13, 16), pancreata were preserved with the two-layer method (oxygenated perfluorocarbon and University of Wisconsin solution) (9, 10). They were digested by the Ricordi method and purified using a COBE 2991 cell processor, as previously described (6, 11–13). Successful islet isolation was defined as islet yields of more than 300,000 islet equivalents (IE), viability more than 70%, and purity more than 30%, based on the original Edmonton protocol (23).
Culture Media
After the final purification, islets were cultured at 22°C in 5% CO2 in islet culture media until shipping. This media consists of CMRL 1066 culture media (500 ml; Mediatech, Manassas, VA) with human serum albumin, 1.5 ml 1 M sodium hydroxide (Sigma, St. Louis, MO), and 1.1 g sodium bicarbonate (Sigma). The media was filter sterilized and stored in at 4°C.
Media Changes
Culture media changes depended on the purity of the islet preparations. Briefly, media were changed on days 1, 2, 3, 4, and 5 (postisolation) if the purity was <40%, changed on days 1, 3, and 5 if the purity was between 40% and 70%, changed on days 1 and 4 if between 70% and 80%, or changed on day 1 only if >80%. All media were also changed once just prior to shipping regardless of the purity.
Islet Cell Count and Viability
Islet number and purity were determined, as described previously (19). Briefly, islets were stained with dithizone dye (dithizone, Sigma, dissolved in DMSO, Burdick and Jackson, Morristown, NJ) and counted under a microscope from at least two samples of 100 μl from 200-ml tubes. Purity was estimated visually at the time of counting islets. Islet cell viability was assessed with using membrane perm-selectivity dye fluorescein diacetate/propidium iodide (FDA/PI, Sigma) under a fluorescence microscope (1). These results were obtained just before shipment from BIIR and just after arrival at Fukuoka University, using the same methods.
Static Incubation
In order to assess islet cell function in vitro, static incubation was performed, as described previously (18). The static incubation was performed pre- and postshipment, at our center and the receiving center, respectively.
To determine islet potency, after overnight culture at 37°C, human islets are subjected to low (2.8 mM) and high (20.0 mM) concentrations of glucose stimulation. The amount of human insulin in the culture supernatant is measured using the Human Insulin ELISA (Alpco, Salem, NH). The islets remaining in the pellet are quantified for DNA using the DNA Quantification Assay (fluorometric). The insulin released is calculated and corrected for DNA concentration before determining the stimulation index. Glucose-stimulated insulin release was expressed as the stimulation index (SI), calculated from the ratio of insulin released after exposure to high glucose over the insulin released in basal condition.
Transportation Bags and Container
Islets in culture media were transferred to cell container bags with 50-cc syringes. We used both gas-permeable bags (Lifecell Tissue Culture Flask; Baxter, Deerfield, IL) and non-gas-permeable bags (Fenwal Transfer Pack Container; Baxter) for shipping the islets. Both gas-permeable and non-gas-permeable transport bags were put in large Styrofoam™ boxes (Fig. 1A), and the lids remained open until just before shipping to prevent the absorption of air.
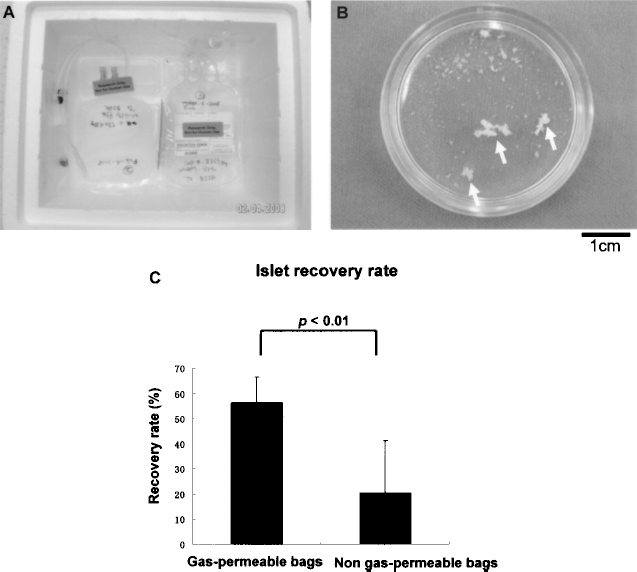
(A) Bags were placed on the bottom of Styrofoam™ boxes and held in position with tape; gas-permeable on the left and non-gas-permeable on the right. (B) Some islets formed clusters (arrows) in two shipments with non-gas-permeable bags and could not be recovered. (C) There was a significant difference in recovery rates of islets from gas-permeable bags and non-gas-permeable bags (p < 0.01).
Shipped Islet Transplantation
We assessed the islet function in vivo using a mouse transplantation model in order to evaluate the efficacy for research and preclinical use. Briefly, postshipping 1,500 IE islets were transplanted into streptozotocin (STZ; Sigma)-induced (180 mg/kg given IV) diabetic nude mice under the kidney capsule. Fasting blood glucose (FBG, mg/dl) levels were measured once every 2 days by a single drop of tail vein blood. Two continuous readings of FBG above 250 mg/dl were considered as a diabetic state.
Temperature Monitoring
As a culture condition, temperature is one of the most important factors. To assess the temperature changes, we shipped an islet container with a temperature recording device (HOBO H08-001-02, Onset Computer Corporation, Bourne, MA).
Statistics
Descriptive statistics were presented as mean ± SE. Pearson's test was used to evaluate correlation and t-test was used to compare two groups. A value of p < 0.05 was considered statistically significant and all p-values reported were two-sided. All analyses were performed in Stat Mate III for Windows.
Results
Islet Isolation
Islets were isolated from pancreata of six cadaveric donors (M/F = 4:2, mean age was 42.8 ± 4.6 years old, mean cold ischemic time was 222.5 ± 18.6 min). The isolation success rate was 100% (6/6). The mean islet IE after final purification was 639,124 ± 47,753 IE, and the mean culture duration until shipping was 93.6 ± 69.1 h (Table 1).
Shipped Islet Summary

Distance of Islet Shipping
We performed a PubMed literature search on islet shipping (Table 2). The distance of our shipment was 11,148.4 km (http://www.cloudwoods.jp/kyoritan/). The distance for our islet shipment was approximately 5 to 13 times farther than other studies.
Reported Distance of Islet Shipping
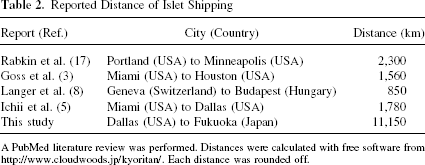
A PubMed literature review was performed. Distances were calculated with free software from http://www.cloudwoods.jp/kyoritan/. Each distance was rounded off.
Shipping Duration
The mean duration of the shipments was 48.2 ± 8.2 h (Table 1). We used FedEx for all islet shipping. No major delay or lost baggage were reported.
Changes in Islet Cell Numbers
The pre- and postshipment islet yields are shown in Table 1. In two cases of shipment with non-gas-permeable bags, the islets clustered (Fig. 1B), and no islets were recovered. Such a phenomenon was not observed with shipments in gas-permeable bags. The mean total recovery rate of the islets (postshipment IE /preshipment IE) was 36.5 ± 22.3%. The islet recovery rates were significantly higher in gas-permeable bags than in non-gas-permeable bags (56.4±10.1% in gas-permeable bags vs. 20.5 ± 20.6% in non gas-permeable bags; p < 0.01) (Fig. 1C). There were no significant correlations between the islet recovery rate and culture duration (Fig. 2A) or between the islet recovery rate and shipping duration (Fig. 2B).

(A) There was no correlation between culture duration before shipping and the islet recovery rate. (B) There was no correlation between shipping duration and the islet recovery rate. Shipping times ranged from 37 to 57 h.
Islet Purity
The mean pre- and postshipment islet purities were 48.8± 13.7% and 34.1 ± 23.1%, respectively, assessed by dithizone staining (Fig. 3A). A significant reduction in islet purity occurred when the islets were shipped in the non-gas-permeable bags (from 50.4 ± 6.4% to 25.9 ± 15.6%; p < 0.05) (Fig. 3B), but not in the gas-permeable bags (from 47.7 ± 18.6% to 40.2 ± 28.2; NS) (Fig. 3B).

(A) Islets after shipment (dithizone dye, original magnification 40x). (B) The postshipping islet purity was significantly decreased in the non-gas-permeable bags. The purity in the gas-permeable bags did not decrease significantly.
Islet Viability
The mean pre- and postshipment islet viabilities were 93.0 ± 4.1% and 90.9 ± 3.5%, respectively (Fig. 4A). Islet viability was not significantly reduced in either the gas-permeable bags (from 95.0 ± 2.9% to 92.5 ± 2.6%; p = 0.18) (Fig. 4B) or in the non-gas-permeable bags (from 91.4 ± 4.6% to 89.7 ± 3.5%; p = 0.12) (Fig. 4B). The two cases in which islets were not recovered were excluded.
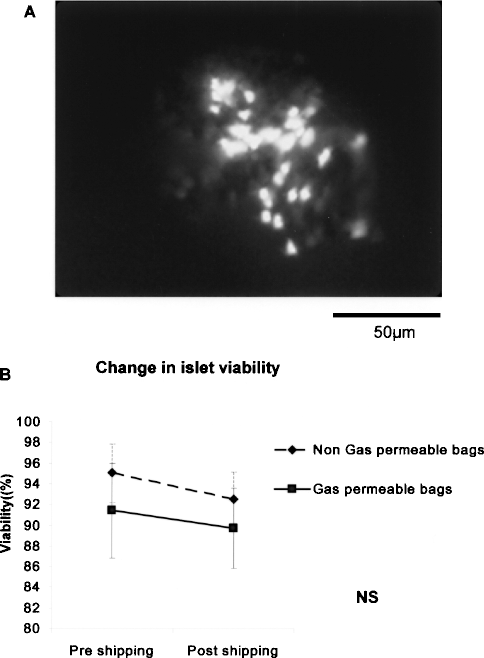
(A) Postshipping islet with FDA/PI dye (original magnification 400x). (B) There was no significant difference in islet viability between the gas-permeable and non-gas-permeable bags.
Results for Pre- and Postshipping Static Incubation
The mean stimulation index (SI) was 3.1 ± 0.9 at preshipping and 2.3 ±1.0 postshipment. The reduction of SI was not significant (two cases in which islets were not recovered were excluded from this calculation) (Fig. 5A). Moreover, there was no significant difference for the SI ratio (postshipping SI/preshipping SI) in gas-permeable bags or in non-gas-permeable bags (excluding the two cases in which islets were not recovered) (Fig. 5B).

(A) There was no significant difference between the preshipping and postshipping mean SI. (B) No significant difference was detected between the SI ratios in gas-permeable and non-gas-permeable bags. SI ratio: postshipping stimulation index/preshipping stimulation index.
Results of Shipped Islet Transplantation
Fasting blood glucose (FBG, mg/dl) converted to normoglycemia at 60 days posttransplantation in one of the three STZ-induced nude mice (33.3%) that received 1,500 IE postshipping islets (Fig. 6). Initially, their FBGs decreased below normal criteria immediately after transplantation; however, they increased after 5 days posttransplantation. After 25 days posttransplantation, FBG of one transplanted mouse reached normal levels again.
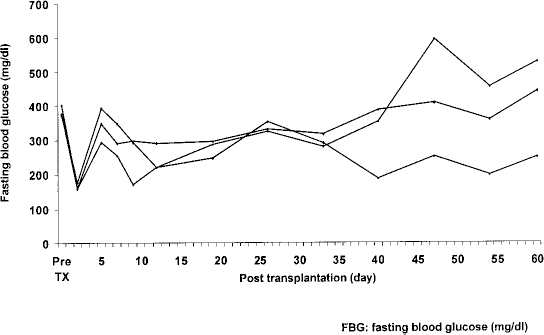
The measurement of fasting blood glucose (FBG) of 1,500 IE transplanted nude mice after shipment. All FBG converted to normoglycemia within 5 days postoperation; however, only one of the three mice achieved normoglycemia at 60 days postoperation.
Temperature Change During Shipment
We preserved islets in the refrigerator just before the shipping after packing; however, the temperature inside the shipping box changed dramatically even though we used the Styrofoam™ box (Fig. 7). Interestingly, the temperature did not change in the US or in the aircraft; however, after arrival in Japan, temperature increased to 35°C. It was found to be difficult to maintain constant temperature in the Styrofoam™ box without temperature modifiers.

The measurement of temperature change in shipped Styrofoam™ box. Temperature gradually increased after departure from our institute, and dramatically increased in Japan according to the log.
Discussion
In this study, we demonstrated that human islets could be transported to centers located more than 10,000 km from an islet isolation center using gas-permeable bags. The islet number was reduced by approximately one third; however, islet viability and functionality were maintained. Transported islets have been already used for several basic and preclinical studies in Japan.
Since the University of Alberta group demonstrated that islet transplantation could reverse diabetes status (23), this treatment has advanced significantly. However, there are several limiting issues with respect to islet transplantation. A severe donor shortage in some countries such as Japan is one of these issues (15). It is extremely difficult to establish a new donor source immediately, making this a serious situation for patients who require ICTx.
The pancreas is not considered a so-called ‘life saving organ” because patients can receive exogenous insulin. Nevertheless, some patients die while on the waiting list for islet or pancreas transplantation in Japan (24). Therefore, the international shipping of islets for clinical use should be seriously considered.
We have shown here that safe and effective islet shipment of over 10,000 km can be done with gas-permeable bags and the existing transportation system. This work demonstrates that institutes in countries that have a shortage of donor pancreata can have access to human islets for basic and preclinical research.
We have previously shown that gas-permeable bags maintain a stable temperature and stable O2 and CO2 pressure during domestic shipment (5). However, there are still possibilities of temperature and pressure change during international transportation. Actually our temperature monitoring demonstrated that sudden temperature change occurred when islets arrived in Japan. Thus, as Rozak et al. reported (21), we may need to consider special containers to ship islets internationally for further improvement of our results.
In our study, there were significant differences in islet recovery and islet purity between gas-permeable bags (Lifecell Tissue Culture Flasks; Baxter) and non-gas-permeable bags (Fenwal Transfer Pack Container; Baxter). However, there was no significant difference in islet viability between these two type bags. This is considered mainly by the differences of the speed of disappearance of the weak or almost dead cells in the bags during transportation. Anoxic conditions using a non-gas-permeable bag accelerate cell death if the cells are not healthy. In contrast, intact islet cells survive in both bags well, showing that cell viability was not significantly different between these two types of bags. The purity decreased in non-gas-permeable bags and this seemed due to the increase in dead cells. According to Ichii et al., there was a significant difference in islet viability between the gas-permeable bags (PL-732 polyolefin Lifecell; Nexell Therapeutics Inc., Irvine, CA) and the non-gas-permeable bags (5). In their experiments even using non-gas-permeable bags, the islet recovery rate was more than 50%; however, the viability was less than 60%. In our case, the recovery rate in a non-gas-permeable bag was less than 30% but viability was more than 80%. This suggested that long-distance transportation might kill all weak islets, allowing only robust islets to survive.
Moreover, consideration should be given to the influence of culture duration before shipment. Many centers have recently reported privilege of cultured islets for ICTx (2, 4, 7), although the culture conditions are still controversial. However, in this study, there was no correlation between culture duration and islet recovery rate.
Viability assessed by FDA/PI and stimulation index of static incubation showed transported islets were highly viable. However, the shipped islets could convert only one third (33.3%) of drug-induced diabetic nude mice to normoglycemia. This low success rate of in vivo study suggested that 1,500 IE were marginal number after international shipping. Further in vivo experiment is necessary to evaluate the functionality before considering clinical application of internationally shipped islets. In addition, it is necessary to perform endotoxin assay and microbiological assessment for clinical purpose.
To conclude, we showed that islets could be shipped internationally over 10,000 km and longer than 48 h in gas-permeable bags. These shipped islets recovered their numbers, viability, and function in a research setting. Further quality assurance would make it possible to use such islets in the clinical setting, especially for the countries like Japan that have donor shortages.
Footnotes
Acknowledgments
The authors thank Drs. Carson Harrod and Steven Phillips for critical review. This research is partly funded by All Saints Health Foundation.
